Abstract
Background:
In healthy human volunteers and in migraineurs, pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38) infusion caused sustained vasodilation of the middle meningeal artery (MMA) and an immediate as well as a delayed headache. All the study subjects experienced facial flushing. Mast cells (MCs) might have a role in the long-lasting effect of PACAP-38 infusion. We hypothesized that in mast cell-depleted (MCD) rats the vascular responses to PACAP-38 would be lesser than in control rats because of a lack of vasodilatory products released during MC degranulation.
Methods:
MCs were depleted by chronic treatment with compound 48/80. The effect of 20 minutes' intravenous (i.v.) infusion of calcitonin gene-related peptide (CGRP), PACAP-38, PACAP(6-38) (PAC-1 receptor antagonist) and PACAP-27 on the diameter of the MMA and on mean arterial blood pressure (MABP) in control and MCD rats was recorded by using the genuine closed-cranial window (CCW) model. Vasoactive intestinal polypeptide (VIP) infusion was given only in control rats. A combination of the histamine H1 receptor antagonist mepyramine (4 mg kg−1 i.v.) and the H2 receptor antagonist famotidine (1 mg kg−1 i.v.) was given 10 minutes prior to PACAP-38 infusion. Increasing doses of PACAP-38, PACAP-27 and VIP were infused through the intracarotid artery (i.c.) in control and MCD rats to see the direct effects of these peptides on MMA diameter change.
Results:
There was no significant change in CGRP-induced MMA diameter increase in control and MCD rats, and the dilated MMA immediately returned back to baseline after stopping the infusion. The delayed MMA dilation induced by PACAP-38 was abolished in MCD and antihistamine (AH)-pretreated rats. Compared to PACAP-38, the PACAP-27 i.v. infusion gave smaller peak dilation of MMA in control rats. In MCD rats, PACAP-27 did not induce any significant dilation. VIP i.v. infusion reduced MABP but did not dilate MMA significantly. PACAP(6-38), which is a potent MC degranulator, also gave a significant delayed dilation of MMA. PACAP-38 i.c. responses (direct receptor mediated response) were not affected by MC depletion. Only the maximum response (%
Conclusions:
The delayed MMA dilatory responses to PACAP-38 infusion were attenuated in MCD and AH-pretreated rats, indicating a role of the MC mediator-histamine in PACAP-38-induced delayed dilation of MA.
Introduction
Migraine is a complicated neurovascular disorder involving multiple brain nuclei, genetic predispositions and effector sites. Recently, it was ranked eighth out of all diseases in years lived with disability, and its global prevalence is 15% (1). Infusion of pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38) in patients with migraine without aura caused an immediate headache and dilation of the superficial temporal artery as well as a delayed migraine-like attack (2). In healthy human volunteers PACAP-38 infusion caused vasodilation of the middle meningeal artery (MMA) for up to five hours after infusion. Furthermore, it induced an immediate headache during infusion as well as a delayed headache (3). Sumatriptan, a migraine-specific drug, relieved the delayed headache in parallel with constriction of the MMA but not of the middle cerebral artery (3). Vasoactive intestinal polypeptide (VIP) infusion in migraineurs caused marked dilation of cranial arteries but did not trigger migraine attacks (4). PACAP-38 immunoreactivity was reported to be increased in the plasma of patients during a migraine attack (5). In a rat closed-cranial window (CCW) model, one-minute intravenous (i.v.) infusion of PACAP-38, PACAP-27 and VIP have dilatory effects on the MMA (6).
In humans, PACAP-38 has an elimination half-life of five to 10 minutes (7). Therefore, the delayed effect of PACAP-38 infusion cannot be attributed to the direct effects of PACAP-38, but more likely to a cascade of events triggered by PACAP-38. Mast cells (MCs) contain neurosensitizing, vasodilatory and vasoconstrictive mediators (8). We have recently shown that PACAP-38 and PACAP(6-38) have a strong degranulatory effect on rat peritoneal and dural MCs (9). Therefore, we aimed to investigate if MC degranulation has a role in PACAP-38-induced prolonged dilation of the MMA in the rat CCW model.
