Abstract
Background
The exact mechanisms responsible for migraine remain unknown, although it has been proposed that changes in brainstem anatomy and function, even between attacks, may contribute to the initiation and maintenance of headache during migraine attacks. The aim of this investigation is to use brainstem-specific analyses of anatomical and diffusion weighted images to determine if the trigeminal system displays altered structure in individuals with migraine.
Methods
Voxel-based morphometry of T1-weighted anatomical images (57 controls, 24 migraineurs) and diffusion tensor images (22 controls, 24 migraineurs) were used to assess brainstem anatomy in individuals with migraine compared with controls.
Results
We found grey matter volume decreases in migraineurs in the spinal trigeminal nucleus and dorsomedial pons. In addition, reduced grey matter volume and increased free water diffusivity occurred in areas of the descending pain modulatory system, including midbrain periaqueductal gray matter, dorsolateral pons, and medullary raphe. These changes were not correlated to migraine frequency, duration, intensity or time to next migraine.
Conclusion
Brainstem anatomy changes may underlie changes in activity that result in activation of the ascending trigeminal pathway and the perception of head pain during a migraine attack.
Keywords
Introduction
The exact neural mechanisms responsible for the initiation and maintenance of head pain associated with migraine remain unknown, although human brain imaging investigations have shown that during a migraine attack, activity increases in brainstem nuclei such as the spinal trigeminal nucleus, dorsal pons, midbrain periaqueductal gray matter, hypothalamus, and higher brain regions such as the thalamus, cingulate and insular cortices (1–3). In addition to activity changes during migraine attacks, a number of studies have also reported changes between migraines, such as altered sensitivity to somatosensory stimuli and indices of brain anatomy as well as changes in brainstem, thalamic and cortical oscillatory activity (4–6). The mechanisms behind how these functional and anatomical changes occur between migraines, the increases in regional brain activity during migraine and how the underlying factors that trigger a migraine attack all interact, remain hotly debated (7).
For some time, cerebrovascular changes that trigger changes in brain activity have been considered the foundation of migraine pathophysiology and critical for the initiation of a migraine event. Although this might be true, an alternative proposal has emerged suggesting that migraine attacks are initiated by changes within one or more regions within the central nervous system, that is, a central “migraine generator” (8,9). It has been proposed that dysregulation of subcortical sites such as the hypothalamus and midbrain may enable brain activity changes that initiate and maintain a migraine attack, i.e. migraine could result from dysfunction of subcortical sites below the level of the diencephalon, leading to an “abnormal perception of basal level of primary traffic” (10). This idea is controversial, and has been the subject of fierce debate. A recent review focusing on the concept of a migraine generator suggested that in order to support this concept, evidence needs to be provided of “post-mortem histopathology showing potential anatomical and/or molecular abnormalities in candidate brainstem nuclei as compared between migraineurs and non-migraineurs” (11).
Whilst post-mortem studies are logistically difficult, it is possible to use anatomical brain imaging techniques to explore the anatomy of the brainstem in living individuals with and without migraine. Whilst several brain morphology studies have shown that migraine is associated with grey matter volume decreases in areas involved in pain processing including the prefrontal, cingulate and insular cortices as well as in the brainstem, none have focused specifically on the brainstem (5,12,13). This is critical, since the small size of the brainstem means that accurate spatial normalization is essential when exploring changes in regional anatomy. Using brainstem specific analysis techniques, we showed that individuals with painful trigeminal neuropathy, an orofacial neuropathic pain disorder, have reduced grey matter volume and free water movement ease (mean diffusivity) in the nucleus that receives direct nociceptor afferents from the orofacial region, the spinal trigeminal nucleus (14). Similar anatomical changes may also occur in individuals with migraine, as well as alterations in areas such as the midbrain periaqueductal gray matter. If such alterations exist, it would raise the possibility that such changes underlie functional changes that may aid in the generation of migraine or in the central transmission of peripherally initiated noxious information.
The aim of this investigation is to use voxel-based morphometry of T1-weighted anatomical and diffusion weighted images to determine if the trigeminal system displays altered anatomy in individuals with migraine. We hypothesize that, similar to painful trigeminal neuropathy, migraine is associated with reduced grey matter volume and mean diffusivity in the spinal trigeminal nucleus. Furthermore, given the mounting evidence of altered descending analgesic control in individuals with migraine, we also hypothesize that migraine is associated with altered anatomy in areas of the brainstem endogenous circuitry such as the midbrain periaqueductal gray matter.
