Abstract
Introduction
The management of medication-overuse headache (MOH) is often difficult and no specific guidelines are available as regards the most practical and effective approaches. In this study we defined and tested a consensus protocol for the management of MOH on a large population of patients distributed in different countries.
Subjects and methods
The protocol was based on evidence from the literature and on consolidated expertise of the members of the consensus group. The study was conducted according to a multicentric interventional design with the enrolment of 376 MOH subjects in four centres from Europe and two centres in Latin America. The majority of patients were treated according to an outpatient detoxification programme. The post-detoxification follow-up lasted six months.
Results
At the final evaluation, two-thirds of the subjects were no longer overusers and in 46.5% of subjects headache had reverted back to an episodic pattern of headache. When comparing the subjects who underwent out-patient detoxification vs those treated with in-patient detoxification, both regimens proved effective, although the drop-out rate was higher in the out-patient approach.
Conclusions
The present findings support the effectiveness and usability of the proposed consensus protocol in different countries with different health care modalities.
Keywords
Introduction
Medication-overuse headache (MOH) is a common disorder that affects as much as 2% of the general population (1–6). MOH management is a challenge because the disease is potentially treatable, but, after treatment, the risk to relapse back to chronic headache (at least 15 days/month) and drug overuse is typically high, with an ample variability ranging from 26% to 60% (7–17). No established consensus for treatment strategies exists, probably as a consequence of differences in the definition of the disorder, of therapeutic approaches, of types of primary headache, and because of study design issues (18).
Withdrawal and/or detoxification from the overused drug(s) is generally considered an important step in the management of MOH patients; however, consensus protocols as well as multicentre studies confirming this general belief are lacking in the literature.
The objective of this study was to define and test a consensus protocol that foresees the following steps: 1) advice to withdraw overused drugs, 2) detoxification from overused drugs and 3) optional prophylactic treatment of primary headache(s) in a large population of MOH patients. It is anticipated that such a protocol, if proven practicable and effective, will improve the efficiency of MOH management across different countries. The protocol has been developed and tested within the activities of the COMOESTAS Project, funded by the European Commission, FP7 within Call 5 ‘Toward sustainable and personalized healthcare’.
Subjects and methods
A consensus protocol (attached file) for the management of MOH was devised by an expert group (CT, RJ, LB, ZK, ML, JAL, GS, GS, GN, PR) during several meetings, following a thorough literature search addressing a list of questions (see attached deliverable ‘D1.5 Development of a common clinical protocol’, also available at https://www.comoestas-project.eu/page.action?id = 6164). The protocol was based on consolidated clinical expertise and publication records of the members of the group, as well as on data from the literature (19–22), and it foresaw in-patient and/or out-patient detoxification programmes associated with optional prophylactic treatment and regular follow-up visits over a period of six months. The protocol was tested according to a multicentric interventional study design which was non-randomised and non-controlled. A total of six centres participated in the study: four located in Europe (Italy, Denmark, Germany, Spain) and two in Latin America (Argentina, Chile), which were asked to enrol at least 50 MOH patients per centre. Because of local regulations, only three of these centres (Italy, Germany and Spain) had the possibility to apply the in-patient regimen. In these three centres the criteria for selecting IN- vs OUT-patient approach were: i) long distance from the hospital and/or ii) patient’s preference. Enrolment started in January 2009 and was completed in December 2010. A total of 489 subjects were screened and 376 MOH subjects (302 females (F), 74 males (M)) were enrolled.
Reasons for non-inclusion in the protocol were:
– Drop-out between V1 and V0 – Patients’ referral with a wrong diagnosis (i.e. post-traumatic headache, chronic cluster headache, etc.). This was particularly true for the centres lacking a long tradition in headache management; – Failure of previous detoxification procedures (which was an exclusion criterion in the present protocol). This was instead particularly relevant for centres with a long tradition in headache management. Characteristics of the population. MO: migraine without aura; MA: migraine with aura; CM: chronic migraine; M: migraine of any type; TTH tension-type headache. aSome patients received multiple diagnoses.
The main characteristics of the study population are given in Table 1.