Methods
Animals
All the animal experimental procedures were performed in accordance with domestic guidelines and regulations for animal care and treatment. The study protocol was approved by the Danish Animal Experimentation Inspectorate (file: 2012-15-2934-00697). Rats were housed in a standard light and dark cycle with free access to food and water. A total of 76 male Sprague-Dawley (SD) rats (Taconic Europe, Tornbjergvej 40, Ejby, Denmark) weighing 270 to 350 g were used. Twenty-one rats were used for the mast cell-depleted (MCD) protocol. Fifty-five control rats were used for intracarotid artery (i.c.) or i.v. infusion of PACAP-38, PACAP(6-38), PACAP-27, VIP and calcitonin gene-related peptide (CGRP).
Drugs
PACAP-38, PACAP-27 and VIP were purchased from CASLO laboratory, Technical University of Denmark, Lyngby, Denmark. All compounds were dissolved in distilled water to 100 μM; aliquots were stored at −20℃. Compound 48/80 was purchased from Sigma Aldrich, Schnelldorf, Germany, and freshly dissolved in saline before use. CGRP (NeoMPS, Strasbourg, France) stock solution 0.5 mg ml−1 was prepared in water and aliquots were stored at −20℃. Mepyramine (16 mg ml−1) and famotidine (4 mg ml−1) solution was freshly prepared in 50% methanol and 50% saline.
Intravital microscopy on the rat CCW model
Rats were anesthetized with pentobarbital (Mebumal® 65 mg kg−1 intraperitonial) and depth of anesthesia was tested by suppression of the hind paw reflex. Anesthesia was continuously supplemented with pentobarbital (Mebumal® 20 mg kg−1 h−1 i.v.) during the experiment. Body temperature was maintained at 37.0 ± 0.5℃ throughout the experiments using an automatic regulated heating plate (Letica HB101, Panlab, Barcelona, Spain). After tracheal intubation, the animal was mechanically ventilated by a respirator (SAR-830/AP, CWE, Ardmore, PA, USA) with a 30%/70% mixture of O2/N2O, a stroke volume of 3.5–4.0 ml and a stroke rate of 70–75. The CCW model experiments were performed as described previously (10). A catheter (Portex, Fine Bore Polythene Tubing, inner diameter 0.4 mm, Astratech, Tåstrup, Denmark) glued to a gel-loading pipette tip was placed in the right carotid artery for infusion of the test substance. Other catheters were placed in the left and right femoral artery and vein for infusion of anesthetic, i.v. administration of drugs, measurement of mean arterial blood pressure (MABP) and sampling of blood for blood gas analysis. Arterial blood samples were collected prior to, during, and at the end of each experiment, for analysis of partial pressure of oxygen (PaO2), carbon dioxide (PaCO2) and pH (ABL520, Radiometer, Brønshøj, Denmark). All values were kept within normal limits (pH 7.35–7.45, MABP 80–130 mmHg and PaCO2 35–45 mmHg). In brief, the animal was placed in a stereotactic frame. Skin covering the dorsal surface of the skull was retracted and the connective tissue and muscle removed, leaving the right parietal bone exposed. The bone was thinned, making a window (10 × 7 mm2) by carefully drilling with a dental drill cooled by application of ice-cold synthetic interstitial fluid (in mM: 108 NaCl, 3.48 KCl, 0.7 MgSO4, 26 NaHCO3, 1.37 NaH2PO4, 1.5 CaCl2, 9.6 Na+ gluconate, 5.55 glucose and 7.6 sucrose) until the artery was clearly visible but still leaving the skull intact. For visualization of the arteries, an intravital microscope (model MZ 16, Leica, Heerbrugg, Switzerland) was positioned above the window. A video dimension analyzer (V94; Living Systems Instrumentation Inc, Burlington, VT, USA) continuously monitored and measured the diameter of the target artery. All data including changes in diameter of the arteries and MABP were continuously collected and simultaneously analyzed by Perisoft (Version 2.5.5; Perimed AB, Järfälla, Sweden). As drilling induces dilation of dural arteries, the rat was left to recover for 30–45 minutes before proceeding with the pharmacological experiments. After the experiments the animals were sacrificed by an i.v. overdose of 1 M KCl.
