Abstract
Objective
To investigate the functional connectivity of the hypothalamus in chronic migraine compared to interictal episodic migraine in order to improve our understanding of migraine chronification.
Methods
Using task-free fMRI and ROI-to-ROI analysis, we compared anterior hypothalamus intrinsic connectivity with the spinal trigeminal nucleus in patients with chronic migraine (n = 25) to age- and sex-matched patients with episodic migraine in the interictal phase (n = 22). We also conducted a seed-to-voxel analysis with anterior hypothalamus as a seed.
Results
All patients with chronic migraine had medication overuse. We found a significant connectivity (T = 2.08, p = 0.024) between anterior hypothalamus and spinal trigeminal nucleus in the chronic group, whereas these two regions were not connected in the episodic group. The strength of connectivity was not correlated with pain intensity (rho: 0.09, p = 0.655). In the seed-to-voxel analysis, three regions were more connected with the anterior hypothalamus in the chronic group: The spinal trigeminal nuclei (MNI coordinate x = 2, y = −44, z = −62), the right dorsal anterior insula (MNI coordinate x = 10, y = 10, z = 18), and the right caudate (MNI coordinate x = 12, y = 28, z = 6). However, these correlations were no longer significant after whole brain FWE correction.
Conclusion
An increased functional connectivity between the anterior hypothalamus and the spinal trigeminal nucleus, as previously reported in preictal episodic migraine, was demonstrated in chronic migraine with medication overuse. This finding confirms a major role of the anterior hypothalamus in migraine and suggests that chronic migraineurs are locked in the preictal phase.
Introduction
Episodic migraine manifests as attacks of headache with distinctive associated symptoms (1), followed by long asymptomatic interictal periods. In some migraineurs, episodic migraine is progressively transformed into a chronic form, with headache on at least 15 days per month, affecting 1–2% of the population (2).
Neuroimaging studies using PET or fMRI have uncovered the brain areas that are activated in migraine attack. Early studies showed activation of the dorsal pons and hypothalamus in the pain phase (3–8). Recent studies showed activation of the hypothalamus within the preictal phase of both triggered (9) and spontaneous attacks (10). Moreover, a study using fMRI in one subject scanned every day over one month and three spontaneous migraine attacks revealed increased functional connectivity of the hypothalamus with the spinal trigeminal nuclei in the preictal phase and with the dorsal pons in the pain phase, suggesting that the hypothalamus was involved in both phases of migraine attack (10).
The role of the hypothalamus in chronic migraine is less well known. In a recent study using fMRI and nociceptive trigeminal stimulation, an increased anterior hypothalamic activation was demonstrated in chronic migraine compared to episodic migraine and healthy controls. A more posterior region of the hypothalamus was bilaterally activated during the pain phase in migraineurs compared to the headache-free phase in migraineurs and controls. The authors concluded that the anterior hypothalamus, which in episodic migraine is involved in the initiation of attack, became more activated during the transition from episodic to chronic migraine, thus lowering the threshold for internal and external inputs to generate the next attack, which then involved more posterior parts of the hypothalamus (11).
The present study was conducted in two parts. In the first part we sought to determine whether an increased connectivity between the anterior hypothalamus and the spinal trigeminal nucleus could be demonstrated in chronic migraine compared to interictal episodic migraine. For this purpose, we used resting state fMRI and a ROI-to-ROI approach.
In the second part, we conducted an exploratory study using seed-to-voxel analysis and the anterior hypothalamus as a seed. Our objective was to identify the functional brain networks associated with migraine chronification.
Methods
Subjects
Twenty-five subjects who met International Headache Society criteria for chronic migraine (1) and 25 age- and sex-matched subjects with episodic migraine with or without aura criteria were included in the study. Episodic migraineurs had fewer than four attacks per month, had no history of medication overuse or chronic migraine, and were not taking a migraine preventive medication. Preventive treatment was accepted in chronic migraineurs if unchanged for 3 months. None of the subjects had chronic neurologic or psychiatric conditions and none had antidepressant, antiepileptic or neuroleptic treatment. Episodic migraine patients were studied in the interictal phase, with at least a 72-hour interval before or after an attack. A mild tension type headache ( < 5/10 on visual analog scale) was accepted in chronic migraine during the imaging session.
