Abstract
Introduction
The objective of this review is to provide an evidence-based discussion of different treatment strategies for medication-overuse headache (MOH).
Method
We searched PubMed for articles discussing the treatment and prognosis of MOH published between 2004 and August 2014. Titles, abstract and articles were reviewed systematically. The level of evidence provided by each study of the included articles was determined according to the American Academy of Neurology Clinical practice guideline manual. We discuss the level of evidence to support the early discontinuation/withdrawal of overused medications, the level of evidence to support the use of preventive treatment, the short- and long-term prognosis, and the outcome according to the class of drug overused in patients diagnosed with MOH.
Results
The initial search resulted in 1313 articles; 68 articles met our inclusion criteria and were discussed. The level of evidence to support early discontinuation of overused medications alone is low due to the absence of controlled studies. Adding preventive medication to early discontinuation led to a better outcome than early discontinuation alone. For patients with chronic migraine (CM) and medication overuse (MO), there are large randomized control trials supporting the use of onabotulinumtoxinA and topiramate without early discontinuation of overuse; however, the evidence is limited since data were obtained from post hoc analysis.
Conclusion
Considering current available evidence and the systemic toxicity of overusing acute headache medication, we suggest discontinuation of the overused medication with the addition of preventive medication. Appropriately sized, randomized controlled trials evaluating the safety and long-term efficacy of preventive medication plus early discontinuation of overuse vs preventive medication alone vs early discontinuation of overuse alone are needed.
Keywords
Introduction
Medication-overuse headache (MOH) is a common and disabling neurological disorder. Its true prevalence is unknown since in some cases, the drug is involved in generating frequent headache, whereas in others, the frequent use of medication is a reflection of frequent headache. Nevertheless, in population-based studies, approximately 1%–2% of the general population is estimated to have MOH (1–3).
Operational diagnostic criteria for MOH were formally introduced in the 2004 International Classification of Headache Disorders (2nd edition) (ICHD-2), revised in 2006 (4–6), and again in 2013 (ICHD-3 beta version) (ICHD-3β) (7). MOH is currently defined as headache occurring on 15 or more days per month developing as a consequence of regular overuse of acute or symptomatic headache medication (on 10 or more, or 15 or more days per month, depending on the medication) for more than three months (4–7). In the ICHD-2, MOH excluded a concomitant diagnosis of chronic migraine (CM). However, according to the ICHD-3β, since it is unknown whether the overuse of medication is the cause or consequence in an individual case, the patient will receive a diagnosis of both CM and MOH when both criteria are met.
Previous ICHD-2 criteria for MOH required the chronic headache to resolve or revert to its previous episodic pattern within two months after discontinuation of the overused medication (4), making early discontinuation mandatory. It was also believed that preventive treatment is ineffective while the patient is overusing acute medications. While discontinuation may increase the responsiveness and is a primary task for all MOH patients (8–11), early discontinuation is complicated by high rates of treatment failure and relapse (12–14). Data from recent well-controlled clinical trials have shown that migraine-preventive medication, especially onabotulinumtoxinA and topiramate, may be effective in patients with CM with medication overuse (MO) even when acute medications are not tapered or discontinued (10,15–20).
There continues to be considerable debate as to whether patients should be initially managed with early discontinuation of the overused medication alone, early discontinuation of the overused medication plus preventive therapy, or preventive therapy without early discontinuation of the overused medication. The guideline from the European Federation of Neurological Sciences recommends the abrupt discontinuation or taper of overused medication (8). The choice of early discontinuation strategy and setting (inpatient or outpatient) depends on the acute medications being overused. This guideline also recommends individualized prophylactic drug treatment be started before or at the first day of early discontinuation therapy, and that patients be followed at regular intervals to prevent relapse of MO. This approach is endorsed by some (21), while others recommend early discontinuation with follow-up in two to three months to determine whether there is a need for preventive medication (22).
The objectives of this review are to determine: 1. The level of evidence to support the early discontinuation/withdrawal of overused medications in patients diagnosed with MOH. 2. The level of evidence to support the use of preventive treatment in patients with MOH. 3. The short and long-term prognosis (remission and relapse rates) in both groups. 4. The short and long-term prognosis in MOH patients according to the class of drug overused (opioids, analgesics, triptans, ergots).
Methods
For this systematic review of the literature, we searched PubMed for English-language articles of humans that were published between 2004 and August 2014. The year 2004 was used as the start date since it was in 2004 that operational diagnostic criteria were first introduced. The following search terms were used: “medication overuse headache,” “medication overuse headache and treatment,” “medication overuse headache and withdrawal,” “medication overuse headache and prognosis,” “medication overuse headache and relapse,” “medication overuse headache and remission,” “medication misuse headache,” “analgesic overuse headache and treatment,” and “analgesic overuse headache and withdrawal.” Also, the reference lists of included articles were searched for additional articles. Articles were excluded if:
(a) medication-overuse headache was not being studied; (b) treatment/discontinuation/detoxification/withdrawal/prognosis/remission or relapse were not included; (c) the article was a case report; (d) the article was a review of or comment on previously published findings; (e) the article focused on rescue therapy for withdrawal headache; (f) the article focused on MOH treatment in children. The titles of all articles resulted from our search were reviewed by one reviewer (CC). The abstracts and all papers in their entirety were further reviewed if the articles were included for this analysis. The initial search resulted in 1313 articles, and 68 met our criteria for inclusion and were thus included in this review (Figure 1). The level of evidence provided by each study was determined according to the American Academy of Neurology (AAN) Clinical practice guideline manual (23), and conclusions were further built based on the suggested verbiage of the same manual.
