Abstract
As brainstem mechanisms and dopaminergic neurotransmission are involved in migraine pathophysiology, we decided to investigate the course of migraine in Parkinson's disease (PD), the paradigm of brainstem dopaminergic disease. We screened 237 consecutive PD out-patients by direct interview to assess the prevalence of lifetime and current migraine. Moreover, we compared the course of migraine in PD patients with that of otherwise healthy age- (± 3 years) and sex-paired migraine controls in a cross-sectional study. PD patients showed a lifetime migraine prevalence of 27.8% and a current migraine prevalence of 13.1%. A positive family history of migraine was less frequent in PD patients than in controls. The frequency of current migraine was significantly lower in PD patients than in controls (47.0% vs. 68.2%; odds ratio = 0.41, 95% confidence interval = 0.19–0.89). Approximately two-thirds of PD patients reported an improvement in or remission of migraine after PD onset. Effects of menopause on migraine course were similar in patients and controls. These findings suggest that PD might somehow shorten the clinical course of migraine. Possible explanations include a prolonged prophylactic effect by chronic dopaminergic therapy or a positive effect of PD pathophysiology, namely nigral degeneration, on migraine mechanisms.
Introduction
Migraine, which is the most common neurological disease, afflicts from 14% to 18% of the general population (1–3). Idiopathic Parkinson's disease (PD) is one of the most frequent neurodegenerative disorders, with a prevalence ranging from 500 to 650 cases per 100 000 individuals over the age of 50 (4).
Interestingly, both diseases share some common functional and biochemical features. Brainstem mechanisms are crucial in migraine pathogenesis, as underscored by recent PET studies (5). Brainstem nuclei, such as trigeminal nucleus caudalis, locus coeruleus and dorsal raphe nucleus, are involved in the central processing and modulation of craniovascular pain, whereas the solitary tract nucleus and the dorsal motor nucleus of the vagus mediate the vegetative phenomena of the migraine attack. In addition, dopamine is considered to have a role in migraine pathogenesis (6, 7). This hypothesis has attracted renewed interest following the demonstration that dopamine D2 receptors in migraine show peculiar functional and genetic features. In particular, specific dopamine D2 receptor polymorphisms seem to be involved in the susceptibility, clinical expression and psychiatric comorbidity of the disease (8–12).
PD is the paradigm of human brainstem dopaminergic disease. In fact, the degeneration of substantia nigra pars compacta is the hallmark of the disease and, despite a variable involvement of other brainstem pigmented nuclei, PD represents a model of selective dopaminergic impairment, as confirmed by the dramatic clinical response to levodopa.
If brainstem mechanisms and dopaminergic neurotransmission are relevant in migraine pathogenesis, the study of migraineurs who subsequently develop PD would provide intriguing information on the role of dopamine and/or brainstem dopaminergic areas in migraine. However, such a prospective longitudinal study is obviously unfeasible.
We therefore decided to study the course of migraine in migraineurs affected by PD compared with that of otherwise healthy age- and sex-paired migraineurs by direct interview in a cross-sectional study. We assessed (i) the lifetime prevalence of migraine (lifetime migraine) in PD patients; (ii) the frequency of current migraine in PD patients with lifetime migraine compared with controls; and (iii) the occurrence of any modification of migraine course after the onset of PD.
Patients and methods
We studied PD patients and otherwise healthy control subjects with lifetime migraine. The presence of lifetime migraine was screened in all subjects during a face-to-face interview by means of the following preliminary questions: ‘have you ever suffered from headache?’, ‘are/were the attacks unilateral in onset?’, ‘are/were the attacks disabling?’, ‘is/was the pain pulsatile/throbbing?’, ‘are/were the attacks associated with nausea/vomiting and/or phonophobia and/or photophobia?’. PD patients with lifetime migraine were consecutively enrolled at our Outpatient Centre for PD and Extrapyramidal Disorders from 1 January 1999 to 31 December 1999. Population controls with lifetime migraine paired for sex and age (± 3 years) with no concomitant diseases or analgesic abuse were consecutively recruited in the same time period from non-consanguineous relatives of neurological outpatients. PD patients were diagnosed according to the UK Parkinson's Disease Society Brain Bank clinical criteria (13). PD severity was rated by using the motor score subset of the Unified Parkinson's Disease Rating Scale (UPDRS) (14) and the Hoehn & Yahr scale (15). Migraine was diagnosed according to the International Headache Society (IHS) criteria (16). Information on current and/or past migraine was gathered by means of a face-to-face interview using a structured questionnaire on family history of migraine, age of onset, presence of aura, attack frequency, location, quality and intensity of pain, associated symptoms, abortive and/or preventive treatment, age of menopause. PD patients were also asked to describe any modification of migraine course (improvement or worsening) that may have occurred after PD onset. All female individuals were asked to report any modification of migraine following menopause. Migraine improvement was defined as a reduction in the attack frequency of > 50%.
