Abstract
Objective
To observe the prevalence and characteristics of premonitory symptoms in Chinese migraineurs and explore their associations with migraine-related factors.
Method
Migraineurs who visited a tertiary headache clinic and one of nine neurology clinics between May 2014 and November 2019 were studied.
Result
Among the 4821 patients meeting the migraine criteria (International Classification of Headache Disorders, 3rd edition), 1038 (21.5%) patients experienced at least one premonitory symptom. The most common premonitory symptoms were neck stiffness, dizziness, yawning and drowsiness. The logistic regression analysis demonstrated that aura, photophobia, aggravation by routine physical activity, triggers, family history, depression, coffee consumption and physical exercise were associated with an increased probability of experiencing premonitory symptoms (p ≤ 0.001). The premonitory symptoms of migraine with and without aura differ in prevalence and most common symptoms. The cluster analysis revealed pairwise clustering of the following premonitory symptoms: Photophobia/phonophobia, concentration change/dysesthesia, loquacity/overactivity, yawning/drowsiness, fatigue/dizziness, and mood change/irritability. The correlation analysis of triggers and premonitory symptoms revealed that temperature change, environment change, sleep disorder, activity and stress were related to multiple premonitory symptoms, and that food, light, menstruation, alcohol and odor were related to special premonitory symptoms (p ≤ 0.001).
Conclusion
The prevalence of premonitory symptoms among migraineurs in China is 21.5%. Some factors influence the probability of experiencing premonitory symptoms. Paired premonitory symptoms in the clustering analysis may share similar origins. Certain triggers associated with multiple premonitory symptoms may induce brain dysfunction; however, other triggers that overlap with corresponding special premonitory symptoms may be premonitory symptoms or a form of premonitory symptom.
Introduction
Migraine is a recurrent primary headache influenced by genetic and environmental factors. Accumulating evidence suggests that migraine is more than a headache and should be treated as a cycling and fluctuating brain dysfunction with at least the following three phases: Prodromal phase, head pain phase and postdromal phase. As these three phases are integrated and occur in succession, the prodromal phase, which is the initial phase of the whole process, is increasingly being emphasized (1–3).
In the International Classification of Headache Disorders third edition (ICHD-3), the prodromal phase is defined as “a symptomatic phase, lasting up to 48 h, occurring before the onset of headache in migraine without aura or before the aura in migraine with aura” (4). During this phase, various symptoms can manifest. Previous studies focusing on premonitory symptoms (PS) have found that some PS correctly predict headache; some continue into the headache phase; and some exhibit patterns associated with brain dysfunction (5,6). Such findings improve our understanding of the neurobiological basis of migraine and can aid the identification of novel therapeutic targets for attack prior to headache (7,8). However, the prevalence of PS varies among studies due to difference in study design and sample number (1,9). Whether other underlying factors influence the result still requires further investigation.
The appearance of PS is believed to represent the beginning of the alternation in brain function (10,11) and is closely associated with trigeminovascular nociceptive system activation. Previous studies have attempted to classify these symptoms functionally; however, the results are inconsistent among studies and limited supporting evidence is available (1,12,13).
In addition to PS, exogenous triggers, such as light and food, have been recognized to warrant investigation before a migraine attack. Studies inducing migraine under natural conditions or with experimental special substances have provided direct evidence of triggers (14–17). However, the close relationships between some triggers and PS, such as those between light and photophobia, food and food craving, and odor and osmophobia, suggest an alternative possibility that triggers may be another form of PS presented during the prodromal phase (1,18). However, whether this relationship is an individual phenomenon or a general one is still uncertain. Thus, further studies of triggers and PS of migraine are needed.
In consideration of the above observation, the aims of our study were to i) ascertain the prevalence and other characteristics of PS in Chinese migraineurs, explore the possible factors influencing the PS prevalence, ii) statistically cluster PS, analyze the feasibility in terms of associated brain function, iii) investigate the correlations between triggers and PS and discuss the possible mechanism.