Flow diagram for systematic review on treatment of medication overuse headache.
Results
The level of evidence to support the early discontinuation/withdrawal of overused medications in patients diagnosed with MOH
We found 27 studies (45 articles) reporting the treatment response to early discontinuation therapy (9,12,14,24,25,29–69). Among them, 19 studies (37 articles) reported the results of Early discontinuation with preventive medication (9,12,24,25,29–45, 53–69).
Three studies discussed the results of Early discontinuation without preventive medication (14,46,47). Another study compared the effect of early discontinuation alone, preventive therapy alone to a control group (48). This latter study is the only one that included a no-treatment control group. Four other studies that focused on other interventions are summarized in the following paragraph (49–52).
Early discontinuation with preventive medication
The method of early discontinuation varied among the articles reporting treatment response from early discontinuation therapy with preventive medication. A protocol usually included inpatient or outpatient discontinuation, intravenous hydration and rescue medication such as promethazine, and medications for withdrawal symptoms such as benzodiazepine, metoclopramide or corticosteroid. Preventive medications were given either at the start of early discontinuation, during hospitalization or at discharge. Most preventive medications were selected according to a patient’s underlying primary headache type. Commonly used prophylactic medications included: sodium valproate, propranolol, topiramate, amitriptyline, flunarizine and selective serotonin reuptake inhibitor (SSRI) (39,53,54).
Andrasik and Grazzi followed a group of patients with CM with MO for five years after a semi-standardized inpatient early discontinuation program combined with prophylactic medications (31,53,55). Seventy-nine percent of 106 patients had completed the initial trial at one year; however, only 53% completed the follow-up at five years. All outcome assessments among the completers, including headache frequency, analgesics used and the Migraine Disability Assessment (MIDAS) score, were significantly improved compared to baseline, at six months, one year, three years, and five years. Headache frequency and analgesic usage lessened over time (headache frequency at baseline, six, 12, 36, 60 months after discontinuation were 26.1, 7.8, 10.5, 11.5, 11.3 days/month), and the MIDAS total score continued to decrease with time, suggesting functional improvement even after five years.
Another study addressed the effect of early discontinuation in patients with treatment-resistant MOH (42). Ninety-eight patients with MOH who had previously been unsuccessfully treated by a specialist were recruited and underwent a structured two-month early discontinuation program with prophylactic medication. At 12 months, they reported a 39% reduction in headache frequency (p < 0.001) and a 63% reduction in medication use (p < 0.001) among the completers.
A recent multinational study involving centers from Europe and Latin America (COMOESTAS project) reported a consensus protocol for the management of MOH advised by an international and multicenter doctors group (24). Patients were included for either inpatient or outpatient early discontinuation/detoxification. Prophylactic treatment was chosen based on the patients’ underlying primary headache and comorbid disorders, and was started on day 1 of early discontinuation. Based on an intention-to-treat analysis, nearly two-thirds of the enrolled 376 patients were no longer overusing medications at the end of the study protocol and nearly half had reverted to an episodic headache pattern (24). Another paper from the COMOESTAS project reported a significant reduction in disability (MIDAS score), and a considerable reduction in number of patients with depression and anxiety after acute medication discontinuation with prophylactic treatment in patients with MOH (25). The limitations of this study were excluding patients with prior discontinuation failures, current prophylactic treatment, and significant psychiatric illness, likely biasing the study toward favorable outcomes (70).
Other studies also reported significant reduction from baseline in headache days, drug intake, and significant improvement in Headache Impact Test-6 (HIT-6) scores, quality of life (QoL), depression and anxiety within one year after treatment with abrupt discontinuation combined with prophylactic medication (29,39,41).
