Abstract
Background
Previous studies have reported a lower migraine prevalence in Parkinson’s disease (PD) patients and improvements in migraine headaches after PD onset, but the clinical association of migraines with PD is unclear.
Methods
We analysed headache and migraine prevalence and clinical correlates in 436 PD patients (mean age, 69.3 ± 7.8 years) and 401 age- and sex-matched controls (mean age, 69.2 ± 8.6 years) in a case-controlled, multicentre study. Migraines were diagnosed by a questionnaire developed according to the International Classification of Headache Disorders, second edition. We evaluated changes in headache intensity, frequency and severity over several years around the onset of PD among PD patients with headaches or migraines, and over the past several years among control subjects with headaches or migraines.
Results
PD patients had lower lifetime (9.6% vs. 18.0%) and 1-year (6.7% vs. 11.0%) migraine prevalences than controls. However, lifetime (38.5% vs. 38.9%) and 1-year (26.1% vs. 26.2%) headache prevalence did not differ between PD patients and controls. After adjusting for gender, timing of the evaluation of headache changes, and recall period, PD patients with headaches or migraines exhibited a pronounced reduction in the intensity, frequency and overall severity of their headaches and migraines after the onset of PD compared with controls with headaches or migraines. PD patients with migraines exhibited a higher rate of depression and higher Pittsburgh Sleep Quality Index and PD sleep scale-2 scores than those without headaches.
Conclusion
While overall headache and migraine severity reduced after PD onset, the presence of migraines was associated with sleep disturbances and depression in PD patients.
Introduction
Parkinson’s disease (PD) is a movement disorder that typically affects people over the age of 50 years. Degeneration of dopaminergic neurons in the substantia nigra of the midbrain causes the characteristic PD motor features, such as bradykinesia, resting tremor and rigidity. In addition, PD patients frequently exhibit non-motor symptoms such as sleep disturbances, cognitive impairment, psychiatric problems, pain and sensory symptoms, which are thought to involve various non-dopaminergic neurons of brainstem nuclei even during the early disease course of PD (1).
Migraines are one of the most widespread disabling neurological disorders, which commonly occur between the ages of 20 and 50 years with a reduced prevalence in those aged over 50 years (2–4). The pathophysiology of migraines involves peripheral and central neurotransmitter systems and changes in various brain regions. Activation of the trigeminovascular system in the brainstem, cortical hyperexcitability, and the involvement of hypothalamic dopaminergic and orexinergic systems have been implicated in migraines (5–8).
Considering the shared involvement that has been reported among neurotransmitters (serotonin, noradrenalin and dopamine), the brainstem and hypothalamic regions in both migraines and PD, we sought to investigate the prevalence of headaches and migraines in PD patients, the temporal relations between headache severity and PD disease progression and the clinical characteristics of comorbid migraines in PD patients. A previous study reported that the frequency of current migraines was lower in PD patients with migraines than in control subjects with migraines, and that two-thirds of PD patients with migraines reported improvement or remission of migraines after PD onset (9). However, the relationship between migraines and PD, including the clinical characteristics that predict migraine co-morbidity in PD patients, has never been studied in a large sample of PD patients. Due to the progressive degeneration of brainstem nuclei in PD, which include the region that regulates pain transmission, we hypothesized that the prevalence of current migraines in PD patients is lower and that the severity of migraines reduces over several years compared with age- and sex-matched controls.
In this multicentre, case-control study, we investigated the prevalence of migraines and their association with the disease course and clinical features of PD patients.
Patients and methods
A multicentre study including eight university hospitals in the Kanto region of Japan was carried out to assess non-motor symptoms (NMS) of PD between September 2014 and April 2016. The Kanto region of Japan consists of seven prefectures including the capital Tokyo, referred to as the Greater Tokyo Area, which has an approximate population of 42.6 million, accounting for one third of the entire Japanese population according to the 2010 Population Census of Japan, Preliminary Counts of the Population and Households.
