Abstract
Background
Post-traumatic headaches are a common sequela of mild traumatic brain injury (concussion). It is unclear whether or how these headaches differ phenotypically from primary headaches.
Objective
Determine whether there is an overarching unobserved latent trait that drives the expression of observed features of post-traumatic headache and other headaches.
Methods
Data from this
Results
Of 1094 soldiers with headaches, 198 were classified as having post-traumatic headache. These headaches were compared to those in the other soldiers (647 without concussion history and 249 with concussion history). Soldiers with post-traumatic headache had greater endorsement of all 12 headache features compared to the soldiers with non-concussive headaches. The confirmatory factor analysis showed good model fit (χ2 (51) = 95.59,
ClinicalTrials.gov identifier NCT01847040
Introduction
Post-traumatic headaches (PTH) are a common sequela of civilian and military concussive injuries (1–7). The International Classification of Headache Disorders (ICHD) (8) provides symptom-based case definitions for headache disorders that have been broadly adopted in epidemiologic studies and clinical trials. The ICHD includes classification criteria for primary headaches such as migraine and tension-type headache – the two most common primary headache disorders – and for secondary headaches such as PTH (8). Due to incomplete understanding of PTH symptomatology, the diagnostic criteria are not as well-developed for these headaches. Moreover, there are no FDA approved therapies specifically for post-traumatic headaches and no evidence-based treatment guidelines (6,9,10). PTH are often treated based on the evidence-base for the headache phenotype they most resemble (10,11) which is most commonly a migraine-like headache (12–14). However, non-migraine features, notably continuous headache, are often reported by these patients (13,15,16).
In a prior epidemiologic study of a cohort of recently deployed soldiers (14), we compared the prevalence of individual headache characteristics (e.g. pulsatility, pain level, presence of aura, etc) in soldiers with PTH to soldiers with non-concussive headaches and found that all features assessed were more prevalent or more severe in the PTH group. We hypothesize that there is a single latent trait that underlies the constellation of observed headache symptoms and features. A latent trait is one that is not observed directly but can be inferred based on other observed data. Examples of latent traits include intelligence, quality of life, and depression. In the present study, we used confirmatory factor analysis to identify and measure this trait (which we named “headache complexity” [HC]) based on the observed prevalence and levels of 12 headache features reported in the same soldier cohort. We used structural equation modeling to compare HC between the soldiers with PTH and those with non-concussive headaches. Finally, we determined whether the HC score had prognostic value in terms of predicting which soldiers would access medical care for headache in the year following return from deployment.
Methods
Study design and setting
Study subjects are from the Warrior Strong Study (NCT01847040), an observational study of recently deployed soldiers returning to two large Army bases (Fort Carson, CO and Fort Bragg, NC) conducted between 2010 and 2015. Details of the study methodology are provided in earlier publications (17,18). Briefly, nearly 25,000 soldiers were screened for a history of mild traumatic brain injury (mTBI = concussion) sustained during their recent deployment. Deployments typically were one year. Screening was conducted by non-uniformed study personnel during routine post deployment health assessments conducted within a few days of return. Screen positive (mTBI+) and screen negative (mTBI−) soldiers were randomly invited to participate, over-sampling screen positive soldiers. Soldiers were excluded if they had been medically evacuated from theater (likely indicates more severe TBI) or if they were returning from areas other than Iraq or Afghanistan (to limit the study population to soldiers who had been deployed to a combat zone). The Warrior Strong cohort consists of 1567 baseline participants (1010 mTBI− and 557 mTBI+). Follow-on interviews were conducted at 3-month, 6-month, and 12-month intervals, by telephone or in-person when possible. Recruitment details including comparisons between the recruited study population and the total screened population are described in an earlier publication (18). The institutional review boards of Fort Carson, CO, and Fort Bragg, NC provided primary study oversight.
Participants
The focus of the present study is the self-administered headache phenotype interview completed at the baseline visit as previously described (14). The analysis sample consists of n = 1094 soldiers with self-reported headaches. The soldiers were grouped based on concussion history and headache attribution into three groups, as illustrated in Figure 1:

Categorization of 1049 soldiers with headache into three groups based on concussion history and headache attribution.
mTBI− (not concussed)
mTBI+/PTH− (concussed, does not attribute headaches to an injury]
mTBI+/PTH+ [concussed, attributes headaches to an injury)
For brevity, these groups are hereinafter referred to as mTBI−, PTH−, PTH+.