The statistical analyses were performed by means of odds ratio (OR) and associated 95% confidence intervals (CI), Student's t- and χ2 tests. All analyses were performed with BMDP New System software.
Results
Two hundred and thirty-seven PD patients were screened. Of these, 66 patients (27.8%) had lifetime migraine (age of PD onset = 57.0 ± 9.8 years, range 32–73 years; UPDRS motor score = 25.1 ± 10.8, range 5–51; H & Y: stage I = 11; II = 36; III = 16; IV = 3). Thirty-one PD patients (13.1%) had current migraine. PD patients were being treated with levodopa (n = 38), dopamine agonists (n = 8), levodopa plus dopamine agonists (n = 17) and deprenyl plus amantadine (n = 3). Sixty-six otherwise healthy population controls paired for sex and age (± 3 years) with lifetime migraine were selected. The clinical characteristics of all subjects studied are summarized in Table 1. PD patients and controls did not significantly differ in age of migraine onset, type of migraine, attack characteristics and age of menopause (49.4 ± 6.4 vs. 47.9 ± 5.1, respectively; P = NS).
Clinical characteristics of Parkinson's disease (PD) patients and otherwise healthy control subjects with lifetime migraine
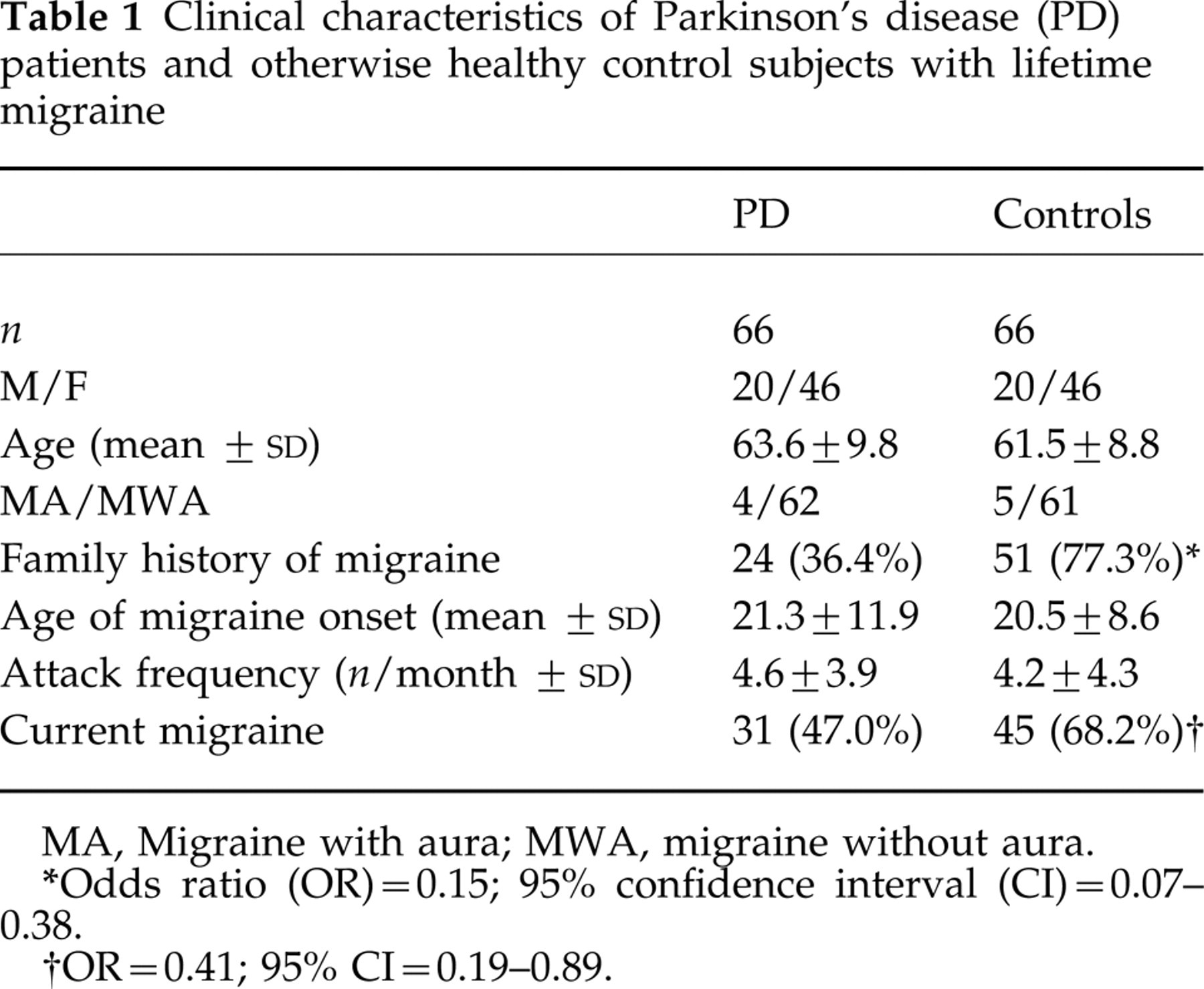
MA, Migraine with aura; MWA, migraine without aura.
