Abstract
Background: Pituitary adenylate cyclase activating peptide-38 (PACAP-38) has been shown to induce migraine in migraineurs, whereas the related peptide vasoactive intestinal peptide (VIP) does not. In the present study we examine the hypothesis that PACAP-38 and its truncated version PACAP-27 but not VIP cause degranulation of mast cells in peritoneum and in dura mater.
Methods: The degranulatory effects of PACAP-38, PACAP-27 and VIP were investigated by measuring the amount of N-acetyl-β-hexosaminidase released from isolated peritoneal mast cells and from dura mater attached to the skull of the rat in vitro. In peritoneal mast cells N-truncated fragments of PACAP-38 (PACAP(6–38), PACAP(16–38) and PACAP(28–38)) were also studied. To investigate transduction pathways involved in mast cell degranulation induced by PACAP-38, PACAP-27 and VIP, the phospholipase C inhibitor U-73122 and the adenylate cyclase inhibitor SQ 22536 were used.
Results: The peptides induced degranulation of isolated peritoneal mast cells of the rat with the following order of potency: PACAP-38 = PACAP(6–38) = PACAP(16–38) » PACAP-27 = VIP = PACAP(28–38). In the dura mater we found that 10−5 M PACAP-38 was significantly more potent in inducing mast cell degranulation than the same concentration of PACAP-27 or VIP. Inhibition of intracellular mechanisms demonstrated that PACAP-38-induced degranulation is mediated by the phospholipase C pathway. Selective blockade of the PAC1 receptor did not attenuate degranulation.
Conclusion: These findings correlate with clinical studies and support the hypothesis that mast cell degranulation is involved in PACAP-induced migraine. PACAP-38 has a much stronger degranulatory effect on rat peritoneal and dural mast cells than VIP and PACAP-27. The difference in potency between PACAP-38- and PACAP-27/VIP-induced peritoneal mast cell degranulation is probably not related to the PAC1 receptor but is caused by a difference in efficacy on phospholipase C.
Introduction
Human provocation studies have been used in different forms for many years in order to elucidate the molecular mechanisms of migraine. Several signalling molecules induce headache in healthy volunteers and migraine-like headaches in migraineurs (1–5). Blockers of nitrogen oxide synthase (NOS) (6) and calcitonin gene-related peptide (CGRP) (7,8) have shown therapeutic efficacy in acute migraine attacks.
Pituitary adenylate cyclase activating peptide-38 (PACAP-38) is a 38 amino acid neuropeptide belonging to the secretin/glucagon superfamily. As well as this most abundant form, it is present in the body in a C-truncated 27 amino acid form (PACAP-27). The PACAPs are structurally closely related to vasoactive intestinal polypeptide (VIP), a 28 amino acid peptide from the same family with 68% sequence identity from the N-terminus (9). VIP and the PACAPs partially share receptors: VIP and both PACAPs all function as agonists on the VPAC1 and the VPAC2 receptors with close to equal affinity. A dedicated receptor for PACAP (PAC1) has nearly the same binding affinity for the two isoforms of PACAP, whereas VIP has affinity 3–4 orders of magnitude lower for this receptor. VPAC1, VPAC2 and PAC1 receptors are all known to activate adenylate cyclase through Gs protein. Furthermore, the PAC1 receptor can activate phospholipase C (PLC) through Gq/11 protein (10).
Although PACAP-38 has been demonstrated to be an effective inducer of headache and migraine (5), infusion of comparable doses of VIP caused only a minimal and transient headache (11,12). This might implicate the PAC1 receptor in migraine pathophysiology, and makes it a putative target for migraine treatment.
For several hours after the infusion of PACAP-38 there are severe skin flushing and dilatation of the superficial temporal artery (5). These effects were not present to the same degree after infusion with VIP (11,12). The pronounced and long lasting flushing suggests the involvement of mast cells. Erythema can be a direct effect of vasodilators, but this effect is usually transient, and diminishes as the drug is eliminated from the circulation. In humans, the half-life of PACAP-38 is 5–10 minutes (13). Return to baseline would be expected within minutes, not hours.