Methods
Subjects
Twenty-five subjects with migraine (four males, mean [ ± SEM] age: 30.2 ± 2.0) and 57 age and gender matched pain-free controls (14 males, age: 28.3 ± 1.3) were studied. All control and migraine subjects were recruited from the general population using an advertisement. There was no significant difference in age (t test; p > 0.05) or gender composition (χ2test, p > 0.05) between the two groups. Migraine subjects were diagnosed according to the criteria laid out by the International Classification of Headache Disorders (ICHD), 3rd edition, sections 1.1 and 1.2 (15). Five migraineurs reported aura associated with their migraines and the remaining 20 reported no aura. All migraineurs were scanned during an interictal period, that is, at least 72 hours after and 24 hours prior to a migraine event. Exclusion criteria for controls were the presence of any current pain or chronic pain condition, current use of analgesics or any neurological disorder. Exclusion criteria for migraineurs were any other pain condition other than migraine or any other neurological disorder. All migraine subjects indicated the intensity (6-point visual analogue scale; 0 = no pain, 5 = most intense imaginable pain) and drew the facial distribution of pain commonly experienced during a migraine attack. In addition, each subject described the qualities of their migraines and indicated any current treatments used to prevent or abort a migraine once started. Informed written consent was obtained for all procedures according to the Declaration of Helsinki seventh revision and local Institutional Human Research Ethics Committees approved the study.
MRI scanning
Subjects lay supine on the bed of a 3T MRI scanner (Philips Achieva, Neuroscience Research Australia, Sydney) with their head immobilized in a fitting 32-channel head coil. In all 25 migraine and 57 control subjects, a high-resolution T1-weighted anatomical image set covering the entire brain was collected (turbo field echo; echo time = 2.5 ms, repetition time = 5600 ms, flip angle = 8°, voxel size 0.8 × 0.8 × 0.8 mm). In addition, in 24 migraine and 22 of the 57 control subjects (no significant difference in age or gender), two high-resolution diffusion tensor imaging (DTI) image sets covering the entire brain were collected using a single-shot multisection spin-echo echo-planar pulse sequence (repetition time = 8788 ms; flip angle = 90°, matrix size = 112 × 112, field of view = 224 × 224 mm, slice thickness = 2.5 mm, 55 axial slices). For each slice, diffusion gradients were applied along 32 independent orientations with b = 1000 s/mm2 after the acquisition of b = 0 s/mm2 (b0) images. Two DTI acquisitions were averaged to improve signal-to-noise ratios. One T1-weighted anatomical and one DTI image set from separate migraine subjects were later removed due to excessive head movement, resulting in 24 migraineurs in each of the brainstem analyses.
Image analysis
Brainstem VBM
Using Statistical Parametric Mapping version 12 software (Wellcome Trust Centre for Neuroimaging, University College London, UK), T1-weighted images were segmented and spatially normalized with a dedicated symmetrical brainstem template, using the SUIT toolbox. Each image was cropped and the brainstem masked before spatial normalization. The subsequent spatial normalization and re-slicing was performed using the “preserve” option, which produced brainstem “maps” of grey matter probabilities modulated by the volume changes due to the normalization (probability × volume), i.e. maps of regional grey matter volume. The maps were re-sliced into Montreal Neurological Institute (MNI) space, and spatially smoothed using a Gaussian filter (3 mm full-width-at-half-maximum). A small smoothing kernel was used to maintain spatial accuracy in small brainstem sites. Significant differences in grey matter between migraine and control subjects were determined using a voxel-by-voxel analysis (p < 0.05, false discovery rate corrected at a voxel level, minimum cluster size 5 contiguous voxels). Age and gender were included as nuisance variables. Clusters of significant difference were then overlaid onto the SUIT brainstem template image for visualization. The location of these clusters was identified using the Atlas of the Human Brainstem by Paxinos and Huang (16).
