Abstract
Background
Many patients with migraines suffer from allergies and vice versa, suggesting a relationship between biological mechanisms of allergy and migraine. It was proposed many years ago that mast cells may be involved in the pathophysiology of migraines. We set out to investigate the relationship between mast cell activation and known neurogenic peptides related to migraine.
Methods
Cultured human mast cells were assayed for the presence of neuropeptides and their receptors at the RNA and protein level. Immunohistochemistry analyses were performed on tissue resident and cultured mast cells. Mast cell degranulation assays were performed and pituitary adenylate cyclase-activating polypeptide (PACAP) activity was measured with a bioassay.
Results
We found that cultured and tissue resident human mast cells contain PACAP in cytoplasmic granules. No other neurogenic peptide known to be involved in migraine was detected, nor did mast cells express the receptors for PACAP or other neurogenic peptides. Furthermore, mast cell degranulation through classic IgE-mediated allergic mechanisms led to the release of PACAP. The PACAP released from mast cells was biologically active, as demonstrated using PACAP receptor reporter cell lines. We confirmed existing literature that mast cell degranulation can also be induced by several neurogenic peptides, which also resulted in PACAP release.
Conclusion
Our data provides a potential biological explanation for the association between allergy and migraine by demonstrating the release of biologically active PACAP from mast cells.
Introduction
The relationship between allergy and migraine was noted many decades ago (1). Many patients with migraines suffer from allergies, and vice versa. Food allergies (2–4), allergic rhinitis (5–8), asthma (6,9–11), and atopic dermatitis (10,12) have all been associated with increases in frequency and severity of migraine attacks. Examples of this association include that the vast majority of allergy patients with “sinus headache” complaints are found to have migraine (13), and individuals with the most severe asthma symptoms have the highest risk for the new onset of chronic migraine among people with episodic migraine (14). Since patients with migraines often have elevated levels of mediators associated with an allergic response, including immunoglobulin E (IgE), histamine, tumor necrosis factor-α, and prostaglandins (8,15,16), it was proposed that mast cells may be involved in the pathophysiology of migraines (17). In addition, it has been shown that immunotherapy (allergy shots), which leads to a decrease in circulating IgE and thus a potential decrease in the high affinity IgE receptor, Fc epsilon RI (FcɛRI)-mediated mast cell degranulation, resulted in a reduction in the frequency of migraines (18). Finally, patients with migraines and known food allergies were protected from migraine signs and symptoms when given the mast cell stabilizer sodium cromoglycate in association with foods previously identified as allergenic (19,20). However, prophylactic treatment for the prevention of acute migraine with leukotriene receptor antagonists (21) or anti-histamines (22,23) have failed to improve outcomes for patients. Overall, even though the biological mechanisms have remained elusive, the evidence suggests that allergic triggers and subsequent mast cell degranulation and release of inflammatory mediators may trigger or exacerbate a migraine.
A number of neuropeptides play essential roles in the activation of the trigeminovascular system, which is believed to be an essential step in the induction of migraine (24). Experimental treatments targeting either calcitonin gene-related peptide (CGRP) or its receptor have been successful in preventing episodic and chronic migraine in recent clinical trials (25–27). Another neuropeptide, PACAP, is also known to induce migraines. Much like CGRP, intravenous administration of PACAP causes brief headache episodes in healthy subjects and migraine-like attacks in individuals with a past history of migraine without aura (28,29). Additionally, plasma PACAP concentrations from migraineurs increase during migraine attacks relative to attack-free periods (30,31) and have been shown to be elevated during episodic cluster headaches (32). PACAP is a member of the vasoactive intestinal peptide (VIP) neuropeptide family and exists in two biologically active forms (PACAP-38 and PACAP-27), with PACAP-38 being the predominant form. Like VIP, the effects of PACAP are mediated through G-protein coupled receptors: vasoactive intestinal peptide receptor type 1 (VPAC1), VPAC2 and pituitary adenylate cyclase-activating polypeptide type 1 (PAC1) (33).
