Abstract
Aim
The aim of this study was to assess biochemical changes in the brain of patients with hemiplegic migraine in between attacks.
Methods
Eighteen patients with hemiplegic migraine (M:F, 7:11; age 38 ± 14 years) of whom eight had a known familial hemiplegic migraine (FHM) mutation (five in the
Results
Patients with hemiplegic migraine showed a significantly lower total N-acetylaspartate/total creatine ratio (tNAA/tCre) in the cerebellum (median 0.73, range 0.59–1.03) than healthy controls (median 0.79, range (0.67–0.95);
Discussion
We found a decreased cerebellar tNAA/tCre ratio that might serve as an early biomarker for neuronal dysfunction and/or loss. This is the first high-spectral resolution 7 tesla 1H-MRS study of interictal biochemical brain changes in hemiplegic migraine patients.
Introduction
Hemiplegic migraine is a rare subtype of migraine with aura, characterized by hemiparesis during the aura phase. There are two subtypes, familial hemiplegic migraine (FHM), which is autosomal dominant, and sporadic hemiplegic migraine (SHM) (1–3). Apart from the hemiparesis during the aura phase, the symptoms of the migraine attacks are similar to the common forms of migraine. Therefore, it serves as a valid model to study migraine pathogenesis (1,4).
So far, mutations in three different genes have been identified in FHM patients. FHM1 is caused by mutations in the
The pathophysiological mechanisms leading to migraine are complex and are only partially understood. Several brain structures are involved in different phases of a migraine attack. The phenomenology of premonitory symptoms suggests an early role for the hypothalamus in the onset of attacks (9). The cortex is involved in the beginning of a migraine attack because of cortical spreading depression (10). The brain stem is implicated through activation of the trigeminovascular system underlying the migraine headache (11).
The aim of this study, first, is to investigate biochemical changes in the cerebellum, hypothalamus, occipital cortex, and pons of hemiplegic migraine patients compared to healthy controls. Secondly, we aim to investigate whether there are differences between FHM1 and non-FHM1 patients. We used 7 tesla (7T) proton magnetic resonance spectroscopy (1H-MRS), which, in theory, leads to a higher spectral resolution resulting in a higher sensitivity and specificity to detect biochemical changes.
Methods
Subjects
Patients with hemiplegic migraine and healthy controls over 18 years of age were included. All patients had attacks that fulfilled the criteria for hemiplegic migraine according to the International Classification of Headache Disorders, third edition (ICHD-III) (3). Mutations in the FHM genes
Study design
Scans were performed in the morning (before 12:00 p.m.). Subjects were not allowed to eat or drink anything but water at least eight hours before the scan. Patients were scanned in an attack-free period of at least three days prior and three days after the study. All subjects underwent a detailed standardized interview and neurological examination (by FB) prior to the scan. Frequency of migraine attacks and the typical symptoms during an attack were recorded. Cerebellar symptoms were scored using the Scale for the Assessment and Rating of Ataxia (SARA), which includes eight items reflecting neurologic manifestations of cerebellar ataxia (12). SARA has eight items that yield a total score of 0 (no ataxia) to 40 (most severe ataxia). Gait (score 0 to 8), stance (score 0 to 6), sitting (score 0 to 4), speech disturbance (score 0 to 6), finger chase (score 0 to 4), nose-finger test (score 0 to 4), fast alternating hand movements (score 0 to 4) and heel-shin slide (score 0 to 4) were all separately rated. Cerebellar ataxia was defined as a SARA score of 1 or more.
Data acquisition
Subjects were scanned on an Achieva 7T whole body magnetic resonance (MR) system (Philips Healthcare, Best, the Netherlands), with a NOVA Medical quadrature transmit and 16-channel receive coil. For 1H-MRS, four separate fixed-sized volumes of interest (VOIs) were placed in the cerebellum (4.3 cc), hypothalamus (1.5 cc), occipital cortex (4.9 cc), and pons (2.1 cc; Figure 1). Planning was performed on axial T2-weighted turbo spin echo images (15 slices; repetition time (TR) 7000, echo time (TE) 100 ms; 0.5 × 0.6 × 2.5 mm3 resolution; 2.8 mm inter-slice gap) covering the brain from base of the cerebellum up to the corpus callosum. For 1H-MRS, single-voxel stimulated-echo acquisition mode (STEAM) was applied with TR/middle interval (TM)/TE = 2000/19/21 ms, bandwidth = 4 kHz, 2048 samples and second-order pencil beam volume shimming. For water suppression, a frequency-selective radio frequency (RF) pulse and gradient spoiling was used and six saturation bands, one on each side of the VOI, were applied to suppress signal from surrounding tissue. All scans included a reference scan without water suppression.