Mast cells are part of the immune system and closely related to basophile granulocytes. They contain a mixture of inflammatory mediators, often called an inflammatory soup, which can be released during degranulation. Some of the primary constituents of the inflammatory soup are histamine, heparin, serotonin, prostanoids and cytokines (14,15). Mast cells are divided into mucosal type mast cells and connective tissue type mast cells. Peritoneal mast cells and dural mast cells both belong to the connective tissue type (16), as do the mast cells present in the skin.
Migraineurs have been shown to have elevated serum levels of histamine (17,18) and tumour necrosis factor-alpha (TNFα) (19), both during attacks and in the interictal phase. Some patients report certain foods as triggers for attacks, and pre-administration of a mast cell stabilizing substance (cromolyn sodium) have shown some efficacy in preventing precipitation of migraine attack after intake of known trigger (20). These findings may indicate mast cell involvement in certain types of migraine.
The aim of the present study was to compare the effects of VIP, PACAP-38 and related peptides on mast cells and to study the signalling pathway of degranulation by blockade of intracellular transduction mechanisms.
Materials and methods
Rat peritoneal mast cells
The procedure for mast cell isolation and mediator release assay were adapted with modifications from Passante and colleagues (21).
Male Sprague-Dawley rats (300–400 g) were exsanguinated under CO2 anaesthesia. 20 ml oxygenated buffer (137 mM NaCl, 2.7 mM KCl, 1.0 mM MgCl2, 0.5 mM CaCl2, 0.4 mM NaH2PO4, 5.6 mM glucose and 10 mM HEPES pH 7.6) was injected in the peritoneum, which was gently massaged for 3 minutes. The peritoneum was opened by midline section, and lavage was removed using a pipette. Cells were sedimented by centrifugation for 5 minutes at 400 g at 13°C. Sedimented cells were resuspended in 10 ml of the same buffer, washed and sedimented by centrifugation three times.
A BSA-Percoll density gradient was mixed in a Falcon tube (162 µl 35% bovine serum albumin (BSA), 8.10 ml Percoll® (GE Healthcare, Little Chalfont, Buckinghamshire, UK), 580 µl distilled water and 1.16 ml salt solution (1.54 M NaCl, 27 mM KCl, 3.8 mM CaCl2)). This gives a Percoll® ratio of 81%. The cell pellet was resuspended in 5 ml of the buffer previously used for washing and layered on top of the Percoll. Cells were separated by centrifugation at 225 g for 25 minutes at 13°C. The density gradient was discarded, and pellet was washed three times in the buffer previously used. The pellet was finally resuspended in 1 ml buffer, 25 µl were stained with 5 µl 0.25% toluidine blue at pH 2 and the number of mast cells was counted under microscope. Cell suspension was diluted to 10 ml with buffer again, sedimented by centrifugation and diluted with RPMI1640 medium without phenol red (Labinova AV, Upplands Väsby, Sweden) to achieve a mast cell concentration of 4 × 105 mast cells per ml (2 × 104 mast cells per 50 µl). Aliquots (50 µl) of cell suspension in media were distributed in a transparent 96-well Nunclon™ plate (Thermo Scientific, Waltham, MA, USA). To three wells was added 50 µl media (blank standard samples) and to three wells was added 50 µl media and 20 µl 1.2% Triton-X100 (Sigma-Aldrich, St Louis, MO, USA) (maximum degranulation samples). To the remaining wells was added 50 µl of test substance dissolved in RPMI1640, tests in duplicate in logarithmically increasing concentration steps. For all experiments, two wells with 10−5 M PACAP-38 were used as an additional reference.
