Abstract
Background
Calcitonin gene-related peptide (CGRP) receptor antagonism is an approach to migraine therapy. The locus of action of antimigraine treatment is not resolved. The objective was to investigate CGRP receptors in the ventrolateral periaqueductal gray (vlPAG) involved in the modulation of trigeminovascular nociception by descending influences on neurotransmission.
Methods
The presence of calcitonin receptor-like receptor (CLR) and receptor activity modifying protein 1 (RAMP1), which form functional CGRP receptors, was investigated. CGRP and its receptor antagonists, olcegepant and CGRP (8–37), were microinjected into the vlPAG while changes of neural responses in the trigeminocervical complex (TCC) were monitored.
Results
Immunoreactivity indicated the presence of functional CGRP receptor components in the vlPAG and adjacent mesencephalic trigeminal nucleus. Inhibition of TCC responses to stimulation of dural afferents and ophthalmic cutaneous receptive fields after microinjection of bicuculline into vlPAG indicated a connection between the vlPAG and TCC neurons. CGRP facilitated these TCC responses, whereas olcegepant and CGRP (8–37) decreased them.
Conclusions
CGRP and its receptor antagonists act on neurons in the region of vlPAG to influence nociceptive transmission in the TCC. This suggests CGRP receptor antagonists may act at loci outside of the TCC and reinforces the concept of migraine as a disorder of the brain.
Introduction
Calcitonin gene-related peptide (CGRP) plays a role in a variety of biologic functions including nociception. It has been widely implicated in the pathophysiology of migraine and other primary headaches (1–3). Clinical trials have shown that the CGRP receptor antagonists olcegepant (4), telcagepant (5), MK-3207 (6) and BI 44370 TA (7) are effective in the acute treatment of migraine. To understand better the mechanism of CGRP receptor antagonist actions in alleviating primary headache disorders, it is important to identify their sites of action within cranial nociceptive systems (2). Because of the widespread distribution of CGRP and CGRP receptors in the brain, it appears possible that CGRP-receptor antagonists have an action at supramedullary structures. We have seen effects of CGRP receptor modulation on second- (8) and third-order (9) nociceptive trigeminovascular neurons, so we have considered the hypothesis that CGRP receptor activation may be outside the direct trigeminovascular system, as for triptans (10).
Calcitonin receptor-like receptor (CLR) and receptor activity-modifying protein 1 (RAMP1), which determines the binding of CGRP receptor antagonists (11), form a complex that can couple to receptor component protein (RCP) resulting in receptor activation and signal transduction after CGRP ligand binding (12). RCP and RAMP1 messenger RNAs (mRNAs) have been found in the periaqueductal gray (PAG), predicting this as a site of functional CGRP receptor expression (13).
Immunohistochemistry better determines the localization of translation products and abundant RCP-immunoreactive cells have been observed in the PAG, especially in its ventrolateral region, suggesting the presence of functional CGRP receptors there. Moreover, brain imaging studies have also implicated the PAG in the pathophysiology of migraine (14), making the pharmacology of that region more interesting in a clinical context.
Because of the critical importance of CGRP in pain modulation, and the prevalence of CGRP and its potential receptor function in the caudal ventrolateral periaqueductal gray (vlPAG), we sought to elucidate whether CGRP and its receptor antagonists have a site of action in this region in relationship to intracranial nociceptive signaling. We extended the localization of the functional CGRP-receptor components RAMP1 and CLR to the PAG by immunohistochemistry and characterized the effect of CGRP-receptor activation and blockade by microinjecting CGRP and its receptor antagonists in this region. We observed modulation of neural activity recorded from second-order neurons in the trigeminocervical complex (TCC) that receive nociceptive input from dural and craniovascular afferents, and convergent input from cutaneous receptor fields of the ophthalmic division of the trigeminal nerve, adopting a systems-level in vivo model (15).
Materials and methods
General
Male Sprague Dawley rats (Charles River Laboratories, Hollister, CA) maintained under standard conditions were used in experiments conducted under anesthesia with terminal euthanasia in accordance with protocols approved by the Institutional Animal Care and Use Committee of the University of California, San Francisco.