MC depletion protocol
In 21 male SD rats, MCs were depleted of their granules using repeated pretreatment with the secretagogue compound 48/80 using a method adapted from Coderre et al. (11). Briefly, the compound 48/80 (0.1% w/v in sterile saline) was injected intraperitonially twice a day (morning and afternoon) for a total of eight doses, starting with 0.5 mg kg−1 injections for the first day, 1 mg kg−1 for the second and 2 and 4 mg kg−1 for the third and fourth days, respectively. Animals were used three to four days later for the intravital microscopy experiments.
Experimental protocols
The MMA diameter was recorded immediately after drilling and after the approximately 30- to 45-minute recovery period. Before the start of any experiment, the baseline diameter of the target artery was measured for at least five minutes to ensure stability. The dilatory capacity of the target artery was tested with one single i.v. dose of CGRP (500 ng kg−1) or one single i.c. dose of CGRP (100 or 500 ng kg−1). To study the effect of a 20-minute infusion of different peptides on MMA diameter over time, control and MCD rats were infused with 0.25 µg kg−1 min−1 of CGRP, and 0.4 µg kg−1 min−1 of PACAP-38, PACAP(6-38) and PACAP-27. VIP (0.4 µg kg−1 min−1) was infused only in control rats. A combination of the H1 receptor antagonist mepyramine (4 mg kg−1) and the H2 receptor antagonist famotidine (1 mg kg−1) was given 10 minutes prior to PACAP-38 (20-minute i.v.) infusion. The antihistamine (AH) drug solution was four times concentrated, allowing a small volume (75 μl for 300 g rat) of drug solution to be infused over two minutes. It has been reported that mepyramine and famotidine in these doses can inhibit histamine-induced MMA diameter change in the rat CCW model (12).
PACAP-38, PACAP-27 and VIP were also administered via i.c. route in half-log increments to get a dose response curve in control and MCD rats. The MMA was allowed to return to baseline between each measurement. In the i.c. cannulation method, the cranial blood supply remains intact. Infused peptides are first distributed to the cranial arteries including the MMA before entering the peripheral circulation. Smaller doses can therefore be used to avoid lowering the systemic blood pressure and subsequent cerebral vasodilation due to autoregulation. First increasing doses of VIP (i.c.) were infused. Thirty minutes after administration of the last dose of VIP, increasing doses of either PACAP-38 or PACAP-27 were infused. In the i.c. protocol, we did not infuse PACAP-38 and PACAP-27 to the same rat. For the i.v. protocol, initially VIP was infused and 10 min after MMA diameter and MABP returned to baseline either PACAP-27 or PACAP-38 was infused. We verified the PACAP-38 infusion effect in another six rats, in which only PACAP-38 was infused. Since PACAP-27 did not dilate the artery significantly in MCD rats, the PACAP-38 infusion was performed in the same rat after a 30-minute interval or longer.
