Abstract
Objective
We performed a systematic review on the comorbidities of familial/sporadic hemiplegic migraine (F/SHM) with seizure/epilepsy in patients with
Methods
We performed a search in MEDLINE and in the Human Gene Mutation and Leiden Open Variation Databases for mutations in the
Results
Concerning F/SHM-Epilepsy1 (F/SHME1) and F/SHME2 endophenotypes, we observed a prevalent involvement of the transmembrane domains, and a strong correlation in F/SHME1 when the positively charged amino acids were involved. The penetrance of epilepsy within the families was highest for patients carrying mutation in the
Conclusion
Among the HM cases with seizure/epilepsy, we observed mutational hot spots in the transmembrane domains of CACNA1A and ATP1A2 proteins. These findings could lead to a better understanding of the pathological mechanisms underlying migraine and epilepsy, therein guaranteeing the most appropriate therapeutic approach.
Introduction
More than a century of investigation has found that seizures and migraine can co-occur in affected individuals and families, and there is much evidence that these diseases also share genetic susceptibility (1). In both epilepsy and migraine with aura (MwA), susceptible brain regions are hyperexcitable and attacks are believed to begin with hypersynchronous neuronal firing. The involvement of this mechanism in migraine without aura (MwoA) is less clear (1). Moreover, the mechanisms responsible for neuronal hypersynchronous activity are also not well understood. However, genetically determined dysfunction of ion transporters seems to indicate a common underlying mechanism for both paroxysmal disorders. In fact, over the last two decades, the mutations in the ion transportation genes
Materials and methods
Search strategy and study selection
We followed PRISMA 2009 guidelines for systematic review and meta-analysis (7). We sought to identify all published studies on the association between HM and seizure/epilepsy in patients or families carrying mutations in one of the HM genes. A search on MEDLINE (Ovid, New York, NY) from September 1993 to January 2016 was performed covering the time from the identification of the first FHM gene (4). No language restrictions were applied. The MEDLINE search strategy included both subject headings (MeSH terms) and text words for the target condition, including: FHM AND epilepsy; FHM AND seizure; HM AND epilepsy; HM and seizures;
Inclusion and exclusion criteria
Published studies and cases from the HGMD/LOVD were included if they reported an association between HM and seizure/epilepsy and their mutations were identified. We excluded review articles, editorials, commentaries, hypothesis papers, letters with no new data, as well as abstracts.
Data extraction
Two review authors independently extracted data; disagreements were resolved by discussion or referring to a third author. We collected data on genotype (DNA mutation), protein modification and patient information related to HM, headache, seizure, epilepsy and other neurological phenotypes, such as intellectual disability (ID), ataxia, and coma.
Data synthesis
For each gene, we selected the mutations associated with HM and seizure/epilepsy, so as to determine the relative frequencies of these mutations among all known mutations for that specific gene. Moreover, we calculated the penetrance (within a family, the percentage of individuals having a gene mutation and exhibiting epilepsy) of HM, seizure/epilepsy and other neurological phenotypes (ID, ataxia, coma). We also plotted the mutations in each protein structure regarding their locations in the transmembrane, extracellular or cytoplasmic domains (Figure 1). We performed a Chi-square test to investigate for associations between mutation location and phenotype.