Patients
This study recruited 490 consecutive patients with PD (age, 69.4 ± 8.0 years; 225 men and 265 women) from outpatient clinics of the participating facilities. After excluding patients with dementia, defined as a Mini-Mental State Examination (MMSE) score lower than 24, and after accounting for missing data, 436 PD patients (age, 69.3 ± 7.8 years; 197 males) were finally included in this study. Bedridden patients or patients who were unable to answer the questionnaire were also excluded from this study. Age- and sex-matched control subjects without any history of neurological or psychiatric diseases (n = 401; age, 69.2 ± 8.6 years; 187 men and 214 women) were recruited from the medical staff of our outpatient clinic, and their friends and family, and from the spouses of PD patients from the outpatient clinics.
Methods
A diagnosis of PD was made according to the UK Brain Bank Clinical Diagnostic Criteria (10): Patients presented the core motor features of bradykinesia and at least one of the following: rigidity, rest tremor or postural instability. Board-certified neurologists experienced in movement disorders evaluated all patients, and brain imaging was performed to exclude atypical Parkinsonian syndrome or vascular parkinsonism. Drug-induced parkinsonism was excluded based on clinical history. Disease severity was rated by Hoehn and Yahr (HY) staging (11). All PD patients completed the Japanese version of the MDS-UPDRS parts II (motor experiences of daily living), III (motor examination) and IV (motor complications) (12).
All participants completed questionnaires regarding their habits, education, sleep status and headaches (lifetime and 1-year prevalence of recurrent headaches unrelated to the common cold, head trauma or alcohol intake). The semi-structured headache questionnaire was developed to differentiate migraines from other types of headache disorders, according to the International Classification of Headache Disorders, 2nd Edition (ICHD-2) (13). The information included all the essential parameters associated with migraines (14): The frequency of past headaches, headache duration, location, quality and intensity, sensitivity to light, noise or smell and the presence of aura worsening due to physical activities, and accompanying symptoms such as nausea and vomiting. A validated self-administered screen for migraines was included (15). Those who reported headaches were asked to classify changes in their headache severity (improved, no change or worsened) and frequency (markedly decreased, decreased, no change or increased) around the onset of PD (approximately 3 years before and after PD onset) for PD patients and over the past several years (defined as the past 5–7 years) for controls. We also used patient global impression (PGI) on headache severity 10-point visual analogue scale (0, much better; 5 no change; 10, much worse) to evaluate changes in overall headache severity around the onset of PD for PD patients with headaches or migraines, and over the past several years for controls with headaches or migraines. PD patients with headaches or migraines were asked to compare changes in headache characteristics over several years around the onset of PD.
The PD sleep scale (PDSS)-2, consisting of 15 individual items for nocturnal non-motor and motor problems, was used to evaluate PD-related sleep problems (16) and PDSS-2 scores ≥ 18 were defined as clinically relevant PD-specific sleep disturbances (17). Daytime sleepiness was measured using the Japanese version of the Epworth Sleepiness Scale (ESS) (18). Excessive daytime sleepiness was defined as an ESS score of 10 or more. The Japanese version of the Rapid Eye Movement Sleep Behaviour Disorder (RBD) Screening Questionnaire (RBDSQ-J) was used, and RBDSQ-J scores of 5 or more were defined as probable RBD (pRBD) (19). Sleep status was assessed by using the Pittsburgh Sleep Quality Index (PSQI), and poor sleep was defined as having a global PSQI score > 5 (20). Restless legs syndrome was diagnosed by assessing four essential features (21). The semi-structured Mini International Neuropsychiatric Interview (MINI) based on the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition, was administered to the participants to assess the presence of minor or major depression (22). The presence of either minor or major depression was regarded as depression in this study. The levodopa equivalent dose (LED) was calculated based on previously reported conversion factors (23).
The study was approved by the institutional review boards of the participating facilities, and written informed consent was obtained from all participants enrolled in the study.
