Abstract
Objective
To determine whether early treatment with sumatriptan can prevent PACAP38-induced migraine attacks.
Methods
A total of 37 patients with migraine without aura were enrolled between July 2018 to December 2019. All patients received an intravenous infusion of 10 picomole/kg/min of PACAP38 over 20 min followed by an intravenous infusion of 4 mg sumatriptan or placebo over 10 min on two study days in a randomised, double-blind, placebo-controlled, crossover study.
Results
Of 37 patients enrolled, 26 (70.3%) completed the study and were included in analyses. Of the 26 patients, four (15%) developed a PACAP38-induced migraine attack on sumatriptan and 11 patients (42%) on placebo (p = 0.016). There were no differences in area under the curve for headache intensity between sumatriptan (mean AUC 532) and placebo (mean AUC 779) (p = 0.35). Sumatriptan significantly constricted the PACAP38-dilated superficial temporal artery immediately after infusion (T30) compared with infusion of placebo (p < 0.001).
Glossary
AUC: area under the curve; cAMP: cyclic adenosine monophosphate; HT: hydroxytryptamine; MAP: mean arterial blood pressure; PACAP: pituitary adenylate cyclase-activating polypeptide; PACAP27: pituitary adenylate cyclase-activating polypeptide-27; PACAP38: pituitary adenylate cyclase-activating polypeptide-38; STA: superficial temporal artery; CGRP: calcitonin gene-related peptide; MO: migraine without aura; MA: migraine with aura.
Introduction
The pathogenesis of migraine is incompletely understood but likely involves activation of the trigeminovascular system (1) and modulation of nociceptive transmission by various signaling pathways (2). Interest in these pathways has led to the identification of signaling molecules responsible for the genesis of a migraine attack (2), and these include calcitonin gene-related peptide (CGRP) and pituitary adenylate cyclase-activating polypeptide (PACAP) among others (3,4). PACAP has garnered considerable attention as both its isoforms, PACAP38 and PACAP27 (5), induce migraine attacks in individuals with migraine (4,6), while healthy volunteers report mild headache at the most (4,7). The mechanism of action is believed to either directly or indirectly involve activation and/or sensitisation of trigeminal sensory afferents and the trigeminal ganglion (8,9). Indirect effects are likely mediated by dilation of extracerebral arteries (10) and/or mast cell degranulation (11,12).
Although it is well-established that PACAP has an important role in the genesis of a migraine attack (13), it remains unknown whether migraine-specific medications abort attacks by inhibition of PACAP-mediated mechanisms. PACAP38 acts via three G-protein-coupled receptors: VPAC1, VPAC2, and PAC1 (14), which are distributed throughout the central and peripheral nervous systems. Activation of either of the three receptors leads to increased cyclic adenosine monophosphate (cAMP) (15). PACAP38, but not PACAP27, also targets the orphan-receptor, MrgB3, which leads to mast cell degranulation (16). Interestingly, flushing and puffing after infusion of PACAP38 lasted longer than after PACAP27 (6), and these reactions may be driven by the binding to this receptor. The PAC1 receptor has been suggested as a novel target for migraine, but in a recent randomised clinical trial (phase 2) monoclonal antibodies against PAC1 failed in preventing migraine (17). This suggests that a new approach for targeting PACAP may be warranted, and future studies could focus on antibodies against PACAP (18). In this context, sumatriptan – a 5-HT1B/1D receptor agonist – is of great interest as it is a well-established migraine-specific abortive medication (19) and there is evidence that suggests that sumatriptan inhibits release of PACAP within the trigeminovascular system (20). It has never been investigated whether sumatriptan can prevent PACAP-induced migraine.
We hypothesised that sumatriptan would prevent PACAP38-induced migraine in patients with migraine without aura. To test this hypothesis, we conducted a randomised, double-blind, placebo-controlled, two-way crossover study.
Methods
Study design and patients
Patients aged 18–60 years were recruited from the Danish Headache Center outpatient clinic and via the online Danish recruitment forum, www.forsoegsperson.dk, between July 2018 and December 2019. Patients had to have a verified diagnosis of episodic migraine without aura (MO) according to the International Classification of Headache Disorders, 3rd edition (21) with an attack frequency of 1–4 attacks per month. No migraine preventive medications were allowed. Use of safe contraceptive methods in women of childbearing potential was required. Exclusion criteria were other primary headaches (except episodic tension-type headache less than 5 days per month), history of serious somatic or psychiatric disease, pregnant or nursing women, daily intake of medication except contraceptives, or drug misuse. Analgesics were not allowed for 48 h. This study was part of a larger parent study that was approved by the Regional Health Research Ethics Committee of the Capital Region (H-17023750) and by the Danish Data Protection Agency. Additional data from the parent study will be published elsewhere. All patients gave written consent after receiving detailed oral and written information. The study was conducted at the Danish Headache Center in accordance with the Helsinki II Declaration of 1964, with revisions until the Brazil conference in 2013 (22). The study is registered at ClinicalTrials.gov (NCT03881644).