The plate was incubated for 30 minutes at 37°C, and centrifuged at 1000 g for 5 minutes at 4°C to stop the reaction. 50 µl of the supernatant of each well (60 µl of the wells with Triton-X) was transferred to a new plate, and to the wells without Triton-X was added 10 µl 1.2% Triton X. To each well was added 60 µl 0.08 M citric acid buffer, pH 4.5 containing 8 mM 4-nitrophenyl-N-acetyl-β-
Degranulation was calculated as percent-wise degranulation compared to 10−5 M PACAP-38. PACAP-38 was found in our pilot experiments to be among the strongest degranulators of a variety of substances tested, so instead of measuring the degranulation as percentage of the N-acetyl-β-hexosaminidase contained in lysed cells, we considered this a better measure of the functional degranulatory capacity of the mast cells.

Transduction mechanism inhibitors against degranulation of peritoneal mast cells
Rat peritoneal mast cells were isolated and experiments were performed as described above, except that the agonist-induced responses were tested in the presence of the phospholipase C inhibitor U-73122 (10−6 M) and/or the adenylyl cyclase inhibitor SQ 22536 (10−5 M). To do this, the mast cells were incubated with the inhibitor for 5 minutes before adding PACAP-38 (10−5 M), PACAP-27 (10−5 M) or VIP (10−5 M).
Degranulation assay of dural mast cells of the rat
Male Sprague-Dawley rats (300–400 g) were exsanguinated under CO2 anaesthesia. The animals were prepared as described earlier (22). Briefly, after decapitation, skin, jaw and cranial muscles were removed from the head. The exposed skull was divided in the midline sagittal plane using a metal saw. Brain halves were carefully removed without disturbing the dura mater. The skull halves were transferred to a beaker of buffer (137 mM NaCl, 2.7 mM KCl, 1.0 mM MgCl2, 0.5 mM CaCl2, 0.4 mM NaH2PO4, 5.6 mM glucose and 10 mM HEPES), and superfused with continuous flow of 500 ml buffer for at least 30 minutes.
The skull halves were transferred to concave moulds of clay lined with Vaseline to prevent leakage, added 350 µl of buffer and placed in a humid heating cabinet at 37°C. The buffer was changed six times at 5 minute interval before starting the experiment.
To establish the baseline for each preparation, skulls was incubated for 2 × 15 minutes with normal buffer (350 µl), saving all aliquots of buffer on ice in Eppendorf tubes for analysis. Preparations were incubated with 350 µl test substance dissolved in buffer for 2 × 15 minutes. All substances were tested at a single, high concentration, 10−5 M.
Samples were centrifuged at 1000 g, 4°C for 5 minutes to sediment any erythrocytes and other cells. Fifty microliters of each supernatant was mixed in duplicate with 60 µl 0.08 M citric acid buffer, pH 4.5 containing 8 mM 4-nitrophenyl-N-acetyl-β-D-glucosaminide in a transparent 96-well plate. The plate was incubated for 2 hours at 37°C, after which the reaction was stopped with 10 µl 1 M NaOH added to each well. The plate was shaken for 5 minutes, and absorbance was measured at 410 nM on a microplate reader (Tecan Infinite M200, Tecan Group Ltd, Männedorf, Switzerland).
Drugs
VIP was acquired from Neo MPS, France and PACAP-38, PACAP(6–38), PACAP(16–38) and PACAP(28–38) were acquired by custom synthesis from Caslo Laboratory ApS, Lyngby, Denmark. All were dissolved in distilled water to 10−4 M, aliquoted and stored at −20°C.
The PAC1 receptor agonist maxadilan, a 61–amino acid peptide with no significant sequence homology to PACAP (23), and the PAC1-receptor-selective antagonist max.d.4, a modified fragment of maxadilan (kindly provided by Dr. Ethan Lerner, Cutaneous Biology Research Centre, Department of Dermatology, Massachusetts General Hospital, Charlestown, Massachusetts, US) were dissolved to 10−4 M in distilled water. U-73122 (Tocris Bioscience, Bristol, UK) was dissolved in ethanol to 10−3 M. SQ 22536 (Tocris Bioscience, Bristol, UK) was dissolved to 10−2 M in dimethylsulfoxide (DMSO). Before use, all antagonists were diluted to final concentration with RPMI1640 medium.