Materials and methods
Participants
This study was conducted in a tertiary headache clinic at the PLA General Hospital and nine other neurology clinics from May 2014 to November 2019. All patients who had been diagnosed with migraine according to the ICHD-3 were invited to participate in the study. The patients who agreed were screened by two qualified and experienced headache experts to exclude possible migraine (Figure 1). The electronic medical records of all patients were collected. The types of aura were recorded as an independent item according to the ICDH-3 for migraine with aura.

Flow chart of the migraineur enrolment.
Standard protocol approvals, registration, and patient consents
The study was approved by the Ethics Committee of the Chinese PLA General Hospital (2020263). The study protocol complied with China’s regulations and Guidelines for Good Clinical Practice. Due to the data-collecting nature of the study, oral informed consent was obtained from the patients before inclusion in the study according to the World Medical Association’s Declaration of Helsinki.
Data availability
We will share anonymized data upon reasonable requests from any qualified investigator. The electronic medical data were all collected by software shared through the “cloud”, rendering the data available online.
Data collection
The multi-clinic data were collected using an electronic structured questionnaire (ESQ) including detailed clinical information and migraine related factors (the details are listed below). This ESQ was available online to all qualified multi-clinic researchers who were responsible for obtaining clinical data. Before we started our enrolment, special training was provided to the doctors at the clinics involved in the study to learn how to complete the questionnaire online to ensure homogeneity in the data entry.
Data related to age, sex, diagnosis, lifestyle factors (alcohol consumption, physical exercise, and tea/coffee consumption), education level, headache characteristics (onset time, course, location, duration, frequency, pain nature, Visual Analogue Scale score, concomitant symptoms, triggers, premonitory symptoms, and mitigation factors) and migraine-related factors (family history, menstrual-related state, sleep state, PHQ-9 scale score and GAD-7 scale score) were collected. Information regarding premonitory symptoms was collected with a structured questionnaire covering the following 25 items: Overactivity, loquacity, mood change, irritability, dysesthesia, drowsiness, fidgeting, concentration change, photophobia, phonophobia, osmophobia, dysphasia, yawning, neck stiffness, food craving, poor appetite, sensation of cold, fatigue, diarrhea, constipation, thirstiness, diuresis, dizziness, edema, and other (any symptoms not included among the other 24).
Statistical analysis
The statistical analyses were performed using SPSS (version 23.0) and R (version 4.0). The continuous data were analyzed using the Kruskal–Wallis test and the Mann–Whitney U test. The categorical data were analyzed using χ2 tests. In the binary multiple logistic regression analysis, the presence of any PS was the dependent variable, and factors, including diagnosis, photophobia, phonophobia, aggravation by routine physical activity, trigger, coffee consumption, tea consumption, physical exercise, family history, education level and depression were regarded as valid PS predictors (p ≤ 0.001). The non-valid predictors were removed one by one from the model. The co-occurrence of PS was tested using Cohen’s kappa with 95% confidence intervals (CIs). A hierarchical cluster analysis was conducted with the Jaccard distance and complete linkage to explore and identify the relationships among PS. Fisher’s exact test was used to analyze the correlations between PS and triggers. A two-sided p < 0.001 was considered statistically significant. Subjects with missing data were excluded from the analyses.
Results
Patient demographics
A total of 10,949 patients initially diagnosed with migraine were invited to participate in the study, and 6717 patients agreed. The electronic data and detailed information of the patients were reviewed by two headache experts. Accordingly, 1896 patients were excluded because 295 patients did not meet the migraine criteria (by the headache experts), 117 patients did not have intact clinic data and 1484 patients were diagnosed with possible migraine (Figure 1). As a result, 4821 migraineurs (1059 males and 3762 females, age 38.7 ± 12.4 years, range 9–86 years) were enrolled in this study. Among the patients, 4107 migraineurs (802 males and 3305 females) had migraine without aura (MO), and 714 patients (257 males and 457 females) had migraine with aura (MA). Moreover, 105 migraineurs (40 males and 65 females) were children (age range 9–15 years).