While migraines may be triggered through a variety of mechanisms, the importance of mast cell activation in migraine remains unknown. Taking the described associations between migraines and allergies into consideration, we hypothesized that allergen-specific IgE antibodies and activation of its receptor FcɛRI on mast cells could be involved. Therefore, we set out to investigate the relationship between mast cells and various neuropeptides by analyzing receptor and mediator expression in human mast cells. We found that human mast cells express negligible levels of the receptors for various neuropeptides; however, they express high levels of PACAP in cytoplasmic granules. Furthermore, mast cell degranulation through IgE/FcɛRI led to the release of PACAP, which we demonstrated to be biologically active by measuring the production of cAMP in PACAP receptor reporter cell lines. While we found no evidence of expression and release of any other neurogenic peptides from mast cells, we did confirm literature reports (34–36) about these peptides causing mast cell degranulation and release of PACAP. Our data provide a potential mechanistic link between mast cells and migraines through the release of the known migraine-inducing factor PACAP.
Materials and methods
Mast cell culture
Human umbilical cord blood-derived mast cells were produced by in vitro differentiation of positively selected cord blood CD34+ progenitor cells (Allcells) as previously described (37,38). Cells were cultured in Serum Free Expansion Media (SFEM Cat. # 09650, Stemcell Technologies) supplemented with Antibiotic/Antimycotic Solution (Thermo Scientific) and growth factors, 200 ng/ml stem cell factor (SCF, produced in-house) and 200 ng/ml interleukin-6 (IL-6, produced in-house); for some experiments, mast cells were differentiated using 200 ng/ml SCF and 200 ng/ml interleukin-4 (IL-4, produced in-house). During the first three weeks, 20 ng/ml Flt3L (Peprotech) was included in the culture media.
Gene array
RNA was extracted from cultured human mast cells using RNeasy Protect Kit (Qiagen) and gene expression profile studies were performed on six replicates using the HG-U133 Plus 2.0 Array representing 14,500 well-characterized human genes according to manufacturer’s instructions (Affymetrix).
Flow cytometry
Cells were stained with anti-neurokinin 1 receptor (NK1R)-APC (R&D Systems), anti-FcɛRI (Biolegend), or anti-VPAC1 and anti-VPAC2 (Alomone Labs) followed by anti-rabbit IgG APC (Invitrogen) in FACS buffer. Propidium iodide was added to the cell suspension to assess cell viability. Multicolor flow cytometry was performed using Fortessa (BD Biosciences), and data were analyzed with FlowJo software (Flowjo Inc).
Immunofluorescence staining
Aliquots of 300,000 mast cells were centrifuged at 400 g for 5 min onto glass slides. Cytospins were dried and fixed with formalin/methanol. For tissue sections, paraffin embedded sections derived from biopsies were dewaxed and rehydrated. Antigen retrieval was performed using Target Retrieval Solution (DAKO) following manufacturer’s instructions. Immunofluorescence staining was performed using mouse anti-tryptase (Clone G3, Millipore), rabbit anti-PACAP (recognizing both PACAP-27 and PACAP-38; LSBio, Cat.#LS-C38027), Alexa Fluor 488 labeled goat anti-mouse IgG (Invitrogen) and Alexa Fluor 594 labeled goat anti-rabbit IgG (Invitrogen). The slides were covered using DAPI Fluoromount-G mounting media (Southern Biotech, Birmingham, AL). Stained slides were examined using a fluorescence microscope (Nikon) and pictures were taken with a Digital Sight DS-Ri1 digital camera.