Odds ratio (OR) = 0.15; 95% confidence interval (CI) = 0.07–0.38.
OR = 0.41; 95% CI = 0.19–0.89.
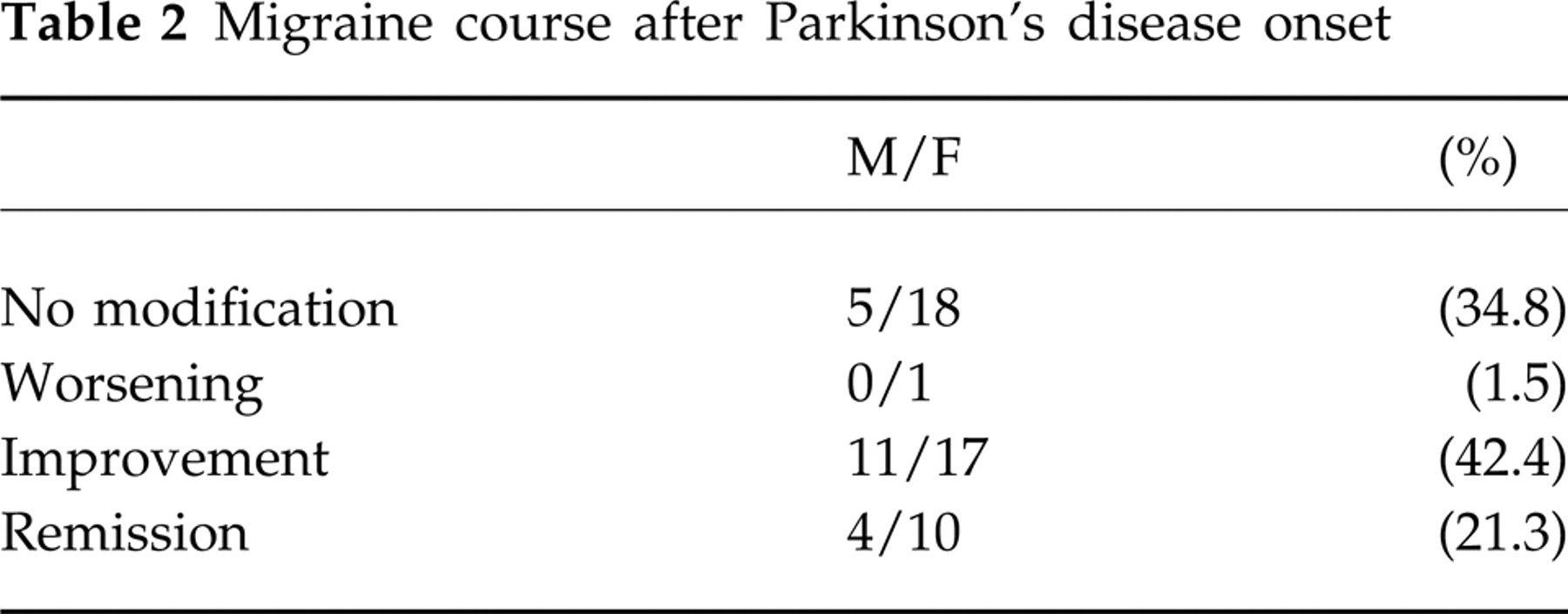
Current migraine was significantly less frequent in PD patients than in controls (Table 1). The majority of PD patients reported an improvement in or remission of migraine after PD onset (Table 2).
Migraine course after Parkinson's disease onset
The effects of menopause on the course of migraine did not differ in PD patients (n = 42) and controls (n = 45) (Table 3). Menopause preceded PD onset in three patients (7.1%); in the remainder, menopause followed PD onset with a time interval of 0–6 years in seven patients (16.7%), 7–12 years in 17 (40.5%), 13–19 years in 11 (26.2%) and > 20 years in four (9.5%).