The figure illustrates the distribution of all the mutations leading to HM in the three protein channels (CACNA1A, ATP1A2, SCN1A). The numbers inside the circles correspond to mutations, which are listed in a rising numerical order in the legend. Mutations associated with HM and epilepsy are shown as black circles, while those associated with HM are shown in white. The reference transcripts used for the 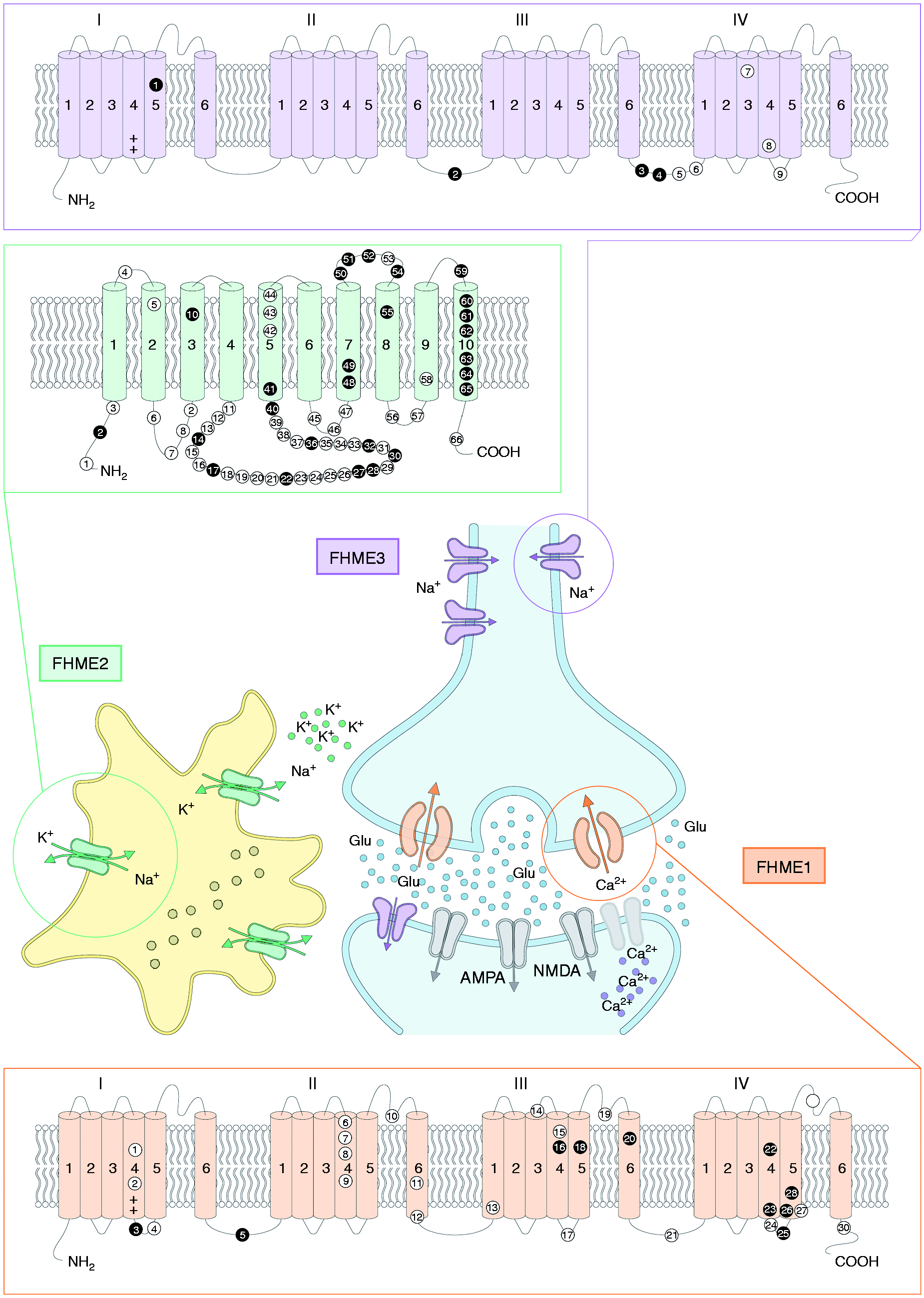
Results
List of the

Abbreviations: FHM, familial hemiplegic migraine; SHM, sporadic hemiplegic migraine; GEFS+, Generalized epilepsy with febrile seizures plus; ID, intellectual disability. “No data” refers to the absence of a detailed clinical description, but the patient had epilepsy.