Representative localization of the regions of interest.
Data processing
MRS data were analyzed with LCModel (Provencher), using the unsuppressed water signal as an internal reference. For each anatomical region, an example of a 1H-spectrum is shown in Figure 2. A basis set simulated for all metabolites under investigation plus lipids and macro molecules was used. Since no data are available of T1 and T2 metabolite relaxation times at 7T, and the absolute concentration of the water in the regions selected is unknown, data are presented as ratio to total creatine (tCre) rather than concentrations. Metabolites under investigation were creatine plus phosphocreatine (tCre), choline (Cho), myo-inositol (Ins), combined N-acetylaspartate and N-acetylaspartylglutamate (tNAA) and combined glutamine and glutamate (Glx). Visual inspection of the spectra was performed retrospectively by two investigators (WMT and JvdG) who were blinded for the diagnosis. Spectra showing clear artifacts, for example due to subject movement or inadequate water suppression, were a priori excluded from further analysis. On some locations the spectral quality for some metabolites was limited for a reliable estimation of the metabolite concentration. Measurements with a Cramér-Rao lower bounds (CRLB, i.e. the estimated standard deviation expressed in percentage of the estimated concentration) smaller than 15% SD were considered reliable estimates of the metabolite concentration.
Example of 1H spectra acquired in the four regions of interest from a male healthy control.
For a complete overview of spectral quality per region and per metabolite, see Supplementary Table 1.
The T2-weighted images were evaluated by a neuroradiologist (MCK), who was blinded for the diagnoses, to identify any major structural anomalies.
Statistical analysis
Statistical analysis of LCModel output was performed with SPSS (SPSS 16 for Windows R16.0.2, SPSS Inc, Chicago, IL, USA). Data were checked on normality of distribution by means of a Kolmogorov-Smirnov test. Equality of variances was checked with the Levene's test as provided by SPSS and the corresponding
Results
Clinical and genetic characteristics of patients
Clinical and genetic characteristics of patients with hemiplegic migraine (
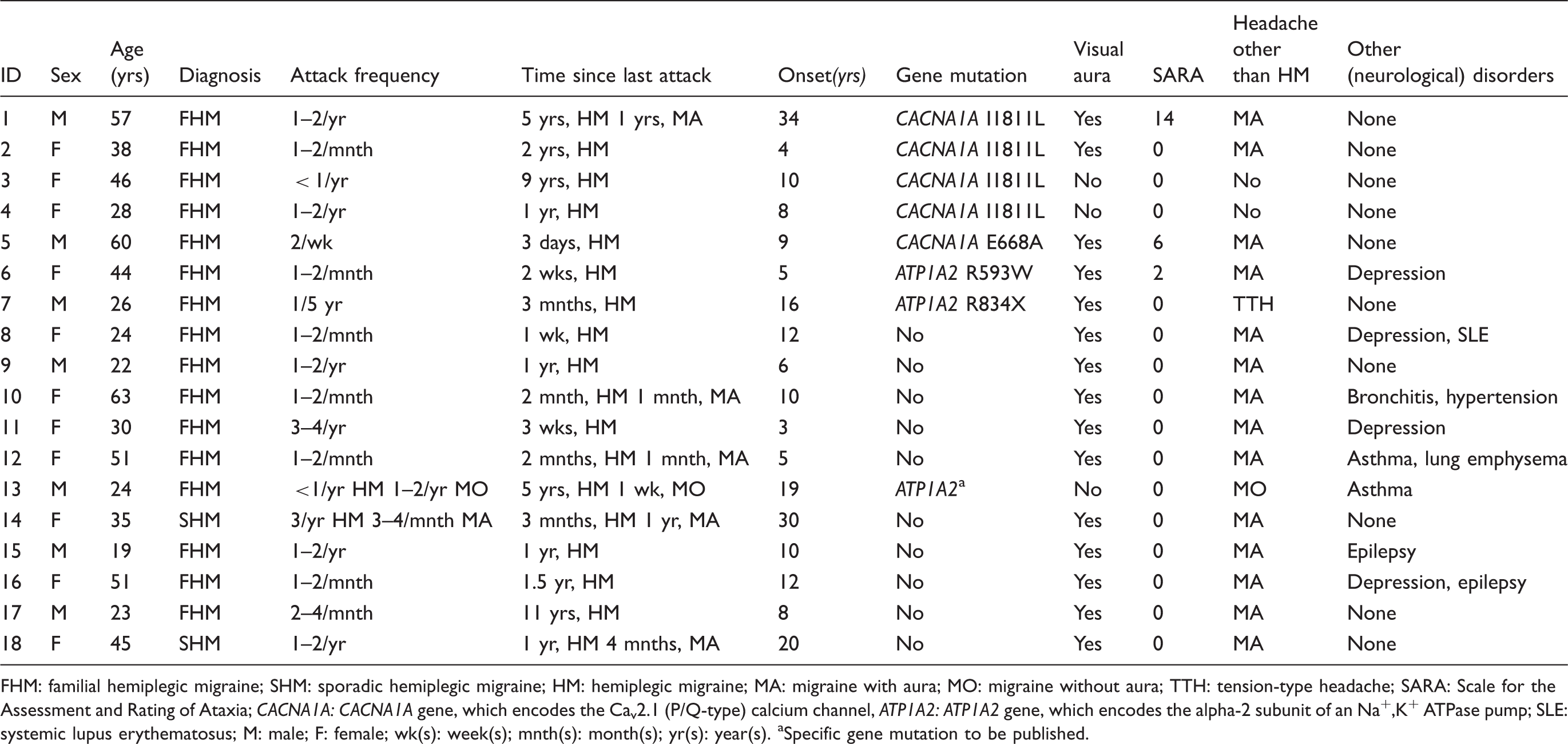
FHM: familial hemiplegic migraine; SHM: sporadic hemiplegic migraine; HM: hemiplegic migraine; MA: migraine with aura; MO: migraine without aura; TTH: tension-type headache; SARA: Scale for the Assessment and Rating of Ataxia;
Metabolite changes
Comparison of metabolite/total creatine ratios in different brain regions of hemiplegic migraine patients and healthy controls.