Early discontinuation without preventive medication
Studies performed to assess the efficacy of early discontinuation treatment usually allowed preventive treatment before or following discontinuation, making it difficult to determine the effect of early discontinuation alone. Zeeberg and colleagues assessed the effect of a two-month medication-free period for 337 patients with probable MOH (14). Levomepromazine or phenergan was allowed as the rescue medication during early discontinuation for the first week, but no other acute migraine medications could be used. Only 64% completed the two-month study. Among the completers, 45% improved, 48% had no changes and 7% experienced an increase in headache frequency (14). The outcomes were affected by the primary headache type and the type of medication overused. Patients with migraine had a significantly larger reduction in headache frequency than patients with tension-type headache (TTH) (67% and 0%, p < 0.001) or patients with both migraine and TTH (37%, p < 0.01). Triptan or ergot overusers improved the most (p < 0.0001). Two other prospective studies evaluated the effect of intensive advice to discontinue the overused medication (46,47). Antiemetics and simple analgesics were allowed for rescue therapy, but preventive medication was not allowed
Comparisons between early discontinuation alone, preventive medication alone, and no treatment (i.e. control)
Hagen and colleagues performed a one-year open-label, prospective randomized multicenter trial including 56 MOH patients who were randomly assigned to: (a) abrupt discontinuation of overused medications with start of preventive treatment after only three months (n = 22); (b) preventive treatment from day 1 without abrupt discontinuation (n = 19); and (c) controls without preventive medication or abrupt discontinuation (n = 20) (treatments were given at five-month follow-up) (48). Intention-to-treat analysis was performed and results that were significant in the between-group analysis included: 1. The prevention group had a significant reduction in total headache index (headache days/month × headache intensity × headache hours) compared with the abrupt discontinuation group (−201 vs −19 at month 12, p = 0.003). 2. The responder rate was significantly higher for the prevention group than controls (41% vs 5%, p = 0.01). 3. Change in days with acute headache medication per month was greater in the abrupt discontinuation group compared with controls (−19.1 vs −6.9 at month 3, p = 0.002, and −18.5 vs −6.1 at month 5, p = 0.001), whereas pairwise comparisons between controls and the prevention group did not achieve significance. At 12-month follow-up, the headache days/month and the reduction in days with use of acute headache medication per month significantly decreased in both the prevention and abrupt discontinuation groups compared with baseline. However, the abrupt discontinuation group had a significant increase in headache hours compared with baseline (1.4 hours, p < 0.009), whereas the prevention group had a decrease of 0.1 hour (p = 0.017) at month 5. While the sample size is small, this is the first randomized study evaluating the efficacy of early introduction of preventive medication compared with abrupt discontinuation therapy and a control group. Early introduction of prophylactic medication without a discontinuation program was an effective way to reduce headache days and total headache burden during the first three months, and the improvement was sustained up to four years (40,48,56). This study is graded class II based on the AAN Clinical practice guideline manual (23) since it’s a randomized controlled clinical trial but less than 80% completed the study.
Other interventions
Studies also reported other treatment methods to be effective and beneficial in early discontinuation and treating MOH patients, including a “brief intervention” scheme (49), self-help group (50) or a multidisciplinary treatment program including behavioral plus pharmacological treatment (51), and short-term psychodynamic psychotherapy (STPP) (52).
Conclusions and recommendations for early discontinuation and for preventive medication when treating medication overuse headache. The evidence class I, II, III IV was based on the “Classification of Evidence Matrix for Therapeutic Questions” AAN (23), Appendix 3. The Conclusions and Recommendations were based on p.13 and Appendix 6 of the same manual “Synthesizing Evidence—Formulating Evidence-based Conclusions.” The suggested verbiages are: Multiple Class I studies: Are highly likely to be effective … Multiple Class II studies or a single Class I study: Are likely effective … Multiple Class III studies or a single Class II study: Are possibly effective … Multiple Class IV studies or a single Class III study: There is insufficient evidence to support or refute the effectiveness …

HIT-6: Headache Impact Test-6; VAS: visual analog scale; CM: chronic migraine; MO: medication overuse; CDH: chronic daily headache.
The level of evidence to support the use of preventive treatment in patients with MOH
Articles investigating a specific kind of preventive treatment are included in this section. Among the 17 studies (23 articles) (15–20,26–28,57,71–83), five studies (10 articles) reported the effect of onabotulinumtoxin (17–19,27,57,71,79,80,82,83), three studies (four articles) discussed the effect of topiramate (15,16,20,72), two for valproic acid (26,75), one for nabilone (73) and one for pregabalin (76). Studies discussing non-pharmacological treatment such as occipital nerve stimulation (28,77,81), occipital nerve block (78), and acupuncture (74) are also included.