List of the
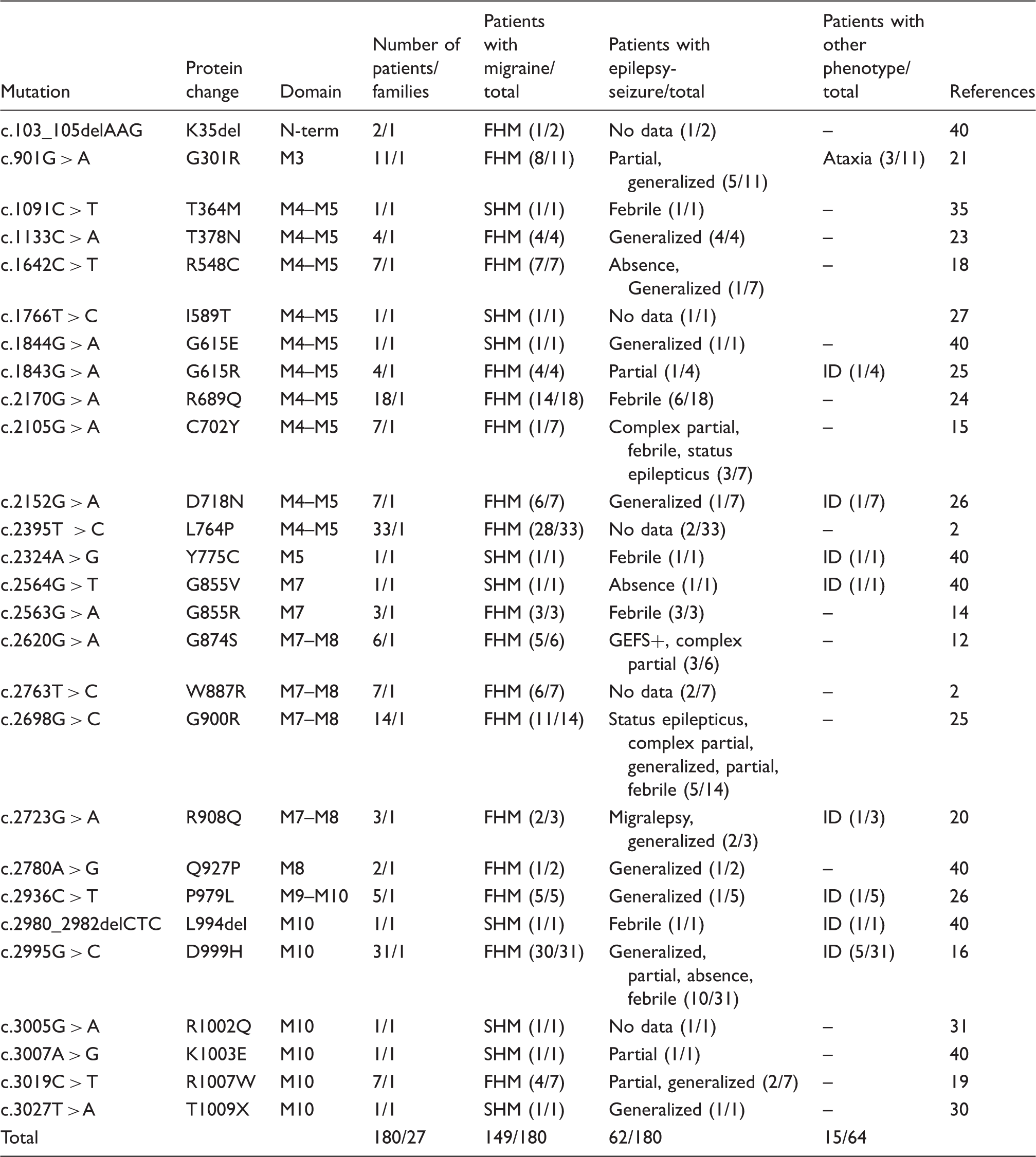
Abbreviations: FHM, familial hemiplegic migraine; SHM, sporadic hemiplegic migraine; GEFS+, Generalized epilepsy with febrile seizures plus; ID, intellectual disability. “No data” refers to the absence of a detailed clinical description, but the patient had epilepsy.
List of the

Abbreviations: FHM, familial hemiplegic migraine; SHM, sporadic hemiplegic migraine; GEFS+, generalized epilepsy with febrile seizures plus; ID, intellectual disability.
F/SHM1 and CACNA1A
Families and patients
Our search found 33 patients belonging to 23 families. In particular, 15 patients belonged to five families and 18 were sporadic cases. Of the former 15 patients, only nine had epileptic comorbidity, so the total number of patients having HM and epilepsy (here defined as F/SHME1, Familial/Sporadic Hemiplegic Migraine and Epilepsy type 1) was 27, nine familial (9/27, 33.3%) and 18 sporadic (18/27, 66.6%) (Table 1).