Experimental design
All patients received an intravenous infusion of 10 picomole/kg/min PACAP38 (Bachem AG, Bubendorf, Switzerland) over 20 min on 2 days separated by at least 7 days. This dose of PACAP38 is known to induce migraine in patients with MO (10,23). Patients were subsequently allocated to receive an intravenous infusion of sumatriptan 4 mg (Imigran® injection, Glaxo Wellcome Operations, Barnard Castle, UK) or placebo (isotonic saline) over 10 min on two separate study days in a randomised, double-blind, placebo-controlled, crossover study. Sumatriptan has been administered intravenously in experimental settings in doses up to 96 micrograms/kg, with the dose of 64 micrograms/kg being most effective (24). This dose corresponds to 3.84 mg in a 60 kg person. For this reason, we chose a standard dose of 4 mg in this study. Intravenous administration of 4 mg sumatriptan has previously resulted in pain relief within 10–30 minutes (24) and was therefore chosen in this study to ensure rapid uptake. Patients were informed that PACAP38 might induce migraine, but timing of migraine was not discussed. Patients were informed that sumatriptan was an anti-migraine drug that may or may not affect PACAP38-induced migraine. Patients reported to the laboratory headache free for 48 h and migraine free for 72 h. Coffee, tea, cocoa, and other methylxanthine-containing foods or beverages, as well as tobacco, were not allowed for at least 6 h before start of the study. Patients were placed in a supine position and on the first study day, a brief medical and neurological examination and 12-lead electrocardiogram was performed before study procedures. Subsequently, a venous catheter (BD Venflon®, Franklin Lakes, NJ) was inserted into both antecubital veins for infusion. Patients were monitored with ECG (Philips, M1971A; IntelliVue, MP5), and oximeter, heart rate and blood pressure were recorded with an auto-inflatable cuff (Philips, Easy Care Adult Long, Ref M4556B; IntelliVue, MP5). If any headache occurred, headache intensity was recorded on a numerical rating scale (NRS) from 0–10 (0, no headache; 1, very mild headache [including a feeling of pressing or throbbing]; 10, worst imaginable headache) (25). Diameter of the frontal branch of the superficial temporal artery (STA) was measured with a high-resolution ultrasonography unit (20 MHz, band with 15 Mhz; Dermascan C; Cortex Technology, Hadsund, Denmark) (26,27) on the side where patients most frequently reported headache, and the right side if equally affected. At each time point, four measurements were performed and the mean of these is reported. All variables were recorded at fixed time points throughout the study (Figure 2).
Facial skin blood flow
Changes in facial skin blood flow were measured by laser speckle contrast imager (Moor Instruments speckle contrast blood assessment, moor Full Laser Perfusion Imager). The whole face was scanned with the laser speckle at a distance of 30 cm from the skin. The speckle was set to a bandwidth of 25 Hz, exposure time to 8.3 ms/pixel, time between images to 5 sec and number of images to a maximum of 5000 images to allow for 4 h recording. Images were marked every 10 min after asking patients to close their eyes and remain still for at least 5 sec in order to obtain correct images. A scaled color-coded live flux image was obtained; high perfusion was imaged by a red color and blue color was low perfusion. The use of laser speckle contrast imaging has previously been described (28).
Facial skin temperature
Thermal pictures of the face were recorded with a high-resolution science grade LWIR camera FLIR A655sc with a 25° lens. The camera records images with 640 × 480 pixel, a temperature range of −40° to 650°C and thermal sensitivity of 0.03°C. Images were recorded with FLIR ResearchIR 4.10. For obtaining the pictures, the camera was fixed in a vertical position at a distance of approximately 50 cm from the face to allow for the whole face to be in the picture. Patients were instructed to lie completely still during measurements. Images were taken every 10 min throughout the experiment. Analyses were performed in the program FLIR TOOLS by manually inserting points on each side of the face corresponding to the facial area of the three branches of the trigeminus (forehead, cheeks and chin). An additional temperature measurement was performed on the tip of the nose (Supplemental Figure 1). The measure point was matched to the size of the iris to adjust for differences in distances from which the pictures were taken.
Rescue medication
Patients were allowed to take anti-migraine medication at any time during the experiment; however, they were not allowed to take sumatriptan or any other triptans before at least 2 h post sumatriptan/saline infusion (until T155 after start of infusion of PACAP38) on either study day to avoid side effects of a double dose of sumatriptan. In case of unpleasant facial puffing provoked by PACAP38, patients could take antihistamine (cetirizine 10 mg) up to three times a day for up to 2 days or up to 100 mg of prednisolone.
Migraine attack criteria and reporting
Criteria for migraine attacks followed the same guidelines as previously used in experimental studies provoking migraine attacks (29). Criteria were based on the IHS criteria for MO (21) and should fulfill either of the following two:
Headache fulfilling criteria C and D for MO according to the IHS criteria. Headache described as mimicking the patient’s usual migraine and treated with acute migraine medication (rescue medication).
PACAP38 infusion was started at T0 and lasted 20 min. Infusion of sumatriptan/placebo started at T25 and ended at T35. As PACAP38 induces immediate headache followed by delayed migraine attacks, we excluded all patients who fulfilled migraine criteria prior to T40 on either of the study days.
Statistical analyses
Incidence of PACAP38-induced migraine attacks (T35 to T720) was reported on each study day and differences in incidence was calculated as categorical paired data using McNemar’s test. For headache intensity (NRS between T35 and T720), area under the curve (AUC) of NRS was calculated according to the trapezium rule (42) and AUC was compared between study drugs using the Wilcoxon signed rank test. Diameter of the STA, facial skin temperature and facial blood flow were evaluated using parametric testing with error bars represent interquatile ranges mixed effects models. We tested for the effects of drug as an interaction on the effect of time. The study was designed using PACAP38 on both days for all subjects, meaning that the effect of time on the measured parameters was actually PACAP38 mediated. For this reason, we tested the outcome variables (STA diameter, facial skin temperature and skin blood flow) in models with time as fixed effect, drug (sumatriptan/placebo) as interaction terms with time and patient as random effects.
