Abstract
Background
The association between migraine and Parkinson’s disease (PD) remains controversial. The purpose of the present population-based, propensity score-matched follow-up study was to investigate whether migraineurs are at a higher risk of developing PD.
Methods
A total of 41,019 subjects aged between 40 and 90 years with at least two ambulatory visits with a diagnosis of migraine in 2001 were enrolled in the migraine group. A logistic regression model that included age, sex, pre-existing comorbidities and socioeconomic status as covariates was used to compute the propensity score. The non-migraine group consisted of 41,019 propensity score-matched, randomly sampled subjects without migraine. The PD-free survival rate were estimated using the Kaplan–Meier method. Stratified Cox proportional hazard regression was used to estimate the effect of migraine on the risk of developing PD.
Results
During follow-up, 148 subjects in the migraine group and 101 in the non-migraine group developed PD. Compared to the non-migraine group, the hazard ratio of PD for the migraine group was 1.64 (95% confidence interval: 1.25–2.14, p = 0.0004). The PD-free survival rate for the migraine group was significantly lower than that for the non-migraine group (p = 0.0041).
Conclusions
This study showed an increased risk of developing PD in patients with migraine.
Introduction
Migraine is a neurovascular disorder characterized by recurrent attacks of throbbing headache associated with autonomic symptoms (1). Previous studies have found that migraine is more prevalent in patients with certain basal ganglion disorders (2), such as Tourette’s syndrome (3), Sydenham’s chorea (4) and restless leg syndrome (5). However, previous studies exploring the association between migraine and Parkinson’s disease (PD) have generated inconsistent findings as, although a cohort study in Iceland reported that migraine is related to an increased risk of Parkinsonism (6), cross-sectional studies found that PD patients have a lower prevalence of migraine (7,8). Moreover, longitudinal evidence in a follow-up study for a temporal relationship between migraine and PD is still limited to the Icelandic study. We therefore performed this large-scale, population-based, propensity score-matched, longitudinal follow-up study in order to determine whether patients with migraine are at an increased risk of developing PD.
Materials and methods
Study subjects and design
This study used data obtained from the National Health Insurance (NHI) claim database from 2000 to 2003 in Taiwan. The NHI program is a single-payer healthcare system with a 97% coverage rate of the whole population in Taiwan at the end of 2003 (i.e. more than 21.9 million persons). We used a propensity score-matched cohort design to investigate the impact of migraine on the risk of developing PD. The study population, including a migraine group and a non-migraine group, were selected from Taiwanese residents in the NHI claim database in 2001. Since the imbalance in demographic and clinical characteristics may introduce confounding effects on the association between migraine and PD, we used propensity score matching to generate comparable cohorts of patients with and without migraine (9,10). All personal identification numbers in the data were encrypted by conversion into scrambled numbers before data processing to ensure individual information confidentiality and to comply with the regulations on personal privacy in Taiwan. As the data used in this study comprised de-identified secondary data released for research purposes and were analyzed anonymously, this study was exempt from full review by the National Taiwan University Hospital Research Ethics Committee and the need for informed consent was waived.
