Abstract
Background
Certain chronic diseases such as migraine result in episodic, debilitating attacks for which neither cause nor timing is well understood. Historically, possible triggers were identified through analysis of aggregated data from populations of patients. However, triggers common in populations may not be wholly responsible for an individual’s attacks. To explore this hypothesis we developed a method to identify individual ‘potential trigger’ profiles and analysed the degree of inter-individual variation.
Methods
We applied N = 1 statistical analysis to a 326-migraine-patient database from a study in which patients used paper-based diaries for 90 days to track 33 factors (potential triggers or premonitory symptoms) associated with their migraine attacks. For each patient, univariate associations between factors and migraine events were analysed using Cox proportional hazards models.
Results
We generated individual factor-attack association profiles for 87% of the patients. The average number of factors associated with attacks was four per patient: Factor profiles were highly individual and were unique in 85% of patients with at least one identified association.
Conclusion
Accurate identification of individual factor-attack profiles is a prerequisite for testing which are true triggers and for development of trigger avoidance or desensitisation strategies. Our methodology represents a necessary development toward this goal.
Keywords
Introduction
The episodic occurrence of debilitating attacks is one of the great unsolved problems of a class of chronic recurrent disease that includes migraine, irritable bowel syndrome, epilepsy and schizophrenia. In many of these diseases occurrence of attacks is largely unpredictable, resulting in significant suffering, interference with daily activities and decreased quality of life for the individual.
Taking migraine, one of the leading causes of disability worldwide (1), as an example, many factors have been suggested as attack ‘triggers’ with varying degrees of supportive evidence based on analysis of population data or individual anecdote (2–5). Furthermore, interactions between triggers appear important and the concept of additive (even synergistic) effects of triggers is a well-accepted hypothesis, although one lacking confirmatory evidence (6). However, whilst useful in identifying which factors may be the most common triggers in the population, this information is of little therapeutic use to individuals with migraine, as there appears to be considerable heterogeneity in trigger profiles among individuals. To date, identification of triggers in individuals has usually been based on retrospective self-report; that is, asking individuals what they think triggers their attacks (7–9). However, self-reporting of triggers has been shown not to be reliable (10). Few studies have identified triggers using a prospective diary approach, as was conducted by Lipton et al., who investigated stress as a trigger (11). The validity of the retrospective approach is limited because of the possibility of selective memory and recall bias, as well as the need for causal explanations for attacks (12). Therefore, an approach is needed that objectively identifies the important triggers at the individual level: this in turn could lead to modifying such triggers with the aim of improving the condition and its effects on lifestyle, health care and other costs (13–15).
The first step in decoding the interaction between an individual’s environment and his or her disease pathology is to track the set of factors that may be of importance in the occurrence of attacks in that individual. Tracking would ideally be (at minimum) daily and would include scoring exposure to environmental (e.g. weather, loud noise, bright light), dietary, physical, and emotional factors, as well as internal factors such as the menstrual cycle.
The second step would be to use N = 1 statistical methods on individual patient data, as opposed to aggregating patient data before the analysis (as is commonly performed). To date, only a single study, restricted to the association between migraine events and weather variables, has applied this individual patient approach (16). In contrast, aggregation of patient data is subject to an observation called Simpson’s paradox (17), which concerns the loss of individual signal after population data aggregation occurs, especially in disease populations with high individual diversity. As an extreme example of Simpson’s paradox, if 10 individuals are each sensitive to 10 different triggers, analysis of aggregated data will not yield anything clinically useful to any of the 10 individuals. Having individual trigger profiles correct in their entirety is critically important in migraine patients because of the belief that multiple triggers may sum, or act in combination, to precipitate attacks (6).
A third and critical step in decoding the interaction between an individual’s environment and individual disease pathology would be to test all triggers that are modifiable for causality in that individual. For example, if a patient is found to have an association between caffeine and migraine attacks, an N = 1 test would be invoked: change the dose or eliminate caffeine from the patient’s diet for a period of time. A consequent reduction in the rate of attack occurrence would confirm caffeine as a trigger.
