Abstract
Background
The present study aimed to evaluate the risk and impact of fibromyalgia in relation to headache frequency in migraine patients.
Methods
This cross-sectional study involved migraine patients from a regional hospital and a tertiary medical center. Diagnoses of migraine and fibromyalgia were made according to the International Classification of Headache Disorders, 3rd edition, and the modified 2016 American College of Rheumatology diagnostic criteria, respectively. Clinical data, including Fibromyalgia Symptoms (FS) scale and revised Fibromyalgia Impact Questionnaire (FIQR), were collected systematically by questionnaires-based interviews. Patients were categorized based on monthly headache day (MHD) cut-offs derived from decision tree analysis based on the chi-squared automatic interaction detection algorithm.
Results
The study involved 2082 migraine patients (1619 female/463 male, mean ± SD age 39.3 ± 12.0 years), including 132 with fibromyalgia (118 female/14 male, mean ± SD age 44.1 ± 12.7 years) (6.3%). Patients were divided into three groups: ≤9 MHDs (n = 924), 10–20 MHDs (n = 745) and ≥21 MHDs (n = 413). The percentage of fibromyalgia increased with headache frequency (p < 0.001). When compared with patients with ≤9 MHDs (2.8%), those with 10–20 MHDs (6.2%) (odds ratio (OR) = 1.90, 95% confidence interval (CI) = 1.11–3.23, p = 0.019) and ≥21 MHDs (14.5%) (OR = 3.68, 95% CI = 2.08–6.49, p < 0.001) were more likely to have fibromyalgia. Patients with more frequent headaches had higher FS and FIQR scores (all p < 0.001 between MHD categories).
Conclusions
There was an independent dose–response association between headache frequency and odds, severity, and impact of fibromyalgia in migraine patients. For migraine patients with a higher headache frequency, the potential risk of comorbid fibromyalgia should not be overlooked given its association with more severe clinical manifestations and greater disability.
This is a visual representation of the abstract.
Introduction
Fibromyalgia is characterized by chronic widespread pain, unrefreshing sleep, physical exhaustion and cognitive impairment. The global prevalence of fibromyalgia ranges from 0.4 to 8.8%, with an average of approximately 2.7%. (1,2) Indeed, it is the second most common rheumatic disorder (3) and the third most common musculoskeletal condition. (1) Fibromyalgia could be associated with significant disability, which could lead to substantial impacts on the quality of life of patients, as well as tremendous socioeconomic burdens. (4) Timely diagnosis is important for early management, which may improve the outcome of such patients. However, fibromyalgia remains under-recognized and under-diagnosed. (5) Identification of patients at risk should be a critical step to optimize the clinical management.
Migraine is the second most prevalent neurological disorder globally and ranks among the top three leading causes of disease-related disability in most countries. (6) An association between migraine and fibromyalgia has been reported in the literature: migraine is one of the commonly reported comorbidities of fibromyalgia and vice versa. (7,8) Migraine is seen in approximately 14–15% of the general population across different regions of the world. (9,10) However, the percentages of comorbid fibromyalgia in migraine and comorbid migraine in fibromyalgia are much higher than the population prevalence of each of the conditions alone, (11–14) which suggest overlaps in pathophysiology. Because migraine shares several risk factors with fibromyalgia, such as depression, sleep problem, stress, obesity, etc., (3,15,16) it could be hypothesized that the association between migraine and fibromyalgia would be stronger in patients with higher headache frequencies.
The primary objective of the present study was to evaluate the risk of fibromyalgia, as well as its severity and impact of symptoms of fibromyalgia, in relation to headache frequency in migraine patients.
