Abstract
Background
There is evidence of altered resting hypothalamic activity patterns and connectivity prior to a migraine, however it remains unknown if these changes are driven by changes in overall hypothalamic activity levels. If they are, it would corroborate the idea that changes in hypothalamic function result in alteration in brainstem pain processing sensitivity, which either triggers a migraine headache itself or allows an external trigger to initiate a migraine headache. We hypothesise that hypothalamic activity increases immediately prior to a migraine headache and this is accompanied by altered functional connectivity to pain processing sites in the brainstem.
Methods
In 34 migraineurs and 26 healthy controls, we collected a series comprising 108 pseudo-continuous arterial spin labelling images and 180 gradient-echo echo planar resting-state functional magnetic resonance volumes to measure resting regional cerebral blood flow and functional connectivity respectively. Images were pre-processed and analysed using custom SPM12 and Matlab software.
Results
Our results reflect that immediately prior to a migraine headache, resting regional cerebral blood flow decreases in the lateral hypothalamus. In addition, resting functional connectivity strength decreased between the lateral hypothalamus and important regions of the pain processing pathway, such as the midbrain periaqueductal gray, dorsal pons, rostral ventromedial medulla and cingulate cortex, only during this critical period before a migraine headache.
Conclusion
These data suggest altered hypothalamic function and connectivity in the period immediately prior to a migraine headache and supports the hypothesis that the hypothalamus is involved in migraine initiation.
Keywords
Introduction
There is growing evidence that the initiation of a migraine requires changes in the central nervous system. Given that many migraineurs report autonomic symptoms including nausea/vomiting and light-headedness (1) and endocrine abnormalities such as appetite and thirst disturbances (2) during the 24-hour period preceding a migraine headache, it has been suggested that migraine initiation involves multiple brain systems, likely centred around changes in hypothalamic function (3–6). Recently, it has been hypothesised that in migraineurs neural tone in brainstem function oscillates between “enhanced” and “diminished” states during which brainstem pain modulatory regions prevent or permit incoming noxious inputs to induce head pain, respectively (7). Alternatively, it might be that altered brainstem sensitivity can generate a migraine headache itself by altering the ascending levels of basal firing traffic.
Regardless of the mechanism, direct evidence supporting a critical role for the hypothalamus is relatively sparse. This sparsity of support is likely due to the challenge in exploring brain function immediately before a migraine, given that one does not know when a migraine headache will occur. Evidence from a single case study in which functional magnetic resonance images (fMRI) were collected each day for a month revealed that during noxious stimuli, the hypothalamus displays increased functional coupling with the dorsomedial pons (dmPons) and spinal trigeminal nucleus (SpV) immediately prior to a migraine headache (8). Furthermore, in a recent brain imaging investigation, we unexpectedly captured brain measures in migraineurs during the 24-hour period immediately prior to and in the 72-hour period following a migraine headache. This allowed us to explore brain function during multiple migraine phases and we found that in the 24 hours preceding a migraine headache, the hypothalamus and brainstem pain modulatory system displays altered ongoing activity patterns characterised by slow waves of increases and decreases in neural activity in the 0.03–0.06 Hz frequency range (9).
While these studies support the idea that hypothalamic function is involved in migraine initiation, it remains unknown if overall activity levels within the hypothalamus change immediately prior to a migraine headache. Furthermore, it is unknown if changes in hypothalamic activity are coupled to connectivity changes with other brainstem regions known to regulate incoming noxious orofacial information. If changes in hypothalamic activity occur, and these are associated with altered connectivity with pain-modulatory regions, it would provide strong evidence that the hypothalamus is involved in the initiation of migraines.
The aim of this study is to extend our earlier investigation by using arterial spin labelling and resting state fMRI to ascertain whether regional brain activity and hypothalamic connectivity are altered in the 24-hour period immediately prior to a migraine headache. We hypothesise that regional cerebral blood flow is increased in the hypothalamus immediately prior to a migraine headache and that this increase will be associated with an increase in the connectivity strength between the hypothalamus and brainstem areas that modulate incoming noxious inputs such as the midbrain periaqueductal gray matter, rostral ventromedial medulla, SpV and the dmPons.