Variables
Demographics
Demographic variables used in the present analysis include age, gender, and race/ethnicity
Headache phenotyping
Headache phenotyping for this analysis is based on headache data collected at the baseline visit, as previously described (14). Briefly, headaches symptoms were assessed with a detailed self-administered questionnaire. The questionnaire is a modification of a validated headache interview developed by Stewart and Lipton and used in prior studies of non-clinical populations (19–21). Features assessed were consistent with International Classification of Headache Disorders (ICHD-3 beta) (22) criteria and included the following 12 items: Unilateral location, photophobia, phonophobia, nausea, exacerbation of headache by routine physical activity, pulsatility, visual aura, sensory aura, pain level, continuous headache, allodynia, and monthly headache days. All but two of the items were captured on a binary scale (absent/present). Pain level was captured on an 11-point scale (0–10) and headache frequency (captured as days per week, per month, or per year) was converted to days per month (0–30).
Headache attribution
Soldiers indicated if their headaches had changed during deployment and if so follow up questions asked if the change was associated with a particular event (e.g. illness, injury, etc). Textual analysis of the perceived cause of headache was done blinded to case status. Headaches that were attributed to an injury were classified as PTH as previously described (14).
Other measures
Medical encounter data for the year following return from deployment was available for 92% of participants. We identified medical encounters for headache (ICD-9-CM 346.x, 339, 307.81, 784, 784.0) and migraine (ICD-9-CM 346.x) within the military healthcare system. We included ambulatory or inpatient medical encounters in any diagnostic position. Self-reported receipt of any rehabilitation services was available at the one-year follow-up visit from 47% of participants.
Statistical analysis
Data management, descriptive summaries, and logistic regression analyses were conducted in SAS 9.4. Latent variable modeling, confirmatory factor analysis (CFA), and structural equation modeling (SEM) were conducted in Mplus version 8.1. Statistical analysis was conducted in three phases.
Phase 1: Measuring headache complexity (HC)
The 12 observed headache features from the baseline interview were used to test the conceptualization of Headache Complexity as a single latent trait using confirmatory factor analysis (CFA). The headache days variable was log transformed for model fitting. Models were fitted to all available non-missing data (no imputation) using diagonally weighted least-squares estimation with mean and variance adjustments (WLSMV), which utilizes a pairwise present approach (23). Model fit was evaluated using the comparative fit index (CFI), the Tucker-Lewis index (TLI), and the root mean square error of approximation (RMSEA), using customary cut-offs for adequate fit of 0.95 or greater for the CFI and TLI and less than 0.08 for the RMSEA (24–28). A single underlying HC factor was evaluated with three pairs of Path diagram representation of final 12-item unidimensional confirmatory factor analysis model.
Phase 2: Soldier group differences in latent headache complexity
Soldier group differences on the HC factor were tested using SEM, where the latent HC factor was regressed on soldier group status (reference coded). Effect sizes from this model were quantified using standard mean differences (SMDs), or Cohen’s d (29,30). Models used all available data.
Phase 3: External validation of headache complexity scores
Predictive validation for the baseline HC score was assessed using the 1-year outcome measures (medical encounters for headache or migraine and receipt of rehabilitation services). External validation of the HC scores was conducted using logistic regression models where one or more medical encounters for headache or migraine by the 1-year follow-up served as dependent variables, and HC scores at baseline was the primary predictor with age, gender, race, and headache group (mTBI−, PTH−, PTH+) serving as covariates, with these covariates selected
Results
Descriptive data
Description of the study population (n = 1094 soldiers with headaches).

Legend
mTBI− Screen negative for concussion.
PTH− Screen positive for concussion, does not attribute headaches to an injury.
PTH+ Screen positive for concussion, attributes headaches to an injury.
Note: Values are based on all available non-missing data; missing data < 1% for all items.
Phase 1: Measuring latent headache complexity (HC)
Results from the single factor CFA model showed good fit to the data, χ2 (51) = 95.59,
Single factor headache complexity confirmatory factor analysis results.