OnabotulinumtoxinA
OnabotulinumtoxinA without early discontinuation
Two phase III, 24-week, double-blind, parallel-group, placebo-controlled studies followed by a 32-week, open-label, single-treatment, onabotulinumtoxinA phase, were conducted by the Phase 3 Research Evaluating Migraine Prophylaxis Therapy (PREEMPT) clinical program at 66 global sites and a pooled analyses of the 56-week program was reported (18). The results showed onabotulinumtoxinA to be an effective prophylactic treatment for CM with or without MO (17–19,71). In PREEMPT studies, patients were randomized to 155–195 U of onabotulinumtoxinA or placebo using MO as a stratifying variable. Of 1384 patients, 65.3% (n = 904) met MO criteria (onabotulinumtoxinA: n = 445, placebo: n = 459). Planned secondary analysis for CM with MO subgroup at week 24 revealed significant between-treatment group mean changes from baseline favoring onabotulinumtoxinA vs placebo for headache days (primary endpoint: −8.2 vs −6.2; p < 0.001) and other secondary endpoints: frequencies of migraine days (p < 0.001), moderate/severe headache days (p < 0.001), cumulative headache hours on headache days (p < 0.001), headache episodes (p = 0.028), migraine episodes (p = 0.018) and the percentage of patients with severe Headache Impact Test-6 category (p < 0.001) (82). All efficacy analyses used the intention-to-treat population including all randomized patients. Of note, despite the overall reduction from baseline in intake across both placebo and onabotulinumtoxinA-treated groups, the intake of acute pain medication (all categories) was similar between the treatment groups. Therefore, the significance of onabotulinumtoxinA treatment across multiple efficacy endpoints could not be attributed to the discontinuation from acute pain medication during the study. The results indicate that onabotulinumtoxinA, without early discontinuation of the overused medication, was effective and well tolerated as headache prophylaxis in CM with MO patients (82). The limitation of applying results from the PREEMPT trial to MOH patients is that the PREEMPT trial was designed for patients with CM with or without MO, instead of specifically for MOH patients, and the results favoring onabotulinumtoxinA in reducing headache days for patients who have CM with MO are from a planned secondary analysis. Furthermore, there were similar reductions in the use of acute migraine medications for patients receiving onabotulinumtoxinA and for those receiving placebo injections. The effect of onabotulinumtoxinA in nonmigraine-related MOH is still unknown.
OnabotulinumtoxinA with early discontinuation
Sandrini and colleagues had evaluated the efficacy and safety of onabotulinumtoxinA as prophylactic treatment for patients with migraine without aura plus medication-overuse headache (80). Sixty-eight patients were treated with standardized inpatient early discontinuation therapy and were randomized in a double-blind fashion to onabotulinumtoxinA or placebo treatment. During the second day of hospitalization, 100 U was given to the onabotulinumtoxinA group. A second injection was given at week 12 if the patient responded to the first injection. A significant reduction was recorded in the secondary endpoint of acute pain drug consumption at 12 weeks in onabotulinumtoxinA-treated patients vs placebo. However, there was no significant difference in the primary efficacy endpoint, the mean change from baseline in frequency of headache days, and no change in the impact on functioning-HIT-6 and MIDAS score. This study supports the use of onabotulinumtoxinA along with early discontinuation therapy. The difference in these results compared to the PREEMPT trials may be due to the lack of a discontinuation intervention in the PREEMPT studies, a much smaller sample size, lower dosage, and fewer injection sites in the Sandrini study (100 U across 16 sites vs 155–195 U across 31–35 injection sites in the PREEMPT trial). The study design met the evidence grading criteria for AAN Class I, however the smaller sample size and not performing intention-to-treat analysis are limitations of this study.
Other nonplacebo-controlled trials reported that onabotulinumtoxinA reduced visits to the emergency department by almost 90% (83), and could reduce the amount of money spent on acute migraine medications in patients with CM with triptan overuse (79).
To conclude, the efficacy of early discontinuation plus onabotulinumtoxinA is superior to early discontinuation alone in reducing acute medication consumption (80), and the efficacy of onabotulinumtoxinA without early discontinuation was demonstrated in the PREEMPT trials (18,19,71,82) for patients who have CM with MO. Large, randomized control trials are needed to evaluate whether onabotulinumtoxinA with early discontinuation or onabotulinumtoxinA alone lead to a better outcome.
Topiramate
Two randomized, double-blind, placebo-controlled parallel-group, multicenter CM trials conducted in the United States (US) and Europe established the efficacy of topiramate in patients who have CM with or without MO (15,16,20). For the US trial, the post hoc efficacy analysis in the subgroup of patients with MO at baseline showed a nonsignificant reduction in mean monthly migraine/migrainous days and migraine/migrainous headaches compared to placebo (10,16,20). In the European trial, 78% patients had MO at baseline. The main results for the MO subgroups (n = 46) were that topiramate 100 mg/day (dosing up to 200 mg/day allowed) without early discontinuation significantly reduced the mean number of monthly migraine days compared to placebo, and the reduction of the mean number of days per month of acute medication intake from baseline did not reach statistical significance compared with placebo (15). There are limitations in these topiramate studies that must be considered when drawing conclusions about the efficacy of topiramate in patients with MO, including: 1) they were not specifically designed for patients who have CM with MO; 2) patients were excluded if they had a primary headache disorder other than CM; 3) the conclusions regarding patients who have CM with MO are from a post hoc analysis; 4) both studies provide Class II evidence because of low completion rates. In the European trial, completion rates for topiramate- and placebo-treated patients were 75% and 52% (15). In the US trial, only 55.8% in the topiramate group and 55.2% in the placebo group completed the double-blind phase of treatment (16).
Another randomized, double-blind placebo-controlled study also reported that topiramate significantly reduced the number of headache days and the mean amount of acute medication taken vs placebo (both p < 0.0001 vs placebo) in patients with CM with MO (72). This later study is graded class II because of a high patient drop-out rate (>20%).