The flowchart of the study is illustrated in Figure 1. Following the baseline visit (visit 0), patients with a history of MOH (headache on at least 15 days/month associated with overuse of one or more classes of symptomatic drugs for at least three months) (23) were asked to fill out a paper diary (24,25) for one month and subsequently return to the centre for visit 1. At visit 1, if anamnestic MOH was confirmed prospectively by the diary data, patients were enrolled in the study, detoxified according to the protocol (see Appendix) and scheduled for a subsequent visit (visit 2) in two months. During visit 2 clinical conditions and diary data were evaluated and the patients were scheduled for the final visit in four months (visit 3). A telephone update was performed halfway between visit 2 and visit 3 (indicated as ‘contact’ in Figure 1).
Flowchart of the study.
Based on the national health care service regulations, detoxification was performed according to one of the following modalities:
– In-patient detoxification, performed either as daily hospitalisation or as regular multi-day hospitalisation (Group 1), with the direct administration of drugs; – Out-patient detoxification with advice on how to perform the detoxification at home and prescription of drugs to be used (Group 2);
The study was approved by the local ethics committees from all centres except from Denmark, where the local ethics committee stated that approval was not necessary because the study did not foresee any new pharmacological treatment. Participants gave informed consent before taking part.
Inclusion criteria are listed below:
Diagnosis of MOH according to the revised diagnostic criteria of the International Headache Society (23); Age between 18 and 65 years; Capability of filling out the headache diary.
Exclusion criteria are listed below:
Current diagnosis of co-existent, significant and complicating medical or psychiatric illnesses; Significant overuse of ‘pure’ opioids (patients overusing combination drugs containing opioids are allowed), benzodiazepines and barbiturates; Overuse of alcohol and other drugs of addiction; Current treatment with migraine prophylactic drugs; Inefficacy of previous adequate detoxification programmes; Inability to provide reliable information about medical history; Pregnancy or breast feeding.
The main outcome measures were:
– Number of headache days/month over the six-month observation period; – Number of days of acute drug intake/month over the six-month observation period.
The primary endpoints where the number of headache days/month and the number of days with symptomatic drug intake/month in each month of the observation period as compared to baseline.
The secondary outcome measures were:
– Percentage of subjects who were non-completers, where ‘non-completer’ is defined as a subject who failed to accomplish the detoxification protocol or did not return for the follow-up visits during the six-month observation period; – Percentage of subjects cured, where ‘cured’ is defined as a subject who no longer overused at the end of the protocol; – Percentage of responders, where ‘responder’ is defined as a subject who reverted to an episodic pattern of headache (<15 days/month of headache) and no longer overused at the end of the protocol; – Percentage of relapsers, where ‘relapser’ is defined as the subject who reverts back to overuse between months 2 to 6, after stopping overusing during month 1; – Number of migraine days over the six-month observation period. Main characteristics of the two study groups. MO: migraine without aura; MA: migraine with aura; CM: chronic migraine; M: migraine of any type; TTH tension-type headache. aSome patients received multiple diagnoses.
Table 2 illustrates the demographic and clinical characteristics of the two study groups.