To add to the complexity, factors associated with migraine attacks may be potential trigger factors, or they may be premonitory symptoms, or it may not be clear whether they are triggers or premonitory symptoms (15). A digital platform that tracks both possible trigger factors and premonitory symptoms, invokes N = 1 analytical procedures, informs individuals about their factor-attack associations, and which may facilitate testing of trigger-attack associations in individual patients has been developed (Curelator Headache™) (13). In developing this digital platform, a detailed 90-day paper-diary database from the PAMINA migraine study was used to develop and test N = 1 statistical methodology for identification of trigger profiles in individuals. The PAMINA study, conducted and published by one of us (CW) (3), was previously used to identify common factors associated with migraine attacks from a population analysis and is the most complete daily migraine trigger database of which we are aware. The factors included in that study were derived from the available literature, based primarily on the patients’ retrospective self-assessment, and represent those factors reported most frequently (2). Use of the PAMINA population-based analysis, and our own population-based multivariate analysis, allows an important comparison with the N = 1 analysis (with post-analysis aggregation) reported in this study.
The aim of the present study was therefore to identify potential trigger factor-attack associations for each individual and describe the inter-individual variation in trigger profiles in the study population. In this article we refer collectively to ‘potential triggers’, bearing in mind the overlap of triggers and premonitory symptoms and the difficulties in differentiating between them for some patients. For example, fatigue was reported to be third in the top-10 migraine triggers (14) as well as the most common premonitory symptom (5). Therefore, our analysis identifies only potential triggers: Understanding causality requires a test (where possible) of the effect on attack occurrence by modifying exposure to the factor.
Methods
Study participants
Pseudonymised data from 327 migraine patients who had participated in the PAMINA study at the Medical University of Vienna, Austria, were reanalysed by Curelator Inc, at the individual level as described below. The transfer of PAMINA study data to Curelator Inc for this retrospective analysis was approved by the Legal Department of the Medical University of Vienna, and an electronic database containing only sequential numbers for differentiating patients was provided. The database did not contain names, initials, dates of birth, or any other identifying information. For the initial PAMINA study, all patients gave informed consent. Since data were anonymised, informed consent from former study participants was not obtained for purposes of the current retrospective analysis.