Results
Degranulation of peritoneal mast cells
PACAP-38, PACAP-27, VIP, PACAP(6–38), PACAP(16–38) and PACAP(28–38) induced a concentration-dependent degranulation of peritoneal mast cells (Figure 1A and 1B). The compounds tested seem to be divided in two distinct groups; the efficient degranulators were PACAP-38, PACAP(6–38) and PACAP(16–38), with pEC50 values between 6.58 and 6.21 and Emax values in percentage of the degranulation induced by PACAP-38 being 69.2 ± 7.1% for PACAP(6–38) and 88.1 ± 7.0% for PACAP(16–38). The other group consisted of weaker degranulators being PACAP-27, VIP and PACAP(28–38), with pEC50 values between 5.49 and 4.78 and Emax values of 30.5 ± 6.4% for PACAP-27, 37.4 ± 7.3% for VIP and 37.9 ± 6.6% for PACAP(28–38).
Degranulation of peritoneal mast cells of the rat presented as degranulation in percentage of PACAP-38 which was the strongest mast cell degranulator tested. A) The effect of the endogenous peptides PACAP-38, PACAP-27 and VIP; B) The effect of the different fragments of PACAP-38. Values are given as means ± SEM of 4–8 experiments.
Maxadilan and max.d.4 showed no mast cell degranulating effects when applied at a concentration of up to 10−5 M.
The experiments did not test concentrations higher than 10−5 M of each peptide, even though the concentration-response curves for the weak degranulators are not complete at this concentration. It was decided not to test higher concentrations, as they would be far higher than concentrations encountered physiologically and thus without clinical relevance. With higher concentrations of peptide, mast cell degranulating effects due to unspecific interaction may also increase, yielding possibly misleading figures.
Antagonism of transduction mechanisms
Potent inhibition of PACAP-38-induced degranulation was demonstrated with the PLC inhibitor U-73122 at a concentration of 10−6 M, which decreased the mast cell degranulation to 32.4 ± 4.4% of the degranulation induced without inhibitor (Figure 2A). The adenylate cyclase inhibitor SQ 22536 at a concentration of 10−5 M caused no inhibition of the degranulation, with values of 88.9 ± 3.5% in the presence of vehicle only and 103.9 ± 11.9% in the presence of SQ 22536. Selective antagonism of the PAC1 receptor with antagonist max.d.4 did not inhibit the response. The degranulation induced by PACAP-27 and VIP was not inhibited significantly by either SQ 22536 or U-73122 (Figure 2B and 2C). For all experiments in this series, N = 5, with peritoneal mast cells extracted and pooled from two animals per experiment.
Degranulating effect on peritoneal mast cells of A) PACAP-38, 10−5 M, B) PACAP-27, 10−5 M and C) VIP 10−5 M in the presence of the phospholipase C inhibitor U-73122 (10−6 M) and the adenylyl cyclase inhibitor SQ-22536 (10−5 M) alone or in combination. Values are presented as amount of degranulation in percentages of degranulation with each peptide alone. Values are given as means ± SEM, n = 5, **: p > 0.01 compared with the vehicle group, Mann-Whitney’s t-test.
Skull model
Using the skull model for studying degranulation of dural mast cells, we found that PACAP-38 was the most efficient degranulator (168.2 ± 18.4% of basal release), with PACAP-27 (122.9 ± 4.3% of basal release) and VIP (104.3 ± 5.8% of basal release) being significantly less potent (Figure 3). The control experiments without peptide gave a result of 80.0 ± 4.6% of basal release. There was a non-significant tendency of PACAP-27 to be more potent than VIP. This order of potency is identical to what is seen for the peritoneal mast cells. Because of the lower throughput of this method, only one, high concentration (10−5 M) was tested for each compound, and the truncated fragments of PACAP-38 were not tested. For this series of experiments, N = 6 for each group: 12 animals and 24 hemisected skulls.
