Abstract
Objective
To investigate which part of the autonomic system is mainly involved and assess the sensitivity of face sympathetic skin response in cluster headache.
Material and methods
The study sample consisted of 19 drug-free cluster headache patients (16 males, three females) and 19 healthy volunteers. Demographic features and pain characteristics were thoroughly identified. Dysautonomic symptoms were evaluated during attack and remission periods of cluster headache patients. Orthostatic hypotension, R-R interval variation and sympathetic skin responses obtained from the face and four extremities were evaluated and the sensitivity of face sympathetic skin responses was assessed in contrast to extremity sympathetic skin responses.
Results
All sympathetic skin responses of face and extremities could be obtained during attack and remission periods. On the symptomatic side, mean latency of face sympathetic skin responses was longer compared to the asymptomatic side and controls (p = 0.02, p = 0.004). There were no differences in latency or amplitude of extremity sympathetic skin responses between symptomatic and asymptomatic sides and controls. No significant relationship was determined between sympathetic skin responses, R-R interval variation, orthostatic hypotension and cluster headache clinical features.
Conclusion
Sympathetic hypoactivity of the face seems to predominate the pathophysiology of cluster headache. Face sympathetic skin responses might be more sensitive compared to extremity sympathetic skin response in demonstrating dysautonomic symptoms in cluster headache patients.
Keywords
Introduction
Cluster headache (CH) has been defined as a primary headache localized unilaterally to the first division of the trigeminal nerve and characterized by autonomic findings that might present as lacrimation, rhinorrhea and chemosis during attacks with seasonal recurrences (1–4). Features of these autonomic disturbances constitute the diagnostic criteria for CH described by the International Headache Society (IHS) in 2004; however, the pathophysiology has not been elucidated entirely. Studies have reported that CH is associated with central or peripheral autonomic dysfunction (5,6). Straube et al. have suggested that there is an imbalance between sympathetic and parasympathetic innervations of the face in CH patients (7).
Other types of primary headaches might also show characteristics of autonomic involvement. For instance, a dysfunction of the sympathetic nervous system has been detected in migraine; however, several studies have presented incongruent data (8–11). Forehead sympathetic skin responses (SSRs) in primary headaches have first been investigated by Yildiz et al., who detected sympathetic hyperfunction during migraine attacks and sympathetic hypofunction afterwards (12). However, few electrophysiological studies have been performed to evaluate the autonomic function in CH patients using the Valsalva manoeuver, orthostatic blood pressure changes (OBPC) or SSRs in extremities (13,14). The circadian rhythm and period between attacks has not been evaluated in CH and SSRs have been determined only in extremities (13).
The objective of this study was to determine which part of the autonomic system is mainly involved in CH by examining dysautonomic parameters including OBPC, R-R interval variation (RRIV) and SSRs of both face and four extremities during attack and remission periods. Additionally, assessment of the sensitivity of face SSR in contrast to extremity SSR was a secondary study objective.
Material And Methods
Subjects
Sixteen male and three female patients (mean age: 38.9 ± 8.9 years) presenting to the headache outpatient’s clinic of our hospital between March and October 2012 and diagnosed with CH were recruited prospectively. The diagnosis of CH was established by IHS 2004 criteria. The control group consisted of 19 healthy subjects (16 males/three females) and mean age was 36.3 ± 8.1years (range from 21 to 49 years). The objective and content of the study was explained to all subjects in detail and written informed consent was obtained from all subjects. The study was approved by the Institutional Ethics Committee and conducted in complete accordance with the Declaration of Helsinki.
All participants were examined by a neurologist and routine blood chemistry was performed. In the patient group, age of disease onset, duration of illness, average number of attacks per year, average duration of cluster attacks in days, average number of attacks per day, duration of pain, seasonal periodicity, circadian rhythms, smoking history and precipitant factors were questioned. Autonomic symptoms of CH attacks such as conjunctival injection, lacrimation, rhinorrhea, periorbital oedema, sweating, miosis, ptosis and agitation were also assessed. A cranial CT or magnetic resonance imaging was performed in all patients to exclude secondary causes of CH.