Valproic acid
One prospective case series reported a significant reduction in mean headache days after two months of divalproex sodium treatment without early discontinuation in 10 patients with CM with MO. However, baseline headache frequency was estimated retrospectively by participants, introducing the potential for significant recall bias (75). This study is graded Class III for the lack of a control group.
Cannabinoids
Nabilone is a synthetic cannabinoid CB1-receptor agonist and its effect in treating intractable MOH had been evaluated in a randomized, double-blind, active-controlled, crossover study in an Italian Medical Center (n = 30) (73). Patients were given oral nabilone 0.5 mg/day for eight weeks, and another eight weeks received ibuprofen 400 mg in a blinded sequence. There was a one-week wash-out period. After 20 weeks’ follow-up, nabilone was significantly superior to ibuprofen in reducing daily acute medication intake, headache intensity, the level of dependence, and changing the QoL indicators (Short Form (SF)-36 Scale). This study concluded that nabilone is efficacious in the treatment of MOH. The study is AAN Class II evidence level since there were more than two primary outcomes specified. Large clinical trials and long-term follow-up are needed to assess the efficacy and safety of nabilone.
Pregabalin
One observational pilot study compared the efficacy, tolerability, and safety of pregabalin (150 mg/d) to topiramate (100 mg/d) during 16 weeks of treatment in patients with MOH (76). Compared to baseline, pregabalin administration without early discontinuation significantly decreased monthly headache frequency, days with acute medication intake, and improved disability. No significant between-groups differences were found in the three outcome measures. Long-term follow-up of this prospective, open-label study is needed.
Non-pharmacological management
A study in 34 patients with CM with or without MO (85% had MO) reported that occipital nerve stimulation (ONS) after at least two months of drug discontinuation significantly decreased the headache intensity, frequency, and triptan/nonsteroidal anti-inflammatory drugs (NSAIDs) usage from baseline (77). However, another study showed that after receiving ONS for refractory head pain, patients with MO had significantly less pain relief at long-term follow-up than those without MO (mean of 28% vs 78%; p = 0.0498) (28). Both studies are Class III owing to lack of a control group and lower completion rate. The use of ONS is not recommended when other less-expensive, less-invasive and more-effective strategies are available.
As for occipital nerve block (ONB), one study reported that MO tripled the risk of ONB failure, and that MO increased ONB failure rate more in migraineurs than in those with occipital neuralgia (78). The use of ONB in MOH patients should be carefully evaluated.
One randomized controlled trial compared the efficacy of acupuncture to topiramate in patients with CM with or without MO. Acupuncture was given in 24 sessions over 12 weeks; topiramate was initiated at 25 mg/day and increased by 25 mg/day weekly to a maximum of 100 mg/day over a four-week titration period followed by an eight-week maintenance period. Data analysis for the MOH subgroup (n = 49) demonstrated a significant improvement in all assessments of efficacy, acute medication consumption, and disability favoring the acupuncture group compared to the topiramate group (74). However, the placebo effect might be higher in the acupuncture group, and other limitations of this study include the small sample size, single-center study, and unblinded evaluations.
The conclusions based on the level of evidence for preventive treatments are listed in Table 1.
The short- and long-term prognosis (remission and relapse rates)
Although clinical trials of preventive medications alone for MOH appear promising, most studies did not report long-term prognosis. Therefore, we present the short- and long-term prognosis of early discontinuation therapy.
Of the articles reporting the outcome of early discontinuation, the completion rate varied from 39% to 93%. In most studies, preventive medication was given during inpatient discontinuation of acute medications or at discharge, and patients continued to use preventive medication at follow-up (30,36,43,58). In one study, the persistence of CDH with MO after four years was associated with a low use of prophylactic medication. The data support the routine use of preventive medications in patients with CDH even in the presence of MO, and also the need for prolonged use in many chronic patients (59).
Here we define remission as without MO at follow-up, and relapse rate was defined as the percentage of patients who successfully discontinued, but then went back to overusing acute medications. Overall, the remission and relapse rates from two to six months after discontinuation were 66%–100% and 13%–34%, respectively (12,35,39,41,46,54,60). At one year, the remission rate was 60%–83% and 17%–43% patients relapsed (12,30,36,37,43,54,61–63). Some studies reported that at three to four years, the remission and relapse rate were 58%–66% and 24%–45% (12,40,56,59).
The first year after discontinuation is the most important period of time that determines relapse. The vast majority (94%) of patients relapsed within the first year after discontinuation (12), and remission at one year was a significant predictor for sustained remission at year 4 (p < 0.005) (59).
Remission and relapse rate after discontinuation.
