Abstract
The parasympathetic nervous system is probably involved in migraine pathogenesis. Its activation releases a mixture of signalling molecules including vasoactive intestinal peptide (VIP) and pituitary adenylate cyclase-activating polypeptide (PACAP), which subsequently stimulate VPAC1, VPAC2 and PAC1 receptors. The objective of the present study was to investigate the in vivo effect of VIP, PACAP-27, PACAP-38, the selective VPAC1 agonist ([Lys15, Arg16, Leu27]-VIP(1–7)-GRF(8–27)) and a PAC1 agonist, maxadilan on rat middle meningeal artery (MMA) diameter using the closed cranial window model. Selective antagonists were used for further characterization of the responses. Reverse transcriptase-polymerase chain reaction experiments were also conducted to determine expression of mRNA of PACAP receptors in the MMA. The results showed that VIP, PACAP-38, PACAP-27 and the VPAC1 specific agonist evoked significant dilations with the rank order of potency; VIP = PACAP-38 > PACAP-27 = [Lys15, Arg16, Leu27]-VIP(1–7)-GRF(8–27). Significant inhibition of dilation was only observed for the VPAC1 antagonist PG97–269 on PACAP-38-induced dilation of MMA. The VPAC2 antagonist PG99–465 and PAC1 antagonist PACAP(6–38) did not significantly block VIP- or PACAP-induced dilation. Expression of mRNA of all three receptors was detected in the MMA. In conclusion, the VPAC1 receptor seems to be predominant in mediating MMA dilation. A selective VPAC1 antagonist may be a candidate molecule in the treatment of migraine headache.
Introduction
Migraine attacks are associated with facial flushing and vomiting together with vasodilation of intracranial arteries. Due to these and other facts, the parasympathetic nervous system has been suggested to be involved in migraine pathophysiology (1, 2). Moreover, release of vasoactive peptides from the parasympathetic nervous system may activate or sensitize intracranial sensory nerve fibres, leading to the perception of pain (3). Trigeminal nerve stimulation activates the parasympathetic nervous system with subsequent release of vasoactive peptides such as vasoactive intestinal peptide (VIP) and pituitary adenylate cyclase-activating polypeptide (PACAP) (3–5). PACAP belongs to the secretin/glucagons/VIP superfamily containing nine peptides in all so far (6) and exerts numerous effects, including dilation of cranial blood vessels (7, 8). Vasodilation of cranial arteries such as the middle meningeal artery (MMA) as well as the middle cerebral artery (MCA) activates trigeminovascular nociceptive pathways (9). PACAP occurs in two biologically active forms (7, 10): PACAP-27 and PACAP-38. Both peptides are derived from the same precursor, with PACAP-38 being predominant in rat brain tissue (7, 11). Although PACAP-27 and the first 27 amino acids in PACAP-38 show homology with VIP (12), PACAP is more potent than VIP in stimulating adenylate cyclase in pituitary cells (13).
VIP and PACAP exert their effects through three G-protein coupled receptors, VPAC1, VPAC2 and PAC1, by activating adenylate cyclase. The VPAC1 and VPAC2 receptors show similar affinity for VIP and PACAP (7, 14), whereas the PAC1 receptor has higher affinity for PACAP-27 and PACAP-38 than VIP, as shown in Table 1.
pKi and pIC50 values for all agonists and antagonists for the three receptors

∗Indicates the pIC50 values.
Numbers in parentheses indicate the corresponding reference number.
NA, not available.
In human studies, infusion of VIP and, even more, PACAP-38 elicits headache in healthy volunteers (15, 16). In vivo studies investigating the pharmacology of VIP and PACAP with respect to their effect on cranial arteries are limited. To address this limitation and based on the headache-inducing potential of these endogenous peptides, the aim of the present study was to: (i) investigate the potential of VIP, PACAP-38, PACAP-27 and additional peptides to increase the rat MMA diameter, using the in vivo closed cranial window model (17); (ii) characterize the receptors with selective agonists and antagonists; and (iii) study messenger RNA expression (mRNA) for VPAC1, VPAC2 and PAC1 receptors in the rat MMA.