Mutation type and its distribution
We found 10 different
Mutation frequency and penetrance of seizures/epilepsy
The overall frequency of mutations associated with HM and seizure/epilepsy was 10/154 (6.5%), where 154 was the total number of mutations reported in the HGMD/LOVD for the
Clinical description
Summary of the results from this review. Abbreviations: GEFS+, generalized epilepsy febrile seizures plus; ID, intellectual disability; HM, hemiplegic migraine; FHME, familial hemiplegic migraine and epilepsy; SHME, sporadic hemiplegic migraine and epilepsy.

F/SHM2 and ATP1A2
Families and patients
We found 180 patients belonging to 27 families. In particular, 171 patients belonged to 18 families, while nine patients were sporadic cases. Of the 171 patients, 53 had epileptic comorbidity, so that the total number of patients having HM and epilepsy (here defined as F/SHME2, Familial/Sporadic Hemiplegic Migraine and Epilepsy type 2) was 62, 53 familial (53/62, 85.5%) and nine sporadic (9/62, 14.5%) (Table 2).
Mutation type and its distribution
We found 27 different
Mutation frequency and penetrance of seizures/epilepsy
The overall frequency of mutations associated with seizure/epilepsy was 27/73 (36.9%), where 73 was the total number of mutations reported in the HGMD/LOVD for the
Clinical description
The epileptic phenotype appeared to be mostly of a generalized type, and in eight families it was reported to be triggered by fever. Ataxia was present in three patients belonging to the same family and was associated with the G301R mutation, whereas ID was observed in both the sporadic (three patients) and familial cases (four patients belonging to four families), with an overall frequency of 11.3% (7/62 patients) (Table 4).
FHM 3 and SCN1A
Families and patients
We found 45 patients belonging to seven families (100% of familial cases). A total of 15 patients had the epileptic comorbidity endophenotype here defined as FHME3 (Familial/Sporadic Hemiplegic Migraine and Epilepsy type 3) (Table 3).
Mutation type and its distribution
We found four different
Mutation frequency and penetrance of seizures/epilepsy
The overall frequency of
Clinical description
L263V was more often associated with generalized epilepsy, while patients with T1174S or Q1489K mutations had focal seizures plus febrile seizures. Finally, the two patients with Q1489H mutation had partial epilepsy with occasional secondary generalization (childhood benign epilepsy), complicated by elicited repetitive daily blindness. There were no reported cases of ataxia or ataxia/ID in these families (Table 4).
Discussion
After having reviewed the cases of patients with HM and seizures/epilepsy carrying mutations in the
Patients with F/SHME1 were mostly sporadic cases (66.6%), while the penetrance of epilepsy in the few familial cases was very high (60%), thus suggesting a strong correlation between specific mutations and the onset of epileptic discharge. Specifically, with the exceptions of S218L, F363S and W1684R, the other seven
The association of
Patients with F/SHME2 were mostly familial cases (85.5%), where the penetrance of epilepsy was quite high (30.9%), suggesting a rather strong effect of some specific mutations in the generation of epileptic discharge. Moreover, regarding the genotype-phenotype correlation, our review evidenced that 73.3% of mutations affecting the transmembrane domains were associated with epilepsy, thus patients carrying such mutations could be much more susceptible to developing epilepsy compared to those having mutations outside the transmembrane domains, for whom this risk appears to be lower (31%). Missense mutations affecting the transmembrane domains can lead to both the perturbation of catalytic site functions and the modification of pump kinetic characteristics (1,2,15,19). Specifically, malfunctioning of the Na+, K+-ATPase pump may lead to neuronal hyperexcitability, which facilitates paroxysmal depolarizing shift (PDS) or CSD, causing seizures or migraine, respectively (1,2,19). This malfunctioning can occur due to three synergistic events: An elevation of extracellular K+ levels, an accumulation of glutamate in the synaptic cleft, or an increase in intracellular Ca2+ concentrations (15). Clinically speaking, epilepsy has been described mainly as generalized and in some cases triggered by fever. The severe cases, which were very rare among FHM2 patients, were instead more frequent (11.3%) among patients with F/SHME2 (Table 4).