General PS information
A total of 1038 (21.5%) migraineurs experienced at least one premonitory symptom (mean 2.1 ± 1.76, range 1–12) during the prodromal phase. Among these PS+ migraineurs, 47.2% reported experiencing more than one PS (Figure 2(a)). The most common PS were neck stiffness (6.9%), dizziness (6.9%), yawning (4.2%) and drowsiness (3.5%) (Figure 2(b)). Among the child migraineurs, dizziness (12.4%), photophobia (7.6%), fatigue (5.1%), poor appetite (4.8%) and attention disorder (4.8%) were the most common PS, and no loquacity, thirstiness, fidgeting, diarrhea, diuresis or food craving were experienced (Figure 2(c)).

Details of the PS experienced by migraineurs. (a) Numbers and proportions of migraineurs who experienced different number of PS simultaneously. (b) Numbers of migraineurs experiencing each PS to the number of migraineurs experiencing any PS (%).From the data we determined the PS spectrum experienced by most migraineurs and its dissimilarity in MA and MO. Orange: migraineurs with aura (MA); blue: migraineurs without aura (MO); green: both. (c) Numbers of child migraineurs experiencing different PS. Compared with adult migraineurs, child migraineurs had a different PS spectrum; they showed no loquacity, thirstiness, fidgeting, diarrhea, diuresis or food craving. (d) Numbers of MA with different auras (orange) and PS experienced individually (blue) (indicated as number to number, %). As show in the figure, although visual aura is the most common aura, MA with aura dyskinesia was experienced at the greatest PS rate (56.5%).
The migraineurs with photophobia (p < 0.001), phonophobia (p < 0.001), aggravation by routine physical activity (p < 0.001), with a trigger (p < 0.001), coffee consumption (p < 0.001), physical exercise (p ≤ 0.001), family history (p = 0.001), thirstiness, fidgeting, and depression (PHQ Total Score > 4) (p < 0.001) were more likely to experience PS. Tea consumption was associated with a low probability of experiencing PS. Moreover, the migraineurs with a trigger and depression were likely to experience more than one PS (p < 0.001) (Table 1).
The number of migraineurs experiencing PS and the mean number of PS experienced.

*Indicates statistically significant (p < 0.001).
In the binary logistic regression analysis, MA (odds ratio, OR = 1.66, 95% CI 1.29–2.12), photophobia (OR = 1.49, 1.22–1.82), aggravation by routine physical activity (OR = 1.56, 1.22–1.99), trigger (OR = 2.31, 1.78–3.02), family history (OR = 1.30, 1.08–1.57), depression (OR = 1.40, 1.08–1.80), coffee consumption (OR = 2.40, 1.64–3.49) and physical exercise (OR = 1.45, 1.15–1.82) were associated with an increased probability of experiencing PS (Figure 3 and Supplemental Table 1).