Methods
Patients
This cross-sectional study involved retrospective analysis of a prospectively collected cohort of consecutive migraine patients recruited at their first visit to the headache clinics of a regional hospital (Taipei Municipal Gan-Dau Hospital) and a tertiary medical center (Taipei Veterans General Hospital). Patients were asked to complete a questionnaire as part of the routine evaluation for headache patients at their first visit, which was followed by evaluation by headache specialists. The diagnosis of migraine was based on the criteria of the International Classification of Headache Disorders, 3rd edition (ICHD-3) (17) and the diagnosis of fibromyalgia was made according to the modified 2016 American College of Rheumatology (ACR) diagnostic criteria (see supplementary material, Table S1). (18) Patients were included if they were (a) aged between 20 and 80 years, (b) fulfilled the ICHD-3 criteria for migraine with aura, or migraine without aura and (c) had medical records available for analysis. The exclusion criteria included (a) headaches with the onset within one month, (b) coexistence of other primary or secondary headache disorder(s) and (c) incomplete questionnaires that precluded further analysis. The study protocol was approved by the Institutional Review Boards of Taipei Municipal Gan-Dau Hospital (No. 1130002906) and Taipei Veterans General Hospital (No. 2024-06-005CC).
Questionnaire
The questionnaire was designed to collect clinical profiles, headache characteristics, medication use, psychological disturbances and sleep quality. The Migraine Disability Assessment (MIDAS) scale was used to evaluate headache-related disability, and patients with a MIDAS score of ≥11 were categorized as having moderate-to-severe disability. (19) The Hospital Anxiety and Depression Scale (HADS) was used to screen for symptoms of anxiety and depression. Depression was defined as a depression score (HADS-D) of ≥11, whereas anxiety was defined as an anxiety score (HADS-A) of ≥11. (20) Sleep quality was assessed using the Pittsburgh Sleep Quality Index (PSQI), with a PSQI score of >5 implying poor sleep quality. (21) Furthermore, the presence of widespread pain and non-pain symptoms of fibromyalgia was assessed using the widespread pain index (WPI) and symptom severity (SS) score, respectively. (22,23) The severity of manifestations of fibromyalgia was evaluated by the Fibromyalgia Symptom (FS) scale, comprising the sum of WPI and SS scores, (22) and the impact of presentations of fibromyalgia was measured using the revised Fibromyalgia Impact Questionnaire (FIQR), which consists of three health domains: physical function, overall impact and symptoms. (24) An FIQR score ≤30, 31–45 and ≥46 indicates remission, mild severity and moderate-to-severe severity, respectively. (25) All migraine patients were asked to complete the WPI, SS and FIQR regardless of whether a diagnosis of fibromyalgia could be fulfilled. The responses to the questions in the questionnaire were verified at subsequent face-to-face interviews.
Statistical analysis
Descriptive data are expressed as the mean ± SD or n (%). Numerical data, such as age, monthly headache days (MHDs), number of days with acute medication use, scores of neuropsychological instruments, etc., between groups were compared using Student's t-test. A chi-squared test was used to evaluate between-group differences for categorical variables. Patients were grouped based on MHDs cut-offs derived from decision tree analysis based on the chi-squared automatic interaction detection (CHAID) algorithm, (26) using fibromyalgia as the dependent variable, and MHD, age, sex, education level, marital status, employment status, aura status, average headache severity, medication overuse, moderate-to-severe disability, depression, anxiety and poor sleep as the independent variables. The FS and FIQR scores were compared among these three groups of patients by one-way analysis of variance, followed by post-hoc analysis with Fisher's least significant difference method. Categorical or ordinal variables among headache frequency categories were compared using the chi-squared test and the chi-squared test for trend. Logistic regression modeling (enter) was carried out to estimate the odds ratios (ORs), as well the 95% confidence intervals (CIs), for the risk of fibromyalgia in relation to headache frequency categories, which were further adjusted for age, sex, education level, marital status, employment status, aura status, average headache severity, the presence of medication overuse, at least moderate disability, depression, anxiety and poor sleep quality. All statistical analyses were carried out using SPSS, version 24.0 (IBM Corp., Armonk, NY, USA). p < 0.05 (two-tailed) was considered statistically significant.
Results
Study participants
During the study period, 4051 headache new patients were screened. After excluding patients with headache onset within one month (n = 199) or other headache disorders (n = 1156), as well as those with incomplete questionnaires (n = 614) (Figure 1), 2082 migraine patients (1619 female/463 mala, mean ± SD age 39.3 ± 12.0 years) were included in the analysis.