Statistical analysis
We planned a study with 400 PD patients and 400 controls. A previous study indicated that the probability of migraine headaches among controls is approximately 0.1 (2). If the true probability of migraine headaches among PD patients is 0.05, we will be able to reject the null hypothesis that the migraine prevalences for cases and controls are equal, with a power of 0.8 (24). The alpha error probability associated with this test of the null hypothesis is 0.05. We used an uncorrected chi-squared statistic to evaluate the null hypothesis. Mann-Whitney U tests or Student’s t-tests were used where appropriate to compare the continuous variables, and chi-squared or Fisher’s exact tests were used to compare the categorical variables between the patients with PD and controls. The demographic characteristics among the PD patients without headaches, those with non-migraine headaches and those with migraines were compared using one-way analysis of variance (ANOVA) for the continuous variables followed by the Bonferroni post hoc test and chi-squared tests for the categorical variables. To analyse changes in overall severity, frequency and intensity of lifetime headaches and migraines, multinomial logistic regression analyses, adjusting for gender and timing of the evaluation of headache changes, were performed (model 2 in Table 1). We estimated timing of the evaluation of headache changes using “age of PD onset plus 3 years” for the PD group and “age when the study was performed” for the control group, while taking into account the difference in the times at which changes in headaches were assessed between PD patients with headaches/migraines and controls with headaches/migraines. To address a difference in recall period between PD patients and controls, we compared changes in headache/migraine characteristics between the PD patients with a disease duration of ≤ 7 years and the controls (model 3 in Table 1). We also analysed the differences in overall headache and migraine severity on the 10-point visual analogue scale between the full PD and control groups and between the subset of PD patients with a disease duration of ≤ 7 years and the control group using analysis of covariance (ANCOVA) adjusted for gender and timing of the evaluation of headache changes.
Two-tailed
Results