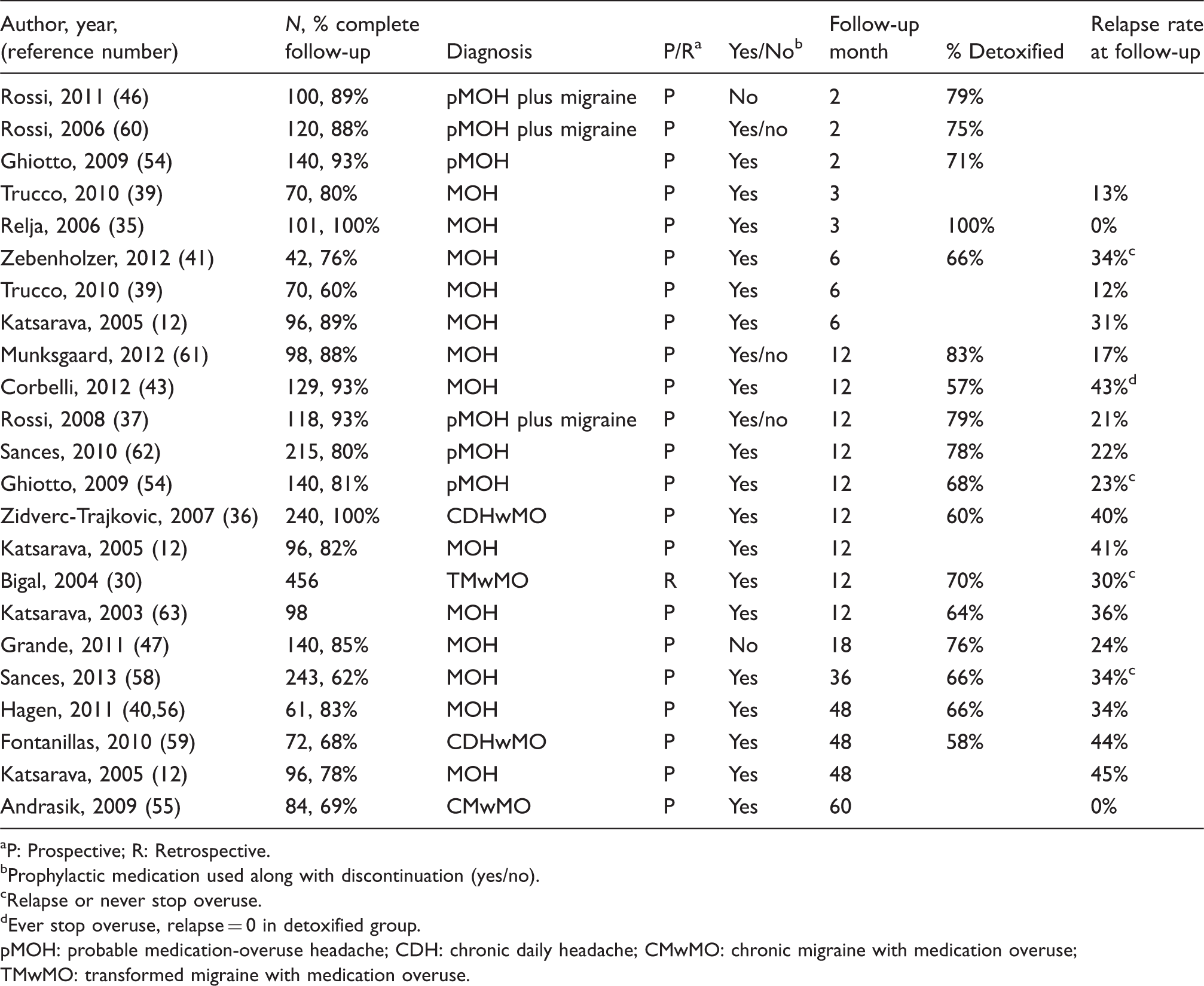
P: Prospective; R: Retrospective.
Prophylactic medication used along with discontinuation (yes/no).
Relapse or never stop overuse.
Ever stop overuse, relapse = 0 in detoxified group.
pMOH: probable medication-overuse headache; CDH: chronic daily headache; CMwMO: chronic migraine with medication overuse; TMwMO: transformed migraine with medication overuse.
Several studies discussed prognostic factors of outcomes after early discontinuation therapy. These factors include:
Primary headache type: Patients with migraine had a significantly greater reduction of headache frequency and lower relapse rate (31.5%) than patients with TTH (91%) or a combination of migraine and TTH (70%) (12). Other studies suggested that the headache type did not influence the prognosis (48,62). Type of medication overused: see discussion below. Psychological profile: Personality profile such as hypochondriasis, depression, paranoia, and fears assessed by the Minnesota Multiphasic Personality Inventory (MMPI)-2 predicted the worst outcome (never stop overusing medication) (58). High Leeds Dependence Questionnaire total score, suggesting substance dependence tendency, was related to a low discontinuation rate (43). Higher total Hospital Anxiety and Depression Scale (HADS) score at baseline was a predictor for poorer response after four years (p < 0.005) (55). Poor baseline QoL, depression and anxiety are associated with poor outcome in terms of headache frequency. Socio-economic variables: being unmarried, unemployed, less frequent consumption of coffee, smoking and alcohol consumption are factors related to negative outcome and relapse (58,62). Baseline disease severity: Several factors that were associated with a greater severity of illness including more severe baseline headache, higher headache frequency, longer duration of migraine, and previous consultations with a greater number of specialists are negative outcome predictors (36,37,47,58,62). Other negative prognostic factors include: greater disability measured by MIDAS score (36), poorer self-reported sleep quality, and high self-reported bodily pain as measured by the QoL tool SF-36 (64).