The primary data collection methods and characteristics of the population in this dataset have been described previously (3). Briefly, patients completed a paper diary every day before bedtime for 90 days to assess a wide range of factors that could be associated with headaches and migraine attacks. They recorded the presence or absence of each factor, but they were not asked to decide if any of the factors were triggers for their headaches or migraine attacks. Of note, patients completed the diary with a high degree of compliance (>80%).
Patients were included in this post-hoc analysis if they completed at least the first 15 study days. Using this criterion, only one patient was excluded and data from 326 patients who had migraine with or without aura, according to the second edition of the International Classification of Headache Disorders (ICHD-2) (18), were used.
Potential trigger factors included in the analysis.
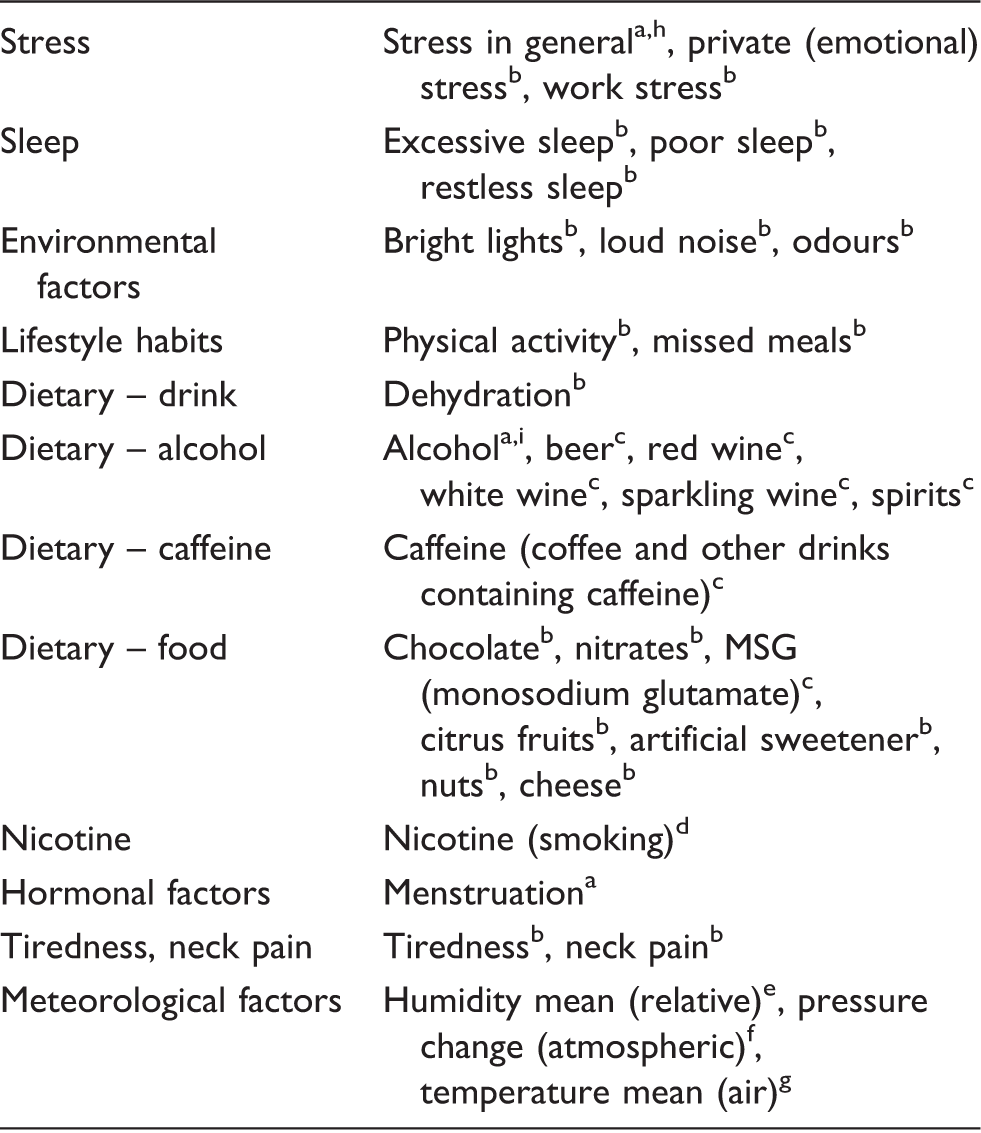
Binary variable: yes/no; bContinuous variable: four-point scale; cContinuous variable: number of glasses, cups or helpings; dContinuous variable: number of cigarettes; eContinuous variable: %; fContinuous variable: hPa; gContinuous variable: oC. hThe ‘stress in general’ dummy factor includes private (emotional) stress and work stress. iAlcohol dummy factor includes beer, red wine, white wine, sparkling wine and spirits.
In this analysis, every episode of headache was classified by an algorithm according to ICHD-3 (20) to differentiate migraine attacks from non-migrainous headaches. Headaches fulfilling criteria for migraine with or without aura were labelled as ‘definite’ or ‘probable’ in the database and both types were classified as ‘migraine attacks’ in the analysis.
Statistical methods
Mean differences were tested using unpaired t-tests or unpaired t-tests with Welch correction, and differences in percentages were analysed using Fisher’s exact tests.
For each patient, the association between factors and migraine events was explored for all factors; univariate associations were analysed by means of Cox proportional hazards models (21). The statistical significance of the association between the factor and migraine events was tested using the Wald test, and the effect of the factor was estimated by means of the hazard ratio (HR). At the end of the 90-day study period, factors were considered as potential triggers if univariate HRs were greater than 1 (HR > 1) and p values less than or equal to 0.05 (p value ≤ 0.05); i.e. they were significantly associated with an increase in the risk of migraine attack occurrence.
A possible approach to differentiating triggers from premonitory symptoms is to exclude the factor data collected on the days when attacks start, the hypothesis being that factors immediately (same day) associated with attacks are more likely to be premonitory signs, whilst those present one or more days before attacks are more likely to be triggers. The PAMINA data were found to be suitable for an analysis that eliminated factor data on the days on which attacks started (termed ‘Day-1 analysis’), but not to also eliminate the day before (i.e. it was not possible to determine significant factors from two or more days before attack start). The Day-1 analysis used the same statistical method described above but excluded the presence of the factor on the day of attack start.