Methods
Subject characteristics
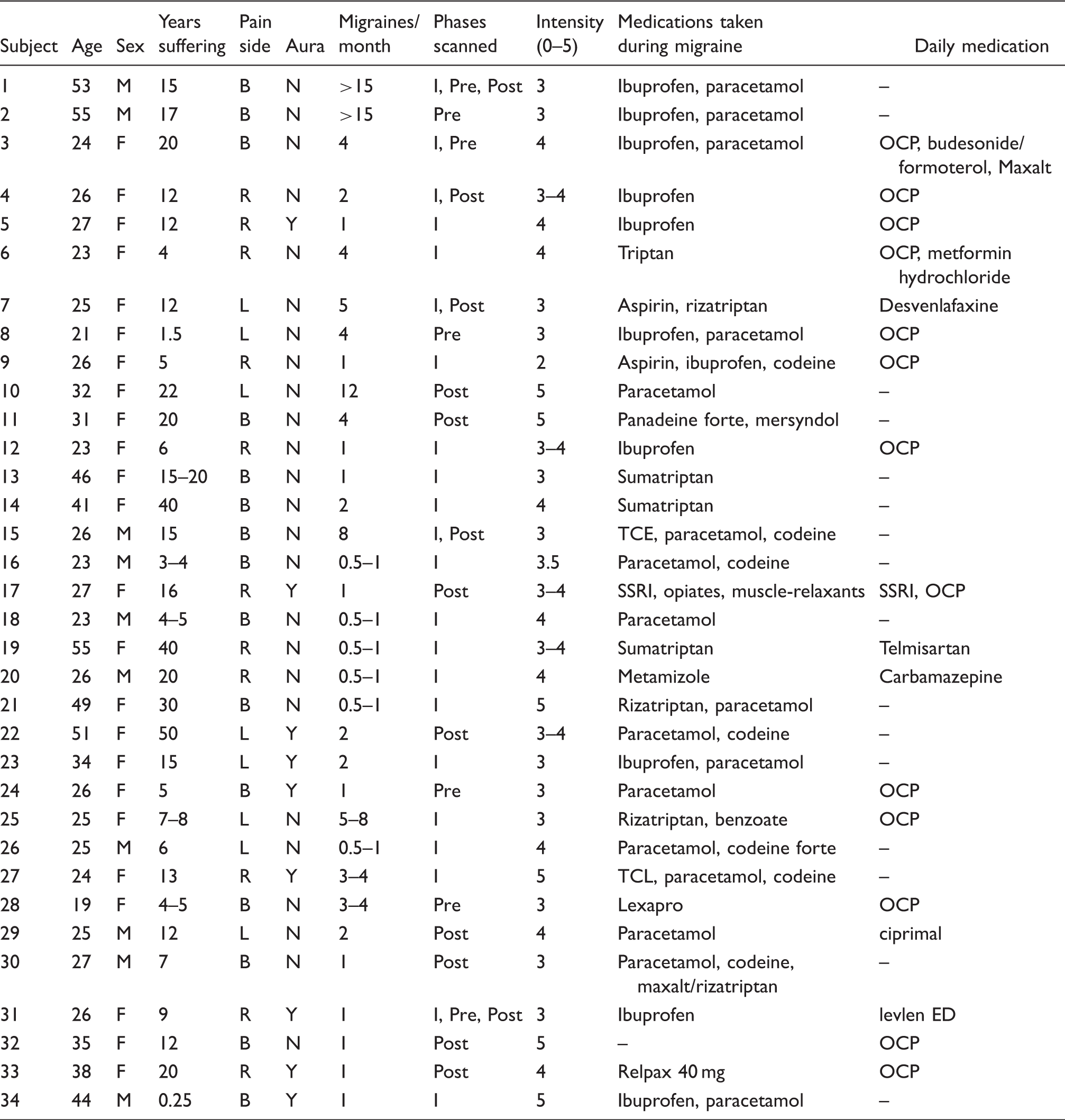
Migraine subject characteristics. M: male; F: female; B: bilateral; L: left; R: right; I: interictal; Pre: 24 hours preceding a migraine headache; Post: 72 hours following a migraine headache; OCP: oral contraceptive pill; SSRI: selective serotonin reuptake inhibitor.
Control subject characteristics. M: male; F: female; OCP: oral contraceptive pill.