The short- and long-term prognosis in MOH patients according to the class of drug overused
Longitudinal studies have suggested that medications containing barbiturates and opioids are associated with the highest risk of developing MOH, while triptans and NSAIDs are associated with lower risk (84–86).
The medications overused have changed significantly in the past 20 years. The relative proportion of patients overusing ergotamine and combination analgesics has decreased, and the number of people using triptans and simple analgesics continues to increase (87). Today, simple analgesics and triptans are the most frequent acute medications taken by patients with MOH (88).
Studies reporting the medication overused and the outcome of different types of drug overused.

Combination medications (COM) containing paracetamol or aspirin combined with caffeine.
Unsuccessfully treated: patients who had not improved after discontinuation or were returned to a pattern of medication overuse within one year.
MOH: medication-overuse headache; NSAIDs: nonsteroidal anti-inflammatory drugs.
Triptan
Triptan overusers account for 10%–40% of the MO population, and the frequency has been increasing over the past 20 years (87). Two months after abrupt discontinuation without preventive medication, triptan overusers had a significantly better outcome than those overusing other acute medications, in which about 50% triptan overusers had a 50%–100% reduction in headache frequency (14). For patients receiving a discontinuation protocol that contained preventive medication, triptan overusers had the best short- and long-term prognosis compared to patients overusing other medications: a. The time elapsed from the beginning of the early discontinuation to the resolution of MOH was the shortest (five days ± 2, p < 0.062) (35). b. The percentage who revert to an episodic headache pattern (78%) is higher than other acute medications at two months after early discontinuation (54) c. The reduction in headache days was the largest (52%, p = 0.022) (42). d. The one- and four-year relapse rates are the lowest. The relapse rates of patients overusing different medications at one year are: triptans (21%), ergots (27%) and analgesics (71%). The four-year relapse rate of triptan overusers remained around 21% (12,63). e. The percentage of patients having moderate to significant pain improvement after discontinuation was the greatest for triptan-related MOH (89).
Ergotamine
Only 3%–13% of MO patients are overusing ergotamine (87). The conflicting outcomes reported may be related to the small patient groups in each study. Some studies showed that ergot overusers had a better outcome with regards to a reduction in headache frequency and remission to episodic headache following discontinuation compared to those overusing other kinds of medication (14). Other studies indicated that ergotamine overuse was related to unsuccessful treatment and persistent overuse (36,59).
Simple and combination analgesic
Simple and combined analgesic overusers account for more than 50% of the MO population (87). More than 60% of analgesic users remained unchanged or got worse two months after abrupt discontinuation without preventive medication (14). The relapse rate was the highest in patients using analgesics compared to triptan and ergot overusers at six months, one year and four years after discontinuation and preventive medication (12). For patients overusing combination analgesics, one study reported a greater reduction in headache days (52%) compared with other medication types (42). However, another study showed a significantly higher relapse rate in patients overusing combination analgesics (33%, p = 0.03) compared to patients overusing other acute medications (62). For simple analgesics, similar controversial results have also been reported (47,61).
Butalbital
Most papers did not discuss butalbital as an independent category; butalbital-containing medication was classified into “combination analgesics” instead.
Opioid
Less than 10% of MO patients overused opioids in the studies reviewed. The actual rate of opioid overuse in the general population is likely to be higher since some studies intentionally excluded patients with opioid overuse. Among studies that included opioid overusers, patients with opioid overuse tend to have a less-favorable outcome with regard to headache frequency, relapse rate, and pain improvement compared to patients overusing other kinds of medications (9,62,89).
Discussion
The treatment of patients with MOH has generated considerable and ongoing debate (90,91). Given the high worldwide prevalence of both CM and MOH, and the enormous disability and suffering associated with both conditions, an evidence-based answer to this clinical question is a major clinical and research priority. This review provides a comprehensive overview of the evidence to support the various treatment strategies employed for the management of patients with MOH.
There is limited evidence to support the strategy of early discontinuation of acute medications without concomitant use of other preventive measures. In fact, in most instances where patients have a long history of overuse and near-daily headache, there is a high non-compliance rate with early discontinuation alone (13,26,48,92) and patients may initially develop several weeks of worsening symptoms such as more severe headache, nausea, vomiting, sleep disturbances, anxiety, and restlessness (93). The majority of studies in this systematic review were actually reporting results of early discontinuation plus preventive therapy, and showed encouraging results in reducing headache frequency and acute medication consumption, improving MIDAS score, QoL, anxiety and depression up to at least one year after discontinuation. However, the lack of control groups is a major limitation of those studies.