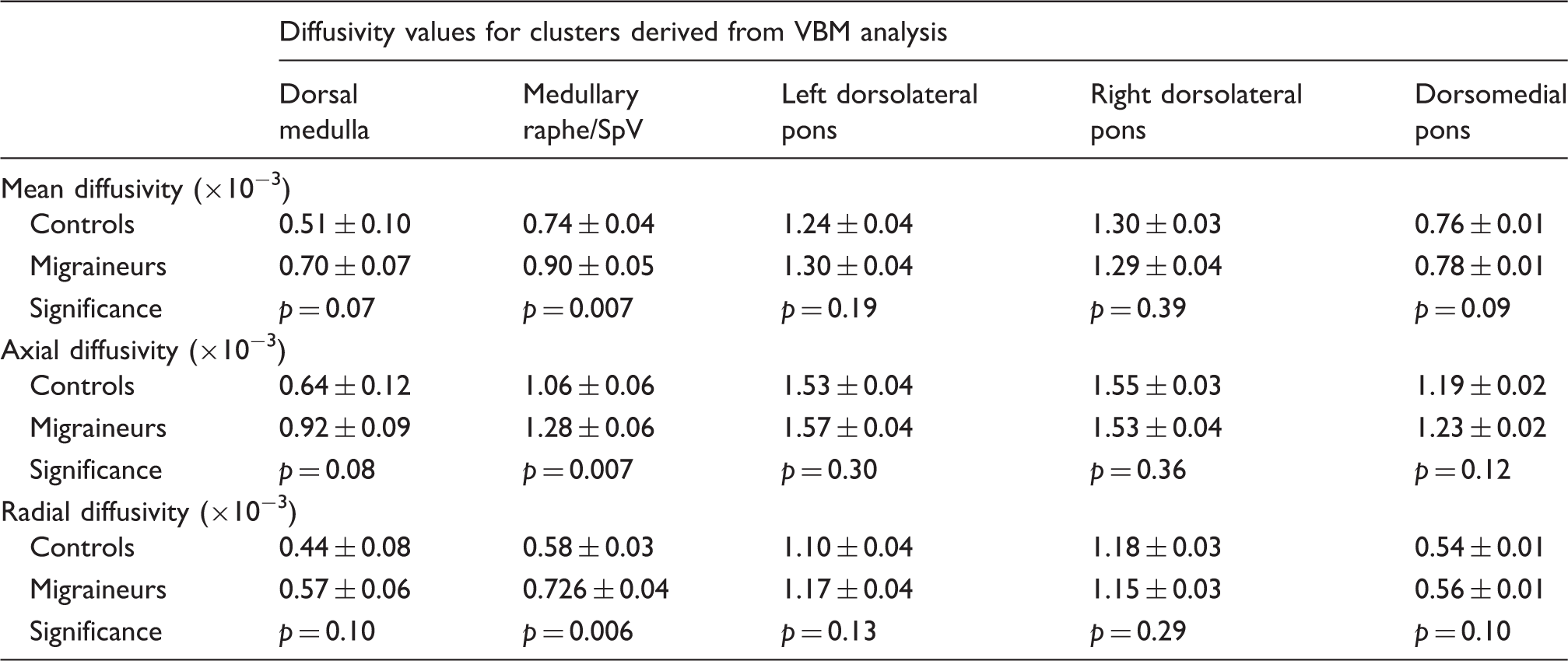
Using SPM's Volumes toolbox, the values at each voxel in each significant cluster were extracted from the grey matter volume maps (probability × volume) and averaged over the entire cluster. These values were plotted to illustrate possible correlations between local grey matter volumes and migraine characteristics, i.e. frequency, years suffering, intensity and days to next migraine (Pearson r, p < 0.05). To determine the potential influence of daily medication use, grey matter volumes in each cluster were compared between the migraineurs taking daily medication and those that were not (two-tailed t-test, p < 0.05, Bonferroni corrected for multiple comparisons). Differences in grey matter volumes in those with and without aura were also determined (two-tailed t-test, p < 0.05, Bonferroni corrected for multiple comparisons). In addition to extracting grey matter volumes, we extracted the mean, axial and radial diffusivity values from each significant cluster. We determined if any of these clusters showed significantly different diffusion values compared with controls, using a two-sample, two-tailed t-test (p < 0.05, Bonferroni corrected for multiple comparisons).
Brainstem DTI
The two diffusion tensor image sets from each subject were realigned based on the b0 images, and the diffusion tensors were calculated from the images using a linear model. Mean diffusivity (MD) whole-brain maps were then derived. These images were then coregistered to each individual subject's T1-weighted image and the brainstem was isolated, spatially normalized and re-sliced to the SUIT template using the parameters derived from the aforementioned T1-weighted SUIT analysis. This process resulted in brainstem maps of diffusion values, spatially normalized in MNI space with raw intensities preserved (non-modulated). The images were then smoothed (3 mm full-width-at-half-maximum) and significant differences in diffusion values between migraineurs and controls determined using a voxel-by-voxel analysis (p < 0.05, false discovery rate corrected at a voxel level, minimum cluster size 5 contiguous voxels). Age and gender were included as nuisance variables. No voxels survived this stringent threshold, so we reduced the stringency (p < 0.001, uncorrected) and performed cluster correction (Bonferroni correction by the number of voxels in each cluster) to assess if more subtle diffusion changes occurred. At this threshold, two clusters emerged and these were subsequently overlaid onto the SUIT brainstem template image for visualization.