At the population level, the associations between factors and migraine events were explored by means of a multivariate Cox proportional hazards model using the Andersen-Gill extension (based on the counting process) and robust variance estimate (22). The automatic factor selection was performed by means of the forward procedure (with p value ≤ 0.05 as selection criterion). The statistical significance of the association between the factors and migraine events was tested using the Wald test, and the effects of the factors were estimated by means of the HR. Factors were considered as potential triggers or warning signs if the HRs from the multivariate Cox proportional hazards model were greater than 1 (HR > 1).
The amount of missing data was very low (<0.8%) and we used the last observation carried forward (LOCF) approach for imputing missing data. All analyses were performed using SAS version 9.3 (SAS Institute Inc, Cary, NC, USA). Given the exploratory nature of this study, p values were not adjusted for multiplicity.
Results
Inter-individual variability
Main characteristics of patients included in the analysis dataset.

Figure 1 shows the percentage of patients according to the number of potential ‘trigger’ factors per patient at the end of the study period. Using our N = 1 methodology, we were successfully able to generate trigger profiles for 285 patients (87.4%). Specifically, these 285 patients had at least one significant factor and on average they had four significant factors (95% confidence interval (CI): 3.6–4.3).
Percentage of patients according to the number of trigger factors per patient.
We were not able to find significant associations in 41 patients. The majority of these patients did not have any (n = 10) or only one or two (n = 21) migraine attacks, insufficient to enable a signal to be detected. Of the remaining 10 patients, seven had ≤4 attacks and/or <30 days of data; only three patients had >5 attacks and ≥85 days of data. On average, we needed approximately five days with migraine and three migraine attacks to detect the first trigger.
Analysis of individual potential trigger profiles: patients with one or more potential triggers.

Potential trigger profile observed in only one patient. bPotential trigger profile shared by at least two patients.
An example of an individual patient trigger profile is shown in Figure 2. Such “Individual Trigger Maps™” are generated by Curelator Headache as a visual tool to aid patients in understanding their potential trigger association profiles. In this patient, artificial sweeteners, physical activity, monosodium glutamate, red wine, menstruation, eyestrain, and neck tension are all associated with migraine attacks. The first five are ‘testable’ potential triggers. Eyestrain is a potential trigger, but difficult to test, or it may be a premonitory symptom. Neck tension is likely a premonitory symptom but could be a trigger and, again, is difficult to test.
Example of an Individual Trigger Map.
Aggregation of individual results
Patients who had at least one significant factor showed a higher mean number of migraine attacks per month per patient than patients with no significant factors (3.2 vs. 1.2; p value < 0.0001). Figure 3 shows the number of significant factors per patient for males and females. A higher proportion of females than males had at least one significant factor (89.6% vs. 75.0%; p value = 0.0049) and females had a higher mean number of significant factors per individual (4.2 vs. 2.4; p value < 0.0001).
Percentage of females and males according to the number of potential trigger factors per patient.
Figure 4 shows the percentage of patients in which each of the factors was significant. The most common was neck tension followed by tiredness, temperature mean, restless sleep, and odours.
Percentage of patients according to each potential trigger factor.
Comparing factors associated with migraine attacks in women and men showed a few statistically significant differences. A higher proportion of females than males had temperature mean (35.1% vs. 18.8%; p value = 0.0297), odours (28.0% vs. 8.3%; p value = 0.0034), bright lights (28.0% vs. 6.3%; p value = 0.0009), loud noise (25.1% vs. 6.3%; p value = 0.0024) and dehydration (20.4% vs. 8.3%; p value = 0.0463) as potential trigger factors. Regarding menstruation, 25% of females with menstrual cycles had menstruation as a significant trigger factor.
Further analysis shows that there are two classes of profiles: (1) unique, i.e. a profile observed in one patient; and (2) shared, i.e. a profile shared by at least two patients. The percentage of patients with unique profiles was higher than the percentage of patients with shared profiles (73.9 vs. 13.5%) (Table 3). Comparison by gender shows that a higher proportion of females than males had unique profiles (70.8% vs. 45.8%; p value < 0.0001) (Table 3). In the group of patients with unique profiles, females had a higher mean number of significant factors per patient than males (5.2 vs. 3.7; p value = 0.0101), whilst males with shared profiles had a higher mean number of significant factors than females (2.4 vs. 1.5; p value = 0.0455).