Anxiety and depression were evaluated using the Hospital Anxiety and Depression scale (maximum score = 21).
Patients with silent brain lesions on MRI were secondarily excluded.
Standard protocol approvals, registrations, and patient consents
Written informed consent was obtained from all patients before participation and the study procedures were approved by the local ethics committee (Comité de Protection des Personnes Sud Ouest Outre-Mer IV).
Image acquisition
lmages were acquired on a 3T scanner Philips Achieva using a 32-channel head coil. Participants were in a supine position with their eyes closed and were instructed not to fall asleep. For the task-free functional imaging we used a T2*-weighted echo planar imaging sequence. All the images were acquired parallel to the anterior commissure-posterior commissure line. For the resting-state fMRI scan, 160 images were acquired with a TR of 3000 ms. The scanning parameters were as follows: TE = 30; acquisition matrix =80mm × 80mm; slices = 45; flip angle = 90°; spatial resolution = 3 × 3 × 3 mm3. A high-resolution 3D MPRAGE structural image was also acquired for each subject. The scanning parameters were as follows: TR = 8.1 ms; TE = 3.7 ms; flip angle = 8°; resolution =1 × 1 × 1 mm3 (no gap); acquisition matrix 240 × 240, slices = 170.
Image preprocessing
Functional imaging datasets were preprocessed using SPM 12 (www.fil.ion.ucl.ac.uk). Functional images were spatially realigned in six directories, normalized to the Montreal Neurological Institute (MNI) T1 template space, and smoothed with 8 mm Gaussian kernel.
fMRI data analysis
ROI-to-ROI analysis
We performed a ROI-to-ROI analysis between the anterior hypothalamus and the spinal trigeminal nucleus. To define our ROIs we drew a spherical ROI around MNI coordinates obtained from previous publications (x = 0, y = 2, z = −6, 3 mm radius for the hypothalamus, Denuelle et al. (5) and x = 0, y = −40, z = −56, 6 mm radius for the trigeminal spinal nucleus, Youssef et al. (12)).
Functional connectivity was analyzed using a general linear model (GLM) as implemented in the first level model of Conn functional connectivity toolbox (11). In order to correct physiological noise (pulsation due to respiration and heartbeat) and noise induced by patient movement we included, as nuisance parameters in the GLM analysis, two regressors extracted from the cerebrospinal fluid and the white matter and the six movement regressors created during image realignment. All the time series were filtered in the range 0.008 to 0.09 Hz.
The average time course was extracted from each ROI and we calculated the connectivity value (Fischer Z scores). We then used previous values in a two-sample t-test.
Seed-to-voxels analysis
To explore the anterior hypothalamus functional connectivity, we conducted a seed-to-voxel analysis between the anterior hypothalamus as a seed and the voxels in the whole brain. An averaged time course was obtained from the seed. Correlation maps were generated for each subject by estimating the correlation coefficient between whole brain voxels and seed time series. The correlation coefficient map was then converted into a Fisher-Z map by Fisher's r-to-z transform to improve the normality. The connectivity maps were then introduced in a group analysis using the random effect as implemented in SPM12 with the General Linear Model. We compared the resting state functional connectivity between chronic patients and episodic patients using a two-sample t-test. The statistical map was thresholded at p < 0.001 and only clusters of more than 10 voxels were retained. Connectivity was assumed significant at p < 0.05 corrected for multiple comparisons using the family wise error rate (FWE).
Clinical data and connectivity strength correlation analysis
Anxiety and depression were evaluated using the Hospital Anxiety and Depression scale (maximum score = 21). Clinical and demographic data were analyzed using non parametric tests, the Mann-Whitney test or the chi-square test as appropriate, implemented by R version 3.2.5. The correlation between the strength of connectivity, expressed by the mean of normalized correlation coefficient in the cluster, and clinical data was studied using the Spearman's rank correlation coefficient.
Results
Subjects
Three patients from the episodic group were secondarily excluded: Two subjects because they had a migraine attack 72 hours after MRI and a third one because MRI showed silent demyelinating brain lesions.
Demographic and clinical characteristics of chronic and episodic migraineurs.