Methods
In vivo preparations
All experiments were performed in accordance with the guidelines and regulations of the Danish Animal Experimentation Inspectorate (file 2004/561–850) on the care and use of laboratory animals. Male Sprague-Dawley rats (n = 73) weighing 350–400 g were used. The details regarding the surgical preparation and intravital microscopy have been described previously (18). Briefly, the animals were anaesthetized with pentobarbital (Mebumal® 50 mg/kg intraperitoneally), which was continuously given intravenously (20 mg/mg/h) throughout the experiment, via an infusion pump. Following intubation, the rat was cannulated in both the left and right femoral vein and artery to be used for infusion of anaesthetic, test substances, measurement of mean arterial blood pressure and sampling of arterial blood for gas tension analyses. The right parietal bone was then thinned, making a window through which the MMA was visualized via a video camera and TV monitor. A video dimension analyser (V94; Living Systems Instrumentation, Burlington, VT, USA) was connected to the camera, which continuously measured the diameter of the MMA by a scan line. Thereby, changes in dimensions arising from vessel constriction or dilation were automatically followed by rapid time resolution and displayed on a digital panel. Simultaneously, changes in mean arterial blood pressure were registered by connecting the catheter to a transducer (Transducer TCM4-7; World Precision Instruments, Sarasota, FL, USA).
Experimental protocols
In the present study operator blinding was not performed due to the experimental set-up. Furthermore, each animal was used as its own control, thus avoiding inter-animal variation. To study a dose-dependent response of VIP, PACAP-38, PACAP-27, maxadilan and VPAC1 agonist, doses of 1, 3 and 10 μg/kg were administered. Responses to the agonists were characterized using antagonist drugs at VPAC1 (PG97-269) (19), VPAC2 (PG99-465) (19) and PAC1 receptors [PACAP(6-38)] (20). For these interaction experiments, the 3 μg/kg dose of the agonists and the 10 μg/kg dose of the antagonists were chosen; these doses are consistent with the high affinity of the drugs at their receptors (Table 1). Since PACAP(6-38) displayed intrinsic efficacy, a dose–response experiment with the doses of 10, 20 and 50 μg/kg of this drug was performed. Thus, for these experiments the protocol was as follows: infusion of 3 μg/kg of the agonist followed 20 min later by 10 μg/kg of the antagonist. After 2 min the agonist (3 μg/kg) was infused again. When values had returned to baseline, 0.5 μg/kg of the potent vasodilator calcitonin gene-related peptide (CGRP) (21) was infused as an internal standard to check the responsiveness of the artery. All drugs were administered intravenously over 1 min followed by a flush of 150 μl isotonic saline in the same catheter. Changes in MMA diameter and in mean arterial blood pressure were measured 1 min prior to (baseline) and 1–2 min after the administration of the agonists and antagonists. In all studies arterial blood samples were analysed prior to, during and at the end of the experiment. The partial pressures of oxygen (PaO2), carbon dioxide (PaCO2) and pH (NPT 7; Radiometer, Br⊘nsh⊘j, Denmark) were kept within physiological limits (pH 7.3–7.4, PaCO2 35.2–42.7 mmHg and PaO2 81.7–127.5 mmHg).
Data presentation and statistical analysis
Changes in MMA diameter and in mean arterial blood pressure were measured and calculated as percentage change from baseline. Mean arterial blood pressure is given in mmHg. All data are expressed as mean ±
Test substances
Rat VIP, PACAP-38, PACAP-27 and PACAP(6-38) were obtained from NeoMPS (Strasbourg, France), together with rat α-CGRP. The selective VPAC1 agonist ([Lys15,Arg16,Leu27]-VIP(1-7)-GRF(8-27)) and the VPAC1 antagonist (PG97-269) (19) were obtained from Phoenix Europe GmbH (Karlsruhe, Germany). The VPAC2 antagonist (PG99-465) (19) was a kind gift from Dr Patric Robberecht, (Universite Libre de Bruxelles, Belgium). Maxadilan, a PAC1 agonist (22), was given by Dr Ethan Lerner (Massachusetts General Hospital, Charlestown, MA, USA). All test substances were dissolved in isotonic saline (0.9% NaCl) and stored at −20 °C. Prior to use the samples were diluted in isotonic saline to the desired concentration. Heparinized saline (10 IU/ml) was used in the arterial catheters to prevent coagulation of the blood.