Whilst it would be interesting to compare patients having migraine with or without aura, this was not possible with the number of patients in this study. Of the 327 patients, 147 patients reported ‘aura’ with some attacks, for a total of 839 attacks with aura. However, using ICHD-2 criteria and a detailed interview, the PAMINA investigators diagnosed only 54 patients as having ‘migraine with aura’ and in these there was a total of 354 migraine days with aura. Similarly, given the small proportion (7%) of patients receiving prophylactic medication, it was not valid to compare them with those not receiving prophylaxis.
Factor-attack profiles when factor data on attack start days are excluded (Day-1 analysis)
The Day-1 analysis showed that, in general, fewer factors per patient were significantly associated with attacks: The all data mean was 4.0 factors per patient (95% CI = 3.6–4.3) whilst the Day-1 analysis mean was 3.0 factors per patient (95% CI = 2.7–3.3). Figure 5 shows the significant factors in the two analyses: notably a number of factors were much less commonly identified in the Day-1 analysis (neck pain, tiredness, restless sleep, odours, bright lights and noise,) indicating that, at least in a proportion of patients, these are more likely to be premonitory symptoms than attack triggers. ‘Missed meals’ was also less commonly identified in this analysis: intuitively it seems likely that a missed meal could be a trigger acting within hours (e.g. missing breakfast triggering a migraine the same day), although it is also possible that missing a meal could be due to loss of appetite, which in turn may be a premonitory symptom.
Significant factors associated with attacks: comparison of analyses which include factors on attack start day or exclude these (Day-1 analysis).
Population analysis
Potential triggers identified in the population analysis.

SE: standard error; CI: confidence interval.
Discussion
In this study, we successfully generated individual ‘potential trigger’ profiles in almost all patients and found that within the study population virtually all patients with complex factor-attack association profiles had unique and different profiles. This finding underscores the need for individual patient discovery and analysis of such profiles as an important step towards management of diseases showing highly individualised pathologies, such as migraine.
Individual-level analysis of factors with significant association
This post-hoc analysis provides, for the first time at the individual patient level, information about the associations between migraine attacks and a broad spectrum of factors which have been suggested as migraine triggers or premonitory symptoms. We found that most (87.4%) patients had at least one significant factor and, on average, there were four significant factors identified per patient. Many of the factors identified are considered potential trigger factors (e.g. weather, stress and dietary elements) but some may be premonitory symptoms (e.g. neck tension). To add to the complexity, for some factors it is unclear if they are triggers or premonitory symptoms; for example, this may apply to chocolate consumption and tiredness. Further, whether a factor is a trigger or premonitory sign may vary from patient to patient. Therefore, our analysis identifies only potential triggers and premonitory symptoms: Understanding causality requires a test (where possible) of the effect on attack occurrence by modifying exposure to the factor. Interestingly, analysis excluding factor data on the days attacks start (Day-1 analysis) reduces the number of significant factors per individual, notably by reducing the proportion of patients for whom factors such as neck pain, exposure to odours, bright lights or noise and restless sleep are identified. It is probable that these factors, in these patients, occurring within 24 hours of attack start, are premonitory symptoms rather than triggers.
Individual patient profiles: Implication for aetiology and treatment
An important observation of our analysis is that 85% of patients who had at least one identified trigger had unique and individual trigger factor profiles. For patients with three or more triggers associated with their attacks, 96% had unique and individual profiles.
The high degree of heterogeneity in potential trigger profiles among individuals is a surprising finding and raises two important possibilities with respect to management and treatment of migraine. First, the list of 33 factors that were tracked in this study was historically derived from aggregate populations and observation of trigger factors most commonly shared in those populations. Given the high degree of intra-individual heterogeneity, it is highly likely that the list of factors studied (Table 1) does not include a potentially wide array of factors less commonly shared in a population but nevertheless important in the composition of individual trigger profiles. In other words, it is likely that individual trigger profiles contain a mixture of common (included in our list) as well as uncommon trigger factors (not included in our list). Therefore it is possible that in our patients, other trigger factors may be significant but were simply not tracked.
Second, the high degree of heterogeneity we found in profiles among individuals is not easily explained by a simple genetic and/or physiological predisposition to trigger factor sensitivity. Instead, it raises the possibility that individual patients may acquire their unique personal trigger profile from longitudinal exposure to their unique personal environment. This would imply that trigger acquisition may be driven by an alteration in associative (e.g. operant or classical) or non-associative (e.g. sensitisation) learning, instead of pre-set physiological predisposition.