Subjects aged between 40 and 90 years who received a diagnosis of migraine (International Classification of Disease, 9th Revision, Clinical Modification [ICD-9-CM] code 346) in at least two ambulatory medical care visits between January 1, 2001, and December 31, 2001, were eligible for inclusion in the migraine group (n = 41,898). The first ambulatory visit with a diagnosis of migraine was assigned as the index visit. Subjects who had a previous diagnosis of PD (ICD-9-CM code 332.0) or secondary Parkinsonism (ICD-9-CM code 332.1) between January 1, 2000, and the index visit (n = 606) were excluded, leading to a total of 41,292 subjects being included in the migraine group at this stage. The pre-existing comorbidities included as covariates in the propensity score-matched analysis were diabetes (ICD-9-CM code 250), hypertension (ICD-9-CM codes 401–405), dyslipidemia (ICD-9-CM code 272), stroke (ICD-9-CM codes 430–438), coronary heart disease (ICD-9-CM codes 410–414) and traumatic brain injury (ICD-9-CM codes 801–804, 850–854). The information on these comorbidities were retrieved from ambulatory medical care and inpatient records of the NHI database in the year before the index visit. The ascertainment of the diagnosis of these medical comorbidities was defined as ≥1 hospital discharge or ≥2 ambulatory visits with a relevant diagnosis code. As socioeconomic status factors, such as geographical region, urbanization level and monthly income, have been suggested to influence the risk of PD (11,12), we included these factors as variables in the propensity score matching. The geographical location of residence of each subject was categorized as Northern, Central, Eastern or Southern Taiwan based on the information obtained from the population household registry. The levels of urbanization were initially classified into seven levels according to the publication of the Taiwan National Health Research Institute (13), with level 1 denoting as the “most urbanized” and level 7 as the “least urbanized” communities. However, levels 5, 6 and 7 were then merged into a single group labeled as level 5 because the numbers of subjects in these levels were relatively small. The level of monthly income of each subject was determined by the insured payroll-related amount and grouped into four levels: NT$0, NT$1–NT$15,840, NT$15,841–NT$25,000 and ≥NT$25,001 (NT$ indicates new Taiwan dollar). NT$15,840, the government-stipulated minimum monthly wage for full-time employees in Taiwan, was selected as the first cutoff point of income. A total of 273 subjects were excluded from the 41,292 subjects in the initial migraine group due to missing information in the household registry, leaving 41,019 subjects in the final migraine group.
The non-migraine group consisted of subjects without a diagnosis of migraine in the 2001 NHI claim database. We assigned the first ambulatory medical care visit during 2001 as the index visit. The exclusion criteria for the non-migraine group were: a previous diagnosis of migraine (ICD-9-CM code 346); and a previous diagnosis of PD (ICD-9-CM code 332.0) or secondary Parkinsonism (ICD-9-CM code 332.1) between January 1, 2000, and the index visit. The information on pre-existing comorbidities and socioeconomic status was obtained as described above. Considering the very large number of subjects in the NHI database, we used a two-stage propensity-score matching method (14). For each subject in the migraine group, we first randomly sampled 30 age- and sex-matched non-migraine subjects who met the above criteria, giving a total of 1,230,570 subjects in the non-migraine group at this stage. In the second stage, we used a logistic regression model including age, sex, pre-existing comorbidities and socioeconomic status as covariates to predict the probability (i.e. propensity score) of migraine. An 8-to-1 digit greedy matching algorithm (9) was then used to select a unique matched control from the non-migraine subjects for each migraine patient according to the propensity score. A total of 41,019 subjects were therefore included in the final propensity score-matched non-migraine group.
Outcome
For each subject in the propensity score-matched migraine and non-migraine groups, all the ambulatory medical care records and inpatient records from their index visit until the end of 2003 were retrieved from the NHI database. For subjects who died during follow-up, the mortality data were obtained from the national mortality registry. The primary endpoint was defined as the date of the first diagnosis of PD in the follow-up period. The case ascertainment for the diagnosis of PD required ≥1 hospital discharge or ≥2 ambulatory medical care visits with a principal diagnosis of PD (ICD-9-CM code 332.0). All subjects were followed from the index visit to the first occurrence of PD, death or end of follow-up (whichever came first).
Statistical analysis
The differences in demographic characteristics and comorbid medical disorders between the migraine and non-migraine groups were examined using the Chi-square test and Student’s t test. The method of standardized differences was used to assess the balance of covariates between the two groups before and after propensity score matching; this method was preferred to hypothesis testing methods since the standardized difference does not rely on sample size (15). An absolute standardized difference of 0 for a covariate indicates no between-group imbalance for that covariate, and values <0.1 indicate an inconsequential imbalance (16). The PD-free survival curves for these two groups were generated using the Kaplan–Meier method, and the difference in survival between the two groups was examined using the log-rank test. Stratified Cox proportional hazard regression with patients matched by propensity score was used to estimate the effect of migraine on the occurrence of PD. An alpha level of 0.05 was regarded as statistically significant for all analyses. The analyses were carried out using SAS 9.4 software (SAS Institute, Cary, NC).