Data analysis
GraphPrism (GraphPad Software Inc, San Diego, CA, USA) was used for statistical analysis. Data are shown as percentage mean ± SEM; a criterion value of
Results
Effects of CGRP infusion on MMA diameter and on MABP
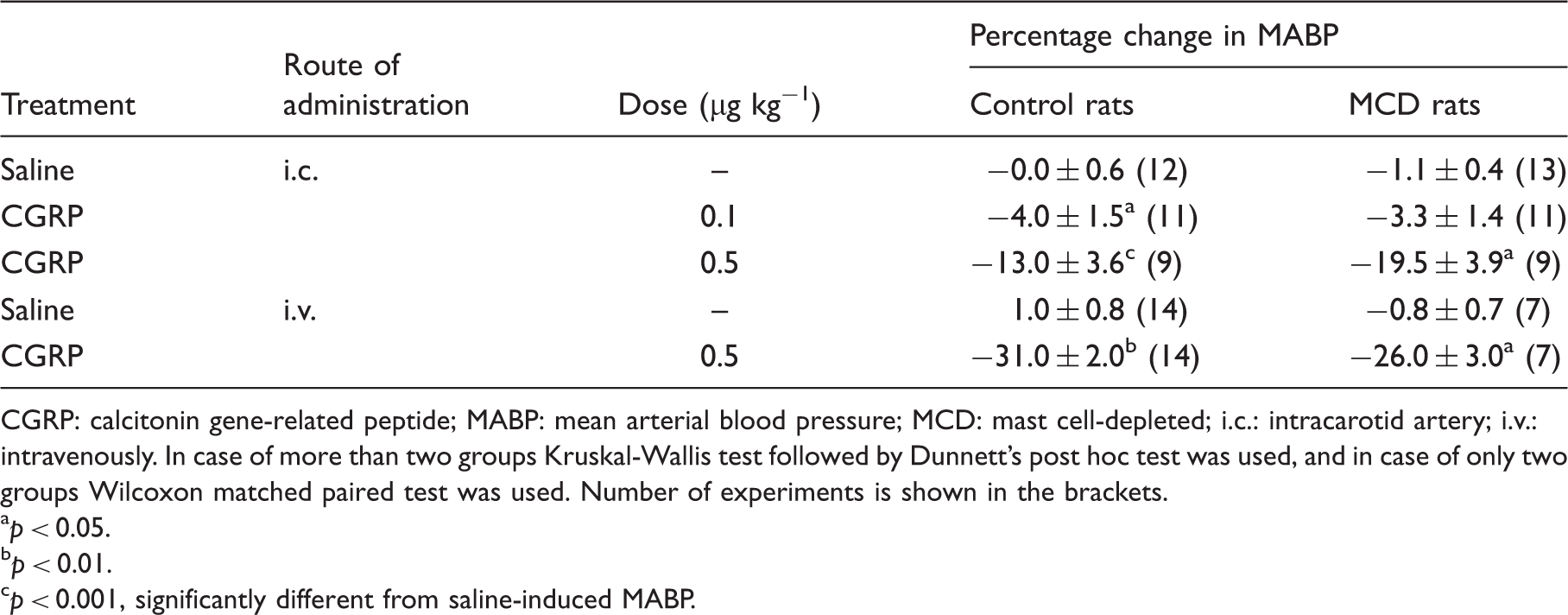
There was no significant difference between dilatory responses of the MMA (Figures 1 and 2) and MABP (Table 1) to the infusion of CGRP (0.1 and 0.5 µg kg−1 i.c. or 0.5 µg kg−1, i.v.; for one minute or 0.25 µg kg−1 min−1 for 20 minutes, i.v.) in control and MCD rats. In control rats a significant increase in MMA diameter that reached a plateau within five to 10 minutes after the start of the infusion was observed. The dilation was constant during the 20-minute CGRP infusion. A decrease in MABP was seen at five, 10 and 20 minutes from the start of the 20-minute CGRP infusion (Table 2). MMA diameter and MABP immediately returned to the baseline after stopping the CGRP infusion. In MCD rats CGRP-induced changes in MMA diameter and in MABP were not statistically different compared to control rats (Figure 2).