For preventive medications, the analyses for MO subgroups were performed as post hoc analyses of randomized clinical trials investigating topiramate and onabotulinumtoxinA for treatment of CM (15,20,72,82). The results suggest that some patients who have CM with MO will have success with immediate initiation of preventive therapy without early discontinuation of the overused acute migraine medication, thereby avoiding the possible discomfort associated with acute medication discontinuation. However, these results must be interpreted with caution since the onabotulinumtoxinA and topiramate studies have limitations when considering outcomes for patients who have MO. In addition, other factors associated with preventive medication use need to be considered, including the cost of such medications, side effects and adverse events associated with preventive therapy, long-term medication adherence, access to care, and availability of specialty care.
Regarding the drug overused, in most of the studies reviewed, butalbital and opioid overusers were relatively rare compared to patients overusing other medications or they were excluded (9,35,36). The topiramate studies included opioid but not butalbital overusers. In the PREEMPT trial, investigators were trained not to enroll patients who frequently used opioids as their acute headache pain medication.
However, there are several studies demonstrating successful discontinuation of patients overusing opioid (89) or butalbital-containing analgesics (35,89). Therefore, we believe that the conclusions of this article can possibly apply to patients overusing opioid or butalbital; however, more studies on the treatment of butalbital and opioid overusers are needed.
There are several pragmatic clinical issues that remain to be addressed within the context of controlled trials. First, the frequency with which acute medications (other than those being discontinued) can be used in patients with moderate or severe headaches during the withdrawal phase is unclear. In addition, the type of acute medication and route of administration that will provide the most effective relief without perpetuating headache in those in whom the MO is the actual cause of frequent headache is also unclear. There is evidence that NSAIDS and dihydroergotamine have the potential to prevent and reverse central sensitization in animal models and/or humans (94). Whether these medications will reduce withdrawal headache and minimize the risk of MOH and relapse is unclear. Second, there would be utility in having controlled studies that compare early discontinuation plus preventive medication vs preventive medication alone. However, there is a medical imperative to reduce or discontinue overused medications due to the potential for systemic toxicity. Therefore, based on the available evidence and sound medical practice, the discontinuation of overused acute medications (regardless of whether the overuse is the cause or consequence of frequent headache) combined with preventive medication and/or other strategies (biobehavioral therapy), should be the standard of care, as already suggested by guidelines from national authoritative organizations (8).
Since not all patients who by an arbitrary definition “over-use” acute medications have MOH, the development of a clinical serologic or imaging biomarker to distinguish those with true MOH will be a priority for future research as these patients may require a different treatment approach.
Conclusion
The level of evidence to support early discontinuation of overused medications alone for the treatment of MOH is low due to the absence of controlled studies. Adding preventive medication to early discontinuation led to a better outcome than early discontinuation alone, and preventive medication alone may be superior to early discontinuation alone. Triptan consumption was significantly reduced compared to other acute medications. Appropriately sized, randomized controlled trials evaluating the safety and long-term efficacy of preventive medication plus early discontinuation vs preventive medication alone vs early discontinuation alone are needed.
Clinical implications
The evidence to support the early discontinuation of overused medications is low because of the lack of a control group in studies. For patients with chronic migraine (CM) and medication overuse (MO), there are large randomized control trials supporting the use of onabotulinumtoxinA and topiramate without early discontinuation; however the evidence is limited since data were obtained from post hoc analysis. Considering current available evidence and the systemic toxicity of overusing acute headache medication, we suggest discontinuation of the overused medication with the addition of preventive medication. Studies on preventive medications plus early discontinuation vs preventive treatment alone vs early discontinuation alone are needed.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
Dr. Chia-Chun Chiang has nothing to disclose. Within the last 12 months, Dr. TJ Schwedt has had no personal financial relationships with companies that manufacture/sell migraine or other headache treatments. He has participated as a site investigator in clinical trials funded by eNeura, Boston Scientific, Alder Biopharmaceuticals, Labrys Biologics, Arteaus Therapeutics, and OptiNose US. Dr. Shuu-Jiun Wang has served on the advisory boards of Allergan, Daiichi Sankyo and Eli Lilly. He has received speaking honoraria from local companies (Taiwan branches) of Pfizer, Eli Lilly and GSK. He has received research grants from the Taiwan National Science Council, Taipei-Veterans General Hospital and Taiwan Headache Society. David W. Dodick MD, has served on advisory boards and/or has consulted for Allergan, Amgen, Alder, Arteaus, Pfizer, Colucid, Merck, ENeura, NuPathe, Eli Lilly & Company, Autonomic Technologies, Ethicon J&J, Zogenix, Supernus, Labrys, Boston Scientific, Medtronic, St. Jude, Bristol Myers Squibb, Lundbeck, Impax, MAP, Electrocore, Tonix, Novartis, Eli Lilly, Teva, Alcobra. Dr. Dodick has received funding for travel, speaking, editorial activities or royalty payments from: IntraMed, SAGE Publishing, Sun Pharma, Allergan, Oxford University Press, American Academy of Neurology, West Virginia University Foundation; Canadian Headache Society; Healthlogix, Wiley, Universal Meeting Management, WebMD, UptoDate, Oregon Health Science Center, Starr Clinical, Decision Resources, Synergy.