The mean disease duration and HY stage were 7.4 ± 5.3 years and 2.3 ± 0.7, respectively. Thirty-four patients (7.8%) were drug naïve, and 227 (52.1%) and 365 patients (83.7%) were treated with dopamine agonists and levodopa, respectively. The mean daily LED was 488.3 ± 360.0 mg. Smoking (17.7% vs. 8.0%,
Table 3 shows the clinical demographics of PD patients classified by no headache, non-migraine headache and migraine headache groups. There was no difference in age, disease duration or HY staging. Compared to the no headache group, the ratio of females was higher in the non-migraine headache and migraine headache groups. The MDS-UPDRS part III score was lowest in the non-migraine headache group. The MDS-UPDRS part II score tended to be higher in the migraine headache group compared with the no headache and non-migraine headache groups. The rates of EDS and pRBD were similar among the groups; however, poor sleeper, PD-related sleep problems and depression were more frequently observed in the migraine group than in the no headache and non-migraine headache groups.
Changes in lifetime headache/migraine frequency and intensity in PD patients and controls.
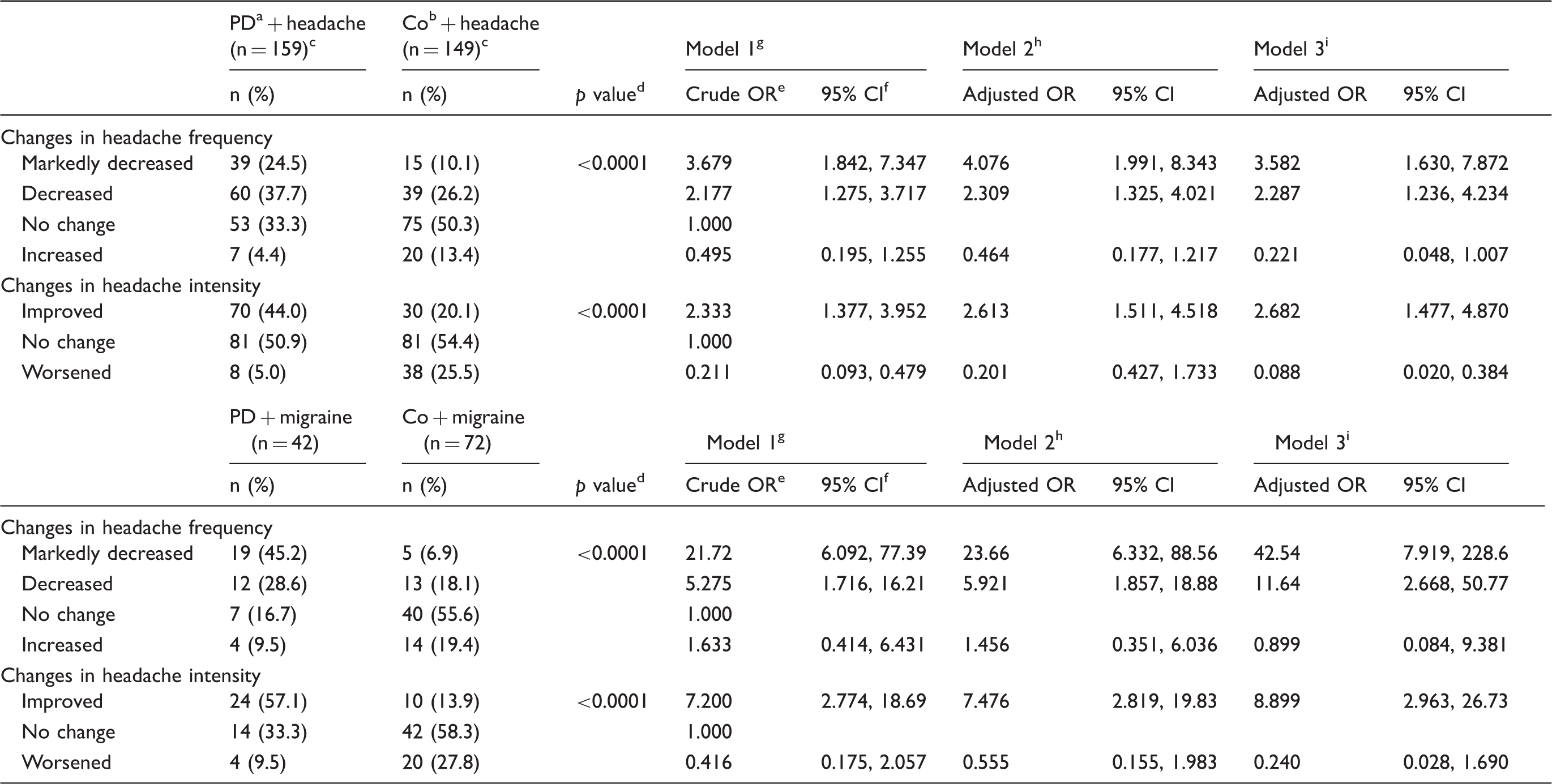
PD: Parkinson’s disease.
Co: controls.
Data are missing for nine PD patients and seven controls.
Based on a Chi-squared test.
OR: Odds ratio.
95%CI: 95% confidence interval.
Model 1 was analysed via multinomial logistic regression.
Model 2 was analysed via multinomial logistic regression after adjusting for gender and timing of the evaluation of headache changes.
Model 3 was analysed via multinomial logistic regression after adjusting for gender and timing of the evaluation of headache changes in the subset PD patients who experienced PD onset within the last several years (≤ 7 years) compared to controls. PD patients with headache corresponded to 95 cases, and PD patients with migraine corresponded to 26 cases.

Changes in overall lifetime headache severity in PD patients and controls.

Changes in overall lifetime migraine severity in PD patients and controls.
Headache prevalence in PD patients and controls.

PD: Parkinson’s disease.
Demographic characteristics among PD patients without headaches, with non-migraine headaches and with migraines.

PD: Parkinson’s disease; MDS-UPDRS: Movement Disorder Society revision of the Unified Parkinson’s Disease Rating Scale; MMSE: Mini-Mental State Examination; LED: levodopa-equivalent dose; EDS: excessive daytime sleepiness; pRBD: probable rapid eye movement sleep behaviour disorder.