Blood pressure, SSR and RRIV were recorded only once in the control group; whereas, all tests were performed twice, i.e. in attack and remission periods, in the patient group. A cluster of attacks of headache lasting for at least2 days with at least one to eight attacks per day was defined as an attack period. An attack was defined as a severe cluster type headache lasting 15 to 180 minutes followed by a short duration of relief until the strike of the next attack within the same cluster. The period between 3 months before and 2 months after an attack was identified as a remission period. Testing could be performed during an attack period in 15 patients. None of the patients had been receiving prophylactic medications in the last 3 months. Both patients and control subjects were instructed not to receive tobacco, tea, caffeine or other food or drugs containing sympathomimetic agents on the day of investigation.
Orthostatic blood pressure changes
Blood pressures were measured at supine position after 10 minutes of resting and upright position after 3 minutes of standing. The orthostatic hypotension (OH) definition of the American Autonomic Society and American Academy of Neurology that required ‘a decrease of ≥20 mmHg in systolic and a decrease of ≥10 mmHg in diastolic blood pressure when a person assumes a standing position’ was used (15).
Sympathetic skin responses
All electrophysiological tests were performed with the Dantec Keypoint EMG/NCS/EP System device. Recordings were obtained at supine position in a quiet, air-conditioned room maintained at 22–24℃. SSRs were recorded from face, hands and feet. Since cluster is a trigeminoautonomic headache, we used the forehead region for face SSR recordings to cover the first division of the trigeminal nerve and compared them with extremity recordings.
For face SSR recordings, active electrodes were placed on the lateral of the frontal bone and reference electrodes were placed approximately 3–4 cm laterally and 2 cm downwards. For extremity studies, responses were acquired with active disc electrodes placed in the middle of palms and soles and reference electrodes placed on the dorsum of hands and feet while the subject was in supine position. Electrical stimulation (0.2 second duration and 30–100 mA intensity) was applied on the median nerve at the wrist on the contralateral of the recording. Filter band pass was adjusted at 0.5–2 kHz, sensitivity was 0.5 mV and sweep speed was 5–10 seconds. In order to overcome habituation, randomized electrical stimulation was applied at a minimum of 30-second and 20-second intervals in extremity and forehead studies, respectively. The onset latency and peak-to-peak amplitude of SSRs were evaluated. Our device automatically calculated mean peak-to-peak amplitudes rather than negative-peak and positive-peak amplitudes. Thus, and also to avoid any confusion in our calculations, we did not use the positive rapid and negative slow components of SSR separately. Ten consecutive stimuli were administered in each forehead and extremity region. Stimulus intensity was increased if no response was obtained. If at least five consecutive 100 mA electrical stimuli could not elicit a consistent change larger than 50 μV from baseline, the result was recorded as absent SSR and SSR was deemed pathological only if no response was recorded.
R-R interval variation
RRIV was recorded after 10 minutes of resting in a supine position and during forced deep hyperventilation (6/second). Active and reference disc electrodes were placed on both palms. Filter band pass was kept at 5--100 Hz, sensitivity was 1 mV and sweep speed was 500 ms. Average of RRIV at rest and deep hyperventilation were termed %R and %D, respectively. According to Oh’s definition, difference (%D–%R) and ratio (%D/%R) of these two values were calculated automatically by Keypoint device (16).
Statistical analysis
All statistical analyses were performed using the Statistical Package for Social Sciences 17.0 (SPSS). Descriptive statistics and quantitative data with normal distribution were compared between the groups with one-way analysis of variation variant analysis, Pearson’s correlation test and independent t test; whereas Kruskal–Wallis test, Spearman’s correlation test and Mann-Whitney U test were performed for nonparametric data. A chi-square test was performed for qualitative data and Pearson’s correlation coefficients were calculated for relations between all independent measures. A p value <0.05 was considered statistically significant.
Results
Clinical characteristics of cluster headache patients.

Spring and/or autumn.
Only three patients (15.8%) had OH during attack period. OH was not found in any healthy controls or patients with CH during remission period.