mRNA expression studies
Three male Sprague-Dawley rats (320–350 g) were anaesthetized with pentobarbital and perfused transcardially with a 300 ml ice-cold buffer solution composed of (mM): NaCl 119, NaHCO3 15, KCl 4.6, CaCl2 1.5, NaH2PO4 1.2, MgCl2 1.2 and glucose 5.5. The brain was removed and the MMA carefully dissected out of the dura mater and submerged in a RNA stabilization solution (RNAlater™; Ambion, Austin, TX, USA). For each mRNA expression study, tissue known to express target mRNA was dissected out in parallel, serving as a control for the target-specific primers and the experimental set-up. Total RNA was purified from the tissues using the RNeasy Mini Kit (Qiagen, Valencia, CA, USA). Yield and purity of the purified RNA were determined spectrophotometrically. Complementary DNA (cDNA) was synthesized from 500 ng purified RNA in a final reverse transcriptase-polymerase chain reaction (RT-PCR) volume of 20 μl, using the Quantiscript Reverse Transcription Procedure (Qiagen). A negative control (no RT added to the RT-PCR mixture) was included for each mRNA study, serving as a control for genomic contamination in cDNA preparations. One microlitre of the cDNA and non-cDNA preparations was mixed with 0.5 μM of the target-specific primer pair, water, RedTaq ReadyMix™ PCR reaction Mix and MgCl2 (Sigma, St Louis, MO, USA). The PCR program used for amplification was as follows; pre-denaturation at 94 °C for 2 min, 40 amplification cycles of template denaturation for 1 min at 94 °C, primer annealing for 45 s at 58 °C and extension for 2 min at 72 °C. The PCR products were loaded on an agarose gel containing ethidium bromide and the PCR products were separated by electrophoresis along with a PCR 100 base pair Low Ladder (Sigma). The gel was photographed and the products were cut out of the gel, purified using the QIAquick Gel Extraction Kit (Qiagen) and sequenced at MWG Biotech (Ebersberg, Germany). Each set of target-specific primers (Table 2) used for the amplification and detection of mRNA was designed from National Center for Biotechnology Information GenBank extracted sequences with the aid of primer design software.
Primers used in the reverse transcriptase-polymerase chain reaction analysis that was designed to cross exon/intron boundaries and/or to anneal to sequences in two different exons

Results
Agonist dose–response experiments
Administration of 1, 3 and 10 μg/kg of VIP evoked dose-dependent dilation of the MMA with a maximum dilation of 62 ± 9% elicited by 3 μg/kg (Fig. 1A). The same doses of PACAP-38, PACAP-27 and the selective VPAC1 agonist were used. This resulted in significant dilations of the artery; for PACAP-38 the values were 22 ± 5, 44 ± 5 and 45 ± 13%, respectively, for each dose and for PACAP-27 13 ± 1, 28 ± 6 and 60 ± 9%, respectively (Fig. 1B,C). The selective VPAC1 agonist caused a dose-dependent dilation (9 ± 3, 33 ± 9 and 63 ± 13%, respectively, for each dose) (Fig. 1D). Finally, identical doses of the PAC1 agonist maxadilan failed to induce significant changes of artery diameter (data not shown). Thus, the apparent rank order of agonist potency was VIP = PACAP-38 > PACAP-27 = VPAC1 agonist >>> maxadilan. In all cases, CGRP produced significant dilator responses that were, in general, larger in magnitude than those evoked by the agonists (Fig. 1). The agonists also produced concomitant decreases in mean arterial blood pressure with a similar rank order of potency: PACAP-38 > VIP > PACAP-27 = VPAC1 agonist >>> maxadilan (Table 3).