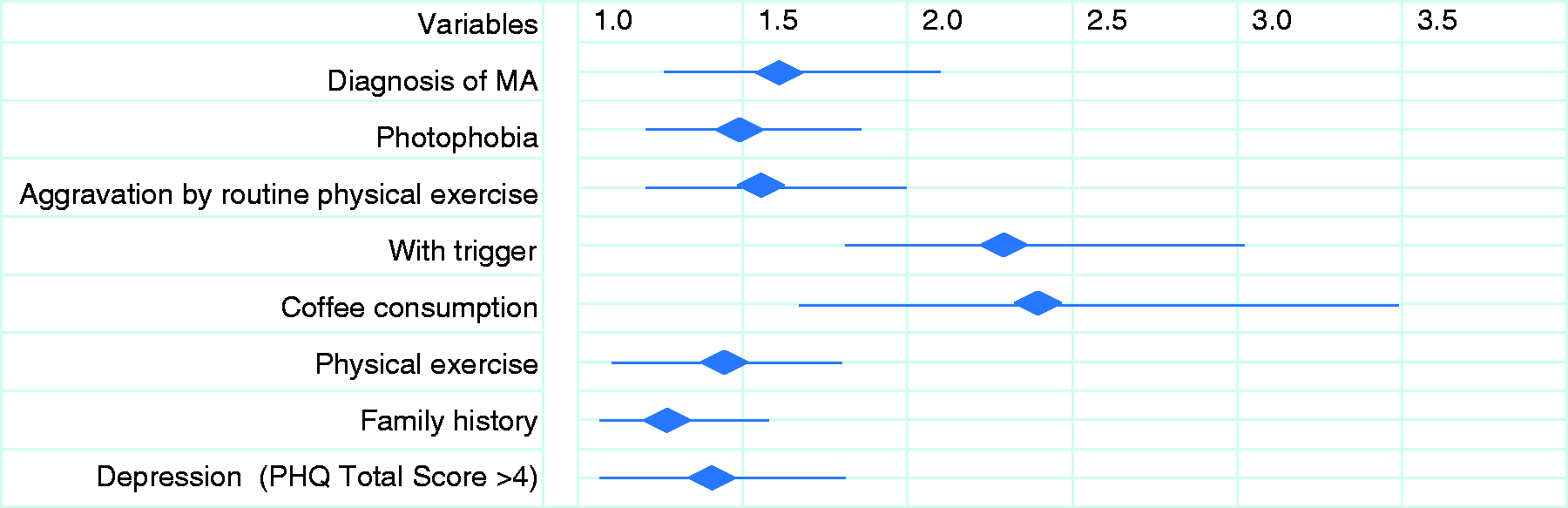
Summary of the statistically meaningful predictors of PS. This figure shows the variables predicting PS and their odds ratios (OR). The detailed results of the binary logistical regression model can be found in Supplemental Table 1.
Comparison of PS between MA and MO
The prevalence of PS significantly differed between MA and MO (30.0%, 214/714 and 20.1%, 824/4107, respectively) (p < 0.001) (Table1). Compared to MO, MA were more likely to experience PS and experience more than one PS (p < 0.001) (Table 1). The most common PS experienced by MO were neck stiffness, dizziness, yawning and drowsiness, whereas the most common PS experienced by MA were dizziness, photophobia, phonophobia and yawning (Figure 2(b)). Although MA with visual aura account for the largest number of MA patients, MA with aura of dyskinesia had the highest PS positive rate of 56.5% (Figure 2(d)).
Cluster analysis of PS
The PS with the highest co-occurrences were photophobia/phonophobia (kappa¼ 0.61, standard deviation (SD) 0.04), concentration change/dysesthesia (kappa¼ 0.37, SD 0.05), and loquacity/overactivity (kappa¼ 0.37, SD 0.11) (Figure 4(a) and Supplemental Table 2). The clustering analysis of PS showed that photophobia/phonophobia, concentration change/dysesthesia, loquacity/overactivity, yawning/drowsiness, fatigue/dizziness, and mood change/irritability were the closest-coupled PS in distance (Figure 4(b)).

Results of the PS analysis. (a) Matrix of the Spearman’s correlation analysis of PS. The PS with the highest co-occurrence are shown in blue squares. Kappa values are given in Supplementary Data Table 2. (b) Clustering analysis of PS. X-axis, individual PS; Y-axis, the distance.
Correlation analysis of PS and triggers
Figure 5 and Supplemental Table 3 show the results of the analysis of the correlations between PS and triggers using Fisher’s exact test. The five columns on the left, including temperature change, environment change, sleep disorder, anxiety and stress, were correlated with multiple PS (≧3, multiple PS-related (MP) triggers), whereas the other triggers on the right, including food, light, menstruation, alcohol consumption, and odor, were associated with specific PS (≦2, special PS-related (SP) triggers) (p < 0.001).