Mast cell degranulation assay
Mast cells were sensitized with IgE anti-DNP chimeric antibody (Serotec) at 1:50 dilution for 4 hr, washed, suspended in Tyrode’s buffer (130 mM NaCl, 5 mM KCl, 1.4 mM CaCl2, 1 mM MgCl2, 5.6 mM glucose, 10 mM HEPES and 0.1% BSA, pH 7.4) and stimulated for 30 min with DNP-OVA (Sigma). Alternatively, a titration of the proteins PACAP-38 (Eli Lilly), VIP (Tocris), Substance P (SP, Tocris), or CGRP (Bachem) was used to activate them. Mast cells were centrifuged for 5 min at 200 g and supernatants were collected. For tryptase and PACAP release, 50,000 mast cells were used. Total release was obtained by freeze/thawing mast cells. Tryptase content in cell lysates and supernatants was measured as previously described (39). Percent (%) release in the supernatants was calculated using the following equation: (Experimental tryptase release – vehicle control tryptase release)/(total tryptase release – vehicle control tryptase release) × 100. PACAP was measured in supernatants using commercially-available PACAP-38 assay following the manufacturer’s protocol (Peninsula Laboratories International, Inc.) with % PACAP release calculated in a similar manner to the tryptase release.
PACAP cAMP assay
Using stably transfected human VPAC2-CHO or PAC1-CHO cells (Eli Lilly), PACAP-induced cAMP levels were measured with a commercially-available kit (cAMP femto 2, Cisbio Assays). A VPAC2 receptor antagonist (Myristoyl-(Lys12, 27, 28)-VIP-gly-gly-thr; PG99-465, Bachem) was used in assays at a final concentration of 5 µM. For cAMP assays, protease inhibitor (cOmplete ULTRA Tablets, Roche) was immediately added to the cell degranulation supernatants and included in the total mast cell lysates.
Statistical analysis
Statistical analyses were performed using GraphPad Prism software (version 6). All graphs show mean ± SD. A p value <0.05 was considered significant.
Results
Neuropeptide receptor expression by human mast cells
Relative receptor expression in cultured mast cells a .

Relative mean RNA expression ± standard deviation from six replicates of cultured mast cells differentiated using SCF and IL-6 on the Affymetrix Array. Values below 200 are considered not expressed.

Neuropeptide receptor expression on human mast cells. Surface expression of FcɛR1, VPAC1, VPAC2, and NK1R was measured by flow cytometry on human mast cells.
Relative mediator expression in cultured mast cells a .

Relative mean RNA expression ± standard deviation from six replicates of cultured mast cells differentiated using SCF and IL-6 on the Affymetrix Array. Values below 200 are considered not expressed.
Human mast cells’ degranulation in response to SP, VIP or PACAP
To further investigate the link between neuropeptides and mast cell degranulation we tested whether various neuropeptides were able to induce degranulation of the mast cells. SP, VIP, and PACAP were able to induce mast cell degranulation in a dose-dependent manner as determined by tryptase release. SP was the most potent inducer, followed by VIP and PACAP (Figure 2). Although VIP and PACAP were comparable in their ability to induce degranulation, it required relatively high concentrations of the peptides to induce this response. Little degranulation was observed when mast cells were treated with CGRP (Figure 2). Although these data confirmed reports in the literature, it is remarkable that mast cells are able to respond to these neuropeptides in the absence of clearly detectable expression of receptors specific for these peptides.
PACAP, VIP, and SP induce mast cell degranulation. Human mast cells were treated with various concentrations of PACAP, VIP, SP or CGRP in triplicate for 30 minutes and tryptase release was measured by enzyme assay. Results show mean percent release ± SD relative to total tryptase levels.