Using SPM's Volumes toolbox, the values at each voxel in each significant cluster were extracted from the mean, axial and radial diffusion maps and averaged over the entire cluster. These values were plotted, and correlations between these diffusion values and migraine characteristics were assessed (Pearson r, p < 0.05). To determine the potential influence of daily medication use, in migraineurs we compared diffusion values in each cluster in those taking daily medication with those that were not (two-tailed t-test, p < 0.05, Bonferroni corrected for multiple comparisons). Differences in diffusion values in those with and without aura were also determined (two-tailed t-test, p < 0.05, Bonferroni corrected for multiple comparisons). In addition, we extracted the grey matter volumes from each significant cluster and determined if these were significantly different to controls, using a two-sample, two-tailed t-test (p < 0.05, Bonferroni corrected for multiple comparisons).
Results
Migraine characteristics
Using a self-report questionnaire in which each migraine subject described the most common location of their migraines over the past 12 months, in 10 of the 25 migraineurs headaches were more common on the right side, in five they were more common on the left, and in the remaining 10 they were most often bilateral. A drawing showing the distribution of pain during migraine attacks is shown in Figure 1a. Migraine subjects most frequently described their migraine pain as “throbbing”, “sharp” and/or “pulsating” in nature and indicated that “stress”, “lack of sleep” and/or “dehydration” most often triggered their migraine attacks (Figures 1b and 1c). The mean estimated frequency of migraine attacks was 18.9 ± 2.3 per year, mean length of time since the onset of migraine attacks (years suffering) 14.6 ± 2.2 years, and mean pain intensity of migraines 3.7 ± 0.1 on a 6-point visual analogue scale. Although 15 of the 25 migraineurs were taking some form of daily medication (mostly the oral contraceptive pill), none of the migraine subjects was taking prophylactic medication prescribed for migraine. See Table 1 for migraine subject characteristics.