MRI scanning
Each subject was laid horizontally into a 3 Tesla MRI scanner (Philips, Achieva) with their head fixed in a restrictive head coil. Subjects were instructed to relax, rest and keeps their minds as clear as possible. A high-resolution 3D T1-weighted anatomical image set, covering the entire brain area, was collected (voxel size = 0.9 × 0.9 × 0.9 mm; turbo field echo; field of view = 250 × 250 mm, matrix size = 288 × 288, slice thickness = 0.87 mm, repetition time = 5600 ms; echo time = 2.5 ms, flip angle = 8°). Next, a series comprising 180 gradient-echo echo planar resting-state functional magnetic resonance (fMRI) volumes, using blood oxygen level-dependent (BOLD) contrast, was collected. Each of these volumes included 35 axial slices that covered the whole brain (field of view = 240 × 240 mm, matrix size = 80 × 78, slice thickness = 4 mm, repetition time = 2000 ms; echo time = 30 ms/slice, flip angle = 90°). Finally, a series comprising of 108 pseudo-continuous arterial spin labelling images (50 axial slices, 54 label/control image pairs, [TE] = 12.7 ms, [TR] = 5310 ms, raw voxel size = 2.4 × 2.4 × 3.0 mm, labelling time = 1650 ms, slice time = 36.6 ms, post label delay time = 1600 ms, background suppression) was collected.
MRI data processing and statistical analysis
Image preprocessing
Using SPM12 (13) and custom Matlab software, all fMRI images in the resting state were realigned, motion corrected and assessed for excessive head movement. We detected no significant movement (greater than 0.5 mm in any direction) and all subjects were used for the subsequent analysis. The effect of movement on signal intensity was modelled and removed. To reduce any potential effects of physiological noise, we used the Dynamic Retrospective Filtering (DRIFTER) algorithm (14), which is a Bayesian method for physiological noise correction. Using the DRIFTER algorithm, a cardiac frequency band of 60–120 beats per minute (+1 harmonic) and a respiratory frequency band of 8–25 breaths per minute (+1 harmonic) were removed. The fMRI images were then linear detrended to remove global signal intensity changes and each subject’s fMRI image set was co-registered to their own T1-weighted anatomical image. The T1 image was then spatially normalized to the Montreal Neurological Institute (MNI) template and the parameters applied to the fMRI image set, resulting in both the T1-weighted and fMRI image sets being in the same location in three-dimensional space. Finally, the images were smoothed using a 6 mm full width at half-maximum (FWHM) Gaussian filter. Cerebrospinal fluid brain maps were created by segmenting the spatially normalised T1 anatomical images, for the purpose of removing any potential influence of signal intensity changes within the cerebrospinal fluid. This resulted in subsequent masking of the fMRI images, leaving only grey and white matter.
For each subject, pseudocontinuous arterial spin labelling images were realigned and co-registered to their own T1-weighted image set. The label and control images were averaged, and using the subtraction method in the ASL toolbox, a mean resting regional cerebral blood flow (CBF) image was created (15). Each subject’s T1-weighted anatomical image was then spatially normalized to the MNI template and the parameters applied to the CBF maps, so that both the T1-weighted and CBF images were in the same location in three-dimensional space. The wholebrain CBF maps were then smoothed using a 6 mm full width at half-maximum (FWHM) Gaussian filter. Using the T1-weighted anatomical image segmentation, a grey matter mask was derived for the purpose of restricting the wholebrain analysis to grey matter. For all significant clusters identified in the analyses of these data, two human brain atlases were used to define anatomical locations (16,17).
Cerebral blood flow; experimental design and statistical analysis
We performed three separate voxel-by-voxel analyses to determine if there were any significant differences in regional CBF between i) controls versus migraineurs during interictal, ii) controls versus migraineurs during the 24-hour period immediately prior to a migraine headache, and iii) controls versus migraineurs during the 72-hour period immediately after a migraine headache. Significant CBF differences between groups were calculated using a two-sample random effects analysis (p < 0.05 false discovery rate corrected, age and gender included as nuisance variables). Resultant significant clusters were overlaid onto an individual T1-weighted anatomical image and rendered onto a glass three-dimensional view of the wholebrain. For each of these clusters, CBF values were extracted from each subject in each of the four subject groups and the mean and standard error of mean (SEM) plotted. Significant differences between controls and each of the other two migraine groups were determined (p < 0.05, two-tailed, two-sample t-test, Bonferroni corrected for multiple comparisons). Differences in CBF values between the groups in the original voxel-by-voxel analysis from which the cluster was derived were not compared to avoid “double-dipping”.
In addition to exploring differences between controls and migraineurs during three phases, for each significant cluster derived from the controls versus migraineurs during the phase immediately prior to a migraine headache, we plotted CBF values against each individual’s time to their next migraine. That is, from the day of scanning, how many days until the next migraine to an upper limit of 30 days. Furthermore, for visualization purposes, mean CBF values for migraineurs with greater than 30 days prior, 30–12 days prior, 11–2 days prior and 1 day prior to a migraine headache were averaged and plotted. Finally, for each significant cluster we derived from the controls versus migraineurs during the phase immediately prior to a migraine headache, we plotted CBF values of three individual migraineurs who were scanned in multiple phases. Two of these individuals were scanned in all three migraine phases.