Human mast cells express PACAP
To understand whether human mast cells could produce various neuropeptides, we looked at their expression by mRNA analysis. We found that VIP, CGRP and tachykinin, the SP precursor, were not expressed (Table 2). In contrast, human mast cells expressed very high levels of PACAP mRNA, at levels similar to histidine decarboxylase, the enzyme that catalyzes the production of histamine. To confirm this observation at the protein level, we evaluated PACAP expression by immunofluorescence. We found that stem cell-derived mast cells stained positive for PACAP within their cytoplasmic granules (Figure 3). To ensure this finding was not an artifact of cultured stem cell-derived mast cells, we analyzed PACAP expression in different sections of tissues from normal human donors. Mast cells were identified in sections of human colon, small intestine and skin by positive tryptase staining (Figure 4(a)). Through concomitant staining we observed that granules of these normal tissue-resident mast cells also expressed PACAP (Figure 4(b)). No staining was observed with the secondary-only antibody control or when the PACAP antibody was pre-absorbed with PACAP-38 (data not shown). These data show that PACAP is prominent in the granules of mast cells and due to its location, suggest that PACAP could be released by signals that induce degranulation of mast cells.
Cultured human mast cells contain PACAP. Representative staining for tryptase (green) and PACAP (red) in cultured human mast cells, with nuclei stained using DAPI (blue). The overlay demonstrates that tryptase and PACAP are present in mast cell granules (magnification: 400×). PACAP co-localization with tryptase in normal human mast cells in skin, colon, and small intestine. (a) Representative pictures of tryptase (green) and PACAP (red) staining in tissue sections of colon, small intestine and skin from normal human donors. Original magnification: 400×. Arrows identify tissue resident mast cells, and boxes identify cells selected for enlargement in (b).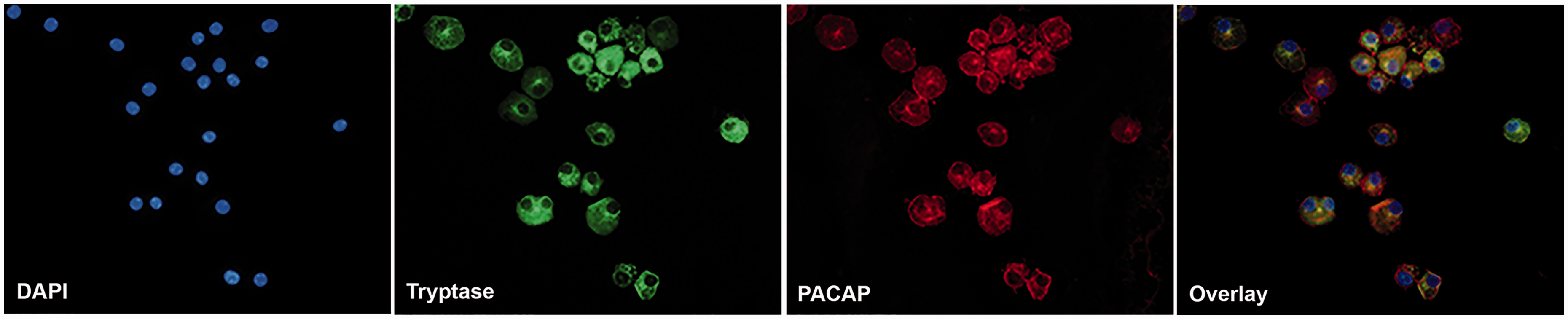

Crosslinking of FcɛRI or SP activation of human mast cell released bioactive PACAP
To understand if PACAP could be released via known mechanisms, we performed classic FcɛRI-mediated degranulation assays on stem cell-derived human mast cells. FcɛRI crosslinking resulted in a dose-dependent release of tryptase, which was used as a positive control for degranulation (Figure 5(a)). PACAP was released from mast cells in a similar dose-dependent manner, consistent with the observation that PACAP and tryptase are located in granules. We hypothesized that degranulation induced by neuropeptides (Figure 2) through non-IgE dependent mechanisms would also lead to PACAP release. Indeed, we found that both tryptase and PACAP were released via SP stimulation in a dose-dependent manner (Figure 5(b)). Moreover, we found that human mast cells cultured with SCF and IL-4 (instead of IL-6) contained the same amount of total PACAP (Figure 5(c)), which was also released in a similar manner following FcɛR1 crosslinking (Figure 5(d)). Together, these data show that mast cell degranulation results in the release of PACAP.