Percent decrease in mean arterial blood pressure in agonist dose–response experiments
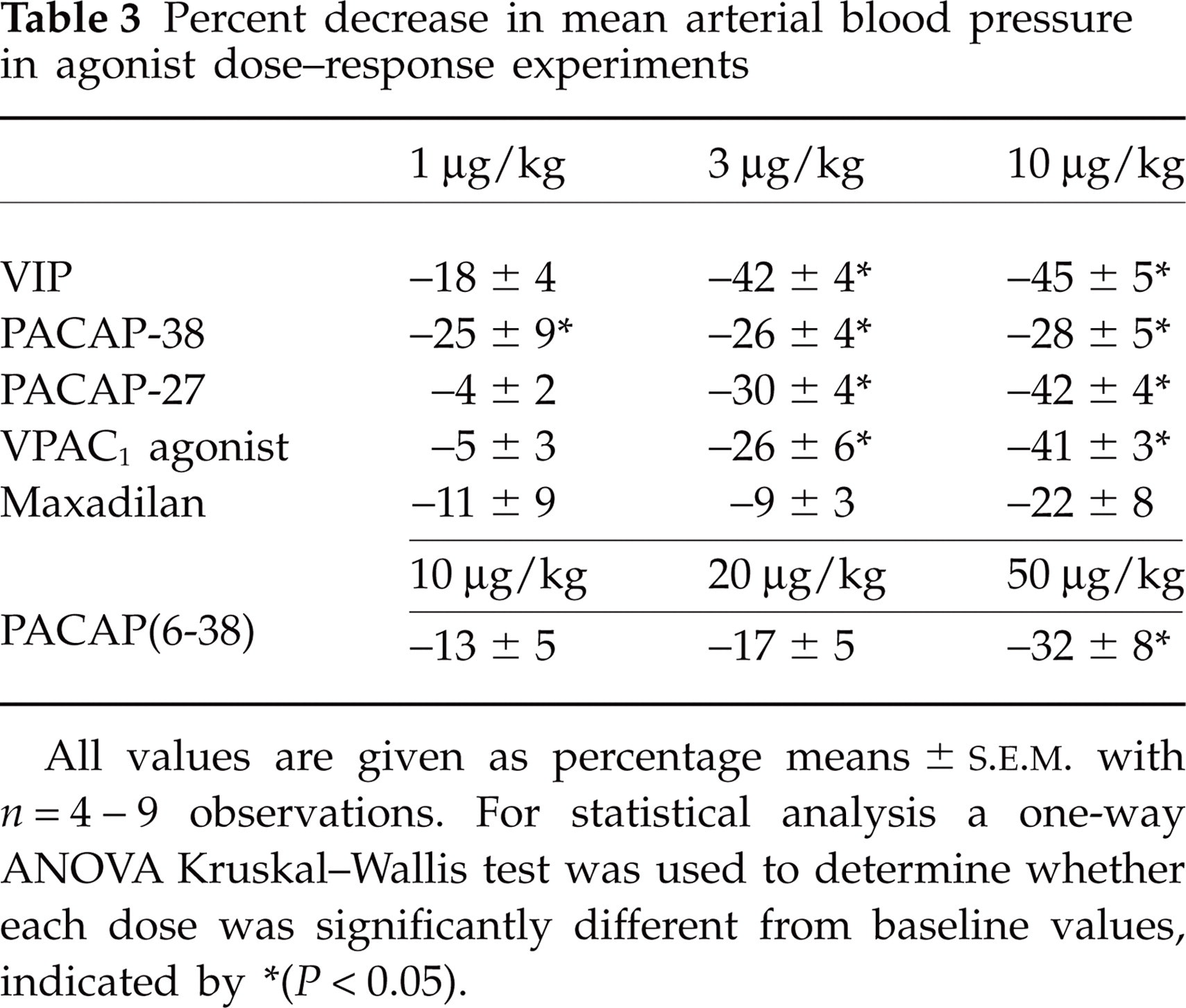

All values are given as percentage means ±

Agonist dose–response experiments illustrating the effect of (A) VIP, (B) PACAP-38, (C) PACAP-27 and (D) VPAC1 agonist [Lys15,Arg16,Leu27]-VIP(1-7)-GRF(8-27) on the MMA diameter. Data are expressed as the mean ±
Interaction experiments
PACAP-38 and PACAP-27 show similar affinity profiles compared with VIP towards the three receptors. Only PACAP-38, due to its predominant form of the two PACAPs in rat brain tissue (7) and VIP, was included in the interaction experiments. Infusion of the VPAC1 antagonist PG97-269 (10 μg/kg) alone had no effect on the investigated parameters. A dilatory effect of 21 ± 2% was observed after the first VIP infusion. The second VIP infusion was preceded by the antagonist, which failed to block the response elicited by VIP (27 ± 10%) (Fig. 2A); the antagonist also failed to block VIP-induced hypotensive effects (Table 4). Thus, the experiment was repeated with a higher dose of PG97-269 (20 μg/kg) against VIP, which also failed to block VIP-induced responses (not shown). PACAP-38 induced a dilation of 53 ± 17%, which was significantly reduced to 26 ± 12% in the presence of 10 μg/kg PG97-269 (P < 0.05, Fig. 2B). The decrease in mean arterial blood pressure induced by PACAP-38 was concomitantly reduced from 32 ± 5 to 15 ± 6%; however, the attenuation was not significant (P = 0.06) (Table 4).