Functional connectivity; experimental design and statistical analysis
The CBF analysis revealed a cluster in the lateral hypothalamus that displayed significant decreases in blood flow immediately prior to a migraine headache. Using this cluster as a seed, we performed functional connectivity analyses to determine whether resting connectivity strengths between the lateral hypothalamus and all other brain regions altered throughout the migraine cycle. Alterations in signal intensity were extracted from the hypothalamic seed individually for each subject, and voxel-by-voxel analyses were performed to determine which brain areas displayed significant signal intensity covariations with this hypothalamic region. After smoothing the connectivity maps using a 6 mm FWHM Gaussian filter, the maps were placed into second level, random-effects analyses to identify significant differences in hypothalamic connectivity strength between controls and each of the migraine groups. To assess changes within small structures in the brainstem, the unsmoothed connectivity maps were masked using a brainstem mask and then smoothed using a 3 mm FWHM Gaussian filter. The resulting brainstem connectivity maps were then placed into second level, random-effects analyses to identify significant differences in hypothalamic-brainstem connectivity strengths between controls and each of the migraine groups. Given we hypothesized that the hypothalamus would display altered connectivity with the midbrain periaqueductal gray, dorsomedial pons, rostral ventromedial medulla and spinal trigeminal nucleus, we created 3 mm radius sphere regions of interests for each of these brainstem sites using the atlas by Paxinos and Huang (17) and our previous publication (9). Following an initial uncorrected threshold of p < 0.001, we applied small volume corrections using these regions of interest (p < 0.05) to reduce the likelihood of Type II errors.
Connectivity strength values were extracted from the resulting significant clusters and mean (± SEM) values were plotted to display connectivity direction. Additionally, connectivity strength values were extracted for all four groups and significant differences between groups were determined (p < 0.05, two-tailed, two-sample t-test, Bonferroni corrected for multiple comparisons). Significant differences between controls and the group from which the cluster was derived during the original voxel-by-voxel analysis were not determined, to avoid “double-dipping”. To explore changes throughout the migraine cycle, we plotted the mean (± SEM) connectivity strengths for the following periods: > 30 days until next migraine headache (n = 10), 30 to 10 days until next migraine headache (n = 7), 9 to 2 days until next migraine headache (n = 5), 1 day until next migraine headache (n = 7). Finally, for each significant cluster we derived from the controls versus migraineurs during the phase immediately prior to a migraine headache, we plotted connectivity strength values of three individual migraineurs who were scanned in multiple phases. Two of these individuals were scanned in all three migraine phases. Supplemental Figure 1 provides an experimental flowchart of the study design described.
Results
Cerebral blood flow
Contrary to our hypothesis, we found only decreases in resting regional CBF in migraineurs compared with controls during the phase immediately prior to a migraine headache (Figures 1(a) and (b), Table 3). Migraineurs displayed significantly reduced CBF in the region encompassing the right lateral hypothalamus, right retrosplenial cortex (RSC) and the left visual cortex. No CBF increases occurred immediately prior to a migraine headache and, furthermore, no significant CBF changes occurred in migraineurs during either the interictal or immediately following a migraine phase compared with controls. Extraction of CBF values from the lateral hypothalamus and visual cortex confirmed that CBF decreases occurred exclusively during the phase immediately prior to migraine headache only (right lateral hypothalamus: controls: 38.7 ± 1.9; interictal: 34.6 ± 2.2; immediately prior to migraine headache: 22.1 ± 2.1; immediately following migraine headache: 32.7 ± 2.4. Left visual cortex: controls: 92.5 ± 4.6; interictal: 83.0 ± 5.0; immediately prior to migraine headache: 49.8 ± 7.7; immediately following migraine headache: 65.0 ± 6.6) (Figure 1(c)). Plots of CBF values for each cluster in individual migraineurs relative to time in their migraine cycle reveal that in these two distinct regions, CBF remained consistent throughout the interictal period, decreasing suddenly immediately prior to a migraine headache. Furthermore, plots of CBF values of these clusters in three individual migraineurs reveals a consistent reduction in the phase immediately prior to a migraine headache compared to other phases (Figure 1(d)).