PACAP release from mast cells through crosslinking of FcɛRI and SP activation. Human mast cells were pretreated with anti-DNP-IgE and then challenged with various concentrations of DNP-OVA in triplicate for 30 minutes. Tryptase release was measured by enzyme assay, and PACAP release was measured by ELISA (a). Mast cells were treated with various concentrations of SP in triplicate for 30 minutes. Tryptase release was measured by enzyme assay and PACAP release was measured by ELISA (b). Total PACAP contained in mast cells grown in SCF and IL-6 (black bars) or SCF and IL-4 (grey bars, (c)) and anti-DNP-IgE/DNP-OVA challenge in triplicate for 30 minutes (d) as measured by PACAP ELISA. Results show mean percent release ± SD relative to total levels.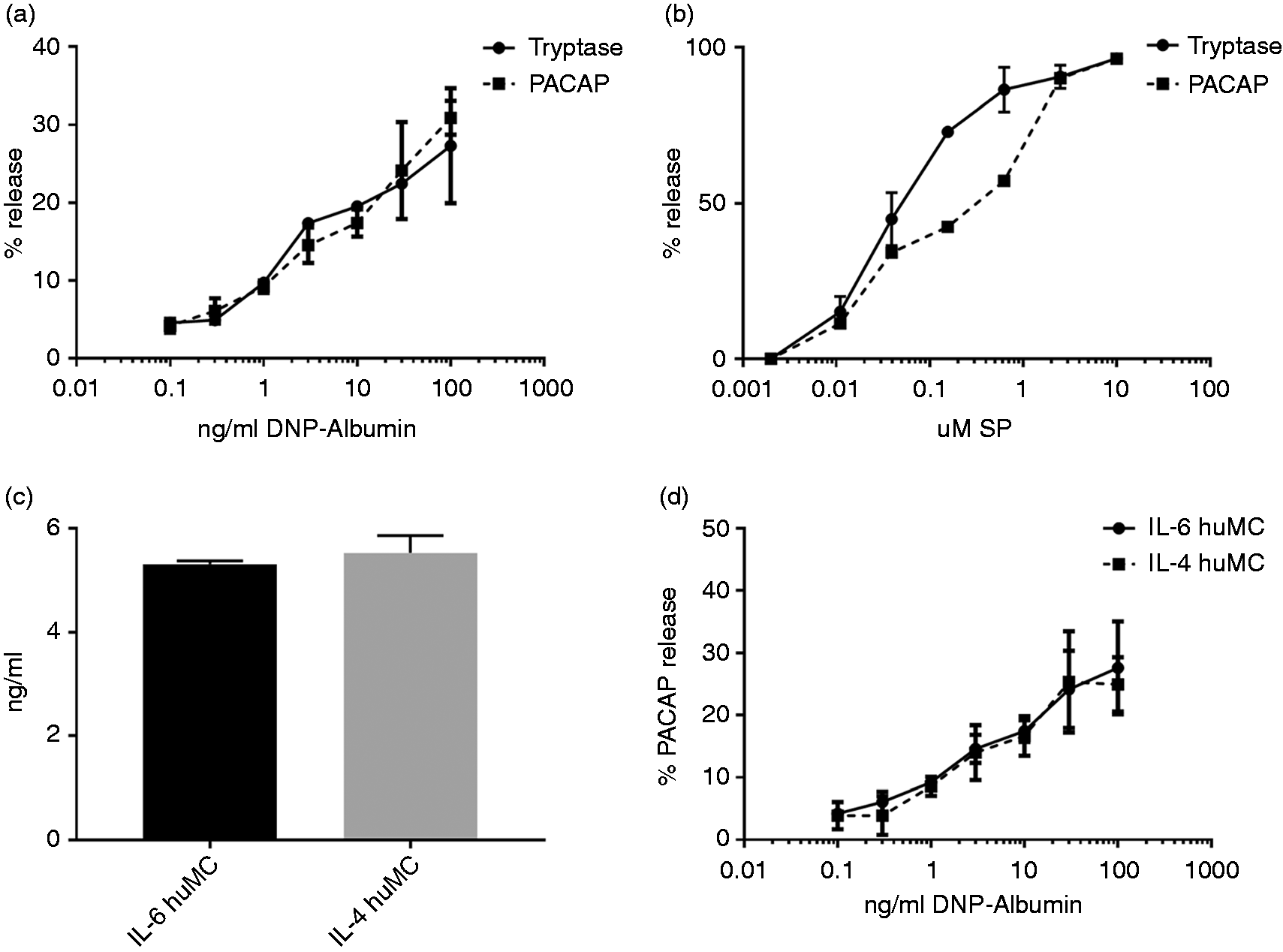
The binding of PACAP to its receptors stimulates adenylate cyclase and subsequently increases the cAMP level in target cells. To determine whether PACAP released from mast cells by degranulation was bioactive, we utilized cAMP reporter CHO cell lines expressing the VPAC2 receptor (CHO-VPAC2) or PAC1 (CHO-PAC1). Stimulation of these reporter cell lines with total mast cell lysates obtained by freeze/thawing of mast cells induced cAMP (Figure 6(a), black bars). Supernatants obtained from mast cells after IgE crosslinking also resulted in cAMP production in the CHO-VPAC2 and CHO-PAC1 cell lines (Figure 6(a), grey bars). Under the same conditions, the CHO parental cell line showed no activity (Figure 6(a)), demonstrating that cAMP production was due to VPAC2 or PAC1 activation. To further demonstrate that cAMP production was mediated through PACAP-specific receptors, CHO-VPAC2 cells were activated under different conditions in the presence or absence of the VPAC2 receptor specific antagonist PG99-465. The antagonist PG99-465 blocked cAMP activity induced by recombinant PACAP, the total mast cell lysate, and supernatant from IgE-treated mast cells (Figure 6(b)). Together, the data demonstrate that mast cells release biologically active PACAP upon degranulation.