Percent decrease in mean arterial blood pressure in interaction experiments

All values are given as percentage means ±

Interaction experiments illustrating the effect of the VPAC1 antagonist PG97-269 against (A) VIP and (B) PACAP-38. (C,D) The effect of the PAC1 antagonist PACAP(6-38) against VIP- and PACAP-38-induced responses, respectively. (E) The effect of the VPAC2 antagonist, PG99-465, on responses to VIP. Each bar is expressed as the mean ±
Infusion of the VPAC2 antagonist PG99-465 per se had no effect on any of the investigated parameters. PG99-465 (10 μg/kg) did not inhibit VIP-induced dilation (43 ± 14% compared with 50 ± 13% without antagonist) (Fig. 2E) or mean arterial blood pressure (Table 4). VIP infusion after PACAP(6-38) (10 μg/kg) resulted in a dilation of 45 ± 16% compared with 41 ± 15% when infusing VIP alone (Fig. 2C). Infusion of PACAP-38 caused a MMA dilation of 53 ± 13%, whereas a second PACAP-38 infusion in the presence of PACAP(6-38) (10 μg/kg) resulted in a non-significant reduction of the dilation to 33 ± 13% (Fig. 2D) as well in mean arterial blood pressure (Table 4). Due to the fact that neither 10 nor 20 μg/kg (data not shown) of PACAP(6-38) inhibited VIP or PACAP-38, but actually showed partial agonistic properties, we chose to perform a dose–response experiment with PACAP(6-38). As 10 and 20 μg/kg had been given in the interaction experiments, a higher dose of 50 μg/kg PACAP(6-38) was administered. All three doses resulted in MMA dilation [28 ± 10, 34 ± 10 and 95 ± 19%∗, respectively, for each dose (∗P < 0.05)]; the corresponding reduction in mean arterial blood pressure is given in Table 3.
mRNA expression studies
mRNA expression of the VPAC1, VPAC2 and PAC1 receptors in rat MMA was analysed by RT-PCR. PCR products of the expected size from both the positive control fractions and rat MMA fractions were sequenced. Alignment of all sequences (250–450 base read lengths) to the target Genbank mRNA sequences resulted in 99–100% identity, verifying the target specificity of the primers. mRNA expression was shown for all three receptors (Fig. 3) in the rat MMA.