¶
Discussion
This multicentre, case-controlled study showed that the lifetime (38.5% vs. 38.9%) and 1-year prevalence (26.1% vs. 26.2%) of headaches did not differ between PD patients and control subjects but revealed lower lifetime (9.6% vs. 18.0%) and 1-year (6.7% vs. 11.0%) migraine prevalences in PD patients than in controls. In contrast, a lower 1-year headache and migraine prevalence was reported in PD patients than in controls (headache 40.8% vs. 69.4%; migraine 28.3% vs. 33.0%) (25). In a cross-sectional study, among 237 PD patients the lifetime prevalence of migraines was 27.8% and the prevalence of current migraines was 13.1% (9). Compared to those studies, headache and migraine prevalence in PD patients in our study was lower (9,25). This difference may have arisen from methodological (questionnaire vs. face-to-face interview) and ethnic differences among the studies. In Japan, a nationwide survey reported that the 1-year prevalences of headaches and migraines in the general population were approximately 40% and 8.4%, respectively (2).
The most important finding of our study is that, although the control group also showed a tendency of reduced headache and migraine prevalence over several years, PD patients with headaches or migraines reported a more pronounced reduction in the intensity, frequency and overall severity of their lifetime headaches and migraines over the few years after disease onset. This difference was significant between groups after adjusting for gender, timing of the evaluation of headache changes and recall period (Table 1 and Figures 1 and 2). This finding is in line with a study reported by Barbanti et al. (9) that 63.7% of PD patients with migraines reported improvements in their headaches after disease onset, although no comparison was made with a control group in the degree of headache improvement. In contrast, in a study including 109 PD patients and 152 non-PD controls, 21% of PD patients and 24% of controls had a history of migraines; an improvement in migraines was observed in 62% of PD patients with migraines after disease onset and in 54% of non-PD controls with senescence, but no difference in improvement in migraines was found between PD patients and controls (26). However, one methodological issue in the previous study is that definition of improvement in migraines was unclear (frequency or intensity), as subjects reported ‘yes’ or ‘no’ as to whether their headaches improved.
In non-PD subjects, migraine severity generally improves as age advances, which is possibly related to a decline in cerebral vasodilator capacitance due to age-related cerebral atherosclerosis and changes in cerebral vasculatures (27). The pathology of PD involves not only the substantia nigra, which has abundant 5-HT1B receptors (28), but also serotoninergic raphe and noradrenergic locus coeruleus nuclei, all of which play crucial roles in pain transmission (29). The contribution of dopaminergic systems to migraines has been suggested based on observations that dopaminergic stimulation can induce premonitory symptoms of migraines and that dopamine receptor antagonists are effective in acute migraine attacks (8). By contrast, in the animal experiments, administration of dopamine and dopamine receptor agonists to trigeminocervical complex neurons inhibited their activation after nociceptive stimulation and lesioning of dopaminergic neurons. All nuclei of the hypothalamus facilitated nociceptive signalling from the trigeminocervical complex, suggesting that dopamine may act directly on trigeminal afferents to regulate firing in the neurons of the trigeminocervical complex (5,30). In addition, a dual orexin receptor antagonist reduced trigeminal nociceptive activity in an experimental migraine model (6), and therefore degeneration of hypothalamic orexinergic neurons in PD (31) may play some role in attenuating migraines in PD patients with headaches. Taken together, these findings suggest that the disease course of PD could modify migraine severity via progressive degeneration of the brainstem, including pain regulatory systems. Another possibility by which headache severity could be improved during the disease course of PD is that chronic dopaminergic therapy reduces pain in PD patients; however, in our study, there was no difference in LED irrespective of headache comorbidities.
Interestingly, van Hilten et al. (32) reported that among five patients with migraines, the course of migraines did not change in those with a family history of migraines after PD onset, whereas migraine severity lessened in those without a family history of migraine. This suggests some genetic differences in migraine comorbidity may play a role in the course of migraines in PD patients. However, Maude et al. (33) reported that the -141C Ins/Del polymorphism in the dopamine D2 receptor gene was not associated with patients with migraines or PD. Conversely, population-based studies have shown that subjects with migraines (34), particularly migraines with aura, were more likely to develop parkinsonian symptoms than subjects without migraines (35).