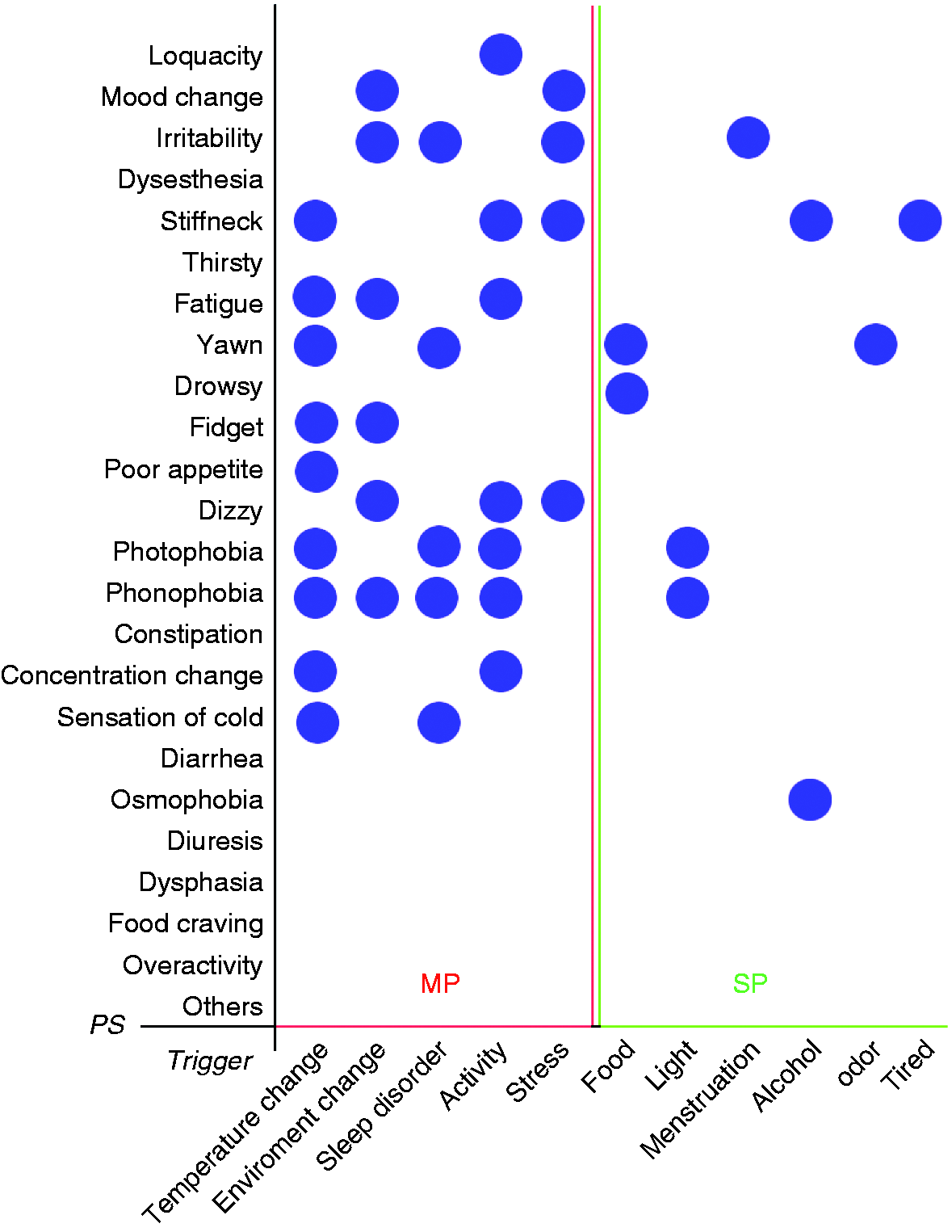
Correlations between PS and triggers as determined using the Fisher’s exact probability method. Blue circles indicate significant correlations (p ≤ 0.001). The five columns on the left including temperature change, environment change, sleep disorder, activity, and stress were correlated with multiple PS (≧3, MP triggers); the remaining triggers on the right, including food, light, menstruation, alcohol, odor and tiredness, were associated with specific PS (≦2, SP triggers) (p < 0.001). The p-values are shown in Supplemental Table 3. MP, multiple PS-related trigger; SP, special PS-related trigger.