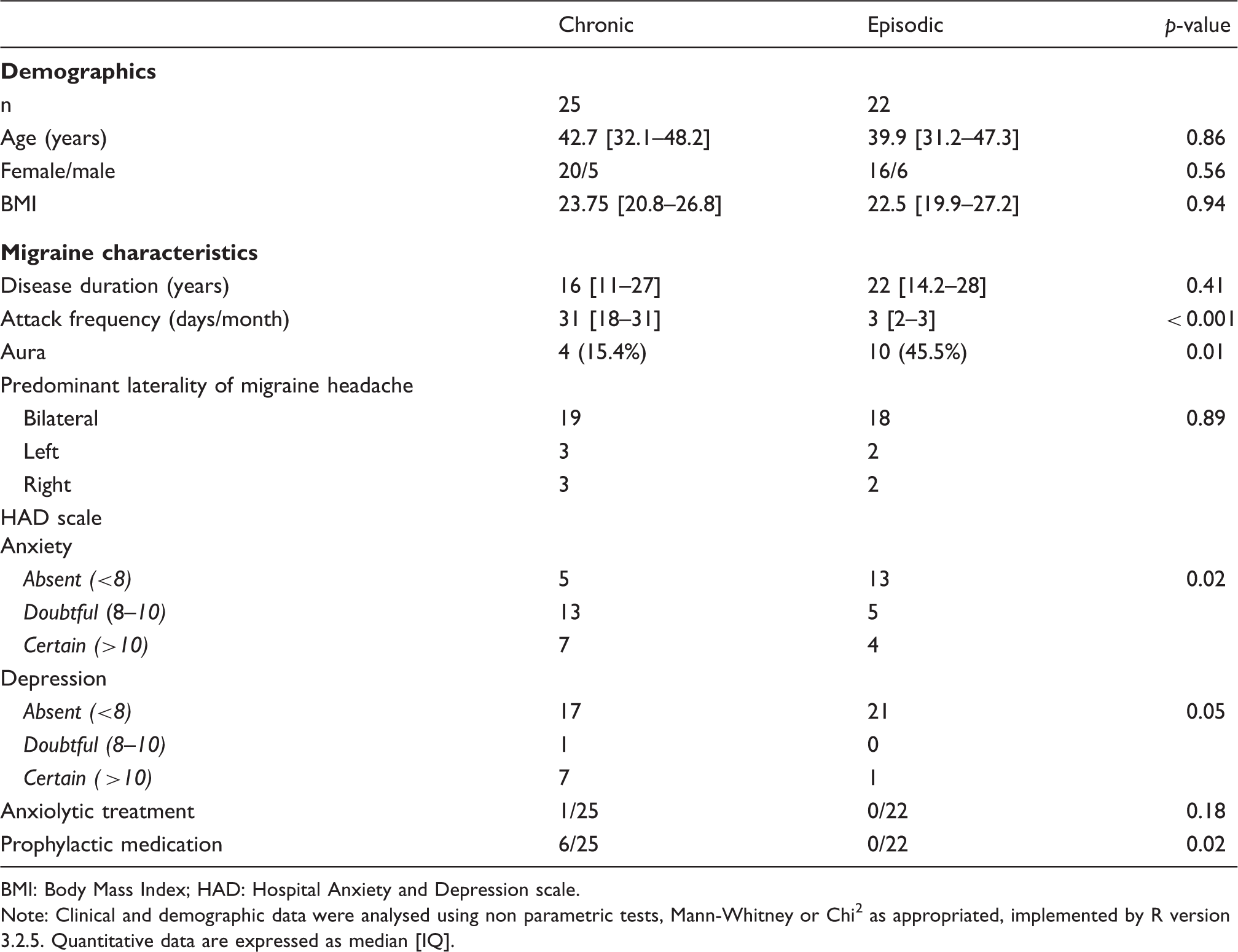
BMI: Body Mass Index; HAD: Hospital Anxiety and Depression scale.
Note: Clinical and demographic data were analysed using non parametric tests, Mann-Whitney or Chi2 as appropriated, implemented by R version 3.2.5. Quantitative data are expressed as median [IQ].
Functional connectivity analysis
Movement extent during imaging was evaluated in the two groups (mean (sd), 0.305 mm (0.155) for the chronic group and 0.187 mm (0.114) for the episodic group).
ROI-to-ROI analysis
We found a significant connectivity between the anterior hypothalamus and the spinal trigeminal nucleus in the chronic migraine group (p = 0.024), whereas these two regions were not connected in the episodic migraine group (p = 0.907). The difference between groups was significant (t = 2.31, p = 0.025) (Figure 1).