Pain distribution, quality and triggers in migraineurs (n = 24). (a) Overlap of individual migraine pain patterns: The white-grey-black scale bar corresponds to the pain overlap; (b) Frequency (percentage of subjects) of words chosen to describe the quality of migraine pain; (c) situations that most commonly trigger the onset of a migraine attack. Migraine subject characteristics. B = bilateral, L = left, OCP = oral contraceptive pill; R = right.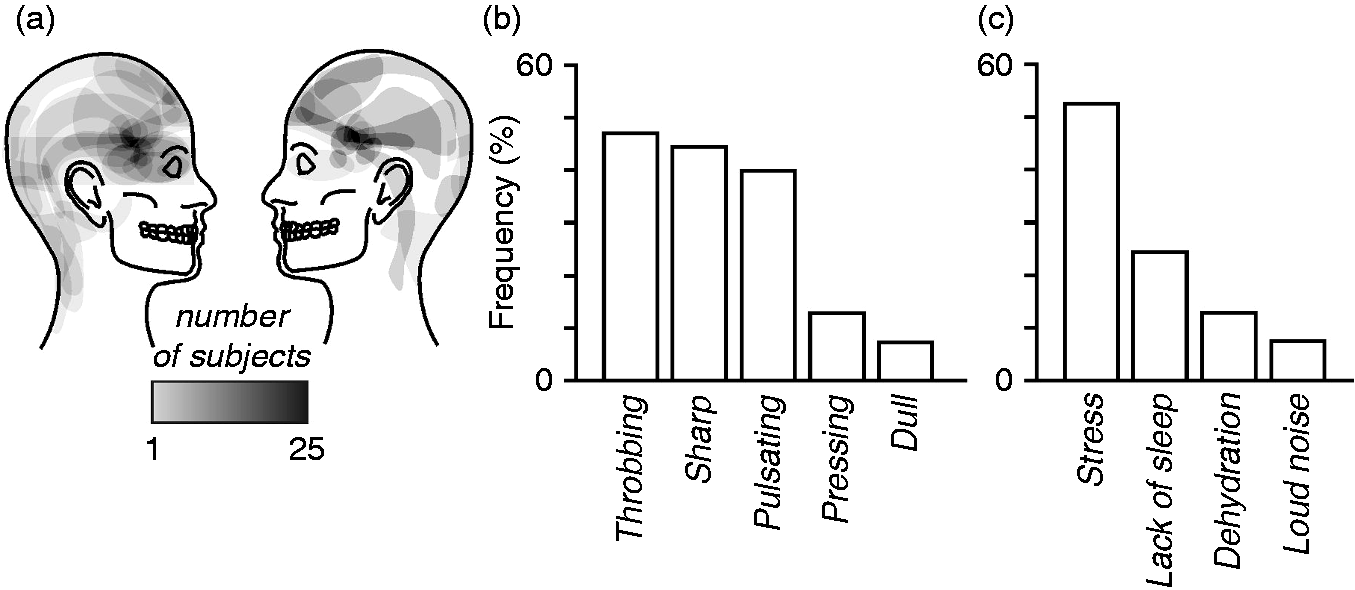

Grey matter volume changes (VBM)
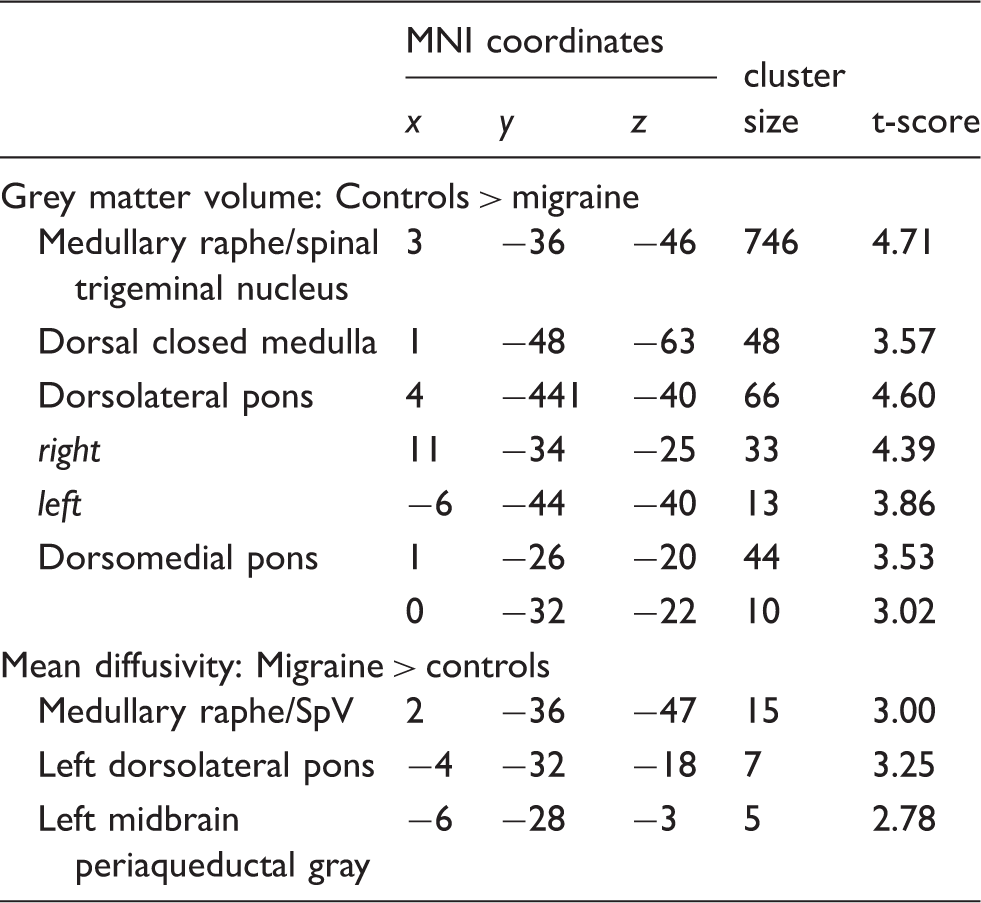
Comparison of grey matter volumes in controls and migraineurs revealed significant regional grey matter volume changes in several regions (Figure 2, Table 2). Migraineurs displayed significant reductions in grey matter volume in the dorsal closed medulla in the region of the nucleus tractus solitarius (controls 0.27 ± 0.01, migraineurs 0.22 ± 0.01, p < 0.001), the region of the medullary raphe/spinal trigeminal nucleus (SpV) (mean ± SEM grey matter probability × volume: controls 0.16 ± 0.01, migraineurs 0.11 ± 0.01, p < 0.001), left and right dorsolateral pons (dlPons) (left: controls 0.27 ± 0.01, migraineurs 0.23 ± 0.01, p = 0.001; right: controls 0.286 ± 0.01, migraineurs 0.23 ± 0.01, p < 0.001) and in the region of the dorsomedial pons (dmPons) (controls 0.26 ± 0.01, migraineurs 0.22 ± 0.01, p = 0.004). In no brainstem region was grey matter volume greater in migraineurs compared with controls.
Regional grey matter volume changes in migraineurs (n = 24) compared with pain-free controls (n = 57) overlaid onto sagittal and axial brainstem template images. Significant grey matter volume decreases are represented by a t-statistic with a cool colour scale. Slice locations are indicated at the upper right of the sagittal and the upper left of the axial slices in Montreal Neurological Institute space. Plots of mean ( ± SEM) grey matter volumes derived from each region are shown to the right. Compared to controls, migraineurs have lower grey matter volumes in the right spinal trigeminal nucleus caudalis (SpV), medullary raphe, dorsal medulla, dorsolateral pons (dlPons) and dorsomedial pons. Montreal Neurological Institute (MNI) coordinates of significant grey matter volume (p < 0.05 false discovery rate corrected) and mean diffusivity (p < 0.001, uncorrected) differences between controls and migraineurs.