PACAP released from human mast cells induces cellular activation through VPAC2 and PAC1. Total mast cell lysate (black bars) and supernatant from FcɛRI-crosslinked mast cells (grey bars) induced cAMP in CHO-VPAC2 or CHO-PAC1 cells compared to mast cell supernatant from cells without IgE (white bars). No cAMP induction was observed in the CHO parental cells (a). Treatment of the cells with VPAC2 specific inhibitor, PG99-465, blocked the activity of PACAP-38, total mast cell lysate and supernatant from FcɛRI-crosslinked mast cells mast cell supernatants in the CHO-VPAC2 cAMP reporter cell line assay (b). All data are presented as mean ± SD (two-tailed unpaired t-test; *p < 0.01).
Discussion
The correlation between allergy and migraine has long been noted, although the precise mechanisms that link allergy to migraine have remained elusive. It is well demonstrated that mast cell degranulation and release of inflammatory mediators may result in more frequent and/or severe migraine attacks. Here, we show that both cultured and tissue-resident human mast cells express PACAP in granules and that bioactive PACAP can be released from mast cells following FcɛRI activation by crosslinking with IgE. These findings provide a rationale for the observed correlation between allergy and migraine. Moreover, we demonstrate that mast cell degranulation and PACAP release can also be induced by certain neurogenic peptides, representing a pseudo-allergic pathway. Since it has been demonstrated that PACAP infusion can trigger migraines (28,29), we propose that this finding provides a potential mechanistic explanation of how mast cell degranulation could contribute to migraines.