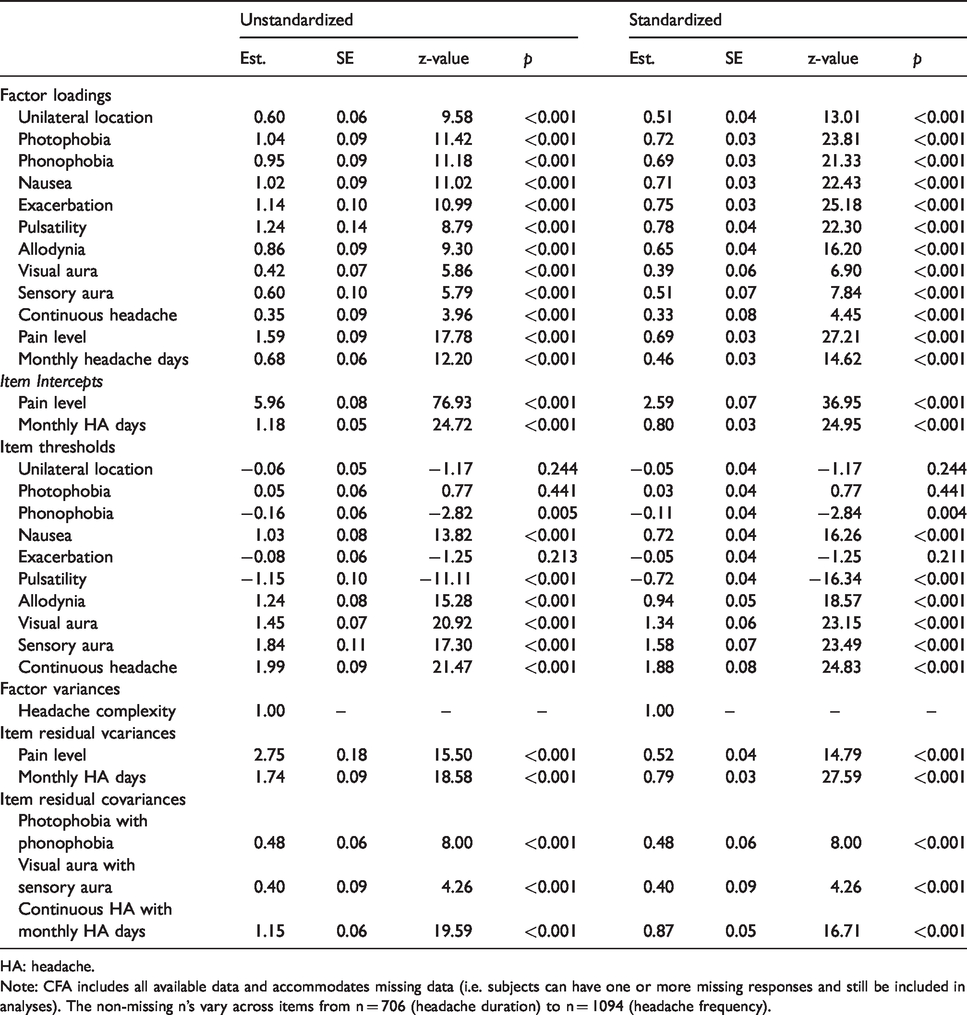
HA: headache.
Note: CFA includes all available data and accommodates missing data (i.e. subjects can have one or more missing responses and still be included in analyses). The non-missing n’s vary across items from n = 706 (headache duration) to n = 1094 (headache frequency).
Phase 2: Soldier group differences in latent headache complexity
Building on the CFA model from Phase 1, an SEM was estimated where soldier headache group predicted HC score as measured by the 12 observed headache features. The model demonstrated good fit, χ2 (73) = 184.60, Visualizing headache complexity effect sizes across soldier groups.
Phase 3: External validation of headache complexity scores
Table 3 provides the results from the logistic regression analyses using baseline HC scores as a predictor of medical encounters at the 1-year follow-up. Overall, results showed that, controlling for age, sex, race, and headache group, higher HC scores predicted increased odds of medical encounters for both headache and migraine. Specifically, a one standard deviation increase in HC score at baseline was associated with almost doubled odds of one or more headache medical encounters (OR = 1.87, 95% CI: 1.49–2.35,
Logistic regression model results: Prediction of medical encounters for headache and migraine in the year following return from deployment.