Diffusion and p values in clusters derived from the voxel based morphometry (VBM) grey matter volume analysis. The grey shading indicates clusters in which diffusivity values were significantly greater in migraineurs than in controls (p < 0.05, Bonferroni corrected). Note that only the medullary raphe/spinal trigeminal nucleus (SpV) displayed a significant increase in diffusivity compared with controls.
Diffusion value changes (DTI)
In addition to changes in grey matter volume, DTI analysis at a more liberal statistical threshold revealed MD differences in two brainstem sites (Figure 3, Table 2). Compared with controls, migraineurs had greater MD in the region of the medullary raphe/SpV (mean ± SEM [ × 10−3] MD: controls 0.75 ± 0.02, migraineurs 0.82 ± 0.02, p = 0.002), left dlPons (controls 0.75 ± 0.01, migraineurs 0.81 ± 0.01, p < 0.001) and left midbrain periaqueductal gray matter (PAG: mean ± SEM [ × 10−3] MD: controls 0.90 ± 0.02, migraineurs 0.96 ± 0.01, p = 0.005). Within these three clusters, both axial and radial diffusion values were significantly greater in migraineurs compared with controls (raphe/SpV: axial: controls 1.03 ± 0.03, migraineurs 1.14 ± 0.03, p = 0.002; radial: controls 0.60 ± 0.02, migraineurs 0.67 ± 0.02, p = 0.003; dlPons: axial: controls 1.36 ± 0.02, migraineurs 1.42 ± 0.02, p = 0.008; radial: controls 0.45 ± 0.01, migraineurs 0.52 ± 0.02, p = 0.007; PAG: axial: controls 1.09 ± 0.02, migraineurs 1.16 ± 0.02, p = 0.008; radial: controls 0.81 ± 0.02, migraineurs 0.87 ± 0.02, p = 0.005). In no region was MD significantly less in migraineurs compared with controls.
Regional mean diffusivity changes in migraineurs (n = 24) compared with pain-free controls (n = 22) overlaid onto sagittal and axial brainstem template images. Significant mean diffusivity (MD) increases are represented by a t-statistic with a hot colour scale. Slice locations are indicated at the upper right of the sagittal and the upper left of the axial slices in Montreal Neurological Institute space. Plots of mean ( ± SEM) MD, axial diffusivity and radial diffusivity derived from each region are shown to the right. Compared to controls, migraineurs show increased mean and axial diffusivity in the region of the medullary raphe/spinal trigeminal nucleus (SpV), dorsolateral pons (dlPons) and midbrain periaqueductal gray matter (PAG). (a) Plots of mean ( ± SEM) MD, axial diffusivity and radial diffusivity derived from clusters displaying significantly lower grey matter volume in migraineurs compared with controls. Note that although all of these regions had significantly reduced grey matter volumes, only the medullary raphe also had significantly increased mean and axial diffusivity; (b) plots of mean ( ± SEM) grey matter volume derived from clusters displaying significantly increased mean diffusivity in migraineurs compared with controls. Only the medullary raphe displayed both grey matter volume and diffusivity changes in migraineurs.

In migraine subjects, MD values in these clusters were not significantly correlated to migraine frequency (raphe/SpV: r = −0.03, p = 0.90; dlPons: r = 0.07, p = 0.72; PAG: r = 0.02, p = 0.91), years suffering (raphe/SpV: r = 0.28, p = 0.19; dlPons: r = 0.29, p = 0.16; PAG: r = 0.08, p = 0.71), intensity of migraine pain (raphe/SpV: r = 0.26, p = 0.22; dlPons: r = 0.32, p = 0.12; PAG: r = −0.17, p = 0.41) or days to next migraine (raphe/SpV: r = −0.29, p = 0.19; dlPons: r = 0.07, p = 0.75; PAG: r = 0.15, p = 0.51). Comparison of those migraineurs taking daily medication with those that were not revealed no significant differences in MD in either of these regions (raphe/SpV: p = 0.49; dlPons: p = 0.92; PAG: p = 0.47). Comparison of those with and without aura also resulted in no significant differences (raphe/SpV: p = 0.88; dlPons: p = 0.77; PAG: p = 0.50). In addition, extraction of grey matter values from each cluster revealed that grey matter was significantly reduced in migraineurs in the medullary raphe (controls 0.13 ± 0.01, migraineurs 0.10 ± 0.01, p = 0.002), but was not different between groups in the dlPons (controls 0.13 ± 0.01, migraineurs 0.12 ± 0.01, p = 0.18) or PAG (controls 0.18 ± 0.01, migraineurs 0.17 ± 0.01, p = 0.44) (Figure 4b, Table 3).