Before data was incorporated in the above-mentioned models, facial skin blood flow was reduced to the mean perfusion rate calculated at each time point between T0 and T240 in each participant on each day. For facial skin temperature, the mean temperature was calculated at each time point between T0 and T240 before inclusion in the final statistical model. Heart rate and mean arterial blood pressure (MAP) is reported as relative changes from baseline.
Calculation of sample size was based on detection of a difference between sumatriptan and placebo in incidence of PACAP38-induced migraine attacks at a 5% significance (two-tailed) with 80% power (30). We assumed that induction rate on the placebo day would be 65% (4,31,32) and proposed that sumatriptan would lead to a 50% reduction in induction of migraine. We estimated that 19 patients should be included in a crossover experiment and therefore aimed to include at least 30 patients to ensure power in case of dropouts.
Primary endpoints were incidence of PACAP38-induced migraine attacks and headache intensity after sumatriptan and placebo. Secondary endpoints were the effect of sumatriptan on STA diameter.
Analyses were performed in R version 3.5.2 with stats and lme4 packages. p-values are reported as two-tailed with a 5% significance level.
Results
A total of 37 patients were enrolled in the study, of whom 26 (70.3%) completed both study days and were included in the final analyses (24 women [92.3%] and two men [7.7%]) (Figure 1). The median age was 26 years (range 20–46) and median weight was 65.5 kg (range 53–100). Median frequency of migraine attacks per month was 2 (range 1–4). A detailed description of usual attacks and headache characteristics on each of the two study days is listed in Table 1.

CONSORT flow diagram for crossover trials adjusted from the CONSORT 2010 statement: Extension to randomised crossover trials.
Clinical characteristics and associated symptoms of usual and PACAP38-induced migraine attacks and/or headaches in the 26 MO patients that were included in final analysis. Red text represents attacks that fulfill migraine criteria.
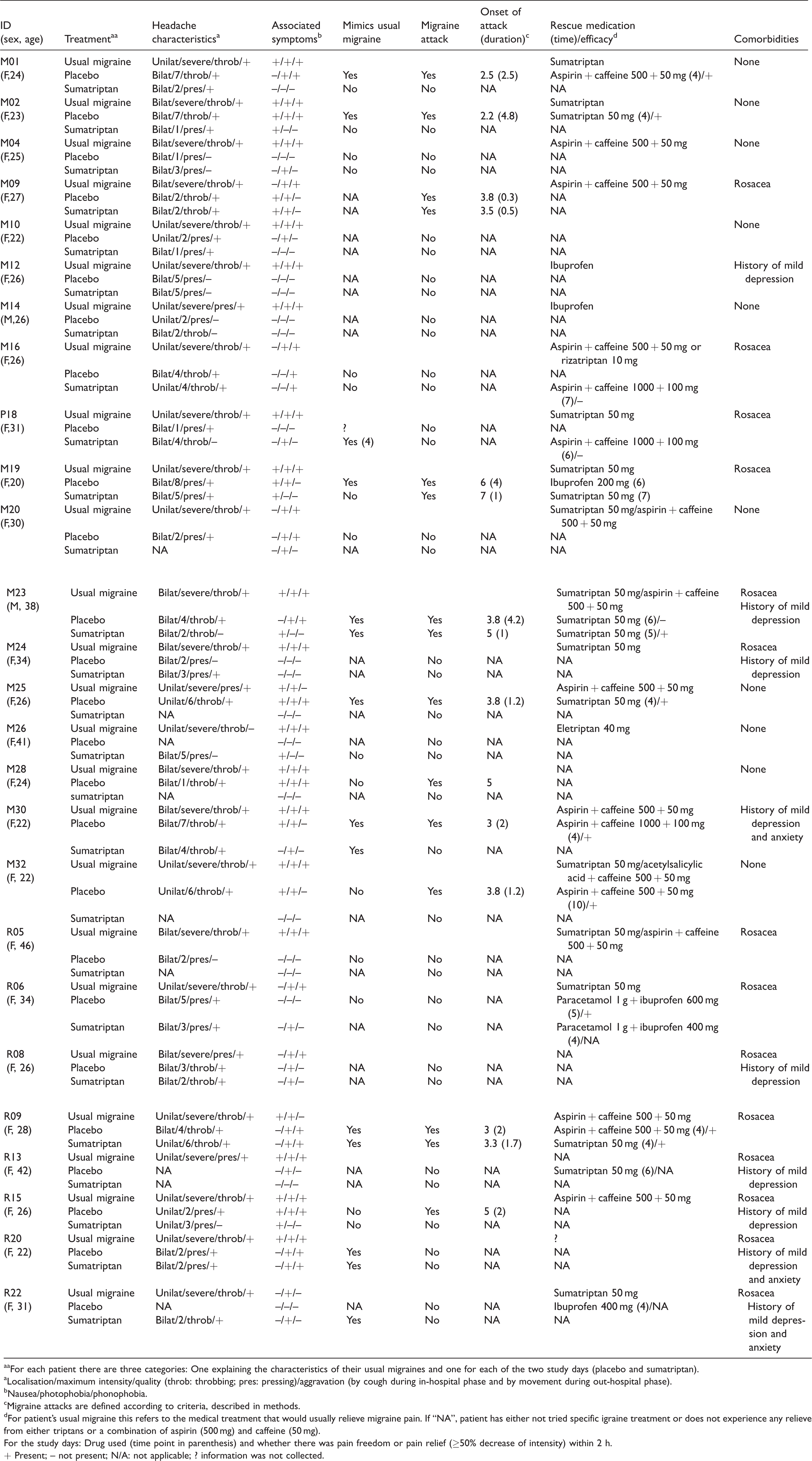
aaFor each patient there are three categories: One explaining the characteristics of their usual migraines and one for each of the two study days (placebo and sumatriptan).