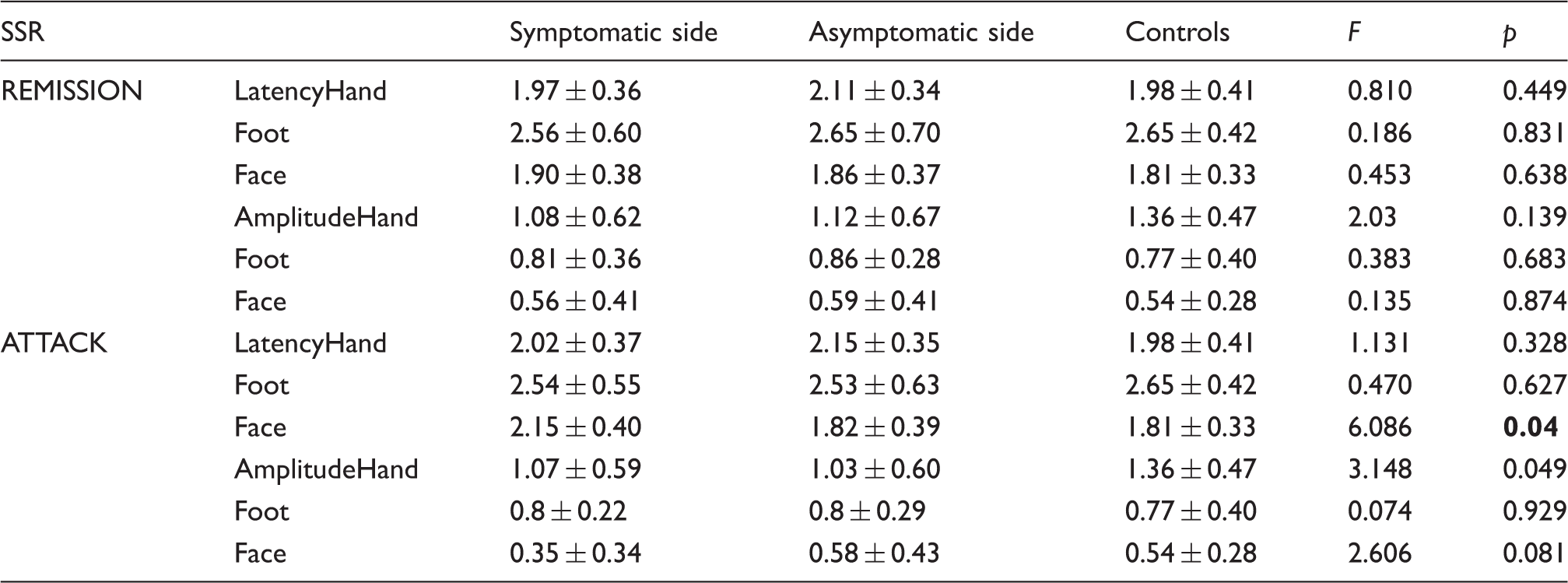
SSR of hands, feet and face were obtained in all controls and patients during attack and remission periods (Figures 1 and 2). There was no statistically significant difference in the mean values for latency and amplitudes of hand, foot and face SSR between controls and asymptomatic and symptomatic sides of CH patients during remission periods (Table 2). During attack periods, face SSR latencies and hand SSR amplitudes were significantly different between symptomatic sides, asymptomatic sides and controls (p = 0.04, p = 0.049, respectively). Post hoc analyses showed that face SSR latencies on the symptomatic side were significantly longer compared to asymptomatic sides and controls (p = 0.02, p = 0.004, respectively). However, hand SSR amplitudes were not significantly different between symptomatic and asymptomatic sides and controls in post hoc analysis (p = 0.74, p = 0.142, p = 0.084, for symptomatic and asymptomatic sides, symptomatic sides and controls, asymptomatic sides and controls, respectively). No significant difference was determined in foot SSR latencies and amplitudes, hand SSR latencies and face SSR amplitudes between symptomatic and asymptomatic sides and controls during attack periods (Table 2). Moreover, face SSR amplitudes were lower and latencies were longer during attack, compared to remission periods (p = 0.070, p = 0.021, respectively) (Figure 3). There was only a weak correlation between periorbital oedema and SSR amplitudes of face (r = 0.457). No significant correlation was determined between the other autonomic symptoms of CH and SSR parameters.