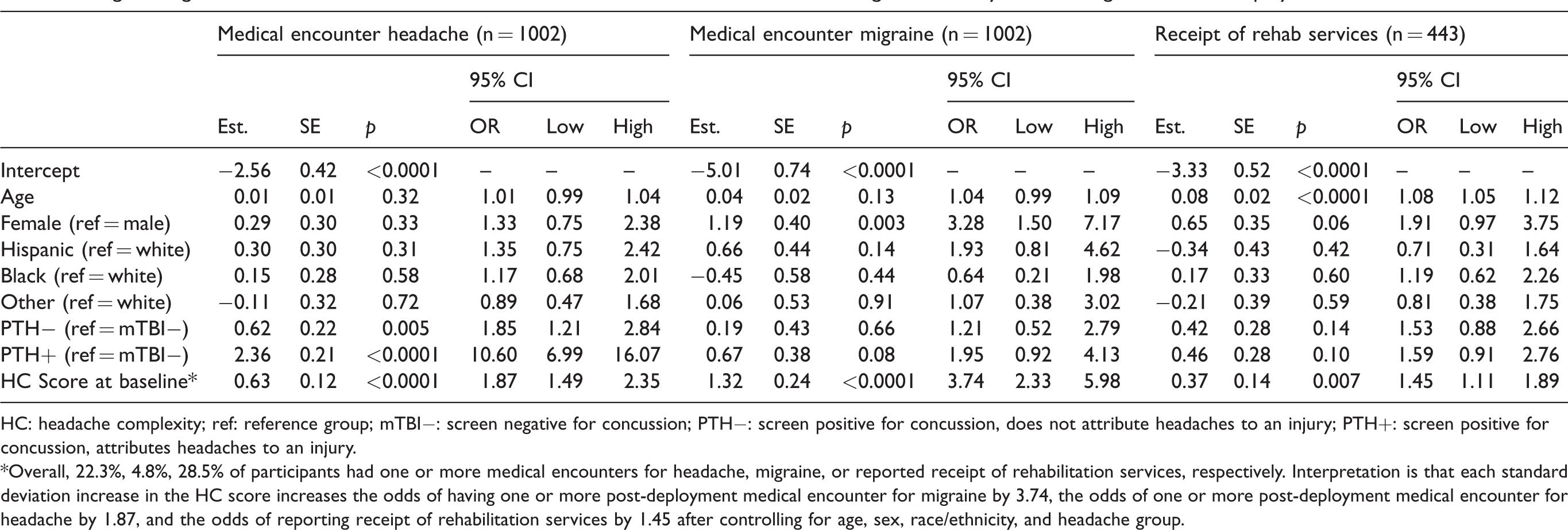
HC: headache complexity; ref: reference group; mTBI−: screen negative for concussion; PTH−: screen positive for concussion, does not attribute headaches to an injury; PTH+: screen positive for concussion, attributes headaches to an injury.
*Overall, 22.3%, 4.8%, 28.5% of participants had one or more medical encounters for headache, migraine, or reported receipt of rehabilitation services, respectively. Interpretation is that each standard deviation increase in the HC score increases the odds of having one or more post-deployment medical encounter for migraine by 3.74, the odds of one or more post-deployment medical encounter for headache by 1.87, and the odds of reporting receipt of rehabilitation services by 1.45 after controlling for age, sex, race/ethnicity, and headache group.
Discussion
PTH are a common post-concussive symptom for injuries sustained in civilian and military environments. The ICHD classification criteria for headaches related to injuries to the head are non-specific, characterizing PTH only in relation to time of onset of headaches since injury and their chronicity (e.g. lasting more than 3 months) (8). There are limited phenotypic data on post-traumatic headaches in non-clinical populations and most prior studies have characterized PTH phenotypes using the relatively crude ICHD classification, finding that the most common PTH phenotype resembles migraine (12–14). A greater understanding of PTH phenotypes, as distinct from primary headaches, may help provide clues to pathophysiology, characterize the natural history of PTH, and inform the design of treatment trials with, potentially, novel inclusion criteria and outcome measures.
In the present study, we took an exploratory approach to consider whether concussive and non-concussive headaches differ on a theoretically-informed single latent trait (HC), defined by a constellation of observed headache features. Although latent traits such as HC are rarely discussed in headache research, they provide valuable insights for clinicians and researchers. Headache can be characterized by a multitude of interrelated symptoms and latent variable modeling provides a parsimonious way to represent and quantify individuals’ levels on the overarching construct (e.g., HC) that gives rise to the observed symptoms. Results suggested that the magnitude of the HC score measuring this trait differed between concussive and non-concussive headaches and predicted the likelihood of accessing medical care for headache – presumably a marker of greater headache severity and impact on healthcare services. There are no other studies to our knowledge that have taken a similar approach to characterize PTH phenotypes, although such studies have been done for primary headaches (31–34).