Immunohistochemistry
Rats (n = 3) weighing 350–400 g were anesthetized with sodium pentobarbital (80 mg/kg intraperitoneally (i.p.), Nembutal; Hospira, Lake Forest, IL) and euthanized by transcardial perfusion with heparinized saline followed by fixative containing 0.5% paraformaldehyde in phosphate-buffered saline (PBS) (pH 7.4). The brain and cervical spinal cord were resected and placed in the same fixative overnight, then cryoprotected with 30% (w/v) sucrose in PBS. Coronal sections (40 µm) were cut through the PAG (Paxinos and Watson, 2005) and caudal spinal cord on a cryostat. Alternate serial sections were collected into PBS, mounted onto slides and Nissl-stained with cresyl violet (Biological Stain Commission certified AcW-25 acetate, Sigma C5042) using conventional methods (16).
The other sections were double stained with primary antibodies specific for CLR and RAMP1 (17). Sections were incubated in a blocking solution (10% normal donkey serum and 0.2% Tween 20 in PBS), then overnight at 4℃ with goat anti-RAMP1 diluted 1:100 in PBS containing 2% normal donkey serum and 0.5% Triton X-100 (dilution buffer) and for two hours with Alexa Fluor 488 donkey anti-goat immunoglobulin G (IgG) conjugate (Invitrogen, Molecular Probes, Eugene, OR) diluted 1:800. The sections were subsequently incubated in blocking solution using 10% normal goat serum and then overnight at 4℃ with rabbit anti-CLR diluted 1:500 and for two hours with Alexa Fluor 568 goat anti-rabbit IgG conjugate (Invitrogen) diluted 1:800 before mounting on glass slides under no. 1 coverslips (Corning Life Sciences, Acton, MA) using antifade hard-set mounting medium containing 4′,6 diamidino-2-phenylindole (Vectorshield, Vector Laboratories, Burlingame, CA). At each step, sections were washed three times with PBS. Fluorescent staining was visualized at room temperature using a Zeiss Axioplan Universal microscope (Carl Zeiss Microscopy, Oberkochem, Germany) fitted with an HBO 50 mercury short-arc lamp fluorescence illuminator and power supply (Carl Zeiss MicroImaging, Göttingen, Germany) and MRc5 Axiocam digital camera (Carl Zeiss Vision, Hallbergmoos, Germany). Filter set 09 (excitation 450–490 nm, dichrotic beam splitter 510 nm, emission barrier 520 nm) was used for Alexa Fluor 488 conjugate (green) and filter set 00 (excitation 530–585 nm, dichrotic beam splitter 600 nm, emission barrier 615 nm) for Alexa Fluor 568 conjugate (red). Plan-Neofluar objectives (magnification/numerical aperture: ×40/0.65, ×5/0.15, ×2.5/0.075) and Pl × 10/25 eyepieces were used. Images were processed using an AxioVision LE software (version 4.6, Carl Zeiss MicroImaging, Jena, Germany) microscope fitted with a mercury arc lamp and an appropriate filter set (Zeiss Axioplan; Oberkochem, Germany). Images were processed using AxioVision LE software (version 4.6, Carl Zeiss MicroImaging, Jena, Germany) and imported into Adobe Illustrator CS3 (version 13.0.1, Adobe, San Jose, CA) for figure layout and to generate TIFFs.
Electrophysiology
Surgery
Rats (315 ± 26 g, range 278–372 g, n = 17) were anesthetized with 3% (v/v) isoflurane (Abbott Laboratories, North Chicago, IL) and then sodium pentobarbital during surgery (Nembutal, Hospira; 60–70 mg/kg i.p. after induction, then 10–20 mg/kg/h as required) followed by α-chloralose dissolved in 45% 2-hydroxypropyl-β-cyclodextrin (18), initially 60–70 mg/kg (intravenously (i.v.)) and then 10–20 mg/kg/h through a femoral venous catheter. Arterial blood pressure was monitored through a cannulated femoral artery (DTX Plus DT-XX; Becton Dickinson, Sandy, UT) and maintained within physiologic limits. During electrophysiologic recordings and test stimuli, the rats were immobilized using pancuronium bromide (Gensia Sicor, Irvine, CA), 1.1–1.6 mg/kg, then 0.8–1.8 mg/kg/h, and artificially ventilated. End-tidal CO2 was maintained between 3.5% and 4.5% and skin and mucous membrane color monitored to confirm adequate oxygenation. Core temperature was maintained at 37.5 ± 0.5℃ using a homeothermic blanket system (Harvard Apparatus). The rat was fixed in a stereotaxic instrument (Kopf, Tujunga, CA) and the incisor bar adjusted until the heights of lambda and Bregma were in a horizontal plane. This flat-skull position was achieved when the incisor bar was lowered 3.9 ± 0.5 mm as consistent with published data (19).