Patient recruitment in the present study.
Clinical characteristics of migraine patients with comorbid fibromyalgia
Among the entire study population, 132 patients were diagnosed as having fibromyalgia (118 female/14 male, mean ± SD age 44.1 ± 12.7 years) (6.3%). Comorbid fibromyalgia in migraine was associated with an older age (mean ± SD age 44.1 ± 12.7 years vs. 39.0 ± 11.9 years, p < 0.001), a higher proportion of women (89.4% vs. 77.0%, p = 0.001), lower percentages of having a bachelor's degree or higher (60.6% vs. 75.1%, p < 0.001) or being employed (62.9% vs. 73.3%, p = 0.013), higher headache frequency (19.2 ± 10.0 vs. 12.4 ± 9.4 MHDs, p < 0.001), greater average headache severity (6.9 ± 2.1 vs. 6.1 ± 2.0, p < 0.001) and more frequent acute medication use (8.7 ± 9.4 days/month vs. 6.8 ± 7.8 days/month, p = 0.029) (Table 1). Furthermore, patients with fibromyalgia had greater headache-related disability (MIDAS: 63.5 ± 70.4 vs. 30.1 ± 43.3, p < 0.001), more severe symptoms of anxiety (HADS-A: 11.5 ± 4.0 vs. 8.4 ± 4.1, p < 0.001) or depression (HADS-D: 9.7 ± 4.5 vs. 6.4 ± 4.1, p < 0.001) and poorer sleep quality (PSQI: 13.6 ± 3.9 vs. 9.6 ± 4.2, p < 0.001). In addition, they had higher FS (17.9 ± 3.8 vs. 7.4 ± 3.5, p < 0.001) and FIQR (44.6 ± 20.3 vs. 23.9 ± 17.3, p < 0.001) scores. On the other hand, there were no between-group differences in body mass index (BMI), marital status and aura status.
Comparisons between migraine patients with and without fibromyalgia.
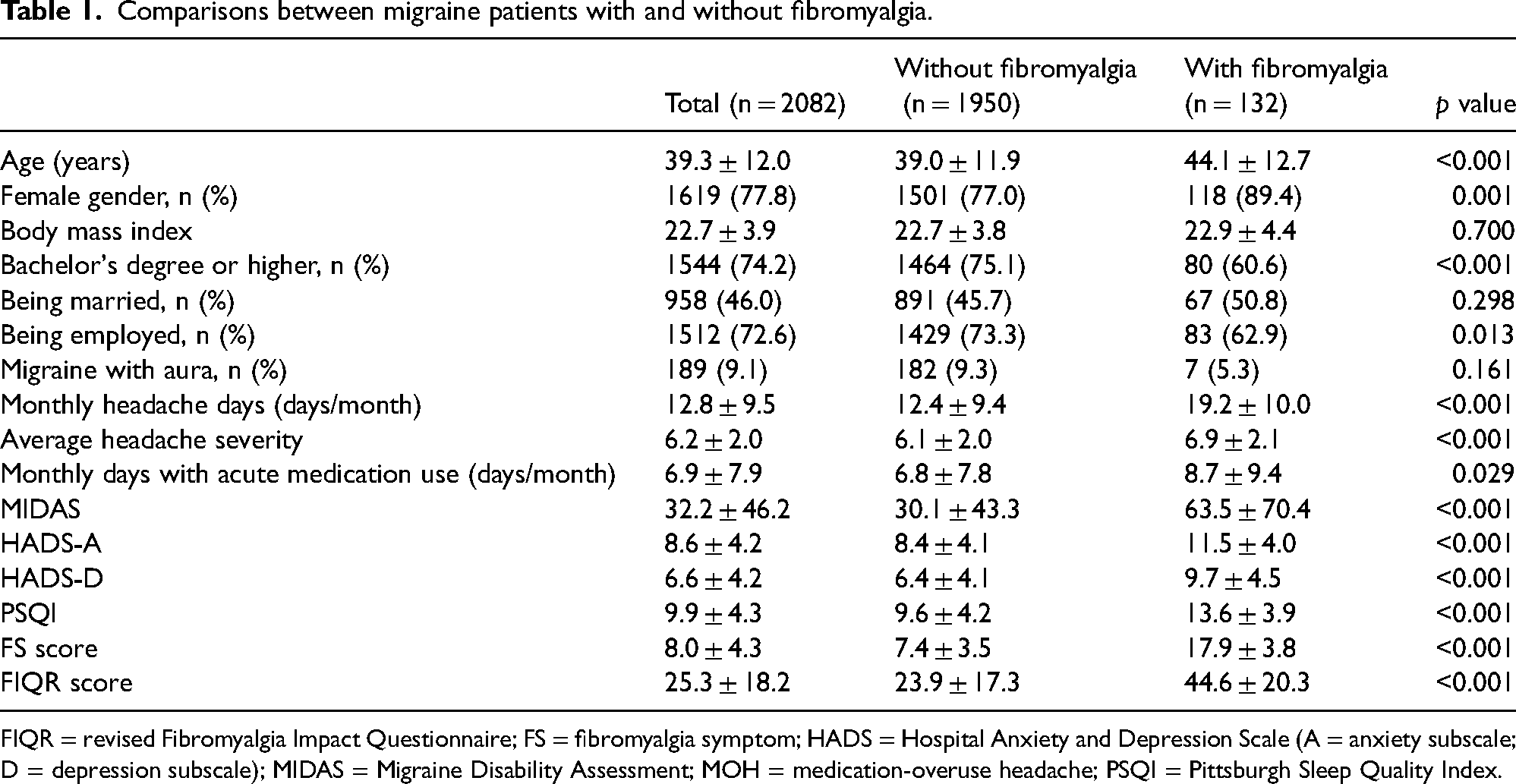
FIQR = revised Fibromyalgia Impact Questionnaire; FS = fibromyalgia symptom; HADS = Hospital Anxiety and Depression Scale (A = anxiety subscale; D = depression subscale); MIDAS = Migraine Disability Assessment; MOH = medication-overuse headache; PSQI = Pittsburgh Sleep Quality Index.