Discussion
The results of this study reveal that migraine is associated with changes in the volume and diffusion properties in a number of brainstem regions. Grey matter volume decreases occurred in the region that receives orofacial noxious afferents and in the dorsolateral and dorsomedial pons. In addition, both reduced grey matter volume and increased free water diffusivity occurred in areas of the descending pain modulatory system, that is, in the PAG, medullary raphe and dorsal medulla in the region of the subnucleus reticularis dorsalis. In migraine subjects, these grey matter volume and diffusivity changes were not influenced by daily medication use nor were they correlated to migraine frequency, duration or pain intensity.
Over the past decade a number of brain morphometric studies have explored grey matter changes associated with migraine (5,13,17–20). These studies report increased cortical volume/thickness in the primary somatosensory cortex, hippocampus and caudate nucleus and reduced volume in areas including the precentral gyrus, cingulate and insular cortices. None of these previous investigations focused their analysis specifically on the brainstem, and although one recent investigation did report a reduction in overall brainstem volume in migraineurs, the authors did not perform a voxel-by-voxel brainstem analysis (21). They did however find that migraine was associated with a significant overall brainstem volume reduction, consistent with the regional grey matter reductions reported in this investigation. Although investigation of the anatomy of higher brain centers is important, given the role of brainstem regions in transmitting noxious information from the cranial vessels and meninges, modulating noxious inputs and even its potential role as a migraine generator, the investigation of brainstem anatomy and function could highlight central changes critical for the generation of migraine pain.
It has been argued for some time that the headache phase of migraine results from activation of nociceptors in brain meninges and large cerebral arteries that are innervated by trigeminal afferents (22). In rodents, afferents innervating the dura mater and the middle meningeal artery terminate primarily in the caudalis and interpolaris divisions of the right spinal trigeminal nucleus as well as in the right upper cervical dorsal horn (23,24). Those afferents innervating the middle cerebral artery project to the trigeminal principal nucleus oralis and interpolaris subdivisions as well as the dorsal horn of the upper cervical spinal cord and the PAG (25). We found that migraine subjects had significantly reduced grey matter volumes in all divisions of SpV. We have recently reported decreased grey matter volumes in the right SpV in individuals with chronic orofacial neuropathic pain following endodontic treatment (14), although in contrast to migraineurs, these subjects also displayed reduced mean diffusivity in the same region.
The nature of the cellular changes underlying volumetric and diffusion changes is unclear. Whilst grey matter volume decrease may result from neuronal loss, it may also result from shrinkage or atrophy of neurons or glia, or synaptic loss (26). In addition, MD increases are a marker of more subtle changes in tissue microstructure, which may result from tissue shrinkage or gliosis (27). Whilst other forms of more constant neuropathic pain conditions are associated with neuronal degeneration in the region of primary afferent synapse, including significant reductions in inhibitory interneurons synapse (28–32) and increased sensitivity (33,34) it remains unknown if a similar phenomenon occurs in migraineurs. Migraine is associated with cutaneous allodynia, which is likely driven by central sensitization within brainstem trigeminal neurons (35,36). Despite this increased sensitivity, it has been shown that during the interictal phase, migraineurs display reduced SpV sensitivity to trigeminal noxious stimulation compared with controls (37). Importantly, the same study showed that SpV activation during noxious stimuli increases to control levels in migraineurs in the 72-hour period prior to a migraine attack, suggesting the sensitivity of the trigeminal system alters over the migraine cycle. It is possible that there are also dynamic changes in regional grey matter volume within the brainstem that underlie this reported change in SpV sensitivity. We speculate that such dynamic changes could, for example, include changes in astrocyte activation and an associated release of gliotransmitters that sensitize trigeminal brainstem neurons, resulting in increased responsiveness to peripheral noxious inputs immediately before and during a migraine attack (38). Interestingly, none of the regional anatomical changes were correlated to migraine properties such as frequency, intensity or duration, which suggests that the changes are not cumulative over time and is consistent with the idea that they may be dynamic in nature and therefore unlikely to be correlated to such indices. Such dynamic changes would be consistent with astrocyte activation.