The middle meningeal artery, its branches, and adjacent dura mater (MMA) were exposed by performing a small craniotomy over the parietal cortex. The vlPAG was accessed through a burr hole in the skull adjacent to the Bregma suture and superior sagittal sinus allowing dorsal entry and thus avoiding tracts of relevance to the pathways studied (Figure 1(a) from Akerman et al. (20)). A C1 partial hemilaminectomy was performed to allow access to the TNC and its C1 transition zone in the TCC.
Microscopy results.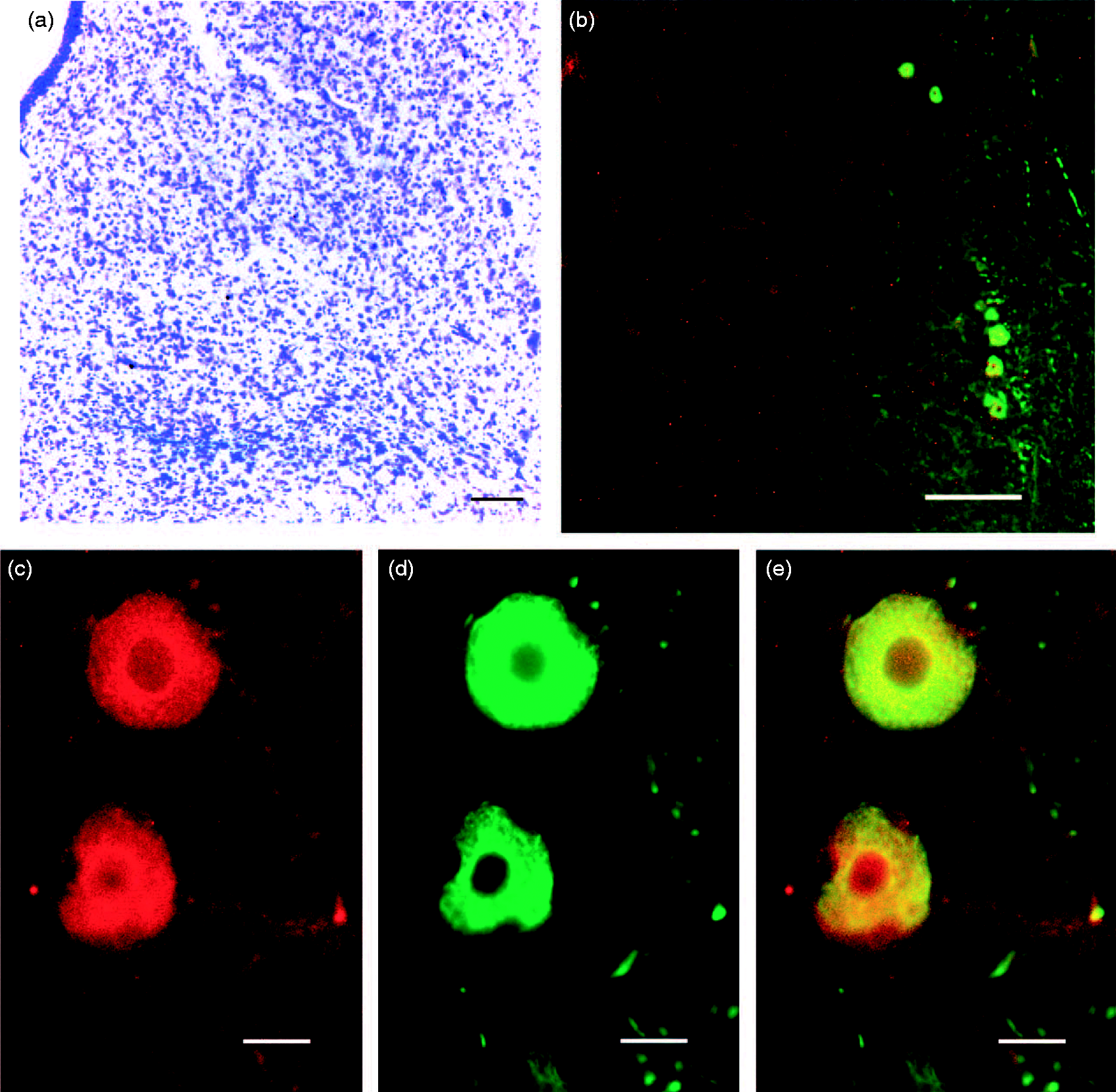
Stimulation and recording
We stimulated the MMA electrically and recorded multiple units in the TCC where we found convergence of input to nociceptive neurons from periorbital cutaneous receptive fields, thus providing a quantitative measurement of craniovascular nociception. The general procedure was similar to that described previously (10,15). In brief, a bipolar stimulating electrode was placed straddling the MMA, just touching the dura mater, and trains of stimuli were applied.
TCC neuron recording
Extracellular multiunit-recordings were made from neurons in C1 (about 3.2–3.8 mm caudal to the obex and 1.2–1.9 mm lateral to the midline) with tungsten microelectrodes (TM33A05; WPI, Sarasota, FL) positioned in 5 µm steps. Signals from recording electrodes were amplified, filtered (from 300 Hz to 10 kHz), fed to a noise subtraction system (Hum Bug; Quest Scientific, Vancouver, BC, Canada); and further amplified to total gain of around × 50–100 k. The signal was passed to a data acquisition system (Power 1401; CED, Cambridge, UK) and to a gated amplitude discriminator, the output of which was fed to the data acquisition system, and to an oscilloscope and audio amplifier to assist with spike discrimination. A storage oscilloscope triggered by the MMA stimulus further assisted with spike discrimination by displaying post-stimulus activity.