Association between headache frequency and risk of fibromyalgia
A CHAID decision tree was generated with 13 terminal nodes and three layers deep (Figure 2). Headache frequency was the most decisive variable at the time of classification, and patients were thereby divided into three categories: ≤9 MHDs (n = 924), 10–20 MHDs (n = 745) and ≥21 MHDs (n = 413).

Tree created by the CHAID (i.e. chi-squared automatic interaction detection) model. HADS = Hospital Anxiety and Depression Scale (A = anxiety subscale); MHD = monthly headache day; MIDAS = Migraine Disability Assessment; PSQI = Pittsburgh Sleep Quality Index.
The percentages of fibromyalgia were 2.8, 6.2 and 14.5% in patients with ≤9 MHDs, 10–20 MHDs and ≥21 MHDs, respectively, and there was a positive correlation (p < 0.001, chi-squared test for trend) (Figure 3A). When compared with patients with ≤9 MHDs, those with 10–20 MHDs (OR = 2.27, 95% CI = 1.39–3.71, p = 0.010) and ≥21 MHDs (OR = 5.87, 95% CI = 3.65–9.45], p < 0.001) were more likely to have fibromyalgia, and there was no sex difference (p = 0.630 for sex-by-MHD-category interaction). There remained a gradient in the odds for having fibromyalgia for patients with 10–20 MHDs (OR = 1.90, 95% CI = 1.11–3.23, p = 0.019) and ≥21 MHDs (OR = 3.68, 95% CI = 2.08–6.49, p < 0.001) compared to patients with ≤9 MHDs, after controlling for age, sex, education level, marital status, employment status, aura status, average headache severity, medication overuse, moderate-to-severe disability, depression, anxiety and poor sleep (Figure 3B).