SSR recordings of face, hand and foot in a control subject. SSR recordings of face, hand and foot in a patient during a left-sided attack. Face SSR values on symptomatic side during attack and remission periods. SSR results during remission and attack periods of cluster headache patients and controls.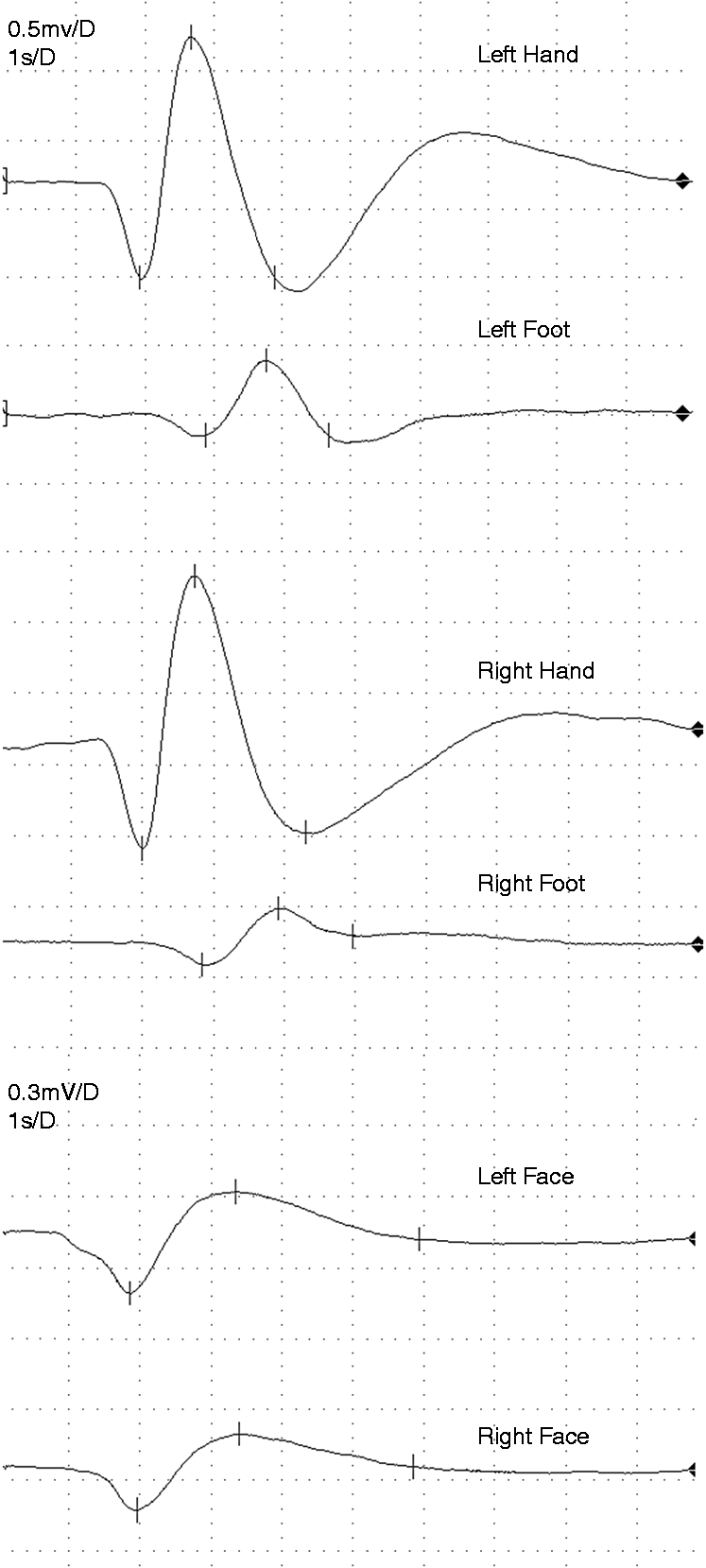


There was no significant difference in RRIV at rest and deep hyperventilation between controls and CH patients during attack periods (p = 0.482, p = 0.152). No significant difference was found in RRIV at rest and deep hyperventilation in either group during remission periods (p = 0.479, p = 0.179, respectively). Similarly, no significant differences were found in RRIV at rest and deep hyperventilation between attack and remission periods of CH patients (p = 0.957, p = 0.409, respectively). There was no significant correlation between RRIV parameters and autonomic symptoms.