Degranulation of mast cells in dura mater after treatment with vehicle, PACAP-38, 10−5 M, PACAP-27, 10−5 M or VIP 10−5 M. Values are presented as amount of degranulation in percentages of the basal mast cell degranulation measured before the initiation of the experiment. Values are given as means ± SEM, n = 5, Mann-Whitney’s t-test. **: significant difference from control (p < 0.01), $$: significant difference from PACAP-27 (p < 0.01), ##: significant difference from PACAP-38 (p < 0.01), @@: significant difference from VIP (p < 0.01).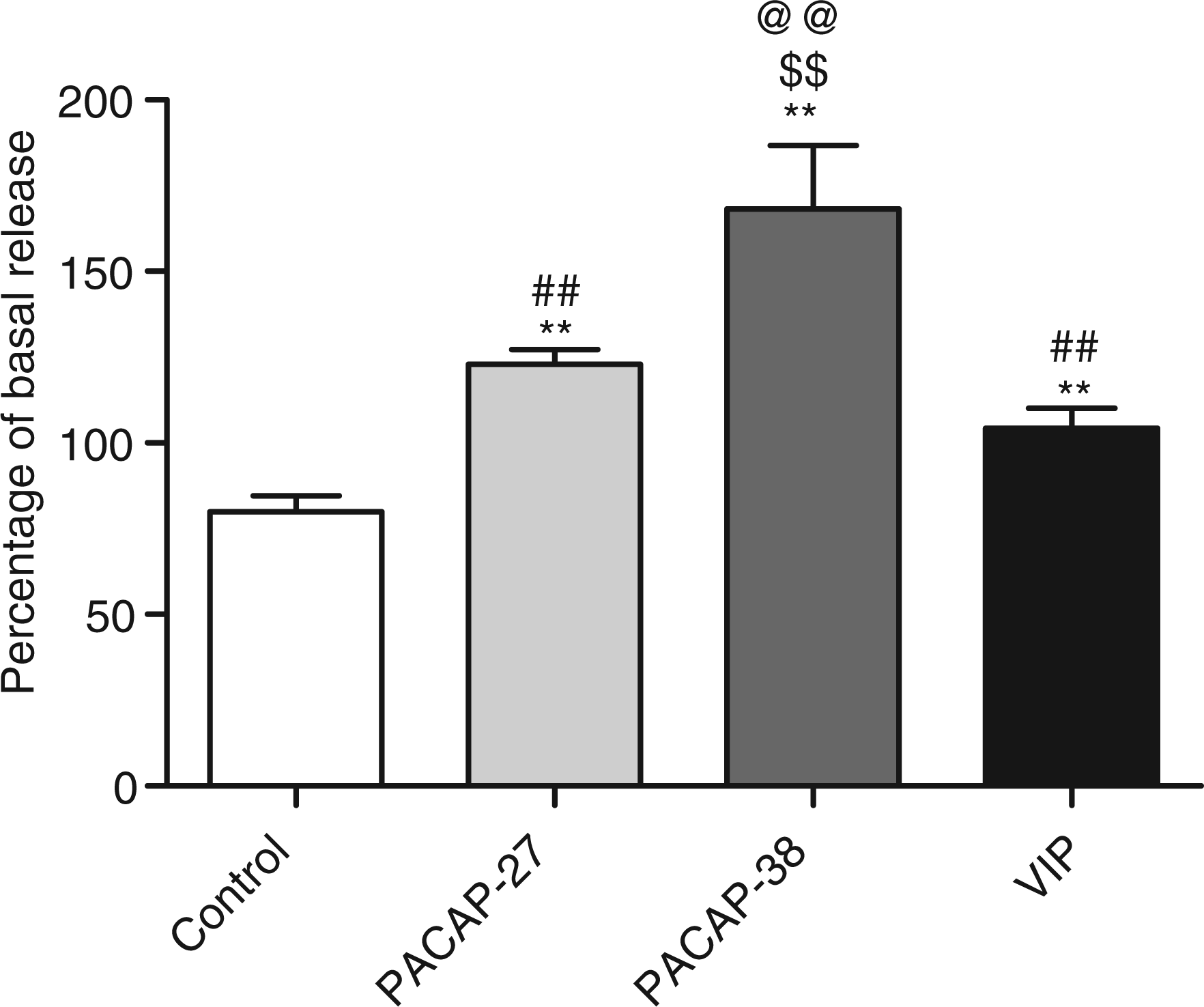
Discussion
Degranulation of rat peritoneal mast cells
The main findings in the present study were that PACAP-38 degranulated peritoneal and dural mast cells significantly more potently than VIP. In humans PACAP-38 causes significantly more migraine-like attacks than VIP. This suggests that degranulation of mast cells may be a mechanism of PACAP-induced migraine. From human studies it was concluded that the migraine provocation might originate from PAC1 receptor activation because PACAP and VIP are roughly equipotent on VPAC1 and VPAC2 receptors. If degranulation of mast cells is important in migraine, the present study suggests, however, that the PAC1 receptor may not be important because a PAC1 receptor agonist was unable to degranulate mast cells. However, the receptor mechanisms of PACAP-induced mast cell degranulation remain unknown.
The low degranulatory potency of PACAP(28–38) indicates that the more potent action of PACAP-38 vs. PACAP-27 is not caused solely by the C-terminus on the longer of the peptides. Part of our findings is supported by previous studies using a slightly different approach (24,25). PACAP(6–38) has been described as having agonistic properties in certain PACAP-related systems (26), while behaving as a full antagonist in other systems. Furthermore, PACAP(6–38) has been shown to have antagonistic action on the VPAC2 receptor (27).
A possible explanation for the difference in potency of PACAP-38 as compared to PACAP-27 and VIP could be the presence of splice variants of the PAC1 receptor, which have been shown to be expressed as a result of alternative splicing of two exons encoding a region in the third intracellular loop (28). It has been suggested that the splice variants have differential coupling