Risk of fibromyalgia in relation to headache frequency categories. Patients with more frequent headache attacks were more likely to have fibromyalgia (p < 0.001) (chi-squared test for trend) (A). Patients in the higher headache frequency categories had greater odds for having fibromyalgia (B). *Adjusted for age, sex, education level, marital status, employment status, aura status, average headache severity, medication overuse, moderate-to-severe disability, depression, anxiety and poor sleep. CI = confidence interval; OR = odds ratio.
Severity and impact of fibromyalgia in relation to headache frequency
Patients with more frequent headaches had higher FS scores (≤9 MHDs: 6.9 ± 3.7; 9–20 MHDs: 8.4 ± 4.3; ≥21 MHDs: 10.0 ± 4.9) (all p < 0.001 between groups) (Figure 4A) and were more likely to have moderate-to-severe impact from clinical manifestations of fibromyalgia as measured by the FIQR (≤9 MHDs: 7.9%; 9–20 MHDs: 13.2%; ≥21 MHDs: 25.7%) (p < 0.001, chi-squared test for trend) (Figure 4B). Patients in the higher headache frequency categories had higher FIQR scores (all p < 0.001) (Table 2). When the three domains of FIQR were analyzed separately, there were positive correlations between MHD categories and the scores in the overall impact domain (all p < 0.001 between groups), symptoms domain (all p < 0.001 between groups) and physical functional domain (p = 0.025 for ≤9 MHD vs. 10–20 MHD, and otherwise p < 0.001 between groups) (Table 2).

Severity and impact of fibromyalgia symptoms in relation to headache frequency categories. Patients in the higher headache frequency categories had higher FS scores (all p < 0.001 between groups, except p = 0.008 for HFEM vs. LFCM, ANOVA followed by post-hoc analysis using the least significant difference method) (A) and were more likely to have moderate-to-severe impact from symptoms of fibromyalgia (p < 0.001, chi-squared test) (B). FIQR = revised Fibromyalgia Impact Questionnaire; FS = Fibromyalgia Symptoms scale.
Comparisons between migraine patients with and without fibromyalgia.

Analysis of variance.
all p < 0.001 between groups.
all p < 0.001 between groups, except p = 0.025 for ≤ 9 MHD vs. 10–20 monthly headache days.
FIQR = revised Fibromyalgia Impact Questionnaire; MHD = monthly headache days.
Discussion
In the present study, migraine patients with a higher headache frequency had a greater risk of fibromyalgia. Furthermore, headache frequencies were positively correlated with the severity and impact of fibromyalgia. In the clinical evaluation of migraine, clinicians should be alerted to the potential risk of comorbid fibromyalgia, especially in patients with ≥10 MHDs. Considering the association between comorbid fibromyalgia and more severe manifestations and clinical impact of migraine, the importance of early identification of patients at risk could not be overemphasized. However, whether such patients could benefit from a more aggressive treatment approach remains to be determined.
One of the major strengths of the present study is its large sample size, with more than 2000 migraine patients being included in the analysis. In comparison, the majority of clinic- or hospital-based studies focusing on fibromyalgia in migraine in the literature recruited less than 150 patients. (13,14,27–30) A large sample size could give a more accurate and comprehensive description of the association. Furthermore, patients were recruited from both a regional hospital and a tertiary medical center, which could help reduce selection bias. In addition, the data were collected systematically by questionnaires and were verified at face-to-face interviews by neurologists, and therefore should be of high quality and reliability. In comparison, cases in certain population-based studies were identified using diagnosis codes, (31,32) and coding inaccuracies could be an important concern. Furthermore, only demographics and clinical diagnoses were included in the analysis of those studies, whereas many of the important clinical manifestations were lacking.