Discussion
During the prodromal phase of migraine, brain function begins to change and various non-pain symptoms manifest (11,19,20). As a result of several studies in recent decades, the prevalence and phenotypes of these symptoms have become well recognized (5,6,9,10,17,18,21–26). However, the results are inconsistent among these studies due to the diversity of study designs and sample sizes of enrolled patients. In this cross sectional research, 4821 Chinese migraineurs from 10 clinics were studied. To the best of our knowledge, this PS study constitutes the largest series of migraineurs to date in terms of both the sample size and study period.
Among these 4821 migraineurs, 21.5% (1038) experience at least one PS, which is lower than the percentage reported in most previous studies (1,26). This difference might be attributable to several reasons. One potential reason involves the sample size. According to statistical theory, the PS prevalence estimated from a sample should approach the population prevalence as the sample size increases (27). As migraineurs in the general population often have mild headache and atypical manifestation in contrast to migraineurs presenting at clinics (9), the incidence of PS in population-based studies is low (12,21), ranging from 8–14%. Our result lies between the estimates from population- and clinic-based studies and, therefore, is consistent with expectation. A second potential reason involves the MA:MO ratio in this study. Compared with MO, MA are more likely to experience PS and experience more than one PS (6,9,12,21). Our data revealed a difference in PS prevalence of nearly 10% between MA and MO. Thus, the lower proportion of MA (14.8%) in this study results in a lower PS incidence. Although there is no direct evidence of a relationship between the MA:MO ratio and PS incidence, studies with higher MA ratios have reported higher PS incidences (9,23). Moreover, the binary multiple logistic regression analysis confirmed the contribution of aura to PS prevalence. Family history and coffee consumption also influence the occurrence of PS according to our results. Family history adequately explained the high PS incidence observed in a previous large sample study, in which a survey of PS in migraineurs and their first-degree relatives was conducted (9). Coffee is the most common dietary trigger of migraine, and caffeine discontinuation improves acute migraine treatment (28,29). Coffee could excite the central nervous system, disturbing sleep and increasing anxiety, and induce vasoconstriction of cerebral blood vessels. Furthermore, coffee withdrawal could induce rebound cerebral vasodilatation (30,31), which is a potential cause of headache (32,33). The present study illustrated the contribution of coffee consumption to the existence of PS, and this factor yielded the highest odds ratio (Figure 3). Interestingly, tea and coffee had opposite effects on PS prevalence. Large data from populations representing coffee/tea consumers are needed to confirm this opposite effect.
Previous studies have demonstrated that premonitory symptoms are a characteristic of typical migraine (9,14,18,22). Therefore, it is easy to understand the result that PS are closely related to manifestations of typical migraine such as photophobia, phonophobia, aggravation by routine physical activity and triggers. The effect of physical exercise on migraine remains a controversial issue, with physical exercise as a regimen belonging to the non-pharmacological treatment field. Recent research indicates that its efficacy relies on individuals’ exercise tolerance, exercise intensity and the speed of exercise increment (34). Thus, targeted personal prescription is expected to be a good preventive treatment for migraine (35). To the best of our knowledge, the relationship between PS and physical exercise has never been previously reported. However, our results suggest that a negative relationship exists between both. The reason for this result may originate from the lack of quantitative analyses of the intensity and intervals of regular exercise. We should pay more attention to the life habits mentioned above, which suggest that possible lifestyle adjustments could prevent PS or even migraine.