(a) Significant differences in regional cerebral blood flow (CBF) between controls (n = 26) and migraineurs during interictal (n = 22), the phase immediately prior to (n = 7) and the phase immediately following a migraine headache (n = 13) in a wholebrain voxel-by-voxel analysis. Note that the phase immediately prior to a migraine exclusively showed significant differences compared with controls. (b) Brain regions in which CBF was significantly decreased (cool colour scale) in migraineurs immediately prior to a migraine compared with controls overlaid onto a mean T1-weighted anatomical image set. Locations of each slice in Montreal Neurological Institute space are indicated at the top right. Note the significant blood flow reductions in the hypothalamus, retrosplenial cortex (RSC) and visual cortex. (c) Plots of mean (±SEM) blood flow in significant clusters extracted from controls and migraineurs during all three phases. There were only significant decreases in regional CBF compared to controls in these clusters in the phase immediately prior to a migraine (*p < 0.05 derived from voxel-by-voxel analysis). To the right are plots of blood flow in individual migraineurs (circles) against days until their next migraine in significant clusters. The white squares represent the mean blood flow values for the periods greater than 30 days prior, 30–12 days prior, 11–2 days prior and 1 day prior to a migraine headache. Note that blood flow in these regions gradually decreased throughout the interictal period as a migraine headache approached. (d) Resting regional cerebral blood flow values extracted from the lateral hypothalamus, RSC and visual cortex in three migraineurs in whom scans were acquired during at least the interictal and immediately prior to migraine headache phases. Migraine subject numbers correspond to those in Table 1. Montreal Neurological Institute (MNI) coordinates, cluster size and t-score for regions in which ongoing blood flow and hypothalamic connectivity were significantly different between controls and migraineurs.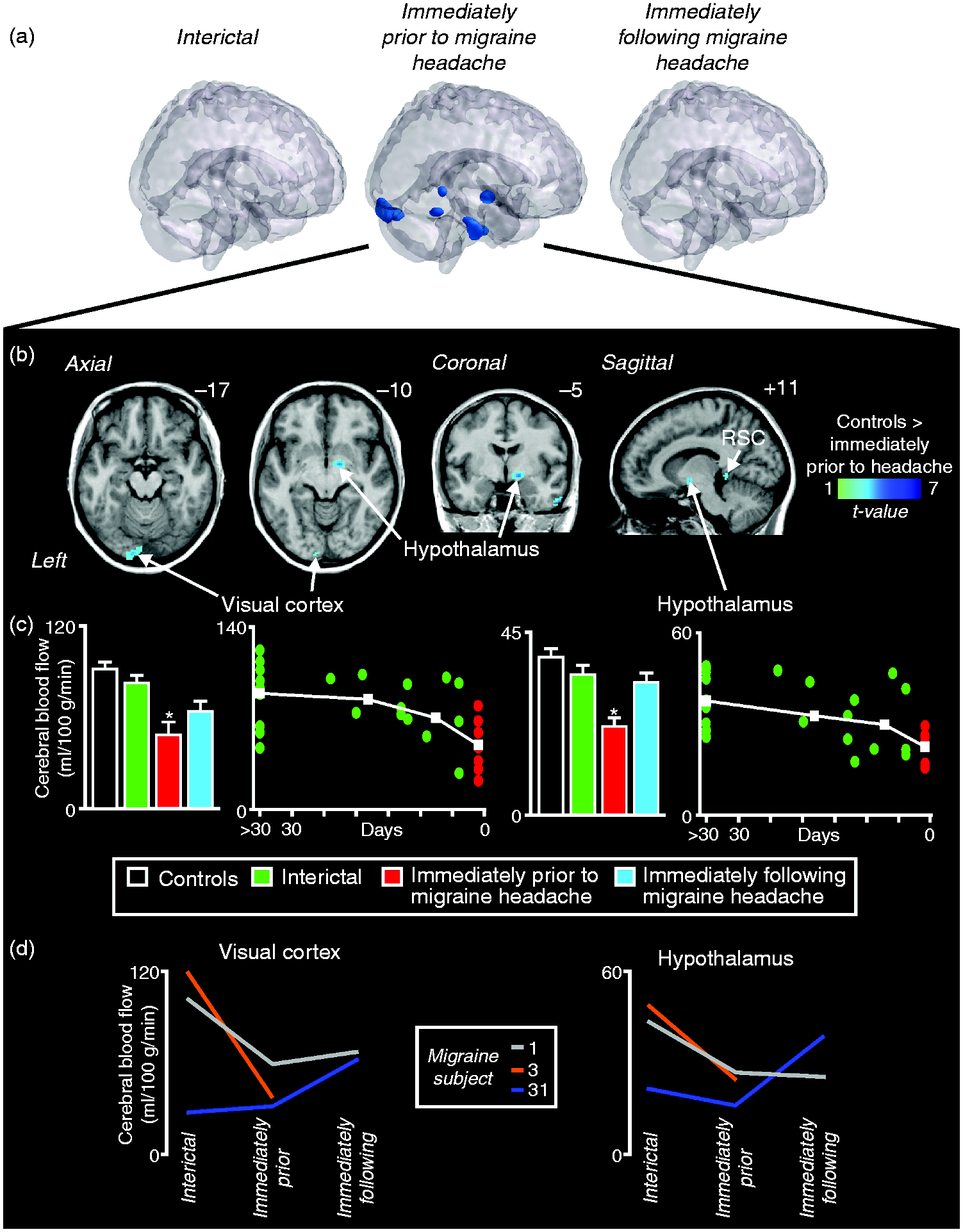

Hypothalamic functional connectivity
Similarly, we found only decreases in lateral hypothalamic connectivity in migraineurs compared with controls during the phase immediately prior to a migraine headache (Figures 2(a) and (b), Table 3). Migraineurs displayed significantly reduced lateral hypothalamic functional connectivity with only two higher brain regions, the right hippocampus and bilateral subgenual anterior cingulate cortex (subACC). Extraction of connectivity strength values from each of these clusters confirmed that decreases occurred exclusively during the phase immediately prior to migraine headache only (mean connectivity strength: hippocampus: controls: 0.10 ± 0.02; interictal: 0.07 ± 0.02; immediately prior to migraine headache: −0.08 ± 0.07; immediately following migraine headache: 0.07 ± 0.03. (a) Significant differences in hypothalamic resting connectivity between controls (n = 26) and migraineurs during interictal (n = 22), the phase immediately prior to (n = 7) and the phase immediately following a migraine headache (n = 13) in a wholebrain voxel-by-voxel analysis. Note that the phase immediately prior to a migraine exclusively showed significant differences compared with controls. Note brainstem changes are not displayed. (b) Significantly decreased connectivity (cool colour scale) in migraineurs immediately prior to a migraine compared with controls overlaid onto a mean T1-weighted anatomical image set. Locations of each slice in Montreal Neurological Institute space are indicated at the top right. Note the significant hypothalamic connectivity decreases with wholebrain regions including the subgenual anterior cingulate cortex and hippocampus. The inset to the left