to second messengers (29). However, in studies on cells expressing four different PAC1 receptor isoforms, PACAP-38 and maxadilan showed the same rank order of binding affinity and potency on activation of adenylyl cyclase and mobilization of [Ca2+]I (30). Furthermore, the PAC1 receptor antagonist max.d.4 did not inhibit the PACAP-38-induced mast cell degranulation.
Studies of transduction mechanisms in peritoneal mast cell degranulation
The PAC1 receptor is positively coupled to adenylyl cyclase and phospholipase C (31). In a study in PC12 cells, PACAP-38 was 100 to 1000 times more potent in increasing inositol lipid levels than PACAP-27 (32). The effect of the two peptides on adenylyl cyclase activity was equipotent. To test the second messenger systems activated by PACAP-38, PACAP-27 and VIP we studied mast cell degranulation in the presence of the adenylyl cyclase inhibitor SQ 22536 and the phospholipase C inhibitor U-73122. PACAP-38-induced but not PACAP-27- or VIP-induced degranulation was potently inhibited by the phospholipase C inhibitor U-73122, whereas the adenylyl cyclase inhibitor SQ 22536 had no effect on mast cell degranulation induced by any of the three peptides. In combination, SQ 22536 and U-73122 proved no more efficient than the phospholipase C inhibitor alone. Thus, PACAP-38 induces degranulation primarily via phospholipase C. A difference in coupling to the second messenger after activation by PACAP-38 as compared with PACAP-27 and VIP can explain the difference in potency between PACAP-38 and PACAP-27/VIP. SQ 22536 has in previous studies been shown to inhibit PACAP-induced responses (33–35), which is in contrast to our findings in peritoneal mast cells. In most studies using SQ 22536 as an inhibitor of adenylyl cyclase, a concentration of 100 µM has been used, which is 10 times the concentration used in our studies. Solubility and vehicle issues caused us to limit the concentration of SQ 22536, as a higher concentration of DMSO in the experiments would have had mast cell stabilizing effects on the experiment according to pilot studies (data not shown). However, a concentration of 10 µM SQ 22536 has been efficient in inhibiting PACAP-stimulated cAMP production (33). Thus, the concentration we used should have been able to inhibit adenylyl cyclase activated by PACAP and VIP.