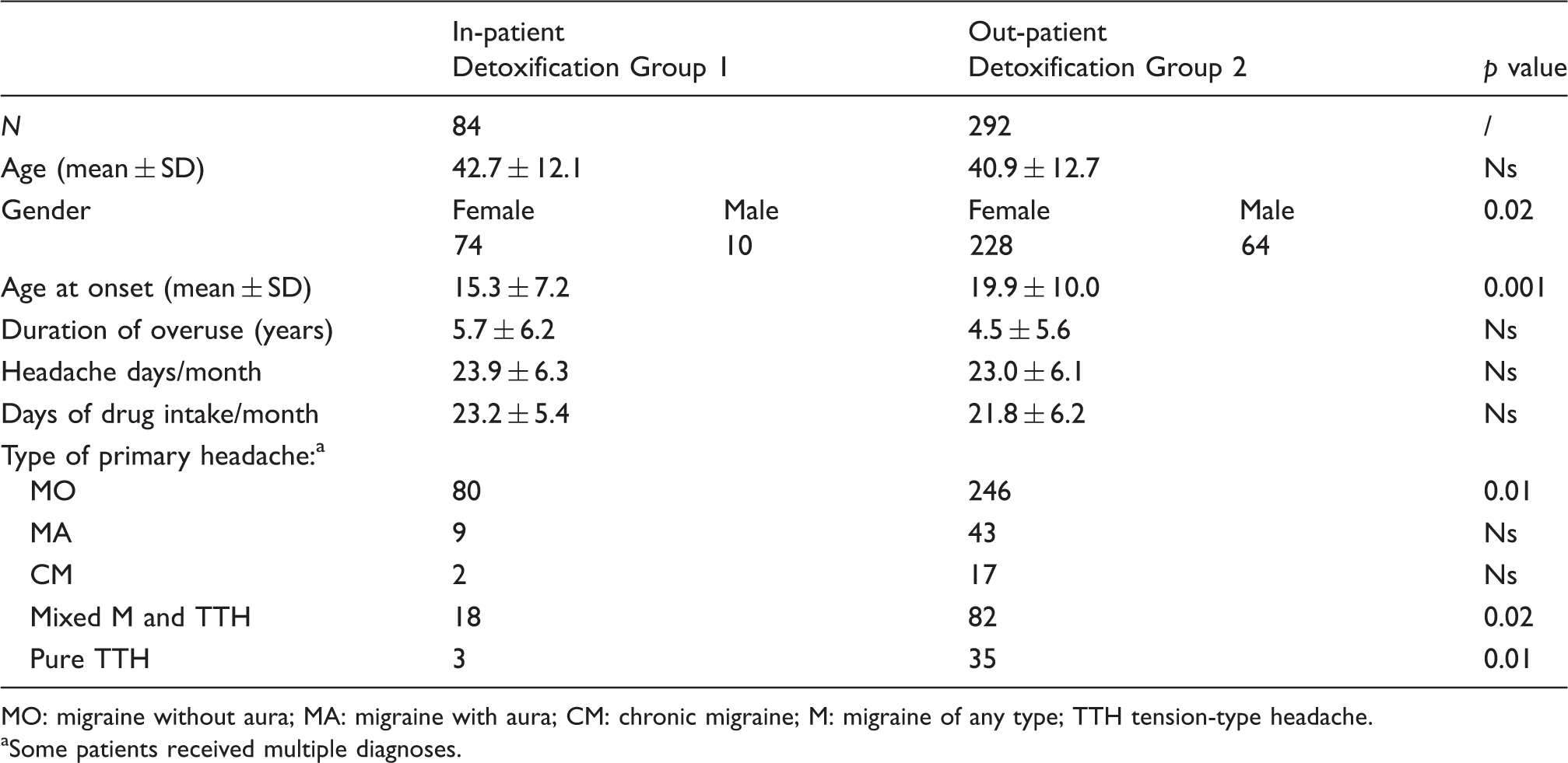
Please note that the diagnosis of chronic migraine was made a posteriori, after the publication of the International Classification of Headache Disorders, third edition, beta (ICHD-III beta) criteria (26), using the evaluation of the diaries compiled during the month preceding detoxification.
Internal validity (integrity) of the data collected in the different centres was optimised by means of the local online collection and update of data on an electronic patient report form, devised within the Comoestas Project, which ensured correctness and completeness of data, while protecting patient privacy with an encoding system. Data were entered in the system by ad hoc trained personnel. As regards external validity (generalisability) of data, this was addressed by the subsequent enrolment of patients who sought attention at headache centres located in different countries, with different health systems and slightly different drug availability.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) for Windows, version 19.0, was used for the calculation. For each qualitative and continuous variable the first step was to assess whether the distribution was normal. In particular we tested: age, age at onset, duration of overuse (years), number of headache days/month at baseline and number of days of acute drug intake/month at baseline (the last two were considered main outcome of the study as previously described). We evaluated ‘skewness’, ‘kurtosis’ and
The data regarding the secondary outcome variables were analysed and presented in terms of intention-to-treat (ITT, patients who were enrolled in the study) and according to per-protocol subpopulations (DT: patients who completed at least detoxification and SC: patients who completed the entire study).