Effect of MCD on CGRP-induced dilation of MMA in a CCW model. Values are given as percentage increase in MMA diameter from pre-stimulation baseline, mean ± SEM. Effect of 0.25 µg kg−1 min−1 for 20 minutes i.v. infusion of CGRP, and 0.4 µg kg−1 min−1 for 20 minutes i.v. infusion of PACAP-27 and PACAP(6-38) on MMA diameter in control rats and MCD rats. Values are given as percentage increase in MMA diameter from the pre-stimulation baseline mean ± SEM. ** Effects of CGRP on mean arterial blood pressure. CGRP: calcitonin gene-related peptide; MABP: mean arterial blood pressure; MCD: mast cell-depleted; i.c.: intracarotid artery; i.v.: intravenously. In case of more than two groups Kruskal-Wallis test followed by Dunnett's post hoc test was used, and in case of only two groups Wilcoxon matched paired test was used. Number of experiments is shown in the brackets. a b c Effects of various agents on mean arterial blood pressure. CGRP: calcitonin gene-related peptide; MABP: mean arterial blood pressure; ACAP: pituitary adenylate cyclase-activating polypeptide; VIP: vasoactive intestinal polypeptide; AH: antihistamine; MCD: mast cell-depleted; min: minutes; ND: not done; NR: not recorded. Kruskal-Wallis test followed by Dunnett's post hoc test was used. Number of experiments is shown in the parentheses. a b c
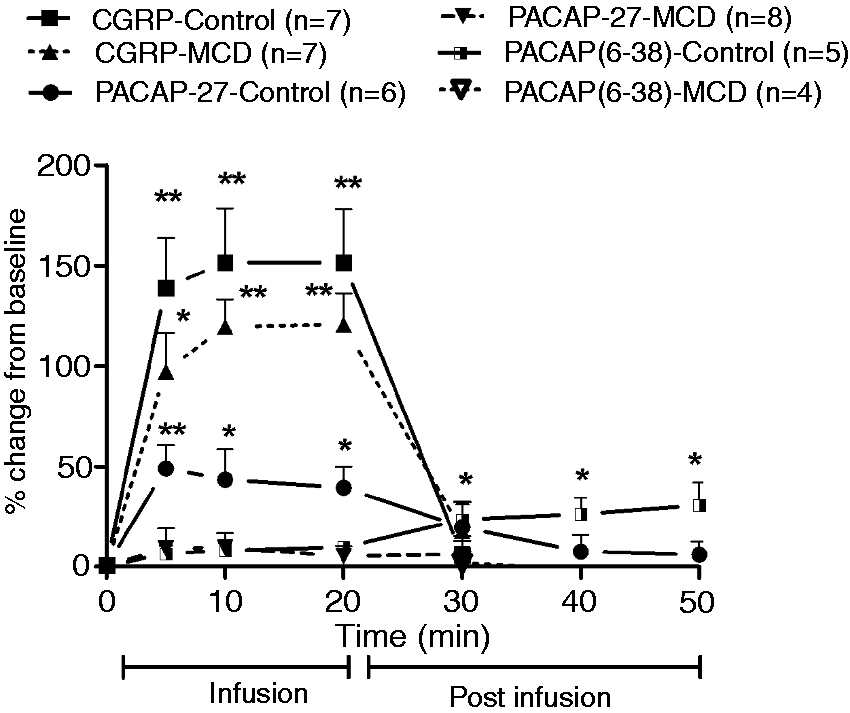

Effect of MC depletion on MMA diameter and on MABP with 20-minute i.v. infusion of PACAP-38