aLocalisation/maximum intensity/quality (throb: throbbing; pres: pressing)/aggravation (by cough during in-hospital phase and by movement during out-hospital phase).
bNausea/photophobia/phonophobia.
cMigraine attacks are defined according to criteria, described in methods.
dFor patient’s usual migraine this refers to the medical treatment that would usually relieve migraine pain. If “NA”, patient has either not tried specific igraine treatment or does not experience any relieve from either triptans or a combination of aspirin (500 mg) and caffeine (50 mg).
For the study days: Drug used (time point in parenthesis) and whether there was pain freedom or pain relief (≥50% decrease of intensity) within 2 h.
+ Present; – not present; N/A: not applicable; ? information was not collected.

Timeline for the 240 min in-hospital phase of the experiment. STA: Superficial Temporal Artery.
Incidence of PACAP38-induced migraine attacks
Four of the 26 patients (15%) developed a migraine attack after treatment with sumatriptan, and 11 patients (42%) developed a migraine attack after placebo (p = 0.016, 95% CI [1.4: inf]). No patients developed attacks only on the sumatriptan day. Fifteen patients (58%) did not develop attacks on either of the two study days.
Incidence and intensity of headache
Nineteen patients (73%) reported headache after treatment with sumatriptan and 23 patients (88%) reported headache on the placebo day. Two patients only experienced headache on the sumatriptan day and five patients only experienced headache on the placebo day. One patient did not experience any headache on either of the two study days. We found no difference in AUC35-720 min for headache intensity over time between sumatriptan (mean AUC 532, 95% CI [322–741]) and placebo (mean AUC 779, 95% CI [454–1104]) (p = 0.35). Figure 3 shows individual headache intensities on the two study days.
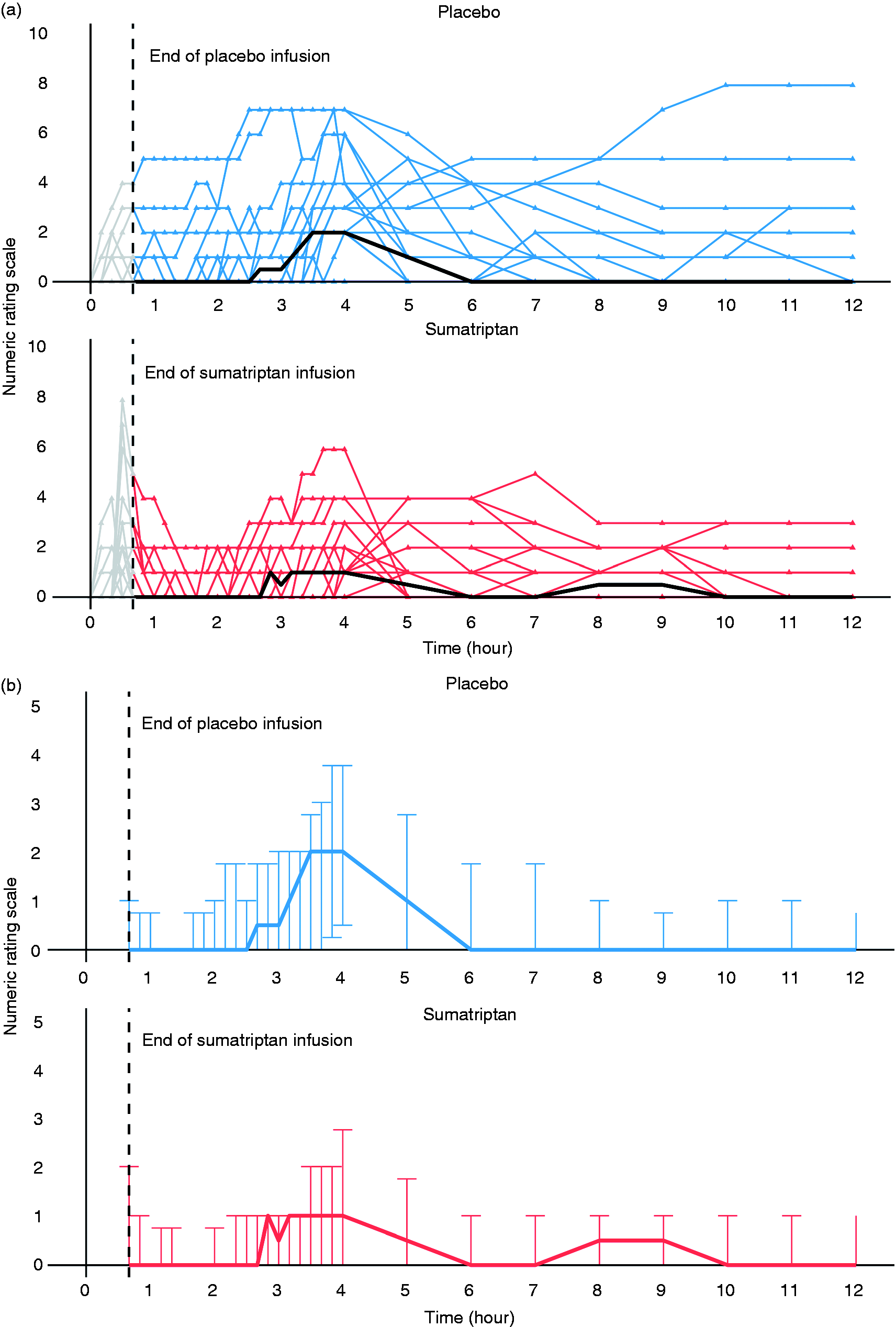
(a) Numerical rating scale (NRS) scores for headache intensity after infusion of PACAP38 on the two study days for the 26 patients included in analysis. Blue lines are headache scores on the placebo day and red lines represent the sumatriptan day. The black lines represent the median headache scores on each day. The grey areas on the left of the graphs represent headache scores prior to T40. (b) Median headache score for all patients on numerical rating scale on the two study days. The blue line represents median scores on the placebo day and the red line represents median scores on the sumatriptan day. Error bars represent interquartile ranges.