Results
Total group
A total of 376 MOH were enrolled (also see Figure 1). Of these, at the moment of enrolment, 168 also suffered from chronic migraine (44.6%) according to the ICHD-III beta criteria. Of the 376 subjects, 321 completed detoxification (85.4%): a total of 266 were also prescribed prophylactic treatment (82.9%), while for the remaining 55 (17.1%) a prophylactic medication was not prescribed because either it was considered unnecessary by the doctor or according to the patient’s preference. The complete dataset was available for 257 out of the 321 patients (80.1%) who completed the detoxification protocol. A total of 119 subjects (119/376, 31.7%) dropped-out from visit 1 to visit 3 either because they did not complete the detoxification protocol (
A marked reduction in both main outcome measures was already observed during the first month of the six-month observation period (M1). The amplitude of reduction tended to further increase over the following five months (M2 through M6) (Figures 2 and 3).
Days of headache during the six-month study period. Data are represented as mean ± standard deviation. M: month. * Days of drug intake during the six-month study period. Data are represented as mean ± standard deviation. M: month. *

No significant differences in the main outcome measures were observed when stratifying patients by prophylactic medication (prescribed/non-prescribed) or by type of prophylactic medication. Similarly, no significant differences emerged when patients were stratified based on the type of acute drug overused.
Secondary outcome measures for the total population.

(a) Intention-to-treat analysis.
(b) Sub-population analysis: Detoxification completed.
(c) Sub-population analysis: Protocol completed.
The analysis of secondary outcome measures in the group of subjects who completed detoxification is illustrated in Table 3(b), which shows that 72.9% of subjects were cured and 54.5% were responders. In the subpopulation of subjects who completed the study (Table 3(c)), the percentage of cured subjects rose to 91.1% and the number of responders to 68.1%.
In-patients vs out-patients
Eighty-four MOH patients were detoxified during multi-day hospitalisation (Group 1, in-patients), while the majority of subjects (292) performed detoxification at home (Group 2, out-patients). In order to control for the different distribution of tension-type headache as primary headache in the two groups (Table 2), we performed the statistical analyses described in Figures 4 and 5 and in Table 4 after excluding the patients whose primary headache was tension type headache (pure or mixed with migraine). In this way, our ITT population was formed by 238 subjects: 64 in Group 1 and 174 in Group 2. Sixty-three patients in Group 1 completed the detoxification (98.4%), while in Group 2, detoxification was completed by 153 of the 174 subjects (87.9%) (Group 1 vs Group 2, Days of headache during the study period in the two groups of patients. IN: patients who were hospitalised for the detoxification. OUT: patients who were detoxified as out-patients (see text for further details). Data are represented as mean ± standard error. M: month. Days of drug intake during the study period in the two groups of patients. IN: patients who were hospitalised for the detoxification part of the study. OUT: patients who were detoxified as out-patients (see text for further details). Data are represented as mean ± standard error. M: month. * Secondary outcome measures for the in-patients (Group 1) and the out-patients (Group 2). (a) Intention-to-treat analysis. (b) Sub-population analysis: Detoxification completed. (c) Sub-population analysis: Protocol completed.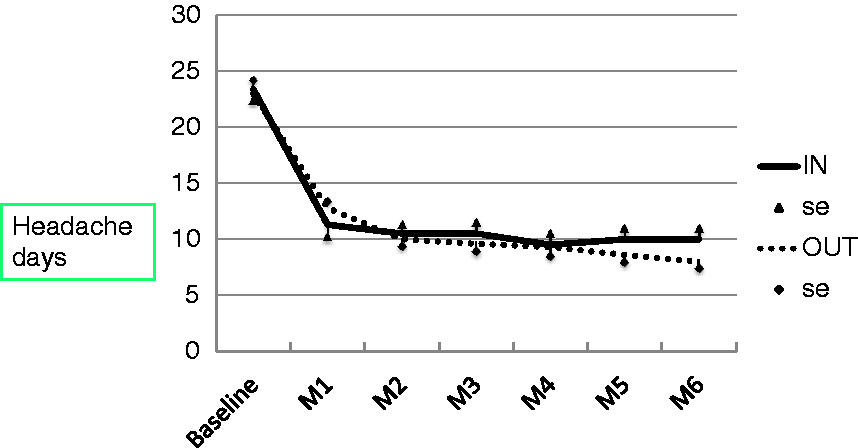


A marked reduction in both main outcome measures was observed already during the first month and persisted over the following five months in both groups (Figures 4 and 5).