In the present cohort of migraine patients, the headache frequency paralleled the risk of fibromyalgia, and there was no sex difference. When compared with patients with ≤9 MHDs, those with 10–20 MHDs and ≥21 MHDs had an approximately two-fold and more than 3.5-fold increase, respectively, with respect to the probability of having fibromyalgia. In the literature, most studies focused on the percentage of fibromyalgia within just one of the following diagnostic entities: episodic migraine (EM), chronic migraine (CM) or transformed migraine (TM). (13,14,29,30) There was inconsistent evidence suggesting that fibromyalgia was more common in CM than in EM. (11,27,33) However, only the percentages of fibromyalgia, based on the 1990 or 2010/2011 ACR criteria, (22,23,34) were reported in those studies, and it is uncertain whether the association could also be attributed to psychiatric comorbidities or other risk factors shared by fibromyalgia and CM. (2,15,35–37) In the present study, there was an association between headache frequency and the risk of fibromyalgia, which remained significant after controlling for potential confounders. More importantly, the present study extended the findings beyond the dichotomy of CM and EM, and identified a dose–response relationship. Indeed, the risk was increased for patients with ≥10 MHDs, among whom a certain proportion belonged to the category of high-frequency EM. Recently, there were discussions on whether the 15-day threshold for CM is appropriate because it may not be able to capture all of the patients with high disease burdens and treatment needs. (38) Our findings might echo these arguments, because the increase in the risk of fibromyalgia was not limited to patients with ≥15 MHDs. Based on our findings, a headache frequency of ≥10 MHDs in migraine patients should prompt the treating physician to actively question the presence of clinical manifestations of fibromyalgia, rather than to just passively wait for self-reports by the patients.
Our findings deserve special attention because comorbid fibromyalgia in migraine was associated with a more severe phenotype and greater functional impact. When compared with those without fibromyalgia, migraine patients with a concomitant diagnosis of fibromyalgia have higher frequency or severity of headache, greater impact or disability, more symptoms of depression or anxiety, poorer quality of life and even increased suicidal risks. (13,27–29,31,33,39,40) Indeed, certain chronic pain conditions, including migraine, fibromyalgia, temporomandibular disorder (TMD), etc., could coexist or coaggregate, and are referred to as chronic overlapping pain conditions (COPCs). (41) COPCs are characterized by pain amplification and psychosocial vulnerability. (41) It was reported that CM patients with pain in more body regions had poorer physical and psychological functions and greater health-care utilization compared to those whose pain was limited to the head region. (42) Interestingly, it was also shown that widespread pain was a predictor of poor functional outcome following treatment for painful TMD. (43) It would be of clinical significance and importance to determine whether comorbid fibromyalgia in migraine could be associated with a poorer treatment response, and whether more aggressive treatment is needed for such patients.
The positive association between headache frequency of migraine and the risk and impact of fibromyalgia identified in the present study could be multifactorial. In addition to overlapping risk factors and clinical phenomenology, pathophysiological mechanisms shared by both conditions could play important roles, especially central sensitization and impaired descending pain modulation. (1,3,15,44) It was reported that migraine patients had lowered pain thresholds and allodynia in cephalic and extracephalic regions, which could be related to sensitization of the second- and third-order neurons of the trigemino-nociceptive pathway, and this phenomenon is more pronounced in patients with more frequent attacks. (15,45–47) Patients with fibromyalgia could have many of the features associated with central sensitization, such as hyperalgesia, allodynia, temporal summation, etc., as well as activation of the pain matrix and loss of gray matter volume in structures involved in pain processing. (1,48–51) Furthermore, it was also found that there were alterations in the descending modulatory mechanisms in migraine and fibromyalgia, indicating an imbalance between the facilitatory and inhibitory mechanisms. (52–54) Other potential mechanisms shared by both conditions include neuroinflammation, increased oxidative stress, etc. (55) On the other hand, although obesity is a commonly reported risk factor for migraine and fibromyalgia, (56,57) the BMIs were actually similar between patients with and without fibromyalgia in the present study. In particular, the average BMI in the present cohort was only 22.7 ± 3.9 kg/m2. It remains to be determined whether this could be attributed to racial or ethnic differences. More studies are needed to understand the underlying pathophysiology, which could pave the way to the development of novel treatments.