Discussion
Our study of the interictal autonomic function in CH demonstrated that face SSR latencies were significantly longer on symptomatic compared to the asymptomatic side during attack periods. Additionally, face SSR amplitudes were lower on the symptomatic side during the attack period; although not to a statistically significant level. Together with the lack of difference either in extremity SSR or parasympathetic parameters of autonomic system, our results suggested that sympathetic hypoactivity might clinically be more prominent and face SSR might be more sensitive than extremity SSR in detecting the autonomic dysfunction in CH.
Trigeminofacial brainstem reflexes are thought to generate pain and autonomic symptoms in CH. Of these, conjunctival injection and lacrimation are related to parasympathetic activation; miosis and ptosis are due to sympathetic hypofunction (17–21). The overall accepted theories of autonomic features of CH have focused on parasympathetic hyperactivation or sympathetic paralysis (22,23). While the provoking factor of this dysfunction is yet unclear, it has been suggested that changes in the trigeminovascular system are not a cause, rather a result. Painful stimuli to cranial tissues stimulate nociceptive receptors and lead to trigeminal parasympathetic vasodilation, lacrimation, salivation and rhinorrhea. During cluster attacks, sympathetic fibres are thought to be injured by a neurogenic inflammation in large intracranial vessels (24). It has been suggested that collateral sprouting of parasympathetic fibres make functional connections with these defective sympathetic fibres and initiate vasodilation once again (25). Moreover, levels of calcitonin gene related peptide, nitric oxide and vasointestinal polypeptide have been found to be elevated during CH attacks and suggested to be associated with trigeminal and parasympathetic activation, respectively (26). Functional imaging studies in spontaneous or nitroglycerin triggered CH attacks also demonstrated the role of the hypothalamus, suggesting an involvement throughout the central pathway rather than a peripheral drive of the trigeminofacial reflex, probably via trigeminohypothalamic pathway in the brainstem (25,27).
Sympathetic skin response has been used to evaluate pre- and post-ganglionic non-myelinated C fibres. Although autonomic functions of CH have been investigated mainly in pharmacological studies, there are also few electrophysiological studies. SSRs have been evaluated in CH patients only in two former studies. Meineri’s study has commented about the presence or absence of SSRs in limbs and the authors have reported no significant change in the presence of SSRs in CH patients in active and remission periods (13). Evers et al. have examined hand SSR in primary headache patients during the interictal period (28) and reported prolongation of hand SSR latencies in CH patients without any change in SSR amplitudes compared to healthy subjects and controls with low back pain. On the other hand, the authors have found no difference in latencies and amplitudes of SSRs on symptomatic sides compared to asymptomatic sides in CH patients, similar to other primary headache patients. Similarly, we also did not find any significant difference in latencies and amplitudes of hand and foot SSRs of CH patients during attack and remission periods.