Superficial temporal artery
PACAP38 dilated the STA immediately after infusion on both study days. On the sumatriptan day, STA was significantly constricted at T35 compared to infusion of placebo (p < 0.001) and remained constricted until T120 (p = 0.004); however, constriction was not significant at T180 and T240 (Figure 4).

Diameter of the superficial temporal artery on the two study days. The black line represents mean diameter on each study day. A decrease in STA diameter is seen on the sumatriptan day immediately after infusion of sumatriptan at time 35 min.
Facial skin blood flow
Facial skin blood flow after PACAP38-infusion increased by approximately 90% at 10 min. Sumatriptan diminished the PACAP38-induced blood flow increase at T40 (p = 0.025) and T50 (p = 0.029); thereafter there was no difference between the two interventions. For details, see Figure 6.
Facial skin temperature
Across all facial areas, the temperature increased by 1.2°C immediately after PACAP38 infusion (T10) (p < 0.001) and remained increased for the remaining in-hospital period (4 h) on both study days. There was no side difference. The most profound temperature increase was on the nose, on which the temperature increased by 2.4°C immediately after infusion of PACAP (T10) (p < 0.001). On the sumatriptan day, the temperature on the nose was 1.1°C lower at T30 (from start of infusion of sumatriptan) than after placebo (p < 0.001) and this effect was apparent until T160. For details, see Figure 7.
Mean arterial blood pressure and heart rate
Infusion of PACAP38 led to a decrease in MAP on both study days. Maximum decrease was reached at 10 min on both study days. On the sumatriptan day, maximum decrease was 10.2%, 95% CI (−13.2, −7.1). This decrease was abolished after sumatriptan infusion, which resulted in an increase from baseline of 6.3%, 95% CI (2.6, 10.1) at T30.
On the placebo day, maximum decrease was 8.9%, 95% CI [−10.6, −7.3]. The PACAP-related MAP decrease was almost abolished after placebo infusion, with a decrease from baseline of 4.2%, 95% CI (−7.0, −1.4) at T30 (for details, see Figure 5).

Percent change in mean arterial pressure (MAP) and heart rate on the two study days. Placebo is marked in blue and sumatriptan is the red line.

Relative changes in facial skin blood flow measured by laser speckle from baseline to T240. A is the mean from all patients, and B is the individual changes in facial skin blood flow.
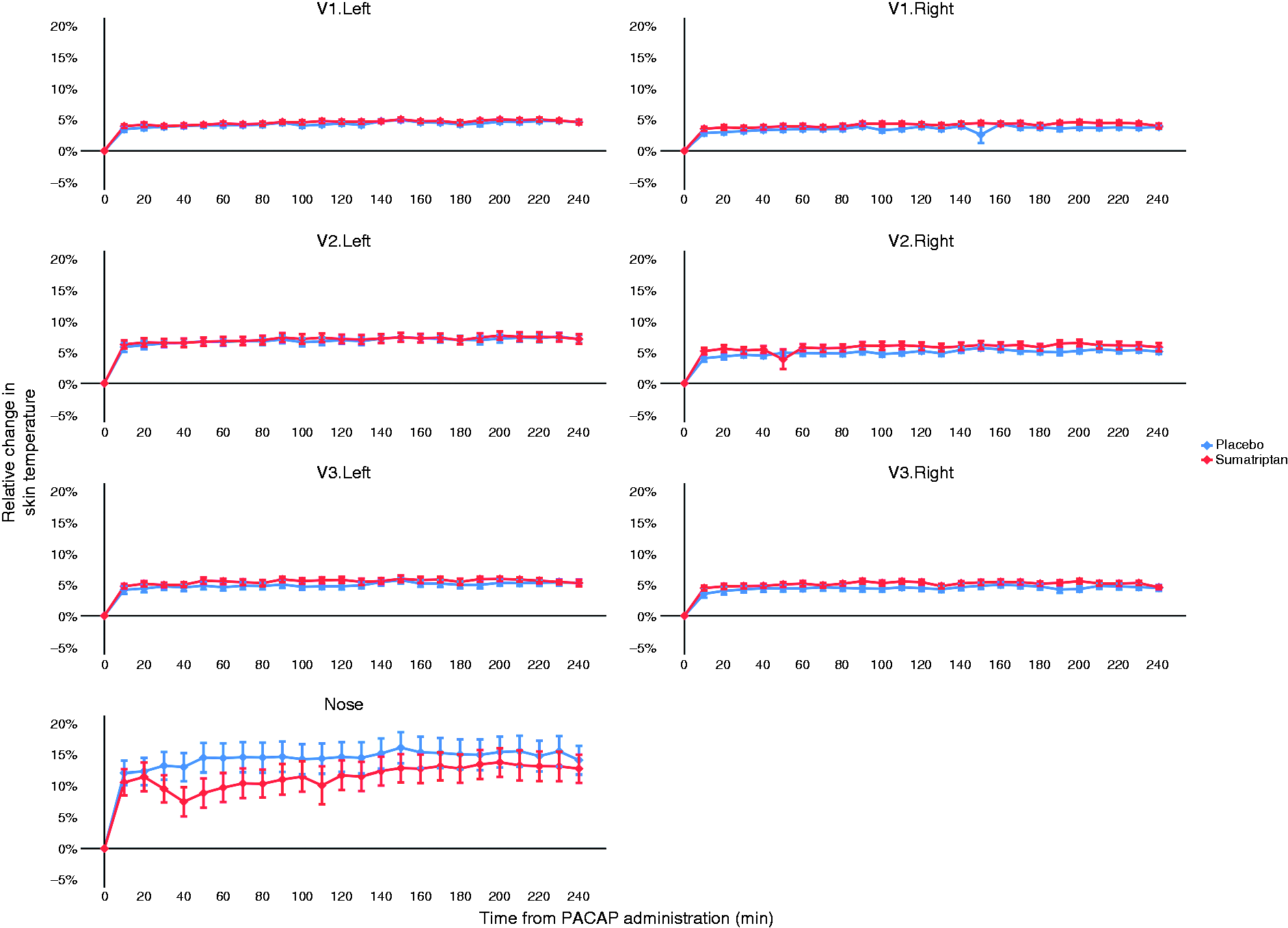
Relative changes in facial skin temperature on three points corresponding to the three branches of the trigeminal nerve (forehead, cheeks and chin), along with the nose. V1: ophthalmic nerve; V2: maxillary nerve; V3: mandibular nerve; R: right; L: left.
Adverse events