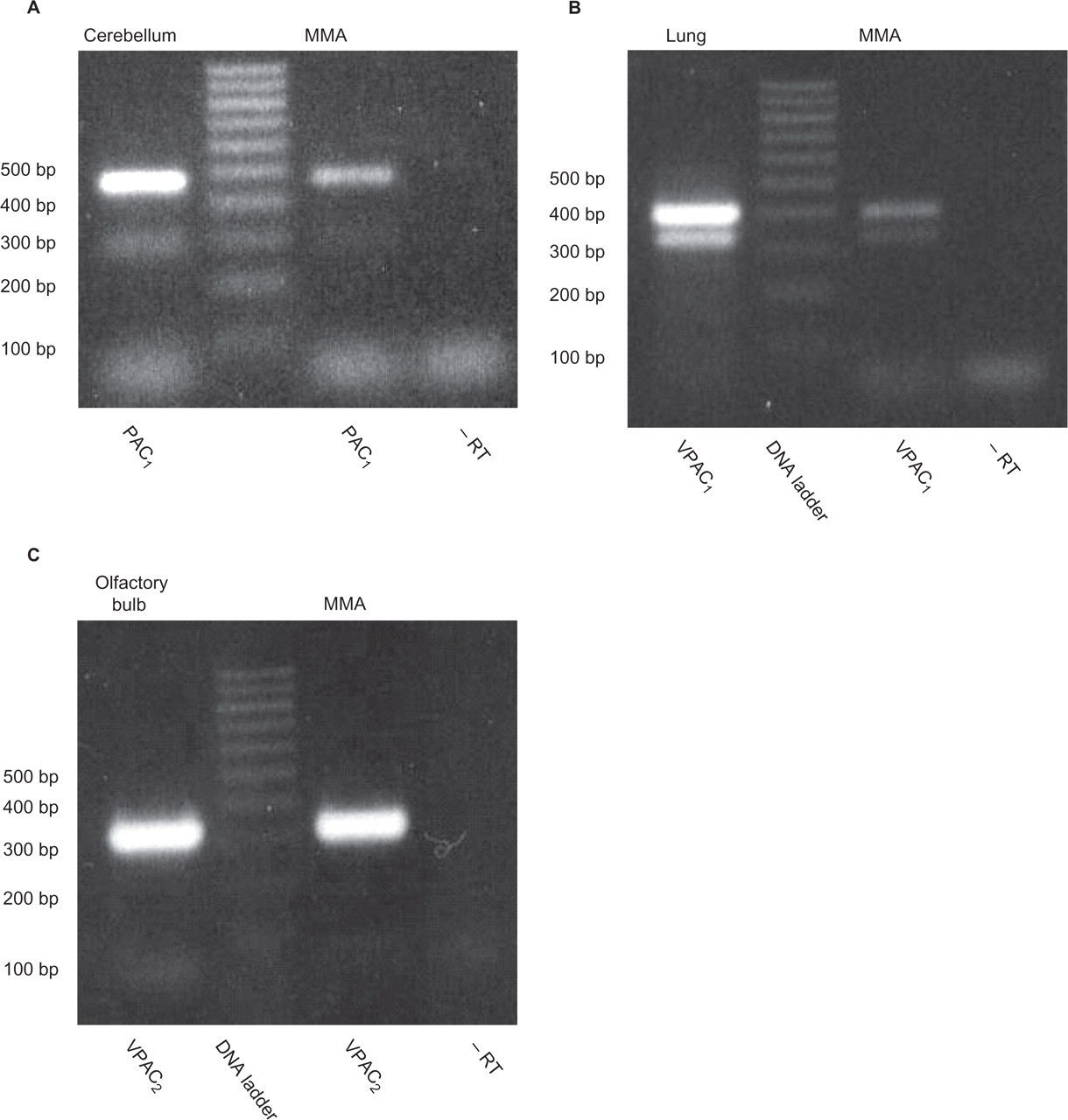
The expression of (A) PAC1 (472 bp), (B) VPAC1 (407 bp) and (C) VPAC2 (333 bp) receptors in rat middle meningeal artery (MMA) demonstrated by reverse transcriptase-polymerase chain reaction (RT-PCR) in lane 3. The following rat tissue cDNA (lane 1) served as positive controls for the designed target-specific primers: (A) PAC1: cerebellum, (B) VPAC1: lung and (C) VPAC2: olfactory bulb. PCR products were not observed in lane 4, where no RT was added to the RT-PCR mixture, both serving as a control for genomic contamination in the cDNA preparation and verifying that target-specific mRNA was detected in MMA samples. The lower additional bands observed in (A) PAC1 and (B) VPAC1 were also sequenced and found not to be splice variants of the receptors, but non-specific products.