Characterization of neurons
Neurons in the TCC with convergent input from the MMA and cutaneous receptive fields on the face were identified in the region of C1. The receptive fields were mapped by applying innocuous (brush) and noxious (pinch) stimuli. We selected wide-dynamic range neurons (WDR) with dermatomes predominantly in the first division of the trigeminal nerve (V1) and with convergent input from the MMA. Cutaneous receptive fields in all three trigeminal innervation territories were assessed.
Substance preparation
(–)-Bicuculline methiodide (BIC), α-CGRP (rat), and CGRP-(8–37) (rat) were freshly obtained from Tocris (Ellisville, MO). We were particularly conscious of the lability of the Cys2–Cys7 disulfide bridge and, to avoid its oxidation, peptides were stored as solids below –20℃ in sealed vials before their reconstitution in saline (United States Pharmacopeial Convention (USP) grade) that had been purged of oxygen by nitrogen sparging. CGRP (316 µg/ml) and CGRP-(8–37) (2 mg/ml) pH < 7; BIC (0.4 mM, pH 5.0–5.5) and olcegepant (2.0 mM BIBN4096BS, pH 6.0–7.0; Boehringer Ingelheim, Biberach, Germany) were dissolved in saline. All drug solutions were freshly prepared and filtered to 0.02 µm. Chicago (Pontamine) Sky Blue (Color Index 24410; Alfa Aesar; Heysham, Lancashire, UK) (2% w/v) was dissolved in 100 mM sodium acetate, pH 6.0.
vlPAG microinjection
A seven-barreled glass capillary assembly pulled to long taper (PMP107; MicroData Instruments, South Plainfield, NJ) was used for the microinjection of drugs. The position of the multibarrel pipette assembly tip was aimed at coordinates for the vlPAG ipsilateral to the spinal recording site (16): 1.36 mm rostral and 4.08 ± 0.29 mm dorsal to the interaural line, and 0.6 ± 0.15 mm from the midline as used by others (10,15).