Out-patient detoxification seemed as effective as in-patient detoxification, performing a little less than in-patient detoxification only for the first month of observation and only as regards the days of drug intake (Figure 5).
As regards the secondary outcome measures, in the ITT analysis (Table 4(a)), a significantly higher percentage of subjects dropped out in Group 2 (Group 1: 12.5%; Group 2: 33.9%,
The analysis of secondary outcome measures in the group of subjects who completed detoxification is illustrated in Table 3(b), which shows that the percentage of cured is above 70% in both groups, with a similar percentage of responders (Group 1: 55.6%; Group 2: 61.8%, NS) and of relapsers (Group 1: 14.3%; Group 2: 7.6%, NS). In the subpopulation of subjects who completed the study, the percentage of cured subjects rose to 83.9 in Group 1 and to 90.4% in Group 2 (Group 1 vs Group 2 NS), with a higher rate of responders in Group 2: 62.5% in Group 1, 75.5% in Group 2 (
Discussion
The findings obtained in the present study demonstrate the applicability and the efficacy of a consensus protocol for managing MOH patients in a large population of subjects from different countries. The association of advice to withdraw overused acute medications, detoxification from overused medication and, in the majority of cases, prophylactic treatment for the primary headache affecting the MOH subjects allowed the removal of overuse in two-thirds of subjects and the reversion of the headache pattern from chronic to episodic in 46% of them.
It is noteworthy that detoxification was performed according to an out-patient regimen in the majority of MOH patients and that this out-patient regimen proved almost as effective as the detoxification obtained according to an in-patient regimen. Altogether, these findings strongly suggest that the protocol for managing MOH that we developed and tested was efficient, low-cost and adaptable to different care settings. Cost analysis of the proposed method will be the subject of a separate, detailed publication.
No general consensus exists as regards the best treatment approach to MOH, mostly because of the lack of published prospective studies aimed at evaluating the efficacy of specific treatment approaches on large populations of MOH patients. Detoxification from the overused drugs is generally considered an important step in the management of MOH. Grande et al. showed in a group of 109 MOH subjects that a brief intervention consisting of simple, unstructured advice to refrain from using acute medications for headache was associated with a reduction in headache index of 24% (27). Bigal et al. showed retrospectively that detoxification proved successful in nearly 70% of a population of 479 subjects with chronic headache and drug overuse (20). Zeeberg et al. showed that detoxification resulted in the improvement of headache, quantifiable in a frequency reduction of 46%, in 45% of 221 MOH patients (28).
Recently, however, the findings of some studies conducted for evaluating the efficacy of topiramate or onabotulinumtoxinA in chronic migraine patients – who were in more than 60% of cases also overusers of acute drugs – were interpreted to suggest indirectly that detoxification might not be a necessary step in the management of MOH, and a debate has been ongoing among experts as to whether the first step in MOH approach should be detoxification or rather it is more time and cost saving to put patients directly on prophylactic medication (29–33). These studies, mostly controlled clinical trials, are not methodologically comparable with the present one – open-label, not controlled nor randomised – although it seems that the size effect of prophylactic medication alone or botulinum toxin A injection may be lower, based on the published data (30–33) than the effect recorded in the present study: –14.3 ± 2.1 headache days per month and –15.5 ± 4.2 days of drug intake at month 6. This, along with the potential risk imposed by the excessive and/or continued use of acute drugs, suggests that discontinuation of overused drugs is a very important step in the management of MOH.