Results
Demographic characteristics and comorbid medical disorders for the migraine and non-migraine groups before propensity score matching.

Data are expressed as n (%) or mean ± standard deviation.
US$1 = NT$34 in 2001.
Demographic characteristics and comorbid medical disorders for the migraine and non-migraine groups after propensity score matching.
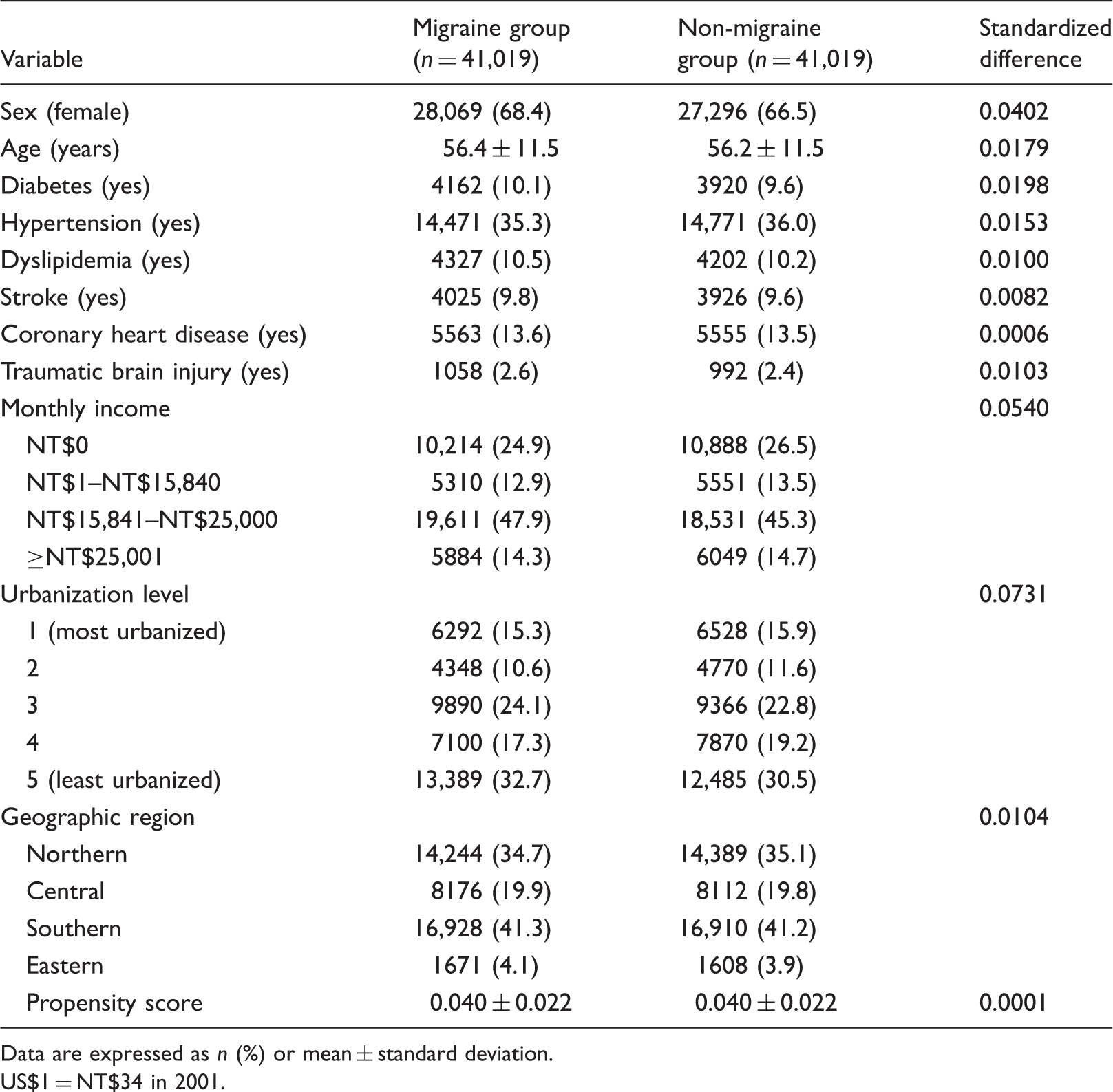
Data are expressed as n (%) or mean ± standard deviation.