There are several potential implications from the present findings as well as questions raised. First, the confirmatory factor analysis identified a single latent trait (HC) that included all observed symptoms, although there was variability in the strength of association for individual symptoms. In other words, the results suggest that a single latent trait/factor adequately represented the data. Furthermore, the single trait was valid across all headache groups, although the HC score was greatest in the group with PTH. This suggests that PTHs differ phenotypically from non-concussive headaches by severity rather than by kind, at least based on the symptoms measured. We hypothesized that a single latent trait would adequately describe the headache phenotype data. While outside the scope of this paper, the one-factor solution was examined within each subgroup and was found to fit the data well. This finding provided us with increased confidence that the parsimonious one-factor solution adequately described all groups examined here and was not unduly influenced by any single subgroup.
As the headache symptoms assessed were mostly, but not exclusively, symptoms of migraine, does this suggest that PTHs are in some sense a type of acquired migraine in susceptible individuals (6,35)? If so, since migraine has a strong familial or genetic component, we would expect to see enrichment of migraine in family members of individuals who develop chronic headache
This study has several strengths. The study population was recruited outside a clinical context, so presumably is more representative of the spectrum of PTH expression compared to a study population recruited from patients seeking medical care. Our soldier cohort included a well-matched comparison group with presumed non-traumatic headaches. While the study population (being primarily young men) may not generalize to the civilian population, headache disorders are relatively understudied and possibly under-diagnosed (39) in men – who are more likely to sustain head injuries than women (40). We characterized headaches using a detailed interview and grouped features based on confirmatory factor analysis rather than using the standardized headache classification. However, our interview was biased in the sense that it included primarily migraine features. Future studies should include a broader range of headache symptoms including novel symptoms. Other limitations include the exploratory nature of the analysis and lack of information on family history or prior migraine diagnosis.
In summary, we used confirmatory factor analysis to determine whether a constellation of observed headache symptoms differentiates between post-traumatic and non-traumatic headaches and predicts accessing medical care for headache. Results suggest that PTHs differ, phenotypically, from primary headaches by severity rather than kind, based on the symptoms assessed. Future research is needed to replicate this measure of HC and generalize findings to other post-traumatic headache populations.
Clinical implications
We hypothesized that headaches of different etiology (post-concussive vs. non-concussive) differ on an overarching latent trait based on observed headache features, which we named “headache complexity”. In a cohort of 1094 recently deployed soldiers, scores based on this latent trait differed between post-concussive and presumed non-concussive headaches and predicted the likelihood of accessing medical care for headache
Footnotes
Acknowledgments
The authors thank active duty service members for their service; the soldier–volunteers who collaborated in this study by donating their time across several waves of data collection; and key study personnel, including Jennifer Johnson, Donna Dennis, Dr Lonnie Nelson, Denise Strasser, Lisa Betthauser, Mary Alice Dale, and Gurvaneet Sahota.
The views expressed in this article are those of the authors and do not necessarily reflect the official policy of the Uniformed Services University of the Health Sciences, the US Public Health Service, the United States Army, the Department of Health Affairs, or the Departments of Defense or Veterans Affairs, or the US Government.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: In the past three years, AIS has served on an advisory board for Allergan and currently receives research support from Eli Lilly. She is an associate editor for
JSM has received funding, research support, salary, or other compensation from Vector Psychometric Group LLC (employment), the American Headache Society (speaking and teaching), Amgen, Inc. (research support),
RJW has received funding, research support, salary, or other compensation from Vector Psychometric Group LLC (employment), the American Headache Society (speaking and teaching), Amgen, Inc. (research support),
RBL is the Edwin S. Lowe Professor of Neurology at the Albert Einstein College of Medicine in New York. He receives research support from the NIH: 2PO1 AG003949 (multiple principal investigators), 5U10 NS077308 (principal investigator), R21 AG056920 (investigator), 1RF1 AG057531 (site principal investigator), RF1 AG054548 (investigator), 1RO1 AG048642 (investigator), R56 AG057548 (investigator), U01062370 (investigator), RO1 AG060933 (investigator), RO1 AG062622 (investigator), 1UG3FD006795 (multiple principal investigators), 1U24NS113847 (investigator), K23 NS09610 (mentor), K23AG049466 (mentor), K23 NS107643 (mentor). He also receives support from the Migraine Research Foundation and the National Headache Foundation. He serves on the editorial board of
LAB has consulted for major league sports teams. KS, HT, and WRC report no disclosures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Primary funding for the study was provided by the Congressionally Directed Medical Research Programs. Additional support was provided by the Center for Neuroscience and Regenerative Medicine, Defense Medical Research and Development Program, the Defense and Veterans Brain Injury Center, and the Military Clinical Neuroscience Center of Excellence.