Metabolites under investigation were creatine plus phosphocreatine (tCre), choline (Cho), myo-inositol (Ins), combined N-acetylaspartate and N-acetylaspartylglutamate (tNAA) and combined glutamine and glutamate (Glx). Median and range of metabolite/creatine ratios and number of included spectra (N) in controls and hemiplegic migraine patients for anatomical regions of interest are shown. P-values refer to results of Mann-Whitney tests, significant results (p < 0.05) are in bold, not corrected for multiple testing.
Number of excluded spectra based on visual inspection (by WMT and JvdG), spectra showing clear artifacts, for example due to subject movement or inadequate water suppression.
Number of spectra for which LCModel was able to etimate a metabolite/total creatine ratio.

Comparison of cerebellar tNAA/total creatine ratios of
In the hypothalamus, the occipital lobe, and pons no significant differences between patients and controls were found for the tNAA/tCre ratio or for other metabolites.
The spectral quality was highest within the cerebellum and occipital lobe, and lower in the hypothalamus and pons. For a complete overview of spectral quality per region and per metabolite, see online Supplementary Table 1.
Discussion
We report the first high-field 7T brain 1H-MRS study that investigated biochemical changes in hemiplegic migraine. We found decreased cerebellar tNAA/tCre in hemiplegic migraine patients compared to controls. tNAA is considered a marker for neuronal integrity because it is synthesized and localized mainly in neuronal mitochondria (13). Our finding suggests cerebellar neuronal loss or dysfunction in hemiplegic migraine regardless of signs of cerebellar dysfunction (like ataxia) or macroscopic atrophy. Therefore, tNAA may serve as an early biomarker for cerebellar dysfunction and/or neuronal loss in hemiplegic migraine.
FHM1 is caused by a mutation of the
Interestingly, the
We studied three other regions of interest implicated in different stages of a migraine attack. The hypothalamus is thought to play an important role in the control of nociception, the initiation of attacks, premonitory symptoms, and the seasonal, menstrual, and circadian periodicity of migraine attacks (25,26). The hypothalamus has never been studied in hemiplegic migraine. Only Cho/tCre, Ins/tCre and tNAA/tCre could be reliably measured and showed no differences between patients and controls. Because of its small volume and the central anatomical location near the air-containing nasopharynx, it is difficult to achieve a high signal to noise ratio (SNR) in the hypothalamus, even at high magnetic fields.