In addition to SpV, we found grey matter reduction and MD increases in the region of the medullary raphe, and MD increases with no grey matter change in the rostral dlPons and PAG. The PAG – medullary raphe – SpV/dorsal horn circuit forms part of a well-described endogenous analgesic brainstem circuitry that can inhibit incoming noxious information at the primary afferent synapse. The PAG region with altered diffusion was located in the region of the ventrolateral PAG column, an area that receives primarily deep noxious inputs and produces passive coping behaviors and opiate-mediated analgesia upon activation (39). Furthermore, experimental animal studies have shown that ventrolateral PAG stimulation inhibits SpV activity evoked by stimulation of the superior sagittal sinus (40), probably via the medullary raphe and in particular the nucleus raphe magnus (41,42). It is likely that altered PAG anatomy is associated with an altered ability of the individual to dampen incoming noxious trigeminal information, which would increase the propensity for migraine attacks.
We also found grey matter volume decreases in other brainstem regions thought to be responsible for endogenous analgesia, such as the dlPons and in the dorsal medulla in the region of the subnucleus reticularis dorsalis (43). We have recently shown that an individual's expression of conditioned pain modulation (CPM) analgesia, i.e., the ability for one noxious stimulus to inhibit another at a distant site, is associated with activity changes within the dlPons and the subnucleus reticularis dorsalis (43). Whilst reduced CPM responsiveness has been shown to occur in many chronic pain conditions (44), it appears that its role in migraine may be more subtle. Several reports suggested reduced CPM ability in migraine (45,46), others found no change (47,48) and one reported a gradual reduction in CPM responsiveness upon repeated noxious stimulation (49). These contradictory results suggest a more subtle role for endogenous pain mechanisms in migraine, and the apparent inconsistencies may reflect varying endogenous analgesic responsiveness across the migraine cycle. It might be the case that endogenous analgesic ability remains at control levels for the majority of an individual's migraine cycle but then reduces immediately prior to a migraine attack, increasing the likelihood of an attack. Consistent with this idea, it was recently reported that during a migraine attack, migraineurs displayed increased grey matter density in areas of the insula and putamen, changes that were not present during the interictal period (50), two regions that also display altered activity changes associated with CPM responsiveness (51).
In addition to anatomical changes in the SpV and brainstem, regions that can modulate trigeminal inputs, we also found decreased grey matter volume in the dorsomedial pons. This change was in the same region shown to be activated during migraine attacks (1,3,52). Furthermore, this region shows increased activation to noxious trigeminal stimulation during a migraine attack compared with that displayed during the interictal period (37). This pontine region encompasses the serotonergic paramedian raphe nucleus, which is consistent with the finding of increased serotonergic receptor availability within this region during migraine attacks (53). It has been shown that the pontine raphe region projects rostrally to the anterior forebrain and appears to be involved in emotional processing (54,55). Although this area may be involved in processing the emotional aspect of pain during migraine, its role is yet to be determined.
There are a number of methodological and subject related limitations to this study. It is known that analgesic and mood altering medications as well as the use of the contraceptive pill can change brain anatomy (56,57). Although only four of the 25 migraineurs were on daily analgesic or mood altering medications, 10 migraineurs were taking the oral contraceptive pill. Despite this, we are confident medication use did not play a significant role in our study, since we found no differences when comparing migraineurs that did and did not take any medication. In addition, since the spatial resolution of human brainstem imaging, particularly DTI, is relatively low, it is difficult to determine the precise location of each cluster with respect to small brainstem nuclei. We have however, ascribed the location of each significant cluster using brainstem atlases and placed the changes into context with respect to the existing human and experimental animal research. Future improvements in MRI spatial resolution will certainly aid in defining specific nuclei and tracts associated with migraine. Finally, we did use an uncorrected threshold for the initial overlay of diffusion differences between migraineurs and controls, which can result in false positives. However we used cluster correction to limit this potential issue and we are therefore confident that the changes reported in areas such as the PAG and medullary raphe are indeed appropriate (58).
Overall, our findings suggest that migraine is associated with anatomical changes within various brainstem structures involved in trigeminal noxious transmission and endogenous analgesia. Whilst our data clearly show that, even between attacks, migraineurs have altered brainstem structure, it remains unknown what effect these changes have on brainstem processing. Evidence of altered brainstem processing at different stages of the migraine cycle raises the possibility of dynamic changes in brainstem structure and function throughout the migraine cycle, which may either trigger or alter the sensitivity of the brainstem so that an external trigger results in a migraine attack. Future investigations exploring brainstem resting activity, evoked activity and anatomy over the migraine cycle may provide evidence supporting such a proposal. If dynamic changes in brainstem function and structure do occur, we may be in a position to modify these changes and potentially prevent the triggering of a migraine attack.
Footnotes
Key findings
We found grey matter volume decreases and diffusivity increases in the brainstem of migraineurs.
These anatomical changes occurred in parts of the trigeminal and endogenous analgesia brainstem systems.
These changes may underlie increased sensitivity to noxious inputs or the initiation of migraine attacks.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants awarded by the National Health and Medical Research Council of Australia.