US$1 = NT$34 in 2001.
The median follow-up time was 31.5 months (interquartile range: 7.6 months). The number of PD events and the hazard ratios (HR) of PD for the two propensity score-matched groups are presented in Table 3. Of the 41,019 patients with migraine, 148 developed PD during 102,645.8 person-years of follow-up, giving an incidence of 1.44 (95% confidence interval [CI]: 1.22–1.69) per 1000 person-years. Of the 41,019 subjects in the non-migraine group, 101 developed PD during 101,259.0 person-years of follow-up, giving an incidence of 1.00 (95% CI: 0.81–1.21) per 1000 person-years. The HR of PD for patients with migraine was 1.64 (95% CI: 1.25–2.14, p = 0.0004) compared to the non-migraine group. Comparison of the PD-free survival curves shows that the PD-free survival rate for the migraine group was significantly lower than that for the non-migraine group (p = 0.0041; Figure 1).
Parkinson’s disease-free survival rates for the migraine group (dotted line) and the non-migraine group (solid line). Number of Parkinson’s disease events and the hazard ratio of Parkinson’s disease for the matched migraine and non-migraine groups. p = 0.0004.

Discussion
In the present study, we found that migraineurs had a higher risk of developing PD than non-migraineurs (HR = 1.64, 95% CI: 1.25–2.14, p = 0.0004). This study is the first large-scale longitudinal follow-up study showing a higher risk of PD in migraineurs in an Asian population. The mechanism underlying the positive association between migraine and PD remains unclear. We offer the following possible explanations.
It has been suggested that migraineurs have impaired serotonin metabolism in the brain that may trigger cranial vasoconstriction and sensitization of neuronal elements (17). Serotonin depletion has been associated with increased cortical neuron sensitivity, vascular responses induced by cortical spreading depression in an animal model of migraine (18). In addition, it has been suggested that migraineurs have chronic dopaminergic hypofunction and dopamine receptor hypersensitivity (19), which may be related to prodromal symptoms (e.g. yawing and somnolence) in migraineurs (19). While progressive dopaminergic denervation in the brain is considered to be the fundamental pathological feature of PD, many studies have demonstrated a lower-brain serotoninergic activity in PD patients (20–22) and an association between serotonergic dysfunction and the development of motor and non-motor symptoms in PD (23,24). Moreover, serotonergic neuron degeneration in the raphe nuclei has been suggested to occur prior to dopaminergic neuron degeneration in patients with PD (23,25). We therefore hypothesize that the link between migraine and PD may be mediated through a shared pathophysiology mechanism, namely dysfunction of the serotonergic and dopaminergic system. Further studies are needed in order to investigate whether the risk of PD is higher in migraineurs with more pronounced prodromal symptoms.
Second, neuroimaging studies have shown that migraineurs have increased iron accumulation in deep-brain structures (26,27). Iron accumulation in the brain may induce increased oxidative stress, which has been linked to a number of neurodegenerative disorders (28,29). Dusek et al. showed that increased brain iron levels are related to some degenerative movement disorders, such as PD, Huntington’s disease, multiple system atrophy and progressive supranuclear palsy (30). In addition, increased iron accumulation in the substantia nigra in PD has been observed in post-mortem studies (31,32). Thus, although it remains unclear whether iron accumulation in the brain has a pathogenic role in migraine and PD, the association between migraine and PD found in our study may be explained, at least in part, by dysfunctional iron metabolism in the brain. As increased iron accumulation in the brain has been associated with a long migraine history (26,27), future studies are required to evaluate whether the risk of PD is higher in migraineurs with a longer history.