Interestingly, behavioural techniques that involve ‘unlearning’ or desensitisation of triggers such as cognitive behavioural therapy (CBT) (23,24) are surprisingly effective at reducing headaches. In fact the CBT studies cited here have been shown to be more effective than trigger avoidance alone, perhaps suggesting that the mechanisms involved in associative or non-associative learning (‘unlearning’) may be aetiologically more relevant than avoidance of activation of physiological pathways.
Taken together, the high degree of individualisation in trigger profiles combined with the observation that triggers may be ‘unlearned’ suggests that at least some triggers may be acquired through learning of personal environment, rather than solely through predisposition of neurophysiological sensitivity. The high degree of inter-individual heterogeneity could at least partially be the result of an individual’s personal environment and associated learning within that unique environment. Much larger studies with thousands, or even tens of thousands, of patients are needed to confirm the degree to which profiles are shared and whether multiple clusters of patients share common trigger profiles. Nevertheless, the considerable inter-patient variability in trigger factor profiles found in this study emphasises the need for initial determination of personal trigger factors as part of an individualised treatment program.
Gender differences
Analysis by patient gender showed marked differences between females and males in the percentages of patients who had at least one significant factor, the mean number of significant factors per subject, and the percentage of patients who had a unique profile. This is partially accounted for by menstruation as a trigger factor, which has been extensively studied (25), but the results also showed that a higher proportion of females than males had significant factors related to environmental factors (odours, bright lights, loud noise), meteorological factors (temperature mean) and fluid intake (dehydration).
Comparison with population-level identification of triggers
Our results indicate that only eight of 33 factors analysed at the population level were identified as potential trigger factors. Menstruation and neck tension increase the risk of migraine occurrence by up to 53% and 49%, respectively. Tiredness, bright lights, loud noise, excessive sleep, restless sleep, and odours increased the risk by <25%. All these factors were identified in at least 10% of patients: With the exception of menstruation and excessive sleep, they were also the most commonly identified in individuals.
Menstruation shows an interesting difference in the aggregated individual analysis vs. population analyses in this study and the original PAMINA study (3). In both population analyses, menstruation had the ‘strongest’ association with migraine; however, in the aggregated individual analysis, it was found as a factor in only a total of 15% of patients and in 25% of females with menstrual cycles. A possible explanation for the decreased importance of menstruation in the N = 1 analysis is that the 90-day study included just three menstrual cycles. If the association with attacks is not 1:1, then this may be missed as an individual factor until more cycles are included.
Our population analysis results are similar to those described by Wöber et al. (3), although there are some differences which are likely due to the different statistical methods used. Notably, menstruation and neck tension are associated with greatest increase in risk in both analyses. Interestingly, in population-level analyses, neither we nor Wöber identified any dietary factors as causing an increase in risk of migraine attacks. However, at the individual level, dietary factors were significantly associated with attacks in some patients, although individual dietary factors were seen in fewer than 10% of patients. Similarly, there was a striking difference between individual-level and population-level analyses of meteorological factors. Neither the PAMINA study (3,26) nor the current population analysis showed a relevant association between migraine and weather. In contrast, the individual analysis found such an association which supports an editorial comment (27) to the PAMINA findings (26) questioning whether ‘so many patients can be wrong’. Overall, our individual-level analysis identified a much wider range of potential triggers than was found in the population analyses, concordant with Simpson’s paradox (17), mentioned in the Introduction. This has great significance to individuals with migraine: it is necessary to analyse their personal data individually to identify their potential triggers and premonitory symptoms as a prelude to introduction of behavioural changes which might reduce the number (and possibly severity) of attacks.
Study limitations
On comparing patients with at least one factor associated with migraine attacks and those with none, there was a significant difference in the mean number of migraine attacks per month per patient (3.2 vs. 1.2). This is expected and points to a study limitation: In a 90-day study, patients with a low frequency of attacks will have a paucity of data for analysis (which requires both days followed by an attack AND days not followed by an attack); hence significant factors will not be identified. In these patients, collection of data over a longer time period that includes the potential for more attacks may well lead to identification of additional trigger factors. Similarly, even in patients with multiple identified trigger factors, longer data collection periods may have allowed identification of additional trigger factors.