Experimental protocol
Inhibition of the neural responses to MMA and V1 receptive field stimulation after microinjections of the GABAA receptor antagonist BIC into the vlPAG was considered the most reliable evidence of a functional projection between neurons in the region of the PAG microinjection and the recorded trigeminal neurons (15) and was used as a search stimulus. The antinociceptive responses to the small amounts of BIC (20 pmol in 50 nl) microinjected into the vlPAG have a rapid onset and were transient and reversible, allowing subsequent testing with other substances (21).
The responses of WDR neurons in the TCC to electrical stimulation of the MMA were monitored before and after microinjecting CGRP, CGRP-(8–37), olcegepant, or saline vehicle control into the vlPAG. These test substances and controls were microinjected with a volume range of 150–250 nl in a random order over a period of 30 to 120 seconds as previously described (22). Responses to stimulation were analyzed using post-stimulus histograms (PSTH), which were determined and analyzed with the assistance of Spike 5 software (Cambridge Electronic Design, UK). Trains of 25 stimuli at 0.5 Hz (0.5 ms duration) were applied at intervals (1, 5, 10, 15, 20, 25, 30 minutes) after interventions, before which a stable baseline ( ± 5%) of at least three PSTH at five-minute intervals was established. Up to 90 minutes was allowed for activity to return to baseline between drug interventions. Effects on trigeminal activity were calculated as a percentage of the mean of three baseline responses before an intervention.
Recording and microinjection sites
At the end of the protocols, Chicago Sky Blue dye solution (250 nl) was microinjected into the vlPAG to estimate the spread of the test substances and mark the microinjection site. The recording sites in the TCC were marked with electrothermolytic lesions. The marked tissue was excised, fixed in neutral buffered 10% formalin, saturated with 30% sucrose (w/v) in PBS, and cryostat-cut serial sections (40 µm) mounted on glass slides. Images of the dye and lesion marks were visualized and processed using AxioVision LE software (Carl Zeiss Microscopy).
Statistical analyses
PSTH responses were normalized and expressed as a percentage of the mean change in regard to baseline response. Data were analyzed using statistical software (SSPS version 20.0; Chicago, IL) with an analysis of variance (ANOVA) for repeated measurements to evaluate changes over time with respect to the stimulation intervals. If Mauchly’s test of sphericity was violated we made appropriate corrections to degrees of freedom according to Greenhouse–Geisser (23). Where applicable, a Bonferroni post hoc procedure was applied for multiple comparisons. At the time of maximal effect, Student’s two-sample t test was used to evaluate statistical significance compared with baseline. Data are expressed as mean ± standard error of the mean (SEM) or mean ± standard deviation (SD) for weight and anatomic location observations; p < 0.05 was considered statistically significant.
Results
Immunohistochemistry
Within the PAG, we found CLR immunoreactivity to appear more intense in more medial locations, while the intensity of RAMP1 immunoreactivity increased ventrolaterally. Neuronal cell bodies in the vlPAG, particularly near its ventrolateral edge, showed colocalized CLR and RAMP1 immunoreactivity (Figure 1). Homogeneous staining was observed in the cytoplasm of positive cells with a spared nucleus. We noted large cells with particularly intense immunoreactive double labeling in the area of the mesencephalic trigeminal nucleus (rMTN). Colocalization of CLR and RAMP1 receptor components suggesting the presence of functional CGRP receptors was prevalent on larger neurons, but there were exceptions, with some cells expressing only RAMP1. A positive control of spinal cord neurons in the trigeminal nucleus at the level of C1 also showed double staining immunoreactivity for CLR and RAMP1, whereas a vlPAG negative control in which incubation with primary antibodies was omitted did not show staining (data not shown).
Electrophysiology
Basic characteristics
All neurons tested were classified as WDR and had cutaneous receptive fields, primarily in the first division of the trigeminal nerve, although some overlap with the second trigeminal division was sometimes observed. Where overlap occurred, the ophthalmic division was the most sensitive to stimulation.