ROI-to-ROI analysis: Anterior hypothalamus and spinal trigeminal nucleus are functionally connected in chronic migraine but not in episodic migraine.
The strength of connectivity was similar in patients with migraine without aura and patients with aura (p = 0.209).
There was no significant correlation between connectivity strength with the spinal trigeminal nucleus and pain intensity during the imaging session in the chronic group (rho: 0.09, p = 0.655). We did not find any correlations between HAD score and fMRI connectivity (data not shown).
Seed-to-voxel analysis
Three regions were more connected with the anterior hypothalamus in the chronic group (Table 2): Spinal trigeminal nucleus (MNI coordinate x = 2, y = −44, z = −62; Figure 2), right dorsal anterior insula (MNI coordinate x = 10, y = 10, z = 18; Figure 3), and right caudate (MNI coordinate x = 12, y = 28, z = 6) (Table 3). However, these differences were no longer significant after FWE correction.
Seed-to-voxel analysis: Functional connectivity between anterior hypothalamus and spinal trigeminal nucleus in chronic and episodic migraineurs. (a) Voxels showing greater hypothalamus functional connectivity (p uncorrected < 0.001) were superimposed in a T1-weight template. MNI coordinates x = 2, y = −44, z = −62 are located at the blue lines' intersection. (b) Graphic represents averaged Z score's boxplot in the cluster. For chronic migraineurs, median was 0.096 [−0.048; 0.247]. For episodic migraineurs, median was −0.065 [−0.211; −0.021]. Seed-to-voxel analysis: functional connectivity between anterior hypothalamus and right insula in chronic and episodic migraineurs. (a) Voxels showing greater hypothalamus functional connectivity (p uncorrected < 0.001) were superimposed in a T1-weight template. MNI coordinates x = 30, y = 10, z = 18 are located at the blue lines' intersection. (b) Graphic represents averaged Z score's boxplot in the cluster. For chronic migraineurs, median was 0.075 [0.017; 0.137]. For episodic migraineurs, median was −0.073 [−0.118; 0.003]. Seed-to-voxel analysis: Higher connectivity in the chronic group than in the episodic group. Note: The statistical map was thresholded at p < 0.001 and only clusters of more than 10 voxels were retained. BA: Broadman area. Normalised correlation coefficient in chronic and episodic groups. Note: The normalized correlation coefficient was averaged in each cluster from each patient. Values are medians (interquartile range).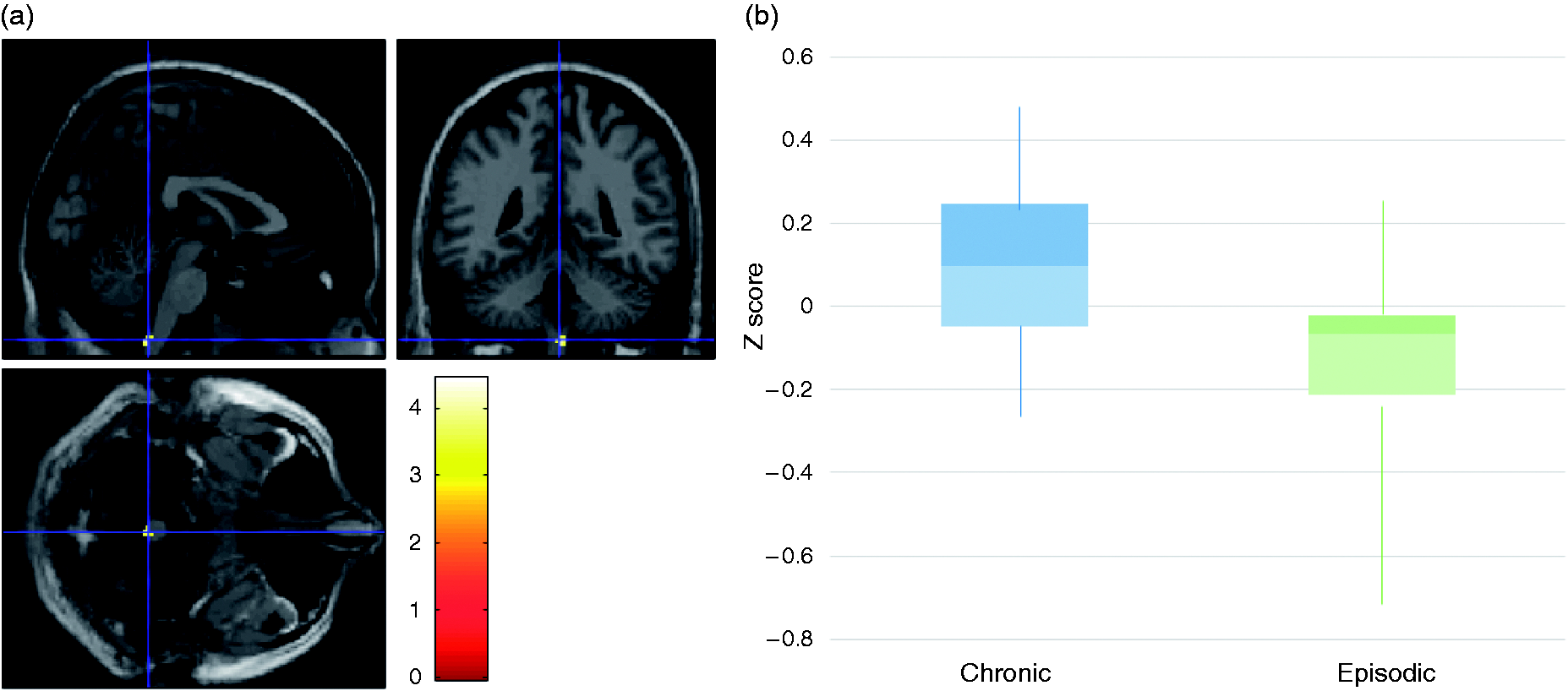



No region was more connected with the anterior hypothalamus in the episodic group.
Discussion