The rate of drop-outs in the present study was above 30%, apparently higher than that reported in other papers on the management of MOH with detoxification and prophylactic treatment (12,21,34), although direct comparison between studies is not granted because of the different duration of follow-ups. Of course, this high rate of drop-outs suggests caution in the interpretation of results, because it may reflect either the lack of compliance due to a good response (i.e. headache markedly improved) to the intervention or the lack of compliance due to a negative effect of the intervention (i.e. lack of improvement, lack of drug tolerability, adverse effects, etc.). However, it is noteworthy that in our population, the drop-out rate was higher in the out-patient setting, where it was associated with the failure to complete properly the detoxification part of the protocol in at least 50% of cases. Conversely, once the detoxification is completed, it seems that the outpatient regimen provides a better performance in terms of responders. This latter finding probably reflects a selection of the most motivated/compliant subjects: i.e. those who successfully complete the detoxification at home, are also more likely to comply with the protocol and with the prescriptions in the following six months; conversely, those who are ‘assisted’ in the in-patient detoxification may face more difficulties in following the prescription during the follow-up at home and probably tend to use more symptomatic drugs. Taken together with the observed slightly better outcome in the in-patient group in terms of percentage of cured, the present findings suggest two important clinical considerations: 1) close monitoring of the patients, especially during the detoxification phase, positively influences the completion of detoxification and the outcome of disease and 2) when a patient fails an out-patient detoxification programme, a second detoxification should be attempted with the in-patient level of care.
Methodological considerations
A major strength of the study is the large group of homogeneous MOH patients, the multinational design, the prospective recording of major variable by means of a specific headache diary and the follow-up time of six months.
A few limitations of the present study must be taken into account in the evaluation of results. First, we selected MOH patients who were naïve to detoxification and who did not overuse opiates. This might of course have influenced positively the outcome of our patients, but it was a necessary step to obtain a homogeneous population of MOH subjects for testing the proposed protocol. Furthermore, these authors believe that complicated MOH subjects, i.e. those overusing opiates, those who have failed previous detoxification protocols or those with clinically relevant psychiatric comorbidity, should be closely followed during the detoxification on an in-patient basis, in agreement with previous reports (35).
Second, patients were distributed to one intervention group during hospitalisation or to the other out-patient basis in a non-randomised way. The resulting disparity of distribution was, however, exactly within the scope of the present study, which relied on the demonstration of the applicability and the usefulness of a clinical protocol for the management of MOH in different settings.
Finally, the lack of a control group might represent a limitation of the study. However, the enrolled patients had had chronic headache for more than five years on average and therefore it seems highly unlikely that the reported improvement might be attributed to any other relevant factor than the tested intervention. Furthermore, inclusion of a control group would pose ethical problems regarding the delay of effective treatment for patients who often have already been waiting for quite some time before being admitted to a headache centre.
Future studies may address these limitations by means of controlled studies which will be aimed at dissecting out the placebo effect associated with the detoxification protocol and the relative effect of detoxification alone vs the effect of prophylactic medications.
Conclusions
Summarising, the present findings demonstrate in a large population of MOH subjects that the adoption of the proposed detoxification protocol in association with optional prophylactic treatment induces a marked improvement in clinical conditions. The protocol is effective when detoxification is performed either as an in-patient procedure or as an out-patient regimen. With this work we provide a tool for the management of MOH that has been tested in several countries, it has proved effective over the six-month observation period and it has proved suitable for different cultural and marketing contests. In more detail, in this large multicentric and multinational study we show that more than 60% (more than 90% in the per-protocol SC analysis) of MOH are overuse free at six months and 46% of them (68% in the per-protocol SC analysis) revert back to an episodic pattern of headache (with a reduction in headache frequency >50%). The in-patient regimen is associated with a lower rate of drop-outs and a higher incidence of subjects who are cured from overuse.
Clinical and public health relevance
Overuse of medication needs to be addressed in medication-overuse headache (MOH). Detoxification is effective in the majority of MOH patients. A consensus protocol for the management of MOH is provided here. Application of this protocol in a large population of MOH patients proved effective.
Footnotes
Funding
This work was supported by COMOESTAS Project – EC contract number 215366 (COMOESTAS) FP7 – Thematic priority ICT.
Conflict of interest
None declared.
Appendix
a
Botulinum toxin was not listed here because the protocol was devised in 2008, before the publication of the PREEMPT studies.