The current study focused on the presence of PACAP in cultured mast cells grown under specific conditions and its release upon induced degranulation. Mast cell phenotypes are not fixed, but rather driven by tissue residency and responses to local stimuli. As such, mast cells can vary greatly in morphological and histological characteristics including basic traits used for conventional classification, such as expression of tryptase and/or chymase-containing granules. In this study, we found that human mast cells cultured with SCF and IL-6 or SCF and IL-4 produced similar levels of PACAP, which was released following FcɛR1 crosslinking Compared to IL-6 cultured mast cells, IL-4 promotes further maturation of human mast cells including decreased c-kit expression, along with increased expression of FcɛR1, intercellular adhesion molecule-1, lymphocyte function-associated antigen-1, and chymase (42,43). Our IHC results demonstrate that mast cells in different locations (colon, small intestine, and skin) all express PACAP in granules. Interestingly, our results differ from a previous report that did not show PACAP expression in human dermal mast cells (44). However, the Seeliger et al. study (44) focused on PACAP expression in tissues from patients with urticaria, whereas our study used samples from non-diseased tissue. It will be interesting to explore the effect of disease state on the expression of PACAP or other mediators in mast cells. Additionally, mast cell activation can be triggered and influenced by a variety of different stimuli and receptors. Mast cells exposed to pro-inflammatory cytokines or SCF often show increased responses to subsequent signals, whereas factors such as heparin or nitric oxide can inhibit mast cell activation (45,46). The presence of factors like these could influence the mast cell response depending upon the tissue environment. An example of this is the observation that rat dura mast cells have been shown to be more responsive to CGRP-mediated degranulation than rat peritoneal mast cells (47). Therefore, it is possible that tissue resident mast cells are shaped by mediators and activation threshold based on their location and microenvironment.
The clinical importance of PACAP and specific high-affinity PACAP receptors in migraine is still unproven; however, infusion of PACAP in the periphery triggers migraine attacks (28,29), suggesting that PACAP receptors in peripheral tissue are important. As such, IgE-mediated degranulation of mast cells leading to PACAP release in the periphery would also be expected to trigger a migraine episode in susceptible individuals, making the presence of PACAP in brain-resident mast cells interesting, but not necessarily critical to the PACAP-migraine relationship. Further complexity of the system is evident, in that the explicit role of the individual high-affinity PACAP receptors in migraine is still uncertain. VIP has a lower affinity for the PAC1 receptor than PACAP and was less effective at inducing migraine-like attacks when infused intravenously in migraine suffers (29), suggesting PACAP interaction with PAC1 could be responsible for the induction of migraine circuits. However, other work done in migraine patients without aura has suggested that both the PAC1 and VPAC1 receptor may partially mediate nociceptive activation of central trigeminovascular neurons (48). Consequently, more studies are needed to understand which PACAP receptors in either the central nervous system or the periphery are involved in driving the pathophysiology of migraine.
Neurogenic inflammation is hypothesized to be a key pathological process in migraines. It has been shown that the trigeminal neurons in humans express CGRP with a lesser amount of SP and PACAP (49,50), and it has been proposed that secretion of these neuropeptides can induce mast cell degranulation and be a contributing factor in migraine (34). Studies have shown that intradermal injections of PACAP into human skin can cause the release of histamine and that PACAP can induce the degranulation of rat mast cells (34,36). As shown elsewhere, neuropeptides including SP, VIP and to a lesser extent, CGRP, can activate mast cells leading to degranulation (35). Our data supports these reports, since we observed that SP, PACAP, and VIP are all able to induce mast cell degranulation, whilst CGRP induced the least amount of degranulation. It has been demonstrated that other peptides, including β-defensins and cathelicidins, can induce mast cell degranulation through MRGPRX2 (51) and therefore are involved in pseudo-allergic responses. Additionally, several other peptides have been identified as MRGPRX2 ligands, including PACAP and VIP (41). Fujisawa et al. have shown that MRGPRX2 is present on human skin mast cells and that lentiviral small hairpin RNA–mediated knockdown of MRGPRX2 resulted in substantial inhibition of SP-induced mast cell degranulation (52). Taken together, these findings suggest that in addition to SP, the other neuropeptides could use MRGPRX2 to promote mast cell degranulation. Additionally, we found high levels of MRGPRX2 and very low expression levels of all the PACAP-specific receptors, including PAC1, which was previously hypothesized to be key in mediating PACAP mast cell activation (34). However, other groups have suggested that PACAP-induced mast cell degranulation is independent of high affinity PACAP receptors. Indeed, it was previously proposed that PACAP acts through a highly promiscuous low affinity receptor, which accepts several positively charged peptides (36). The abundant levels of MRGPRX2 mRNA found in human mast cells, along with the recent reports on the physiological role of MRGRPX2, support the idea that MRGRPX2 is this promiscuous receptor mediating neuropeptide-induced mast cell degranulation.