Previous studies have focused on the most common PS. Although the PS investigated vary among studies, the results are largely consistent, with the most common PS including fatigue (appeared in nine studies), mood change (seven studies), neck symptoms (six studies), photophobia (five studies), yawning (four studies), difficulty in concentration (three studies), increased activity (two studies), appetite change (two studies), gastrointestinal symptoms (one study) and facing change (one study) (Table 2). In the present study, neck stiffness, dizziness, yawning and drowsiness were the most common PS, which partially differs from previous work. It should be noted that the MA:MO ratio influences the “most common PS” identified because the most common PS typically differ between MA and MO groups (1,6,12,18,24). This discrepancy reflects the heterogeneity of MA and MO and implies a separate derivation of both subtypes.
The PS prevalence studies in recent decades.

NM: not mentioned.
# Children were selected with at least one premonitory symptom.
PS have been classified in previous studies to investigate their underlying mechanism (1,12,13). However, classifications based on personal experience lack objective standards. In recent years, hierarchical cluster analysis has become increasingly valuable in high-dimensional data exploration and is widely used in genomics and biomedical research (39,40). This analysis reveals the internal structure of data and groups observations based on their degrees of similarity. The vast data collected from a large sample of patients facilitated the statistical analysis in the present study, allowing us to group PS for the first time. The results revealed pairwise clustering of the following PS: Photophobia/phonophobia, concentration change/dysesthesia, overactivity/loquacity, drowsiness/yawning, dizziness/fatigue, irritability/mood change, food craving/thirstiness and dysphasia/osmophobia; this finding suggests that members of a pair may share a common origin or have similar functions. Some pairs, including photophobia/phonophobia, overactivity/loquacity, drowsiness/yawning, and irritability/mood change, have been identified in neuro-imaging and functional studies (41–44). Therefore, we infer the remaining pairs. The dysphasia and osmophobia pair has been proven to be more strongly associated with MA than MO, and we infer the cortex to be the source of both not only because of the short distance between them in the clustering results but also because of the neighboring territory of their function centers in the cortex. Thirstiness is considered to be involved in regulating homeostasis of body water and is a signal of water demand. If water is regarded as a special type of food, thirstiness could be a form of food craving.
It has been proposed that triggers, at least partially, may be another form of PS because: i) daily life triggers cannot reliably induce migraine (15); ii) experimental evidence is lacking for most reported triggers (1); and iii) some PS are closely related to specific triggers, such as photophobia with light and osmophobia with odor (14,18). Based on our results, we speculate that two subtypes of triggers may exist: MP triggers and SP triggers. MP triggers may activate brain dysfunction, are associated with multiple PS and cause patients to be vulnerable to migraine under the corresponding state (45,46). They might disturb brain function by altering the rate of energy production by the mitochondria, the membrane properties of the mitochondria, calcium load and transmitter excitability (47); therefore, these triggers might produce a similar trigger effect in other brain-dysfunction diseases, such as epilepsy and vertigo (48,49). In contrast, SP triggers affect only patients who are already in the prodromal phase. They induce a transition from the prodromal phase to the headache phase, allowing patients to experience headache attacks in a short time. The meanings of the association between SP triggers and their special PS are still unknown. We prefer to interpret them as two different forms of the same dysfunction. Typically, both natural light photophobia and the high-intensity sunlight trigger during the pre-headache phase reflect a dysfunction of light perception (either a deceased threshold of photoreception or increased photosensitivity), which, during the prodromal phase, can lead to PS (photophobia) being misidentified as a trigger (light). Our results enrich the evidence in support of point 3 and are supported by prior studies (2,18). If this speculation is correct, the true prevalence of photophobia is higher than previously estimated. For example, in our study, 259 migraineurs classified as trigger+/PS− would correctly be classified as photophobia PS+.
Our study recruited 105 child migraineurs, and 25% of these patients reported at least one PS; this percentage is lower than that reported in prior studies (24,26). Compared with adult migraineurs, child migraineurs seem to exhibit a slightly different PS spectrum (24–26). Among the child migraineurs, eliciting descriptions of PS is preferable to using structured questionnaires to reveal the pathogenesis of epileptic symptoms associated with migraine.