shows the hypothalamus seed used for the analysis. (c) Plots of mean (± SEM) hypothalamic connectivity signal in significant clusters extracted from controls and migraineurs during all three phases. There were only significant decreases in functional connectivity compared to controls in these clusters in the phase immediately prior to a migraine (*p < 0.05 derived from voxel-by-voxel analysis). To the right are plots of hypothalamic connectivity signal in individual migraineurs (circles) against days until their next migraine in significant clusters. The white squares represent the mean blood flow values for the periods greater than 30 days prior, 30–12 days prior, 11–2 days prior and 1 day prior to a migraine headache. (d) Resting connectivity values extracted from subgenual anterior cingulate cortex and hippocampus in three migraineurs in whom scans were acquired during at least the interictal and immediately prior to migraine headache phases. Migraine subject numbers correspond to those in Table 1.
While brainstem-specific analysis also revealed only decreases in connectivity, the scope of these significant changes was more extensive (Figures 3(a) and (b)). Migraineurs had significantly reduced lateral hypothalamic connectivity immediately prior to a migraine headache compared with controls in the midbrain periaqueductal gray matter (PAG), dorsomedial pons (dmPons), right spinal trigeminal nucleus (SpV) and rostral ventromedial medulla (RVM). Extraction of connectivity strength values from each of these clusters confirmed that decreases occurred exclusively during the phase immediately prior to migraine headache only (mean connectivity strength: PAG: controls: 0.18 ± 0.02; interictal: 0.18 ± 0.04; immediately prior to migraine headache: −0.01 ± 0.06; immediately following migraine headache: 0.13 ± 0.05. (a) Significant differences in hypothalamic resting connectivity between controls (n = 26) and migraineurs during interictal (n = 22), the phase immediately prior to (n = 7) and the phase immediately following a migraine headache (n = 13) in a brainstem voxel-by-voxel analysis. Note that the phase immediately prior to a migraine exclusively showed significant differences compared with controls. (b) Significantly decreased connectivity (cool colour scale) in migraineurs immediately prior to a migraine compared with controls overlaid onto a mean T1-weighted anatomical image set. Locations of each slice in Montreal Neurological Institute space are indicated at the top right. Note the significant hypothalamic connectivity decreases with brainstem regions including the midbrain periaqueductal gray (PAG), dorsomedial pons (dmPons), rostral ventromedial medulla, and the spinal trigeminal nucleus. The inset to the left shows the hypothalamus seed used for the analysis. (c) Plots of mean (± SEM) hypothalamic connectivity signal in significant clusters extracted from controls and migraineurs during all three phases. There were only significant decreases in functional connectivity compared to controls in these clusters in the phase immediately prior to a migraine (*p < 0.05 derived from voxel-by-voxel analysis). To the right are plots of hypothalamic connectivity signal in individual migraineurs (circles) against days until their next migraine in significant clusters. The white squares represent the mean blood flow values for the periods greater than 30 days prior, 30–12 days prior, 11–2 days prior and 1 day prior to a migraine headache. (d) Resting connectivity values extracted from rostral ventromedial medulla, spinal trigeminal nucleus, dorsomedial pons and periaqueductal gray in three migraineurs in whom scans were acquired during at least the interictal and immediately prior to migraine headache phases. Migraine subject numbers correspond to those in Table 1.