Migraine course after menopause

χ2 (for two columns and four rows) = 4.86; P = NS.
A positive family history of migraine was less frequent in PD cases than in controls (Table 1). Of the 24 PD patients with a family history of migraine, 11 had current migraine.
Discussion
This is the first study of migraine in PD using the IHS diagnostic criteria. We document a lifetime migraine prevalence of 27.8% and a current migraine prevalence of 13.1% with a female to male ratio of 2:1 in a PD population with a mean age of 63.6 years. We found a lower frequency of current migraine in PD patients than in otherwise healthy controls with lifetime migraine. This difference was not related to either migraine characteristics and age of menopause or different effects of menopause on migraine course in the two groups. Approximately two-thirds of PD patients reported an improvement in or remission of migraine after PD onset.
Epidemiologic data on migraine in the elderly are sparse and conflicting. A lifetime prevalence of migraine of 21% has been reported over the age of 56 years (17), whereas current migraine over the age of 60 ranges from 1.3% to 15.2% in males and from 8% to 10.4% in females, depending on the author (18–21). The only previous survey of headache in PD patients, conducted before the introduction of the IHS criteria, reported a current migraine prevalence of 19.3% at a mean age of 66 years (22). Another study performed on a very small number of PD patients with current migraine (n = 5) suggested that migraine course was favourable only in PD patients without a family history of migraine (23). We cannot confirm this finding as almost half of the PD patients without a family history of migraine had current migraine in our study. Moreover, we have no unequivocal explanation for the lower prevalence of family history of migraine in our PD migraineurs.
Our study has some limitations. This is not a population-based study, as PD patients were recruited in a tertiary-referred centre and controls were enrolled from non-consanguineous relatives of neurological outpatients. Furthermore, given the study design we cannot exclude artefacts due to recall bias. Both factors might account, for example, for the relatively high attack frequency in PD patients and controls. Moreover, we cannot exclude the occurrence in our PD sample of some potential risk factors for migraine such as middle-low social level and/or self-reported poor health. In addition, though we excluded migraineurs with concomitant diseases and/or drug abuse, it could be argued that the lower prevalence of current migraine among PD patients in our study might be related to a prompter ascertainment and treatment in these patients of possible concomitant undiagnosed diseases (such as hypertension) which may contribute to the persistence of migraine in the elderly. However, though this bias cannot be excluded, it is known that other exacerbating factors for migraine, such as depression, are more frequent in PD than in healthy controls.
Nevertheless, despite all these drawbacks, this study suggests that PD may somehow shorten the clinical course of migraine. Two hypotheses may be considered. The first is that chronic therapy with levodopa and/or dopamine agonists might act as prolonged migraine prophylaxis, a hypothesis which is, however, debatable. Although this may apply to dopamine agonists (whose combined dopaminergic and anti-serotoninergic effects are responsible for the prophylactic effect in migraine), a high degree of efficacy on migraine of pure dopaminergic stimulation with levodopa would be surprising. It is noteworthy that the dramatic improvement in migraine with levodopa in one PD patient (24) was not confirmed in a subsequent pilot study (25). Moreover, the beneficial effects of dopaminergic therapy would better explain the improvement in migraine in some patients than the remission of the disease in the others. The second hypothesis is an alternative and intriguing explanation according to which PD pathophysiology, namely nigral degeneration, might somehow positively interfere with migraine pathophysiology. This hypothesis is supported by recent findings on the involvement of the substantia nigra (SN) in migraine. The SN contains the highest concentration of binding sites for 3H-sumatriptan in the brainstem (26). Moreover, the SN and red nucleus are bilaterally activated after the occipital cortex both during the visual aura of migraine (27) and in daily chronic headache (28), as studied by T2-weighted magnetic resonance imaging (MRI). Finally, the SN and ventral tegmental area actively control cerebral microcirculation and the permeability of the blood–brain barrier via dopaminergic fibres that terminate in close contact with penetrating arterioles and cerebral capillaries in the cerebral cortex (29).
Little is known about the mechanisms responsible for the lower prevalence of migraine with increasing age. The favourable effect of prophylaxis, increased mortality in migraineurs, cohort effect and spontaneous remission have been considered (30). It has also been suggested that the physiological ageing of the dopaminergic system may be involved in the decrease in migraine prevalence with age (23). On this basis, we cannot exclude that the pathological ageing of the dopaminergic system in PD might account for the improvement of migraine in these patients.
Footnotes
Acknowledgements
We thank Dr Antonio Bisceglia and Dr Francesco Cesarino for patients' referral.