PACAP-38 infusion (0.4 µg kg−1 min−1 for 20 minutes) in control rats induced a slowly developing dilation of MMA that reached its maximum response at 20 minutes after the start of the infusion. After stopping the infusion at 20 minutes, there was a slow decline in dilation over the next 30 minutes. The MMA diameter was significantly ( Effect of 0.25 µg kg−1 min−1 for 20 minutes' i.v. infusion of CGRP, and 0.4 µg kg−1 min−1 for 20 minutes i.v. infusion of PACAP-38, PACAP(6-38), PACAP-27 and VIP on MMA diameter in control rats. Values are given as percentage increase in MMA diameter from the pre-stimulation baseline, mean ± SEM. *** Effect of PACAP-38 infusion (0.4 µg kg−1 min−1 for 20 minutes, i.v.) on MMA diameter in control, MCD and antihistamine (AH) (combination of mepyramine (H1, 4 mg kg−1 min−1) and famotidine (H2, 1 mg kg−1 min−1)) pre-treated rats. Values are given as percentage increase in MMA diameter from the pre-stimulation baseline mean ± SEM. ***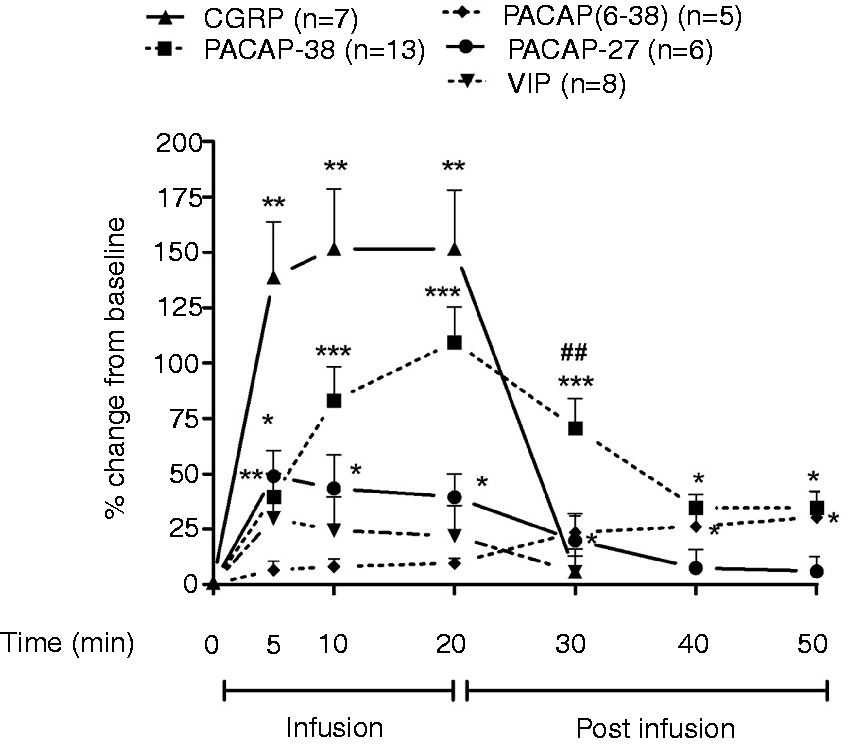

Effect of pretreatment of AH drugs on PACAP-38-induced changes in MMA diameter and in MABP
Pretreatment with a combined mepyramine and famotidine solution reduced the maximum PACAP-38-induced changes in MMA diameter as well as the delayed response. MMA came back to prestimulation baseline at 50 minutes from the start of the 20-minute i.v. PACAP-38 infusion (Figure 4). MABP was significantly decreased at the 10-minute time point (Table 2).
Effect of MC depletion on MMA diameter and on MABP with 20-minute i.v. infusion of PACAP(6-38)
PACAP(6-38), a PAC-1 receptor antagonist, did not change MMA diameter (Figures 2 and 3) during infusion. But it gave a sustained delayed dilation of MMA at 30, 40 and 50 minutes from the start of the 20-minute infusion. MABP was not changed at any time point during and after the infusion (Table 2). In MCD rats, PACAP(6-38) infusion did not cause a significant increase in MMA diameter (Figure 2).