Our study was in two parts. Using a ROI-to-ROI analysis, we first sought to determine whether an increased functional connectivity between the anterior hypothalamus and the spinal trigeminal nucleus could be demonstrated in chronic migraine compared to episodic migraine in the interictal phase. In the second part, we used whole brain seed-to-voxel analysis to further explore the functional connectivity of the anterior hypothalamus in chronic migraine.
ROI-to-ROI analysis
In the ROI-to-ROI analysis, the anterior hypothalamus was functionally connected with the spinal trigeminal nucleus in the chronic migraine group and not in the episodic interictal group.
An increased functional connectivity of the hypothalamus with the spinal trigeminal nucleus has been previously reported in the prodromal phase of episodic migraine (10). We report for the first time a similar increased connection in chronic migraine with acute medication overuse. This finding is a strong argument for the role of the hypothalamus on the nociceptive trigeminal pathway in the chronification process.
All chronic migraineurs had acute medication overuse. In the 3rd edition of the ICHD (1), coexisting acute medication overuse is no longer an exclusion criterion for the diagnosis of chronic migraine and acute migraine medication overuse is acknowledged as one of the most important chronification factors (2,13).
Most patients with chronic migraine had a tension-type headache when scanned. However, the strength of the connection between the hypothalamus and the spinal trigeminal nucleus was not correlated with the pain intensity, suggesting that the anterior hypothalamus was not directly involved in the pain processing but rather in the preictal processing.
Previous studies have documented increased activation of the spinal trigeminal nucleus and of the hypothalamus in the preictal phase of episodic migraine. Stankewitz et al. reported that the trigeminal nuclei activation by trigeminal nociceptive stimulation in migraine patients depended on the time point at which patients were scanned during their individual migraine interval, with a higher activation in patients scanned in the 72 hours before the next attack than in those scanned in the interictal and ictal phase (7). The role of the hypothalamus in migraine attack generation was later suggested by the study of the same episodic migraineur scanned with fMRI every day over 30 days and three spontaneous migraine attacks (10). The activity of hypothalamus and trigeminal nucleus in response to trigeminal nociceptive stimulation were increased in the 24 hours before the next attack. Furthermore, an increased functional coupling between the hypothalamus and the spinal trigeminal nuclei was demonstrated in the preictal phase, whereas the hypothalamus was coupled to the dorsal pons in the ictal phase.