We could not analyse the causality of potential triggers. This could be tested by modifying or eliminating exposure to these for a test period to determine if there is an effect on attack frequency, thereby establishing (or eliminating) causality. Curelator Headache plans to include such guided testing. Establishing which factors are true triggers and those for which modifying exposure leads to a reduction in frequency of migraine attacks would help patients better self-manage their condition. Determining factors that are premonitory symptoms may be helpful to research into underlying mechanisms causing attacks (28). Knowledge both of triggers and premonitory symptoms may be useful in predicting the occurrence of individual migraine attacks.
The findings of this study may also be limited by the fact that we have analysed retrospectively a pre-existing database. However, we believe this to be a valid way to develop and test the algorithms and programs required to analyse at the individual level the factors related to, and which may be triggers of, migraine attacks. The number of patients included in this database was adequate to allow us to quantitatively analyse at the individual level the associations between factors and migraine attacks. Furthermore, the study period completed by patients (approximately 90 days) allowed the convergence of models and, therefore, the results appear valid. The observed differences between men and women should be confirmed in future analyses with a greater number of males.
Finally, in two cases (stress in general and any alcohol consumption) factors were generated by collapsing data with finer granularity: This we considered acceptable given the data available. One factor not analysed, because it would require further derivation from a ‘collapsed’ factor, is letdown from stress, recently shown to be a trigger by Lipton et al. (11).
Conclusion
In this study, we have used an N = 1 analytical approach to understand the composition of ‘potential trigger’ profiles associated with migraine attacks in individuals. We found individual trigger profiles contain an average of four trigger factors associated with increased risk of attacks and a very high degree of heterogeneity between individuals.
A common but unproven hypothesis in migraine is that combinations of triggers act to precipitate migraine attacks in individuals when a ‘threshold’ is reached. To our knowledge, for the first time, this study attempts to quantify this hypothesis by showing that multiple factors are associated with the occurrence of migraine attacks in individuals. However, further study at the individual level is needed to show which single or combinations of factors are true triggers, that is those for which modifying exposure leads to a reduction in the number (and/or severity) of attacks.
Approaches to elucidate an individual’s triggers through associative N = 1 analytical techniques, as opposed to patient self-reporting of triggers (with potential for recall bias), are a critical and necessary prerequisite to management of triggers through either avoidance or CBT techniques. The findings of this study improve markedly the knowledge about potential triggers and premonitory symptoms for migraine attacks at the individual level and provide a method to determine these in individuals, the need for which was discussed by Spierings et al. (13). When applied in a digital platform this method may provide an effective tool for improved (self-)management by people with migraine and by their clinicians as part of a personalised treatment program.
Clinical implications
Correct identification of migraine triggers in individuals may allow more effective (self-)management of the condition. Such identification can be performed only by prospective, daily collection of information about potential triggers, which includes behavioural, dietary, or environmental factors. This requires daily data collection about behaviours and migraine attacks and N = 1 statistical analysis. The profile of significant factors associated with migraine attacks is shown to be highly individual: on average, each migraine patient has four identifiable potential triggers.
Footnotes
Acknowledgements
For additional support with manuscript editing, software programming, and design of visualizations, we thank: Gabriel Boucher, Marc Albert Fañé, Sandy Bodner, Rosa Maria Costas Rodriguez, Jordi Pellat Massó, Ferran Martin, Martín Ferrari, Jordi Roses and Julie Callahan. We also thank the collaborators of the PAMINA study: Çiçek Wöber-Bingöl, Werner Brannath, Karin Schmidt, Magdalena Kapitan, Ernest Rudel and Peter Wessely.
The original PAMINA database is held by Dr Wöber, who should be contacted regarding access. The analysis and statistical models described in this paper are held by Curelator Inc. Contact Dr Donoghue regarding access.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The infrastructure required for the analysis described in this paper was funded by Curelator Inc, Cambridge, MA, USA. Drs Donoghue, Peris and Mian are employees of Curelator Inc. Drs Wöber and Torres are paid consultants of Curelator Inc. Dr Wöber has received personal fees from St Jude, Allergan and Pfizer not related to this study.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