On the sumatriptan day, 16 patients (62%) reported facial puffing, and of these, nine patients reported taking an antihistamine and/or prednisolone to counteract the puffing (mean time to intake = 6.9 h, range [4–16 h]). On the placebo day, 18 patients (69%) reported puffing, and eight of these patients reported to take antihistamine and/or prednisolone to counteract the puffing (mean time to intake = 7.0 h, range [4–16 h]). Four patients experienced symptoms attributable to orthostatic hypotension (e.g. light-headedness) by the end of the experiment. Symptoms resolved within 30 min in all cases. There was no difference between early treatment (sumatriptan or placebo) and presence of orthostatic hypotension.
Discussion
The major outcome of the present study was that sumatriptan reduced the proportion of PACAP38-induced migraine attacks along with counteracting PACAP38-induced dilation of the STA. Consistent with STA findings, sumatriptan temporarily reduced facial skin blood flow and temperature on the nose. These data suggest that early treatment with sumatriptan can prevent migraine-inducing effects of PACAP38 and that this effect is associated with the vasoactive effect of sumatriptan. In our opinion, there are four conceivable ways in which sumatriptan may prevent PACAP38-induced migraine: Neuronal inhibition, vascular inhibition, mast cell inhibition and inhibition of CGRP signaling.
Neuronal inhibition
Sumatriptan directly inhibits release of neuropeptides relevant to migraine. In humans, administration of sumatriptan can reduce levels of PACAP measured in the external jugular vein during spontaneous migraine attacks (20), and in rodents, prolonged administration of eletriptan (another triptan) reduces brain mRNA transcription of PACAP (33). Sumatriptan may also attenuate trigeminal-induced protein extravasation in the dura mater (34,35), inhibit activation of trigeminal afferents regardless of vascular effects (36) and block c-fos expression (a marker of neuronal activity caused by chemical stimulation of the meninges) in the trigeminal nucleus (37). We recorded a short-lasting exacerbation in headache immediately following infusion of sumatriptan, but not placebo (Figures 3(a) and (b)). Transient exacerbation of headache is one of the common side effects of sumatriptan (38,39) and is also reported in other experimental provocation studies (40,41). Preclinical studies suggested that sumatriptan might alter the physiological properties of meningeal afferents, producing transient worsening of headache (38). Interestingly, sumatriptan was able to enter the brain only when disrupting the blood-brain barrier (BBB) with infusion of a hyperosmolar mannitol solution (42). Another study reported that sumatriptan reduced 5-HT1B receptor binding across brain regions involved in pain modulation, although the relation of this finding to its anti-migraine action is unknown (43). Of note, studies of spontaneous migraine attacks suggested that the BBB was intact during attacks with and without aura (44,45). It has been shown that triptans inhibit synaptic transmission between first and second trigeminovascular neurons in the medullary dorsal horn (46). Collectively, these observations suggest that sumatriptan may prevent PACAP38-induced migraine by inhibition of nociceptive transmission within the trigeminal system.