Our hypothalamic ROI, using the coordinates activated in the ictal and preictal phases of episodic migraine (5,10), was located in the anterior part of the hypothalamus. A recent study using fMRI and nociceptive trigeminal stimulation reported a more pronounced activation of the anterior hypothalamus in chronic migraineurs compared to episodic migraineurs and healthy controls, whereas a more posterior part of the hypothalamus was activated during headaches. The authors concluded that the anterior hypothalamus could play a role in attack generation and migraine chronification, while the posterior part was involved in the pathophysiology of headache (11). Our finding in chronic migraine showing an increased connectivity of the anterior hypothalamus with the spinal trigeminal nucleus as reported in the preictal phase of episodic migraine, and independent of pain intensity, further supports the role of the anterior hypothalamus in migraine initiation and chronification.
Seed-to-voxel analysis
In the seed-to-voxel analysis, the anterior hypothalamus showed heightened intrinsic connectivity in the chronic migraine group compared to the episodic group with the spinal trigeminal nucleus and two other unexpected areas: The right dorsal anterior insula and the right caudate nucleus. It should be noted that these correlations were no longer significant after FWE correction. These findings should thus be only considered as hypothesis generating pending confirmation by further studies.
Heightened hypothalamic connectivity with the right dorsal anterior insula
A confirmed connectivity of the anterior hypothalamus with the right dorsal anterior insula could offer an interesting and new perspective in migraine pathophysiology. The right dorsal anterior insula may simply be a nociception modulator (14), but a complex role in migraine is most likely. The dorsal anterior insula is an area of integration of sensory information from the posterior insula to emotional and cognitive valuation (15,16). A meta-analysis of functional neuroimaging activation studies using social-emotional, sensorimotor, olfacto-gustatory and cognitive tasks showed an overlap of all tested functions, except somatosensation and motion, on the dorsal anterior insula (17). Due to this highly integrative function, the right dorsal anterior insula is thus in a position to mediate sensory, autonomic, emotional and cognitive inputs that may contribute to the chronification of migraine.
Some recent studies also suggest a role of the right dorsal anterior insula in migraine. Migraineurs compared to healthy controls using fMRI had increased functional connectivity of the primary visual and auditory cortices to the right dorsal anterior insula, potentially constituting the neuroanatomical framework for phono- and photophobia in migraine (18). Migraineurs show lack of cortical thinning in the insula by age in contrast to healthy subjects (19), possibly reflecting hyperexcitability of this area in migraine. The dorsal insula moreover plays a role in the parasympathetic-sympathetic balance, whose disequilibrium could trigger migraine attacks (22). For all these reasons, and in spite of a lack of significance with our statistical methods, further studies on the connectivity between anterior hypothalamus and right dorsal anterior insula in migraine are warranted.
Heightened hypothalamic connectivity with the right caudate
Heightened hypothalamus connectivity with the caudate nucleus has already been observed in episodic migraine (20); however, its role remains to be determined.
The caudate nucleus seems to be involved in migraine attacks (21) and chronification (22) because this nucleus receives sensory input from the trigeminovascular neurons (7,23). Compared to low frequency migraineurs, high frequency migraineurs have differences in caudate functional connectivity and morphology (22).
Conclusion
In conclusion, chronic migraine differs from interictal episodic migraine by a heightened connectivity of the anterior hypothalamus with the spinal trigeminal nuclei complex similar to that previously shown in preictal episodic migraine. Our study confirms a major role of the anterior hypothalamus in migraine and suggests that chronic migraine patients are locked in a preictal state. This finding may offer a new perspective in chronic migraine pathophysiology and treatment.
Another interesting finding in our study, needing confirmation by specific studies, is a functional connectivity between the anterior hypothalamus and the right anterior dorsal insula in chronic migraine patients.
Footnotes
Clinical implications
A major role of the anterior hypothalamus in migraine is confirmed by its functional connectivity with the spinal trigeminal nucleus in chronic migraine with medication overuse but not in interictal episodic migraine. This connectivity is similar to that seen in the preictal phase of episodic migraine and could be a therapeutic target.
Another finding in chronic migraine with medication overuse is a possible connectivity of the hypothalamus with the right dorsal insula, deserving further studies.
Acknowledgements
The authors would like to acknowledge Dr V. Lauwers-Cances for her help in the methodology, all the TONIC INSERM team for their dedication, and JNLF association for its support.