The present large population-based cohort study showed a temporal association between migraine and PD. Such a temporal relationship is important for establishing a causal connection. However, this study is subject to the following limitations. First, there may be concern about the diagnostic accuracy in this study, since the diagnosis of migraine, PD and medical comorbidities in our study was completely determined by ICD codes from the NHI claim database. However, to improve the diagnostic validity and quality of care, the Bureau of the NHI has organized different audit committees that randomly sample the claim data and review the medical records regularly. Accordingly, the NHI claim database is an established research database and has been widely applied in a variety of biomedical research fields (33). In addition, we used case ascertainment algorithms that required at least two ambulatory medical care visits with a diagnosis code of migraine, and such case definitions may be expected to provide adequate diagnostic accuracy. Nevertheless, migraineurs with only one medical care visit would not be captured by this case definition. This would reduce the sensitivity in the diagnosis of migraine and may lead to an attenuation of the results. Similarly, the comorbidities may also be under-ascertained, since they were identified based on medical records. It is possible that patients with mild comorbidities (e.g. mild head injury) did not seek medical consultation and were not well captured in the NHI database.
Second, since both migraine and PD are neurological disorders, ascertainment bias may occur, as patients receiving medical care for migraine may be more likely to receive a diagnosis of PD. We therefore performed a sensitivity analysis using spinocerebellar disease (ICD-9-CM code 334) as a control neurological disorder. During the follow-up period, the HR of having a diagnosis of spinocerebellar disease for the migraine group was 0.90 (95% CI: 0.35–2.33, p = 0.8256) compared to the non-migraine group. The lack of positive association between spinocerebellar disease and migraine suggests that the increased risk of PD in migraineurs is not likely to be explained by ascertainment bias.
Third, information pertaining to migraine subtypes (migraine with or without aura) was incomplete, because most of the diagnostic codes of migraine were encoded using only the three-digit ICD-9-CM category number (i.e. code 346) without the subcategory number in the NHI claim database.
Fourth, due to the inherent limitations of the NHI database, information on lifestyle factors, such as smoking, alcohol intake and obesity, was unavailable, which may influence the interpretation of our findings; we therefore matched the migraine and non-migraine groups for socioeconomic, geographic and clinical characteristics in order to generate comparable cohorts. However, although propensity score matching can control for various baseline characteristics that may influence the link between migraine and PD, this matching procedure could potentially lead to over-adjusting and attenuation of the HR estimates for the effects of migraine on the risk of PD. For example, if the matching variable is a mediator in the causal pathway between migraine and PD, matching would lead to attenuation of the HR estimates.
Fifth, the present study recruited subjects aged between 40 and 90 years who received a diagnosis of migraine in 2001. Since most people experience their first migraine during adolescence, it is reasonable to assume that migraineurs aged between 40 and 90 years reflect prevalent migraine. Moreover, since the present study used NHI database data from 2000 to 2003, the median follow-up time was only 32 months, and therefore the long-term effects of migraine on the risk of developing PD cannot be evaluated. In addition, the status of PD diagnosis before 2000 was not available in our study, which may also affect the interpretation of our findings. Finally, it is uncertain whether our findings can be generalized to other ethnic groups, as most residents in Taiwan are of Chinese ethnicity.
In conclusion, this population-based, propensity score-matched, longitudinal follow-up study shows that Asian migraineurs have an increased risk of developing PD. Further studies are needed in order to explore the underlying mechanism.
Clinical implications
This population-based, propensity score-matched, longitudinal follow-up study investigated the risk of Parkinson’s disease (PD) in patients with migraine. This study shows that Asian migraineurs were at a higher risk of developing PD.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: this work was supported by the Department of Health, Executive Yuan, Republic of China (DOH93-TD-M-113-030, DOH94-TD-M-113-004 and DOH95-TD-M-113-002).