Cells responded to stimulation of the MMA afferents with mostly short latencies consistent with conduction velocities of A-fiber afferents: 5–30 ms for the distance between the MMA and the TCC recording site. However, when higher stimulus intensities were used they sometimes elicited long latency responses (30–100 ms) consistent with C-fiber afferent activation. To prevent temporal summation or “wind-up” of neurons recorded in the TCC, we avoided excessive stimulation of cutaneous receptive fields and of the dural and perivascular afferents by restricting stimulation voltages (currents) to those just supramaximal for eliciting A-fiber responses: in the range 8–24 V depending on the inherent resistance across the bipolar stimulating electrode. Recording sites at C1 were at depths corresponding to Rexed laminae III and IV (Figure 2).
Receptive fields of recording sites in the trigeminocervical complex (TCC).
Pharmacology
Bicuculline
Microinjection of BIC into the vlPAG
A maximal inhibitory effect of –35 ± 7% (F6,30 = 10.4; post hoc: p = 0.009; n = 6) with respect to baseline was observed at five minutes of BIC microinjection (Figure 3). This activity increased to values that were not significantly different from baseline. A decrease in blood pressure was observed in some rats. This decrease of 10–20 mmHg over 60–100 s after microinjection of BIC is characteristic of activation of the vlPAG (15). The blood pressure decrease was considered indicative of micropipette tip placement in the vlPAG, whereas a blood pressure increase suggested a more dorsal tip location and a need to plunge the micropipette deeper (26).
Effects of CGRP and CGRP receptor antagonists in the PAG.
CGRP and CGRP receptor antagonists
Microinjection of CGRP into the vlPAG
CGRP-induced facilitation was shown by increased neuronal responses to electrical stimulation of the MMA afferents occurring within minutes of the microinjection and was observed for response latencies corresponding to Aδ fibers. A maximum increase of + 32 ± 5% above baseline (F6,30 = 2.9; post hoc: p = 0.020; n = 6) was observed at 10 minutes of CGRP injection.
Microinjection of olcegepant into the vlPAG
Olcegepant is a CGRP receptor antagonist. When microinjected into the vlPAG, olcegepant inhibited the excitability of the TCC neurons to the MMA afferent stimulation with a maximum response of –25 ± 6% (F6,54 = 10.4; post hoc: p < 0.001; n = 10). A maximum response was observed at one minute after olcegepant microinjection and was sustained at a plateau for 30 minutes.
Microinjection of CGRP-(8–37) into the vlPAG
CGRP-(8–37) is a CGRP receptor antagonist. When microinjected into the vlPAG, CGRP-(8–37) inhibited the excitability of the TCC neurons to the MMA afferent stimulation with a maximum response of –23 ± 12% (F6,30 = 3.5; post hoc: p = 0.092; n = 6). A maximum response was observed 15 minutes after CGRP-(8–37) injection, although the excitability of the TCC neurons was decreased from the first minute.
Control microinjections
Microinjection of saline into the vlPAG
Saline vehicle control microinjections did not have a significant effect on TCC activity over 30 minutes’ recording (maximally increased 3 ± 1%; F6,36 = 1.1; post hoc: p = 1.000; n = 7), indicating that the observed test substance effects were not the result of mechanical or vehicle effects.
Microinjection of BIC outside the vlPAG
For five WDR neurons BIC was injected in areas unrelated to descending inhibition outside the PAG and did not affect the elicited responses or spontaneous activity in second-order neurons (p > 0.05).
Discussion
We report on the coexistence of potentially functional CGRP receptor complex components, CLR and RAMP-1, using immunohistochemical methods, and facilitation of trigeminal nociceptive activity after microinjection of CGRP into the region of the caudal vlPAG. The effect is similar to what was seen after microinjection of the P/Q calcium channel blocker agatoxin (15) and implicates both CGRP and the PAG in migraine. The CGRP receptor antagonists, olcegepant and CGRP-(8–37) inhibited trigeminal nociceptive activity, as seen with the 5-HT1B/1D receptor agonist naratriptan in the same region (10). The data suggest both descending inhibitory and facilitatory influences from the PAG targets neurons of the TCC and involve CGRP-mediated components.