Discussion
This is the first in vivo study to investigate the effect of selective agonists and antagonists for the VPAC1, VPAC2 and PAC1 receptors in rat MMA. In addition, the presence of mRNA encoding for all three receptors in MMA was shown using RT-PCR.
Although all physiological values were within their limits, approximately 10% of rats did not respond to the treatment. To avoid drawing the conclusion that non-responding was due to a lack of response to the drug, we used the potent vasodilator CGRP as an internal standard. CGRP responses also provided yardsticks to check variability in amplitude of responses induced by the peptides. Thus, the amount of dilation to VIP/PACAP and their agonists must be compared with CGRP responses. For example, comparing the VIP-induced responses of the dural artery in the dose–response study (Fig. 1A) with the VIP-induced response in the VPAC1 antagonist experiment (Fig. 2A), an approximately threefold variation was observed. However, this variation is similar to that of the CGRP-induced response in the same experiments and can thus be ascribed to variability among the rats.
VIP and PACAP are present in the parasympathetic nerve fibres surrounding human temporal (23, 24) and middle cerebral arteries (25). In the initial experiments PACAP-27 and PACAP-38 induced dose-dependent dilation of the MMA with no significant difference in their maximal effects. A similar concentration-dependent relaxation has also been shown in isolated rat intracerebral arterioles (26). Both PACAPs produced dose-dependent dilation of piglet pial arteriolar diameter when given topically (29 and 36%, respectively) (27). Femoral arterial blood flow in dogs was three to four times higher after PACAP-38 than after VIP or PACAP-27 (28). Thus, the activity of the two peptides varies between species and is tissue dependent. In line with a previous study on haemodynamic effects evoked by intravenous PACAP-27 in rats (29), we observed a dose-dependent decrease in mean arterial blood pressure during infusion of VIP, PACAP-27, PACAP-38 and the selective VPAC1 agonist. Hypotension caused by the agonists may be partially or wholly responsible (via autoregulation) for the observed dilation of the pial artery, but not of the MMA (30, 31). The dilatory effect of PACAPs and VIP in the MMA in the present study was therefore a direct pharmacological effect. Since VIP and PACAPs produced dilation in the MMA (Fig. 1A–C) as well as hypotensive responses (Table 3) with a similar rank order of potency (see Results), and they all display high affinity at VPAC1 receptors (Table 1), it is likely that both responses involve activation of VPAC1 receptors. This is supported by the ability of the selective VPAC1 agonist to elicit similar meningeal dilator (Fig. 1D) and hypotensive responses (Table 3).
Systemic administration of 10 pmol/kg/min (approximately 0.045 μg/kg/min) of PACAP-38 for 20 min in healthy human subjects was given, resulting in a total dose of 200 pmol/kg (approximately 0.9 μg/kg) (15). Remarkably, the peptide administration resulted in headache associated with parasympathetic features such as sustained skin flushing and an increase in middle cerebral artery diameter in 11 out of 12 subjects (15). The dose of PACAP-38 used in the present study was approximately three times higher than that used in the human study. The need for a higher dose in rat compared with man can be explained by the half-life of PACAP-38, which is 0.82 min in rats (32) compared with 3.5 min in man (15). The dilatory effect of VIP in MMA has been shown also in human MMA (33). In the present study, VIP dilated the artery with a peak at 3 μg/kg. In healthy human volunteers, VIP (8 pmol/kg/min for 25 min; total 200 pmol/kg, approximately 0.675 μg/kg) induced a short-lasting increase in middle cerebral artery diameter and dilation of the superficial temporal artery together with a mild and short-lasting headache (16). In addition, a similar study in migraineurs receiving VIP resulted in marked dilation of cranial arteries (34). The VIP dose we utilized was approximately four times higher than that used in the human study (0.675 μg/kg), which is explained by VIP's half-life being approximately 2 min in man (35) compared with only 0.44 min in rat (36).