Finally, regarding 852
Therapeutic implications
The efficacies of antiepileptic drugs (AEDs) in migraine and epilepsy have been reported to be mediated by interactions involved at multiple sites. In fact, these AED targets have more molecular sites in the brain, which are able to alter either voltage-operated ion channels or ligand-gated channels (53–55). Among the drugs widely used for epilepsy, topiramate is the most effective for migraine when compared with valproate and gabapentin, whereas lamotrigine has been reported to be effective for MwA in open studies, but has not been demonstrated to be effective for MwoA (56). The effectiveness of some antiepileptic drugs (topiramate, valproate and lamotrigine) and other migraine prophylactic drugs for CSD threshold have been evaluated only in different animal studies (57,58), so the therapeutic effect of these drugs in migraine patients is not clear. Other relevant targets for migraine pathophysiology could also be effective in migraine but, even in this case, evidence has derived only from animal models, and therein these findings cannot be extrapolated to humans (59–61).
Sodium valproate has been reported to reduce HM attacks, epileptic seizures and severe FHM attacks in patients suffering from both conditions (18), while evidence regarding the efficacy of topiramate is limited in HM (62) whereas lamotrigine has been reported to improve MwA in open-label studies, but not for MwoA in controlled trials vs a placebo or vs an active drug (63). This drug has a proven suppressive effect on CSD, which could explain its selective action on MwA (58). To date, the choice of preventive therapeutic strategies for HM has been based upon recommendations for common migraine, employing empirical strategies formulated on a limited number of FHM case reports. Conversely, AEDs including phenytoin, oxcarbazepine and vigabatrin block voltage-gated sodium channels and GABAergic transmissions, thereby reducing hyperexcitability mechanisms, but they are inefficacious in migraine (64). Despite these limitations with AEDs, the use of some anticonvulsivants, considered efficacious for both migraine and epilepsy, could be reasonable choices for treatment for HM and complex phenotypes, particularly HM and epilepsy. The evidence regarding the use of AEDs in literature is scarce. Therefore, clinicians should consider prescribing AEDs to HM patients having mutations associated with a marked susceptibility to developing epilepsy, instead of other anti-migraine drugs.
Conclusion
This review found that, in HM, mutations associated with seizure/epilepsy are not only frequent in S/FHM3 (44.4%) and S/FHM2 (40.9%) but also in S/FHM1 (33.3%), where in the latter the penetrance of epilepsy had the highest value (60%). Moreover, regarding the genotype-phenotype correlation, F/SHME1 and F/SHME2 had mutational hot spots at the protein levels. In fact, we observed prevalent and statistically significant involvement of the transmembrane domains for S/FHME1 (70%) and S/FHME2 (41%). In the former condition, the strongest genotype-phenotype correlation was observed when positively charged amino acids were involved in the S4 segments, while in the latter condition about 73% of mutations affecting the transmembrane domains were associated with seizure/epilepsy. These findings provide a better understanding of the pathological mechanisms underlying migraine and epilepsy and, if confirmed, these results would necessitate the recognition of an F/SHME subgroup.
Footnotes
Clinical implications
In hemiplegic migraine, the relative number of mutations associated with seizure/epilepsy was elevated not only for
Concerning F/SHM-Epilepsy1 (F/SHME1) and F/SHME2 endophenotypes, we observed a prevalent involvement of the transmembrane domains, and a strong genotype-phenotype correlation in F/SHME1 when the positively charged amino acids were involved.
The few
Our results suggest the presence of mutational hot spots at the protein levels rather than a genotype-phenotype correlation at least for FHME1 and FHME2.
Author contributions
Conception/design: PP, CC, CP, SP. Collection and/or assembly of data: CS, PP, BC. Data analysis and interpretation: PP, CS, CC, SP. Manuscript writing: PP, CC, CML, CP, SP; Figures and tables: CC, CS, PP, CP. Revisions of manuscript: PP, CC, BC, CML, CP, CS, and SP. All authors provided important intellectual content and critically revised the final version of the manuscript.
Acknowledgement
We thank Dr Saviz Yaghmai for his precious support in preparing the illustration.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: PC receives/received research support from Biogen, Lundbeck, Merck-Serono, Sanofi-Aventis and UCB Pharma. He also receives/received support from Ricerca Corrente and Ricerca Finalizzata IRCCS Fondazione Santa Lucia. All other authors report no disclosures. This study is not industry-sponsored.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