PAG influences on trigeminovascular nociception are regulated by presynaptic GABAergic and glutamatergic, serotonergic effects (10), orexinergic inputs (27), and cannabinoid receptors (20). Our new findings support a hypothesis for the involvement of the caudal vlPAG region in trigeminal pronociception after CGRP release in this area and a potentially novel site of action for CGRP receptor antagonists. An important issue that arises from clinical trials is the extent to which CGRP receptor antagonists effective in migraine enter the brain. Positron-emission tomography (PET) studies suggest such entry is modest, although not zero (28), although the studies have been small (28). The affinity of the clinical drugs and the doses employed suggests there is a complex question not yet completely understood (2).
Deletion of the CGRP gene prevents the development of hyperalgesia in mice (29), while in wild-type animals, levels of CGRP in the brain and cerebral spinal fluid (CSF), and blood have been reported to be raised in models of head pain (30). CGRP has been demonstrated to be pronociceptive in the dorsal horn of the spinal cord, where it facilitates glutamatergic neurotransmission (8), and when administered intracerebroventricularly (29). These pronociceptive effects can be blocked by CGRP-(8–37) (8,31–33) and olcegepant (8,31,33). In contrast, CGRP was apparently antinociceptive after other intracerebroventricular administration and in other parts of the brain (for example, see Bates et al. (34)), an effect that could also be antagonized by CGRP-(8–37). Some of this difference may simply be placement of injections within the PAG. As an example, in studies noted above, microinjections made in the rostral PAG, before the beginning of the ventrolateral column, were analgesic (35). Taken together the data suggest a complex role for supraspinal CGRP systems in the modulation of nociception.
Functional CGRP receptors, i.e. CLR and RAMP-1, seemed curiously located to the region of the rMTN. This region receives input from at least serotonergic and dopaminergic neurons in limbic structures and the dorsal raphe nucleus that also target the PAG (36). However, the functional significance of these inputs remains largely unknown (37). The rMTN may control trigeminal motor activity through serotonergic and dopaminergic mechanisms (36).
In summary, our data show CGRP and its receptor antagonists microinjected into the region of the caudal vlPAG modulate the activity of neurons in the TCC that receive meningeal, craniovascular and facial input. We found immunoreactivity for CLR and RAMP1 colocalized in the region of the caudal vlPAG, suggesting the presence of functional CGRP receptors here and notably on neurons in the adjacent region of the rMTN. Together, this suggests central descending mechanisms from these regions for the action for CGRP and its receptor antagonists in the control of head pain. A fuller understanding of the pronociceptive effects of CGRP and the regions in which it is active could have significant clinical implications for the refinement of antinociceptive therapies designed to treat migraine.
Article highlights
Calcitonin gene-related peptide (CGRP) mechanisms have been implicated in migraine, and their blockade has established antimigraine effects. Potentially functional CGRP receptors, calcitonin-like receptor (CLR) and receptor activity modifying protein 1 (RAMP1), are identified in the ventrolateral periaqueductal gray (vlPAG). Direct injection of CGRP receptor antagonists into the vlPAG inhibits nociceptive trigeminovascular activation.
Footnotes
Conflict of interest
PPR, RJS and ARC have nothing to declare.
Dr Goadsby reports grants and personal fees from Allergan, eNeura and Amgen, and personal fees from Autonomic Technologies Inc, Bristol-Myers Squibb, AlderBio, Pfizer, Zogenix, Nevrocorp, Impax, Dr Reddy, Zosano, Colucid, Eli Lilly, Medtronic, Avanir, Gore, Ethicon, Heptares, Nupathe, Ajinomoto and Teva, outside the submitted work.
Funding
This work was supported by the Sandler Foundation.
Acknowledgments
The authors are grateful for the generous gifts of antisera 844 and 3155 from Merck Research Laboratories, West Point, PA, and olcegepant from Boehringer Ingelheim, Germany. We thank Michele Lasalandra for technical assistance with the histology and Drs Knight, Holland, and Akerman for their helpful advice. Dr Pozo-Rosich received a travel grant from the Headache Study Group of the Spanish Neurological Society.
PPR, RJS and PJG were involved in the design, conduct, and analysis of the study, as well as manuscript preparation.
ARC was involved in the conduct and analysis of the study, as well as manuscript preparation.