Effect of MC depletion on MMA diameter and on MABP with 20-minute i.v. infusion of PACAP-27
A significant increase in MMA diameter was seen at five, 10 and 20 minutes of the PACAP-27 infusion (0.4 µg kg−1 min−1 for 20 minutes) in control rats. The MMA dilation reached its maximum at five minutes and was not as sustained as seen for PACAP-38 (Figures 2 and 3). In control rats a significant decrease in MABP was observed at five and 10 minutes as compared to the respective basal MABP. In MCD rats, a significant decrease in MABP was observed only at five minutes, as compared to the respective basal MABP (Table 2). Furthermore, PACAP-27 infusion did not cause a significant increase in MMA diameter in MCD rats (Figure 2).
Effects of 20-minute i.v. infusion of VIP on MMA diameter and on MABP
VIP infusion (0.4 µg kg−1 min−1 for 20 minutes) induced only a slight nonsignificant increase in MMA diameter (Figure 3). This response immediately returned to baseline after stopping the infusion. A significant decrease in MABP was observed at five, 10 and 20 minutes compared to basal MABP in control rats (Table 2). Because VIP infusion did not give a significant increase in MMA diameter in control rats, we did not infuse VIP in MCD rats.
Effect of i.c. PACAP-38, PACAP-27 and VIP infusion on MMA diameter and on MABP
PACAP-38, PACAP-27 and VIP caused a dose-dependent dilation of the rat MMA and decreased MABP (Figure 5) in control and MCD rats. Compared to the control rats, the maximum dilatory effect of PACAP-27 was significantly lower in the MCD rats (Table 3). There was no significant difference in the potency (pED50) of these peptides in MCD rats as compared to control rats regarding percentage change in MMA diameter and MABP (Table 3). PACAP-38 i.c. responses (direct receptor mediated response) were not affected by MC depletion. The highest dose of these peptides induced a lower effect on MMA dilation compared to the previous dose, which gives maximal dilation.
Effects of the increasing doses (i.c.) of PACAP-38 (a), PACAP-27 (b) and VIP (c) on MMA diameter and on MABP in a rat CCW model. Effects of i.c. infusion of various peptides on MMA diameter and on MABP. i.c.: intracarotid artery; MMA: middle meningeal artery; MABP: mean arterial blood pressure; PACAP: pituitary adenylate cyclase-activating polypeptide; VIP: vasoactive intestinal polypeptide; a

Discussion
The long-lasting flushing observed in parallel with a migraine-like headache after PACAP-38 infusion in humans (2,3) set the stage for us to study if PACAP-38-induced vasodilation of MMA could be mediated via degranulation of MCs. We hypothesized that the vascular responses to PACAP-38 in MCD rats would be less than in control rats because of a lack of vasodilatory products released during MC degranulation. To mimic the situation in the human studies, we infused PACAP-38 and the other substances i.v. over 20 minutes. To show the direct effect of PACAP-38, PACAP-27 and VIP on MMA, we gave a one-minute i.c. infusion of the substances. Our results show a lack of change in response to one-minute i.c. infusion of PACAP-38 in MCD rats compared to control rats. The attenuation of the delayed MMA dilatory responses to a prolonged PACAP-38 infusion in MCD rats and in AH-pretreated rats indicates a role of MC degranulation in the delayed phase of the PACAP-38-induced dilation of MMA.
We found that i.c. infusion of PACAP-38, PACAP-27 and VIP induced maximum dilations at 10-fold lower doses compared to a previous i.v. infusion study with these peptides (6). In pressurized isolated MMA segments, pmolar concentration of PACAP-38 caused PAC-1 receptor mediated dilation (14). This response was abolished by the PAC-1 antagonist PACAP(6-38) (14). In the CCW model, a one-minute i.v. infusion of high doses of the PACAP(6-38) (10–50 µg kg−1) gave a dose-dependent decrease in MABP and increase in MMA diameter (6). But it was not able to inhibit dilatory responses of PACAP-38. For the long-term infusion of PACAP(6-38), we have used a 25 times lower dose (per minute dose) of PACAP(6-38), which did not change MMA diameter and MABP during the 20-minute infusion. However, 20 minutes after termination of the infusion a significant sustained increase in MMA diameter was observed. This delayed vasodilation of MMA was completely abolished in MCD rats, which further supports the involvement of MCs as PACAP(6-38) is a potent MC degranulator (9). Although PACAP-38 and CGRP have similar elimination half-life, only PACAP-38 infusion has shown sustained effects. This is probably due to the fact that CGRP cannot degranulate MC (15).