Vascular inhibition
PACAP38 is a potent vasodilator of both extracerebral (STA and middle meningeal artery (MMA)) and intracerebral (middle cerebral artery (MCA)) arteries (4,31). The importance of vasodilation in PACAP38-induced migraine remains unclear. Targeting mechanisms that lead to cAMP release leads to dilation of extracerebral arteries and migraine attacks in a high proportion of patients, which can be inhibited by sumatriptan (29,47). Sumatriptan inhibits the cAMP and protein kinase A (PKA) signaling pathway (48–51), and administration of subcutaneous sumatriptan in PACAP38-induced migraine has been found to attenuate headache along with constricting STA and MMA, but not MCA (31), suggesting a peripheral site of action of sumatriptan. In the present study, intravenous sumatriptan constricted the pre-dilated STA during the first 2 h, probably via the 5-HT1B receptors. 5-HT1B receptors are present in both cerebral and extracerebral vessels (52), on presynaptic nerve terminals on blood vessels, in the spinal dorsal horn, and trigeminal ganglion, co-localised with CGRP (53,54). PACAP and CGRP are also co-localised in many nerve fibres in the peripheral nervous system including in the trigeminal ganglion (55,56). It is possible that presynaptic 5-HT1B receptors located on PACAP-mediating nerve terminals in cranial blood vessels or in nerves in the trigeminal ganglion could be responsible for the effects observed after sumatriptan treatment, but further anatomical studies are needed. Given that prolonged dilation may activate perivascular afferents leading to initiation of an inflammatory response (57) with prolonged thickening of the vessel wall (58), it could be speculated that sumatriptan prevents PACAP38-induced migraine by counteracting dilation of extracerebral arteries following PACAP38 infusion, affecting both STA, facial skin blood flow and facial skin temperature. Release of PACAP by sensory nerve endings in the skin may contribute to flushing and neurogenic inflammation (59,60). Intradermal injection of PACAP38 leads to localised blood flow increase, flare and wheal, suggestive of both neurogenic inflammation and mast cell degranulation (61). In skin, PACAP induces angiogenesis and upregulation of CD31+ cells, which is unrelated to mast cells, supporting the notion of neurogenic inflammation (62). The increased blood flow and facial skin temperature induced by PACAP38 in our study was attenuated, but not entirely blocked by sumatriptan, consistent with our STA findings. Overall, these data suggest a peripheral site of action for both PACAP38 and sumatriptan.
Mast cell inhibition
Mast cells are present in the human (63) and rodent (64–66) dura mater, where they are mainly found in relation to blood vessels and sensory nerves (64–67). In rodents, experimentally induced mast cell degranulation can promote activation of these dural neurons (68). PACAP38 promotes mast cell degranulation (12,16,69), whereas another isoform, PACAP27, which also induces migraine attacks (6), failed to promote significant mast cell degranulation in a rodent model (16). The PACAP38-induced mast cell degranulation seemed to be mediated by the orphan-receptor, MrgB3, which was only activated by PACAP38, but not by PACAP27 (16). Interestingly, both in mast cell-depleted and antihistamine pre-treated rats, PACAP38 was not able to induce lasting MMA dilation (70); however, pre-treatment with the H1-antihistamine, clemastine, only partly prevented PACAP38-induced migraine in humans (25% reported migraine attacks) (71). In rodents, sumatriptan can inhibit experimentally induced mast cell degranulation of the dura mater (64) and inhibit mast cell mediated c-fos expression in the trigeminal nucleus caudalis (10,46). Based on these data, we cannot exclude that sumatriptan inhibits mast cell degranulation in the periphery and thereby prevents PACAP38-induced migraine.
Inhibition of CGRP signaling
It is possible that early sumatriptan administration prevents PACAP38-induced migraine through inhibition of an intracellular pathway. PACAP38 initiates cytoplasmatic signaling cascades that eventually lead to intranuclear signaling (72). Activation of PACAP38 receptors upregulates cAMP, which activates PKA (73–77). By phosphorylation of the cAMP responsive element binding protein 1, PACAP38 might upregulate transcription of other signaling neuropeptides including CGRP (78), which has a firmly established role in migraine. Preclinical data demonstrated that PACAP38 caused a significant concentration-dependent increase in CGRP release from trigeminal nucleus caudalis (78), which may be one of the mechanisms for PACAP38-induced migraine. However, infusion of PACAP38 in patients with MO did not lead to an increase in plasma CGRP (79), though a local increase in CGRP (e.g. in the dura mater) has not been investigated and therefore cannot be excluded. Based on the above findings, the mechanism for PACAP38-induced migraine seems peripheral although a central mechanism of action cannot be excluded; however, further investigation would be needed to confirm this.
Strengths and limitations
Strengths of this study include a large sample size – in terms of human migraine provocation studies, double-blind placebo crossover study design, and the use of a well-established experimental migraine model. To avoid carry-over effect of study medications, we had a minimum of 1 week between experimental days. Limitations included a low proportion of migraine attacks even on the placebo day. The migraine induction rate after placebo treatment (42%) was ∼16% lower than in previous placebo-controlled studies with PACAP38 without pre-treatment (lowest induction rate 58%) (65). It is possible that anticipation of early treatment might have led to a high placebo response (i.e. a lower migraine induction rate than previously). This is supported by a similarly low migraine induction rate of 45% on the placebo day in another study using PACAP38 in combination with pre-treatment (H1-antihistamine) (63). The migraine induction rate on the sumatriptan day in our study was 15.4%. Previous studies provoking migraine with different agents reported mean placebo-responses of 12%, 95% CI (6–20%) (40,41,53,62,65–69). Therefore, migraine induced on the sumatriptan day in our study may be the result of a placebo response and sumatriptan might in fact be more efficient in preventing PACAP38-induced migraine than our results indicate. To ensure 100% bioavailability of sumatriptan, we chose the intravenous formulation. There is no evidence that intravenous sumatriptan should have more vasoactive properties compared to other formulations, although it is possible that sumatriptan-induced side-effects may lead to potential unblinding of the participants. However, the blinding during the study was handled meticulously, we did not see any complications after intravenous sumatriptan, and the above-mentioned placebo response on the no-treatment day suggests that this was not an issue. In our study, sumatriptan was administered very early, and sometimes even before start of migraine and/or headache. In the clinical context, sumatriptan is usually most effective when taken soon after the start of headache (23), suggesting that our results reflect the usual efficacy of sumatriptan in the clinical setting. Patients were allowed to take rescue medication in the form of antihistamine or prednisolone, which could have affected duration of flushing and puffing. However, we found no difference in how many took rescue medication, or time to intake of either antihistamine or prednisolone on each study day, and we believe that this did not affect our results.