It should be noted that MRGPRX2-mediated degranulation requires relatively high concentrations of peptides, and thus may depend on very specific local circumstances to be biologically relevant. A recent study (53) showed differential activation, timing, and degranulation patterns from mast cells after crosslinking of FcɛR1 or through MRGPRX2 activation. High concentrations of neuropeptides were required to trigger mast cell degranulation, whereas the threshold for FcɛRI activation was relatively low. However, degranulation through these pseudo-allergic pathways occurred more swiftly than degranulation induced by FcɛR1. In our study, we demonstrate that both types of stimulation induced PACAP release, suggesting that mast cells can be involved in the induction of migraines through classic IgE-mediated allergic responses as well as through pseudo-allergic mechanisms. Interestingly, an increase in migraine incidence has been reported in diseases associated with activated mast cells that occur in the absence of elevated serum IgE, such as interstitial cystitis and irritable bowel syndrome (54). Based on this, we could speculate that the various pathways that lead to mast cell degranulation all cause the release of PACAP and may thus increase the probability of a migraine attack.
Migraine remains a complex disorder with multiple potential causes. Recent clinical trial results demonstrate a role for the CGRP pathway (25–27), since its blockade resulted in a high degree of efficacy in significant groups of patients. In our study, we found no direct relationship between mast cells and CGRP, suggesting that migraines involving mast cell activation, either via classical IgE or pseudo-allergic pathways, involve a different pathophysiological mechanism. Interestingly, although SP can induce mast cell degranulation, a number of NK1-receptor inhibitors failed to alleviate the signs and symptoms of migraine when tested in clinical trials (55,56) when these were tested as abortive treatments. Overall, and based on our results, we speculate that SP can induce degranulation via MRGPRX2 leading to PACAP release, and this might be the actual link between SP and migraines rather than NK1 receptor activation. However, at this time, clinical evidence that neutralization of either PACAP or its receptors will provide relief to patients suffering from migraines is lacking. Therefore, the clinical relevance of PACAP in migraine is still unknown, but the current findings provide a potential biological mechanism of how mast cell activation and subsequent PACAP release could be linked to migraine.
Footnotes
Article highlights
In the investigation of the role of mast cells in migraine pathology, we found that human mast cells contain PACAP in their cytoplasmic granules.
Bioactive PACAP can be released from mast cells by inducing degranulation.
This finding provides a potential explanation linking mast cell activation to migraine through the release of PACAP.
Acknowledgements
The authors would like to acknowledge Tatiyana Shiyanova, Linda Schirtzinger, Montanea Daniels, Cynthia Winings, John E Brandt, and Narayanan B Perumal for technical assistance, and Ching-Yun Veavi Chang for statistical assistance. Authors’ roles: Study design: AJO, APM, KWJ, MPJ, and RJB. Study conduct: AJO, SMM, and DD. Data analysis: AJO, SMM and DD. Data interpretation: AJO, SMM, APM, MPJ, KWJ and RJB. Draft of manuscript: AJO and RJB take responsibility for the integrity of the data analysis. All authors approve the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