In MCD rats, i.v. infusion of PACAP-27 did not give any significant increase in dilation and i.c. infusion of PACAP-27 gave a lesser peak dilation. PACAP-38, PACAP(16-38) and PACAP-27 released histamine from rat peritoneal mast cells (RPMCs). In this assay PACAP-38 and PACAP 16-38 were the most active peptides, followed by PACAP-27 (16). It has been shown that PACAP-38 has a much stronger degranulatory effect on rat peritoneal and dural MCs than VIP and PACAP-27 (9). Although PACAP-38 and PACAP-27 bind to the same receptor, their potency to trigger intracellular cascades might be different and it has been suggested that PACAP-38 acts on a non-PAC-1, vasoactive PAC-1 (VPAC-1) and VPAC-2 receptor to cause degranulation of peritoneal MCs (9).
Compound 48/80 has a direct excitatory effect on enteric neurons and visceral afferents and it was suggested that functional changes measured in tissue or animal models may involve an MC independent effect of compound 48/80 (17). Chronic treatment with compound 48/80 might have released endogenous PACAP from sensory neurons and might desensitize its receptors, which can be responsible for the decrease in PACAP-27-induced vasodilation observed after both i.c. and i.v. infusions. Since PACAP-38 i.c. response (direct receptor mediated response) was not affected by MC depletion, it is not possible that reduction in the dilatory response to PACAP-38 in the MCD rat was due to desensitization or any other nonspecific effect of compound 48/80. Furthermore, the lack of difference in dilation of CGRP in normal rats as compared to MCD rats further confirms that the treatment with compound 48/80 did not cause a reduction in dilatory response due to damage of the vascular smooth muscle cells.
It has been shown that serum levels of tumor necrosis factor-alpha (TNFα) (18) and histamine (19,20) were elevated in migraine patients. Interestingly, a study in children suffering from migraine showed a correlation between the number of migraine attacks and the levels of the MC degranulation marker tryptase in urine (21). Furthermore, the MC stabilizer sodium cromoglycate prevented migraine induced by food triggers (22). Histamine induces headache in control subjects and migraine in migraineurs (23,24). MC degranulation releases histamine (25,26). The rate of histamine elimination from MCs is slow (27). In rats, histamine causes dilation of dural arteries via endothelial H1 receptors and H2 receptors located on the smooth muscle cells (28). Histamine-induced MMA dilation was completely abolished by the i.v. nitric oxide synthase (NOS) inhibitor
In humans, dilation of cranial arteries alone is not enough to cause an immediate headache (29,30). PACAP-38 infusion-induced vasodilation of dural arteries might not be the only cause of migraine-like headache. MC mediators released by PACAP-38 infusion in humans might be responsible for long-lasting flushing and delayed migraine-like attacks. We also speculate that PACAP-38 released during migraine attacks from sensory and parasympathetic nerve endings can degranulate MCs present around dural arteries. Released MC mediators can further sensitize sensory neurons to cause a prolonged migraine attack.
Article highlights
Pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38) infusion caused sustained dilation of the middle meningeal artery (MMA) in a rat closed-cranial window model. Prolonged MMA dilation induced by PACAP-38 was abolished in mast cell-depleted rats. Histamine released from mast cells was responsible for the delayed effect of PACAP-38 infusion.
Footnotes
Funding
This work was supported by an International Mobility Grant (Faculty of Health Sciences, University of Copenhagen), Candy's Foundation, Lundbeck Foundation and the A.P. Møller Foundation for the Advancement of Medical Science.
Conflict of interest
None declared.