Discussion
Our data provides evidence that immediately prior to a migraine headache, regional ongoing activity decreases within the lateral hypothalamus. In addition, this activity decrease is associated with a reduction in resting lateral hypothalamus connectivity with brainstem structures known to modulate incoming orofacial noxious inputs. These results corroborate the idea that altered hypothalamic function plays a key role in migraine initiation, possibly through descending connections to brainstem sites that regulate orofacial pain processing pathways.
Whilst numerous investigations have explored resting regional cerebral blood flow (CBF) in migraineurs during the interictal phase, few have explored regional blood flow during the premonitory phase though prodromal symptoms have long been associated with a drop in CBF (18). In direct contrast to our results, Maniyar and colleagues recently reported significant increases in blood flow during the premonitory phase, triggered by intravenous administration of nitroglycerin, in the same hypothalamic region where we found blood flow decreases; that is, the right lateral posterior hypothalamus (19). While we used similar subject numbers to those of Maniyar and colleagues, the major difference is that our subjects were explored immediately prior to a spontaneously occurring migraine and not one that was triggered by nitroglycerin. The reduction in CBF was further corroborated by individuals who were scanned during multiple phases, revealing a consistent decrease in CBF in the period immediately preceding a migraine headache. Furthermore, the same posterior hypothalamic region displays greater activation during pain processing in ictal episodic and chronic migraineurs compared to controls, suggesting that changes in hypothalamic ongoing as well as noxious processing occur immediately prior to and during a migraine headache (6). Although triggering a migraine may change the underlying nature of overall activity, the posterior hypothalamus appears critical in the initiation of both naturally occurring and triggered migraines and continues to display altered sensitivity throughout the headache phase.
We also found significant decreases in connectivity between the lateral hypothalamus and the same brainstem sites where Schulte and May reported pain-related connectivity increases during the premonitory phase in an individual migraineur; that is, the spinal trigeminal nucleus (SpV) and dorsomedial pons (dmPons) (8). While the directions of these changes are different, our connectivity changes were determined with subjects at rest and not during noxious stimulation and thus underlie changes in resting brainstem function. Functional studies have also displayed increased connectivity between the hypothalamus and brain regions involved in regulation of autonomic functions during the interictal phase (20), as well as the hypothalamus and dorsal rostral pons during the ictal phase (21). Numerous studies have shown dmPons activation during a migraine headache (22–26) and, more recently, early activation of the dmPons during the premonitory period (19). Indeed, we have shown altered resting ongoing activity patterns in the dorsal pons, periaqueductal gray (PAG) and SpV during the premonitory phase (9). The PAG can modulate nociception by relaying in the rostral ventromedial medulla (RVM) activating “on” or “off” cells, before descending to send projections to the SpV (27–30), and there is growing evidence that the PAG is critically involved in migraine initiation (23). For example, electrical activation of the ventrolateral PAG (vlPAG) inhibits nociceptive activity in the SpV following electrical stimulation of the dura mater (31), migraine-like headaches can be evoked by direct electrical stimulation of the PAG (32) and migraineurs display altered anatomical changes in the PAG, RVM and SpV (10,33). It has been proposed that dysregulation of “on”-“off”-cell activity within the RVM is involved in the initiation of migraine (3) and that modulation of the PAG-RVM-SpV pathway can either initiate a migraine by enhancing basal ascending noxious activity or alternatively provide an environment by which a trigger can evoke a migraine headache (7). Interestingly, we recently found that although pain sensitivity decreased immediately prior to a migraine headache, SpV activation increased at similar pain intensities and this was coupled with reduced RVM-SpV functional connectivity (11). This suggests that SpV sensitivity does indeed increase immediately prior to a migraine headache and, irrespective of the underlying mechanism, our connectivity data supports the idea that the PAG-RVM-SpV pathway is involved in migraine initiation and that this pathway is likely under the influence of the lateral hypothalamus.