Conclusions
Sumatriptan prevented PACAP38-induced migraine attacks. This effect was associated with sumatriptan’s ability to counteract PACAP38-induced arterial dilation. We suggest that sumatriptan prevents PACAP38-induced migraine by modulation of trigeminovascular mechanism in the very beginning of the migraine cascade.
Key findings
In this randomised cross-over trial of 26 patients with migraine without aura, an early treatment with sumatriptan prevented pituitary adenylate cyclase-activating polypeptide-38-induced migraine. Migraine prevention was associated with immediate constriction of the superficial temporal artery by sumatriptan. Migraine initiation by pituitary adenylate cyclase-activating polypeptide-38 and prevention with sumatriptan involve trigeminovascular modulation; however, possible interplay from non-vascular targets cannot be ruled out.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102420975395 - Supplemental material for Early treatment with sumatriptan prevents PACAP38-induced migraine: A randomised clinical trial
Supplemental material, sj-pdf-1-cep-10.1177_0333102420975395 for Early treatment with sumatriptan prevents PACAP38-induced migraine: A randomised clinical trial by Nita Katarina Frifelt Wienholtz, Casper Emil Christensen, Ditte Georgina Zhang, Hande Coskun, Hashmat Ghanizada, Mohammad Al-Mahdi Al-Karagholi, Jens Hannibal, Alexander Egeberg, Jacob P Thyssen and Messoud Ashina in Cephalalgia
Footnotes
Additional contributions
The authors especially thank all patients that participated in the study. We also want to thank Dr. Anne Luise Haulund Vollesen and Dr. Thien Phu Do for helping with blinding, laboratory technicians Lene Elkjær, BS (Danish Headache Center and Department of Neurology, Rigshospitalet Glostrup, University of Copenhagen) and Winnie Grønning, BS (Danish Headache Center and Department of Neurology, Rigshospitalet Glostrup, University of Copenhagen) for expert assistance, and colleagues at the Danish Headache Center who helped make it possible to conduct experiments. No compensation was received for these contributions.
Role of the funder/sponsor
The funders had no role in the design and conduction of the study; design, collection, management, analysis, and interpretation of data; preparation, review, or approval of the manuscript, and decision to submit the manuscript for publication.
Data availability statement
Data are available from corresponding author upon reasonable request.
Author contributions
MA had full access to all data in the study and takes responsibility for the integrity of the data and the accuracy of data analysis.
Concept and design: NKFW, CEC, AE, JPT, MA.
Acquisition, analysis, or interpretation of data: All authors.
Drafting of the manuscript: NKFW, CEC, MA.
Critical revision of the manuscript for important intellectual content: All authors.
Statistical analysis: NKFW, CEC, MA.
Funding obtained: NKFW, MA.
Administrative, technical, or material support: CEC, HG, MA.
Supervision: CEC, AE, JPT, MA.
Data availability statement
Data are available from corresponding author upon reasonable request.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: NKFW has acted as an invited speaker for Novartis and received a travel grant from the Kgl Hofbundtmager Aage Bang Foundation. CEC received personal fees from Teva and acts as consultant for Teva. MAA has acted as an invited speaker for Novartis and received a travel grant from ElectroCore. HG declares no conflicts of interest.
AE has received research funding from Pfizer, Eli Lilly, the Danish National Psoriasis Foundation and the Kgl Hofbundtmager Aage Bang Foundation, and honoraria as consultant and/or speaker from Almirall, Leo Pharma, Samsung Bioepis Co., Ltd. Pfizer, Eli Lilly & Co, Novartis, Galderma, Dermavant, Bristol-Myers Squibb, and Janssen Pharmaceuticals.
JPT has attended advisory boards for Sanofi-Genzyme, Eli Lilly & Co, Pfizer, Abbvie, and Union Therapeutics, and received honoraria as a speaker from LEO Pharma, Regeneron, Abbvie, and Sanofi-Genzyme, and has been an investigator for Sanofi-Genzyme, Eli Lilly & Co, LEO Pharma, Pfizer, and Abbvie.
MA is a consultant, speaker or scientific advisor for Alder, Allergan, Amgen, Eli Lilly, Lundbeck, Novartis, and Teva, primary investigator for current Allergan, Amgen, Eli Lilly, Lundbeck, Novartis and Teva trials. MA has no ownership interest and does not own stocks of any pharmaceutical company. MA serves as associate editor of Cephalalgia, associate editor of Headache, associate editor of the Journal of Headache and Pain. MA is President of the International Headache Society.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from Novo Nordisk Foundation (NNF170C0029698), Augustinus Foundation (17-2523) and Lundbeck Foundation. JH is supported from the Danish Biotechnology Center for Cellular Communication.
Role of the funder/sponsor
The funders had no role in the design and conduction of the study; design, collection, management, analysis, and interpretation of data; preparation, review, or approval of the manuscript, and decision to submit the manuscript for publication.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