The present study has some limitations. First, there could be concerns about generalizability. However, the study involved patients from a regional hospital and a tertiary medical center, and the study population could reflect a wide range of migraine patients. Second, symptoms of anxiety and depression, as well as sleep quality, were evaluated using neuropsychological instruments, rather than by formal psychiatric interviews. However, the neuropsychological instruments used in the present study have been well validated, and have been widely used in clinical studies of migraine and fibromyalgia. (19–22,24) Finally, as a cross-sectional study, only associations, rather than causal relationships, could be demonstrated. Further studies of a longitudinal design are needed to determine the nature of the identified association.
In conclusion, there was a dose–response relationship between headache frequency and risk of fibromyalgia in migraine patients, independent of potential confounders. Furthermore, there were similar trends for the severity and impact of clinical manifestations of fibromyalgia. For migraine patients with a higher headache frequency, the potential risk of comorbid fibromyalgia should be taken into consideration with respect to its association with a more severe phenotype and greater disability. However, further studies are needed to determine whether proper management of migraine could help alleviate the risk or disability of fibromyalgia and whether comorbid fibromyalgia could have an impact on the treatment outcome of migraine.
Headache frequencies were positively and independently correlated with the odds of comorbid fibromyalgia in patients with migraine. Migraine patients with more frequent headaches had greater severity and impact of clinical symptoms of fibromyalgia. Symptoms of fibromyalgia should be proactively screened for in migraine patients with a higher headache frequency.
Supplemental Material
sj-docx-1-cep-10.1177_03331024251317486 - Supplemental material for Association between headache frequency and risk for fibromyalgia in patients with migraine
Supplemental material, sj-docx-1-cep-10.1177_03331024251317486 for Association between headache frequency and risk for fibromyalgia in patients with migraine by Yen-Hui Liao, Yi-Shiang Tzeng, Shih-Pin Chen, Yu-Hsiang Ling, Wei-Ta Chen, Shuu-Jiun Wang and Yen-Feng Wang in Cephalalgia
Footnotes
Declaration of conflicting interests
WTC has received honoraria as a speaker from Allergan/AbbVie, Hava Bio-Pharma, Orient EuroPharma, Pfizer and Viatris. He has received research grants from the Taiwan National Science and Technology Council, and Taipei Veterans General Hospital. SJW has received personal fees as an advisor or speaker from Allergan/AbbVie, Orient EuroPharma, Pfizer and Percept; and has been the PI in trials sponsored by Allergan/AbbVie, Lundbeck, Novartis, Pfizer and Orient EuroPharma. He has received research grants from the Taiwan National Science and Technology Council and Taipei Veterans General Hospital. YFW has received personal fees as an advisor or a speaker from Allergan/AbbVie, Boehringer Ingelheim, Chugai, Daiichi-Sankyo, Eli Lilly, Hava Bio-Pharma, Lundbeck, Novartis, Orient EuroPharma, Pfizer, Sanofi, Teva, UCB and Viatris. He has received research grants from the Taiwan National Science and Technology Council, and Taipei Veterans General Hospital. YHLiao, YST, SPC and YHLing reported no relevant disclosure.
Ethical statement
The study protocol was approved by the Institutional Review Boards of Taipei Municipal Gan-Dau Hospital (No. 1130002906) and Taipei Veterans General Hospital (No. 2024-06-005CC).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supportive by Taiwan National Science and Technology Council [110-2321-B-010-005, 111-2321-B-A49-004, 111-2314-B-075 -086 -MY3, 111-2321-B-A49-011, and 112-2321-B-075-007 (to SJW) and 109-2314-B-075 -054, 110-2314-B-075 -041 -MY3, and 113-2314-B-075 -049 -MY3 (to YFW)], Taiwan Ministry of Health and Welfare [MOHW112-TDU-B-211-144001 (to SJW)], and Taipei Veterans General Hospital [V108C-092, V109C-096, V110C-111, V111C-161, V112C-078, V113C-123, and V112D67-003-MY3 (to YFW)]; this work was also supported by the Brain Research Center, National Yang Ming Chiao Tung University from The Featured Areas Research Center Program within the framework of the Higher Education Sprout Project by the Ministry of Education (MOE) in Taiwan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