Tract tracing investigations have shown that the PAG, more specifically the vlPAG column, receives projections from lateral hypothalamic regions (30,34) and activation of the lateral hypothalamus can produce analgesia, likely mediated by the PAG (35). Although altered hypothalamic function may reflect general processing of noxious stimuli, given the connectivity and blood flow changes that occur before the individual experiences head pain, it is more likely the lateral hypothalamus influence over the PAG is involved in altering the sensitivity of orofacial pain processing circuits. It is possible that this lateral hypothalamus-PAG interaction involves orexins, which are peptides synthesized solely by neurons within the lateral hypothalamus (3,36,37). Hypothalamic orexinergic neurons project broadly to many brain regions including the PAG (38) and there is experimental animal evidence that orexins can facilitate or inhibit nociceptive processing and possibly provide a level of tonic drive over the ascending pain system (39). Interestingly, we have recently reported that immediately prior to a migraine headache, the pattern of resting activity within the hypothalamus, PAG and SpV alters, displaying significant increases in infra-slow oscillatory power (9). We previously proposed that these infra-slow oscillatory changes may result from increases in transient astrocyte synaptic modulation since calcium waves in activated astrocytes oscillate at similar infra-slow frequencies (40,41). It is possible that the combination of transient astrocyte activation in addition to altered decreasing modulation by the lateral hypothalamus, potentially through the release of orexins, results in altered orofacial pain circuitry firing that either generates head pain itself from altered basal firing or alternatively allows for an external trigger to generate head pain.
Whilst our results provide evidence of neural changes prior to the initiation of a migraine headache, there are several limitations that need to be considered. Firstly, the precise localization of clusters to specific brainstem nuclei is difficult. Indeed, while we are convinced that the changes we report encompass the areas described, they also extend to surrounding voxels that may be involving other functional nuclei. Secondly, as it is not possible to predict when an individual’s next migraine headache will occur, it is difficult to collect MRI scans during the 24-hour period immediately prior to a migraine headache. Essentially this occurs by chance and as a consequence the group numbers for this phase are relatively low compared with other migraine phases, and increasing the sample size of the group immediately prior to a migraine headache would add veracity to the current data and our interpretation. While we managed to explore only two migraineurs during all three phases and one during interictal and immediately prior to a migraine headache phase, these three migraineurs did display changes similar to those of the group results. Future studies in which individual migraineurs are followed over the course of a month are needed to verify the changes reported in this investigation.
Key findings
Resting blood flow decreases in the lateral hypothalamus in the phase immediately prior to a migraine headache. Resting functional connectivity strength decreases between the lateral hypothalamus and regions of the pain processing pathway including the midbrain periaqueductal gray, dorsal pons, rostral ventromedial medulla and cingulate cortex, in the phase immediately prior to a migraine headache. Hypothalamic function and connectivity is altered immediately preceding a migraine, supporting the role of the hypothalamus in migraine initiation.
Supplemental Material
CEP911623 Supplemental Material1 - Supplemental material for Altered regional cerebral blood flow and hypothalamic connectivity immediately prior to a migraine headache
Supplemental material, CEP911623 Supplemental Material1 for Altered regional cerebral blood flow and hypothalamic connectivity immediately prior to a migraine headache by Noemi Meylakh, Kasia K Marciszewski, Flavia Di Pietro, Vaughan G Macefield, Paul M Macey and Luke A Henderson in Cephalalgia
Supplemental Material
CEP911623 Supplemental Material2 - Supplemental material for Altered regional cerebral blood flow and hypothalamic connectivity immediately prior to a migraine headache
Supplemental material, CEP911623 Supplemental Material2 for Altered regional cerebral blood flow and hypothalamic connectivity immediately prior to a migraine headache by Noemi Meylakh, Kasia K Marciszewski, Flavia Di Pietro, Vaughan G Macefield, Paul M Macey and Luke A Henderson in Cephalalgia
Footnotes
Acknowledgements
We would like to thank the volunteers involved in this study.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants 1032072 and 1059182 awarded by the National Health and Medical Research Council of Australia.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