We also examined the occipital cortex, the anatomical source of visual migraine aura. Several 1H-MRS studies in migraine also investigated the occipital cortex (27). One 1H-MRS study found decreased occipital NAA levels (which became even more pronounced upon visual stimulation) in migraine with aura patients but not in migraine without aura compared with controls (28). Two recent studies in migraine without aura patients before and after visual stimulation did not show any differences compared to controls (29,30). Increased occipital lactate levels have been reported in migraine with aura (31,32), but not in migraine without aura (29,30), suggesting that a possible deficit in energy metabolism is more marked in migraine with aura patients. The only other 1H-MRS study in FHM1 patients did not show biochemical changes in the occipital cortex (16). A multimodal imaging study on four FHM2 patients also did not find any significant biochemical differences when compared to controls (33). We also did not find any differences in the occipital cortex, although 83% of our hemiplegic migraine patients also experienced visual auras during their attacks. The discrepancy with the earlier finding in migraine patients with visual aura might be explained by the fact that we performed the measurements interictally in patients with low attack frequency.
No studies have previously investigated the pons, which harbors the trigeminal nucleus caudalis that plays a key role in the trigeminovascular system. Because of the small size of the VOI and the deep central location, only Cho/tCre and tNAA/tCre could be reliably measured and showed no differences between patients and controls.
This study was performed in a unique group of hemiplegic migraine patients. FHM can serve as a valuable monogenetic model for the common forms of migraine because, apart from the hemiplegia, the other symptoms during aura and headache phase are similar to the common forms, and patients often have non-hemiplegic attacks as well. The advantage of studying hemiplegic migraine patients is that they belong to a homogenous group with severe aura symptoms including hemiplegia. We included patients with and without a known gene mutation because we assumed that the pathophysiological mechanisms leading to hemiplegic migraine would be more or less similar.
A possible limitation of our study is the low attack frequency of the included patients: more than half had an attack frequency of less than one or two per year. Most likely this obscured dynamic biochemical changes. Ictal measurements during different stages of the attack will solve this problem but can be challenging. A technical limitation of our study is the lack of a volumetric anatomical scan, which prevented us from correcting for partial volume effects that might potentially confound our results when we would use water as our internal reference. Therefore we chose to use total creatine as the internal reference instead of water since concentration ratios are not affected by water scaling and are less sensitive to relaxation and partial volume effects. The metabolite/total creatine ratio is widely used and is considered a reliable indirect measure.
A strength of our study is that it is the first that used high-field 7T MRS to investigate the in vivo biochemistry in the brain of migraine patients. Theoretically, it is possible to measure metabolites more accurately in smaller VOIs. However, the data presented in this study were acquired some years ago, at that time the 7T MRI system was just operational and therefore we were not able to exploit all the theoretical advantages. Furthermore, the spectral quality was limited in the hypothalamus and pons but this seems inevitable for these deep cerebral structures with a small VOI, and location near the air-containing nasopharynx that causes field inhomogeneity (a common issue in high-field imaging). Because high-field MRS is a relative new technique, it is subject to rapid development and ongoing technical improvements and will allow us to distinguish, for example, glutamate (34). In conclusion, our study showed a decreased cerebellar tNAA/tCre ratio in hemiplegic migraine, which may serve as an early biomarker for cerebellar dysfunction and/or neuronal loss.
Clinical implications
We conducted the first high-field 7 tesla brain proton magnetic resonance spectroscopy (1H-MRS) study investigating biochemical changes in hemiplegic migraine. We found a decreased cerebellar N-acetylaspartate/total creatine (tNAA/tCre) ratio, which might serve as an early biomarker for neuronal loss and/or dysfunction in hemiplegic migraine. It is one of few studies performed in a unique group of hemiplegic migraine patients.
Footnotes
Funding
This work was supported by grants from the Netherlands Organisation for Scientific Research (NWO) (MDF: 903-52-291, Vici 918.56.602; GMT: 907-00-217, Vidi 917-11-319) and by a grant from the Centre for Medical Systems Biology (CMSB) established by the Netherlands Genomic Initiative/Netherlands Organisation for Scientific Research (NGI/NWO).
Conflicts of interest
R Zielman reports support for conference visits from Menarini and Allergan. MD Ferrari reports grants and consultancy or industry support from Medtronic, Menarini, and Merck and independent support from NWO, ZonMW, United States National Institutes of Health (NIH), European Community, and the Dutch Heart Foundation. GM Terwindt reports grants and consultancy/industry support from Merck and Menarini, and independent support from NWO.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