Skull model
To investigate whether mast cells in the dura mater respond to PACAP-38, PACAP-27 and VIP as in peritoneal mast cells, we performed a series of experiments in the dural skull model. As only one concentration of peptide could be examined in each skull we choose to use 10−5 M PACAP-38, PACAP-27 and VIP. At this concentration there was a significant difference in the amount of degranulation of peritoneal mast cells to PACAP-38 as compared to PACAP-27 and VIP. In the skull model we found that PACAP-38-induced mast cell degranulation was significantly stronger than mast cell degranulation induced by PACAP-27 and VIP, consistent with the order of potency observed in peritoneal mast cells. This finding suggests that peritoneal mast cells can be used as a model for the effects of PACAP on dural mast cells. Given that the mast cells are not purified in this model, but the entire dura mater is exposed to test substance, it is not impossible that other cells in the preparation contribute with release of N-acetyl-β-hexosaminidase. This enzyme is not unique to mast cells, but present in the lysosomes of multiple tissues. Lysosomes containing the enzyme have been found in smooth muscle cells (36).
Relevance for migraine
We hypothesized that infusion of PACAP-38 induces headache via degranulation of mast cells and found that PACAP-38 is a potent degranulator of rat peritoneal and dural mast cells. If mast cell degranulation is involved in migraine, the dura mater is the most likely site of such mechanism. Involvement of extracranial/peripheral mast cells cannot, however, be excluded. A partial systemic degranulation would liberate large amounts of vasodilatory and proinflammatory substances, eventually reaching the cranial circulation. TNFα and interleukin (IL)-6 cross the blood–brain barrier via facilitated uptake mechanisms (37). In turn, these may interact more or less directly with the cerebral pain pathways. Apart from working directly in the CNS, IL-6 may induce cerebral vascular endothelium to upregulate prostaglandin E2 production through the cyclooxygenase-2 (COX-2) pathway (38). Histamine may have a more direct headache-inducing effect through interaction with the cranial vasculature.
The headache observed after PACAP-38 infusion is divided in two phases; a pain starting almost immediately after start of infusion, and a delayed phase headache starting multiple hours after the end of infusion (39). This may be explained by the immediate headache being caused by degranulating mast cells secreting prefabricated vesicles containing histamine, prostaglandin E2 and other inflammatory substances. After activation, the mast cells may start de novo synthesis of other compounds not preformed, such as TNFα and IL-6 (40), triggering migraine attacks or precipitating a second phase of headache after the time required for synthesizing and releasing these compounds.
We also found that PACAP-27 was between 10 and 100 times less potent as a mast cell degranulator than PACAP-38 was. As far as is known, no headache provocation studies in either healthy volunteers or migraineurs have yet been performed with PACAP-27. If PACAP-38 and PACAP-27 have the same potency order in human mast cells and if degranulation of mast cells in the dura mater is a main cause of PACAP-38 induced headache, PACAP-27 should cause significantly less headache than PACAP-38.
Conclusion
PACAP-38 has a much stronger degranulatory effect on rat peritoneal and dural mast cells than VIP and PACAP-27, and this effect is conserved in N-truncated smaller fragments of the peptide. This may point to mast cell degranulation as a major contributor to PACAP-38 triggered migraine. The difference in potency between PACAP-38- and PACAP-27/VIP-induced peritoneal mast cell degranulation may be caused by different efficacy on phospholipase C.
Footnotes
Acknowledgements
This study supported by the Lundbeck Foundation via the Lundbeck Foundation Center for Neurovascular Signaling (LUCENS), Novo Nordisk Foundation, The Illum Foundation, The Foundation for Neurological Research, Familien Hede Nielsens Foundation and Candy's Foundation.