Third, we observed several migraine-associated clinical features in PD patients with migraines, including female predominance and increased rates of poor sleep, PD-related sleep problems and depression, relative to non-headache PD patients. A previous study related the predominant side of headaches to the ipsilateral side of the initial motor symptoms, but UPDRS part III scores (motor scores) did not differ between patients with or without headaches (25). However, unlike our study, which incorporated MDS-UPDRS part II, III and IV as well as detailed assessments of sleep (PSQI, PDSS-2 and RBDSQ-J) and depression, to the best of our knowledge no study has correlated the presence of migraines with sleep disturbances and depression in PD patients.
Our study has several limitations. First, we performed a case-controlled study and nearly all the patients were treated with dopaminergic drugs. Information on treatment for headaches and migraines was not obtained. In this setting, the recall bias and the effects of dopaminergic drugs and headache treatments on headaches should be considered. Although we analysed the headache changes between PD patients and controls via multinomial logistic regression after adjusting for gender, timing of the evaluation of headache changes and recall period, information bias could not be avoided because to estimate “timing of the evaluation of headache changes” we used age of PD onset plus 3 years for the PD group and age when the study was performed for the control group, and to adjust for the recall period between the PD and control groups, only the subset of PD patients were included. Further prospective population-based studies are needed to confirm our findings. In the PD group, some patients with a disease duration < 3 years who reported no improvement in headaches may experience improvements in their headaches in coming years upon additional observation, which could induce bias toward non-significance. However, we included the PD patients with a disease duration < 3 years in the analysis to prove our hypothesis that headaches/migraines may diminish a few years after PD onset due to degenerative changes in the brain region involved in pain regulation in PD. Second, we made every effort to recruit age- and sex-matched control subjects from the medical staff and their family and friends. However, they were not examined by neurologists and were not derived from the same source population as the PD patients. Third, migraine diagnosis was made based on questionnaires, although a migraine screening questionnaire was included that was based on the ICHD-2 criteria. Interestingly, changes in the frequency and intensity of migraines in PD patients have been observed in prospective studies. Additionally, a large population-based study may clarify whether the development of PD in patients with migraines alters headache characteristics.
In conclusion, we observed in this multicentre, case-controlled study that the lifetime and 1-year prevalences of migraines were significantly lower in PD patients than in controls and that, while overall headache and migraine severity lessened after the onset of PD, the presence of migraines was associated with sleep disturbances and depression in PD patients.
Article highlights
PD patients had lower lifetime and 1-year prevalences of migraines than controls. The lifetime and 1-year prevalences of headaches did not differ between PD patients and control subjects. PD patients with migraines exhibited a higher rate of depression and insomnia than those without headaches. PD patients with headaches or migraines reported a pronounced reduction in the intensity, frequency and overall severity of their headaches and migraines over a few years after the onset of PD.
Participating investigators of Kanto NMPD
Taro Kadowaki1, Ayaka Numao1, Tatsuya Yamamoto2, Taku Hatano3, Yuka Watanabe4, Tomoyuki Miyamoto5, Hideki Shimura6 and Takao Urabe6
1Department of Neurology, Dokkyo Medical University, Tochigi, Japan 2Department of Neurology, Chiba University Graduate School of Medicine, Chiba, Japan 3Department of Neurology, Juntendo University School of Medicine, Tokyo, Japan 4Department of Neurology, Dokkyo Medical University Nikko Medical Center, Tochigi, Japan 5Department of Neurology, Dokkyo Medical University Koshigaya Hospital, Saitama, Japan 6Department of Neurology, Juntendo University Urayasu Hospital, Tokyo, Japan
Footnotes
Acknowledgements
The authors thank Dr. Yuji Watanabe, Dr. Hiroaki Fujita, Dr. Takeo Matsubara, Dr. Hirotaka Sakuramoto, Ms. Sanae Tani and Ms. Kaori Soma, Department of Neurology, Dokkyo Medical University, for help with data entry.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