Autonomic dysfunction in primary headache patients has been investigated by different electrophysiological studies, such as SSR and RRIV. In CH, sympathetic and parasympathetic findings (lacrimation, rhinorrhea, chemosis, sweating, flushing etc.) mainly occur in the face (29). Therefore, recording SSR from the face rather than extremities might be more elucidative in the evaluation of autonomic dysfunction in CH. In the literature, there are two SSR studies recording only from extremities, but there are no face SSR studies in CH patients (13,28). Additionally, Yildiz et. al. have investigated autonomic skin responses in normal subjects with electrodes placed on the forehead, upper lip, post-auricular area and neck and suggested that any autonomic dysfunction could be demonstrated more easily if face and neck regions were used to record SSR (30). In a successive study, authors have investigated forehead SSR in migraine patients and found sympathetic asymmetric hypofunction in attack and interictal periods and sympathetic hyperfunction in post-attack periods (12,30). Likewise, we chose the method of Yildiz et al. to examine autonomic involvement in CH and evaluated SSR of the forehead region to cover the first division of trigeminal nerve in CH patients. Our results demonstrated that face SSR amplitudes were lower and latencies were longer on symptomatic sides during attack compared to remission periods. Possible explanations of the altered face SSR might include decreased sympathetic response or sweat gland capacity, more uniform response or stronger tonic sympathetic activity, as well as peripheral effects of sympathetic activation and altered sweat gland functioning due to sprouting of parasympathetic efferents. Few comparable studies have previously been performed that record SSR from face and neck. Sariahmetoglu et al. have studied forehead SSR in contrast to extremity SSR to demonstrate autonomic involvement in Parkinson’s disease (PD) and reported that forehead SSR is more sensitive than extremity SSR in determining autonomic nervous system dysfunction early in PD due to either preferential degeneration of autonomic nuclei projecting to face or shorter peripheral course (31). Contrary to the degenerative process in PD, a functional change is known to occur in CH attacks. Examining face SSR during the attack should be a better indicator of any sympathetic involvement; however, the intenseness of pain in CH and ethical reasons allowed us to perform examinations only in the attack period, rather than during the intensely painful attack itself.
Moreover, RRIV was studied for the electrophysiological evaluation of parasympathetic involvement in CH patients and no significant changes were determined during attack and remission periods. Meineri et al. have also evaluated parasympathetic cardiovascular affection during active compared to silent periods (13) and found no changes in extremity SSR and postural blood pressure. Contrary to our study, Meineri et al. have concluded that parasympathetic impairment was more prominent than orthosympathetic derangement.
Evaluation of the systemic autonomic dysfunction has provided inconclusive results in CH patients in literature. DeMarinis et al. have investigated ECG (Holter) and blood pressure variations and found that blood pressure is increased and heart rate is decreased in all CH patients during attack period. Authors have reported that sympathetic system seems to be involved in the initial development of the attacks, pursued by the activation of parasympathetic system (32). On the other hand, Meineri et al. have reported that parasympathetic but not sympathetic involvement is prominent in CH patients. (13). Van Vliet et al. have studied cardiovascular autonomic function tests and not found any difference between attack and non-attack periods (14). Similarly, we found that only three patients had OH during the attack period. Therefore, our results suggested to us that sympathetic hypoactivity of the face predominates the pathophysiology of the autonomic disturbance in CH, rather than a systemic involvement. However, it should also be noted that further studies involving more extensive systemic autonomic function tests are needed to verify our suggestion.
Our study had certain limitations including the small sample size and examination of the patients during the cluster periods only. Since the excruciatingly severe pain of cluster attack prevented us from performing the study examinations during an attack, patients were investigated outside of an attack during the cluster period.
Consequently, we believe that evaluation of facial autonomic dysfunction may help illuminate the pathophysiology of CH. In this study, we did not find any significant changes in SSR recordings from extremities, but observed prolongation in latencies and decrease in amplitudes in face SSR recordings and therefore suggested that SSRs recorded from the face during attack periods might be more sensitive than extremity recordings for demonstrating dysautonomic symptoms in CH patients. Although we infer that sympathetic hypoactivity is clinically more prominent in CH, it is also possible that improvement of the methods of evaluation of parasympathetic involvement might provide different results. Recent studies have suggested sphenopalatine ganglion (SPG) as the major efferent peripheral parasympathetic structure in CH and proposed this structure as the main target in treatment (31–33). We believe, if examination of the SPG had been possible by electrophysiological methods, it might help us to better understand the pathophysiology of CH.
Clinical implications
Sympathetic hypoactivity is clinically more prominent in cluster headache. Sympathetic skin responses recorded from face are more sensitive than those recorded from extremities for demonstrating autonomic dysfunction involved in the pathophysiology of CH. Electrophysiological evaluation of sphenopalatine ganglion should be the next step in understanding the pathophysiology of CH.
Footnotes
Funding
This work received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