Limitations
This research may involve memory bias, as the PS are recorded from patients’ retrospective statements rather than prospective headache diary records. However, prospective records increase the possibility of exaggerating subtle changes when people feel unwell, especially when they have pain and pain-related unstable states such as anxiety (50). Only symptoms stably implanted in memory (due to constant repetition) are reliable for research. Furthermore, we did not list all of the PS reported in the structured questionnaire, such as facial autonomic symptoms experienced in experimentally induced migraine (17) and symptoms unique to children, especially episodic symptoms related to migraine (25,26). Instead, we provided a space on the questionnaire where the respondents could write any PS that they experienced that were not included in the list. Additionally, because the samples sizes of the triggers, such as sexual activity, coughing and physical exercise, were too small to analyze independently, they were grouped together into the category “activity”, which may have pooled SP triggers with MP triggers and increased bias. Finally, the hypothesis we proposed to explain the correlations between triggers and PS requires verification, which is the direction of our future research.
Conclusion
In the present study, we detailed the PS of 4812 migraineurs enrolled from 10 clinics in China. We demonstrated that the PS prevalence among Chinese migraineurs was 21.5%, and the most common PS were neck stiffness, dizziness, yawning and drowsiness. MA, trigger, depression, family history, coffee consumption, physical exercise and typical migraine features (mild to severe headache, accompanied by photophobia/phonophobia and aggravation by routine physical activity) were associated with more PS experiences. Furthermore, MA, trigger and depression were associated with experiencing more than one PS. The strongest co-occurrence of PS was observed for photophobia/phonophobia, followed by concentration change/dysesthesia, and loquacity/overactivity. The clustering analysis clustered the following PS into pairs: Photophobia/phonophobia, concentration change/dysesthesia, loquacity/overactivity, yawning/drowsiness, sensation of weak/dizziness, and mood change/irritability. Based on the correlations between PS and triggers, the triggers were categorized into the following two types: MP triggers and SP triggers. Among the SP triggers, light may be a form of photophobia in PS. Although this study has some shortcomings, it has the strengths of including the largest sample of patients enrolled in a PS study and data collection from multiple clinics.
Clinical implications
The prevalence of premonitory symptoms (PS) among migraineurs in China is 21.5%. The most common PS were neck stiffness, dizziness, yawning and drowsiness. Photophobia/phonophobia, concentration change/dysesthesia, loquacity/overactivity, yawning/drowsiness, fatigue/dizziness, and mood change/irritability appeared pairwise in the cluster analysis. There may be two types of trigger according to the analysis of the correlations between triggers and PS.
Supplemental Material
sj-pdf-1-cep-10.1177_0333102421997850 - Supplemental material for Premonitory symptoms in migraine from China: A multi-clinic study of 4821 patients
Supplemental material, sj-pdf-1-cep-10.1177_0333102421997850 for Premonitory symptoms in migraine from China: A multi-clinic study of 4821 patients by Xiaolin Wang, Ziming Yin, Yajun Lian, Yanmei Xu, Yajie Li, Jiale Liu, Qun Gu, Fanhong Yan, Zhaoli Ge, Yu Lian, Dongmei Hu, Sufen Chen, Rongfei Wang, Xiaoyan Chen, Jing Liu, Mingjie Zhang, Ye Ran, Ping Zhou, Jingyi Ma, Xudong Lv, Zhao Dong and Shengyuan Yu in Cephalalgia
Footnotes
Ethics approval and patient consent
The study was approved by the Ethics Committee of the Chinese PLA General Hospital. The protocol of the study complied with China’s regulations and guidelines for Good Clinical Practice. Oral informed consent was obtained from the patients before inclusion in the study according to the World Medical Association’s Declaration of Helsinki. The author has received permission to cite any personal communications and there is no figure nor video from any recognizable patient in the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by grants from the National Natural Science Foundation of China (81771200, 81671077), and Beijing Natural Science Foundation Essential Research Project (Z170002).
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