Since both VIP and PACAP show similar affinity for the VPAC1 receptor (Table 1), we used a specific VPAC1 antagonist, PG97-269, against VIP and PACAP-38. PG97-269 significantly inhibited the dilation induced by PACAP-38. In contrast, neither 10 μg/kg nor 20 μg/kg of PG97-269 had any effect on VIP-induced responses. In fact, the 20 μg/kg elicited VIP response resulted in an increase in dilation in two of five rats. A slight blockade was found in the remaining three animals. In vitro PG97-269 has shown antagonistic properties in an assay involving VIP-, PACAP-38- and PACAP-27-induced cAMP production in cells expressing the human VPAC1 receptor (19). Species and/or differences in receptor composition may account for this unexpected result. The results suggest, however, an important role for the VPAC1 receptor in mediating PACAP-38 vasodilation in vivo in rat.
The VPAC2 receptor antagonist PG99-465 failed to inhibit the VIP-induced responses. In vitro PG99-465 decreased the ability of VIP and PACAP-38 to induce lipolysis in rat adipocytes (37); however, a [cAMP]i assay revealed that PG99-465 acted as a partial agonist on the human VPAC2 receptor (19). In the same study, PG99-465 was described as a full agonist on the human VPAC1 and PAC1 receptors (19); thus, selectivity and potency for this receptor is questionable.
In our hands, the PAC1 agonist maxadilan, a 61 amino acid peptide extracted from the saliva of the sand fly, did not elicit any significant increase in MMA diameter or reduction in mean arterial blood pressure. However, previous in vivo studies are very limited and dealt only with its ability to induce erythema in rabbit skin (38). Moreover, in vitro studies comprise receptor-ligand studies of established cell lines and primary cultures presenting binding of maxadilan in brain membrane fractions from rat, rabbit and human tissue (39). Thus, previous studies do not explain our observations; however, the negligible effect cannot be attributed to a lack of PAC1 receptors, as we observed mRNA expression of this receptor in our RT-PCR studies. The PAC1 receptor possesses at least four splice variants in the rat (40) and up to 14 in humans (41). Thus, maxadilan might not be active on all the PAC1 splice variants and the meningeal arteries could be equipped with one of these.
PACAP(6-38) has been described as the most potent antagonist of the PAC1 receptor in a cAMP stimulation study of human neuroblastoma cells (42). PACAP(6-38) did not inhibit the vasodilatory effect of VIP or PACAP-38. An in vivo cranial window study of piglet pial arterioles showed that topical application of PACAP-38 and PACAP-27 co-applied with PACAP(6-38) caused statistically significant inhibition of the effect. In the same study PACAP(6-38) (10−5 M, approximately 40 μg/ml) was not vasoactive per se (27), in contrast to this study, where 10, 20 and 50 μg/kg (approximately 0.14, 0.29 and 0.71 μg/ml, respectively) of PACAP(6-38) elicited dose-dependent dilation. However, taking the lack of response of maxadilan together with the lack of antagonistic effect of PACAP(6-38) into consideration, we believe that PAC1 receptor might not play a significant role in MMA dilation in rats.
Our RT-PCR expression studies have demonstrated the presence of mRNA of all three receptors. In line with this, mRNA expression for all three receptors has been demonstrated in human cerebral arteries, including the perivascular nerves and in trigeminal, otic and sphenopalatine ganglia (43). The VPAC1 receptor has, furthermore, been shown in rat smooth muscle cells from cerebral arteries and arterioles (14, 44). There is a close structural homology between rat and human among the VPAC1, VPAC2 and PAC1 receptors (45).
In order to delineate further the pharmacology of these receptors and their role in migraine, the development of more selective and potent agonists as well as antagonists for these receptors is clearly crucial. RT-PCR revealed mRNA expression of VPAC1, VPAC2 and PAC1 receptors. Lack of activity of the PAC1 agonist maxadilan and antagonist PACAP(6-38) point to the negligible role of this receptor. VPAC receptors, especially the VPAC1 receptor, seem promising targets to be further evaluated in preclinical models of migraine.
Footnotes
Acknowledgements
This study was supported by The Lundbeck Foundation as part of the Lundbeck Foundation Centre for Neurovascular Signalling (LUCENS) and by Copenhagen County's Research Council, Danish Agency of Science, Technology and Innovation, the Danish Research Council, the Augustinus Foundation, the Family Hede Nielsen Foundation and the Beckett Foundation.
