Abstract
Abstract
Background and Objectives
Increased sensitivity to light and patterns is typically associated with migraine, but has also been anecdotally reported in cluster headache, leading to diagnostic confusion. We wanted to assess whether visual sensitivity is increased ictally and interictally in cluster headache.
Methods
We used the validated Leiden Visual Sensitivity Scale (L-VISS) questionnaire (range 0-36 points) to measure visual sensitivity in people with episodic or chronic cluster headache: (i) during attacks; (ii) in-between attacks; and in episodic cluster headache (iii) in-between bouts. The L-VISS scores were compared with the L-VISS scores obtained in a previous study in healthy controls and participants with migraine.
Results
Mean L-VISS scores were higher for: (i) ictal vs interictal cluster headache (episodic cluster headache: 11.9 ± 8.0 vs. 5.2 ± 5.5, chronic cluster headache: 13.7 ± 8.4 vs 5.6 ± 4.8; p < 0.001); (ii) interictal cluster headache vs controls (5.3 ± 5.2 vs 3.6 ± 2.8, p < 0.001); (iii) interictal chronic cluster headache vs interictal ECH in bout (5.9 ± 0.5 vs 3.8 ± 0.5, p = 0.009), and (iv) interictal episodic cluster headache in bout vs episodic cluster headache out-of-bout (5.2 ± 5.5 vs. 3.7 ± 4.3, p < 0.001). Subjective visual hypersensitivity was reported by 110/121 (91%; 9 missing) participants with cluster headache and was mostly unilateral in 70/110 (64%) and ipsilateral to the ictal pain in 69/70 (99%) participants.
Conclusion
Cluster headache is associated with increased ictal and interictal visual sensitivity. In contrast to migraine, this is mostly unilateral and ipsilateral on the side of the ictal pain.
Introduction
Increased sensitivity to light and patterns is a recognized feature of migraine, occurring in up to 90% of patients during attacks and, less known, in 60% of patients in-between attacks (1–5). Increased sensitivity to light has also been anecdotally reported in cluster headache, sometimes leading to diagnostic delay (6,7). The prevalence, severity, possible laterality, and timing (ictal, interictal, inter-bout) of visual hypersensitivity in cluster headache are unknown (8,14,15).
The Leiden Visual Sensitivity Scale (L-VISS) questionnaire was developed and validated to quantify self-reported visual sensitivity to light and patterns among participants with migraine (5). The L-VISS questionnaire has been used to quantify visual sensitivity in migraine subtypes, visual snow and chronic pain (5,9,10). Here, we use the L-VISS questionnaire to assess visual sensitivity in patients with episodic cluster headache (ECH) or chronic cluster headache (CCH) during and in-between attacks and bouts, to estimate its prevalence, severity and possible laterality, and to compare it with that in healthy controls and people with episodic or chronic migraine during and outside attacks.
Methods
Design
The study consisted of several steps. The initial study population consisted of 264 patients with ECH (n = 158) or CCH (n = 106) from the Leiden University Cluster Headache Neuro Analysis Programme (LUCA), who had previously participated in a study on the prevalence of visual aura in cluster headache (11). These patients were all sent a validated and highly sensitive screening questionnaire to diagnose and exclude people with comorbid migraine and/or eye disorders (i.e. blindness, macular degeneration, cataract) that could influence L-VISS scores (12). Patients who, based on their responses and an algorithm based on the IHCD III Criteria, were suspected of having comorbid migraine and/or severe eye disease, were called to verify these diagnoses by telephone (13). If one or both diagnoses were confirmed, the patients were excluded from further investigation. Four reminders were sent out and non-responders were contacted by phone.
Remaining participants with verified ECH or CCH, with or without visual aura, but without comorbid migraine and/or serious eye disorders, then received the L-VISS Questionnaire. This is a 9-item questionnaire about light and pattern sensitivity (score range 0–36 points), designed to quantify self-reported visual sensitivity and previously validated in people with migraine (5). A 5-point Likert-type response scale was used per question: Not at all (0 points), Slightly (1 point), Moderately (2 points), Severely (3 points) and Very severely (4 points). The questionnaire also included a few additional questions about whether participants felt they experienced visual hypersensitivity and, if so, whether this was bilateral or unilateral, and contralateral or ipsilateral to the ictal autonomic symptoms and pain (always, mostly, no preferred side). Participant were instructed to complete the L-VISS questionnaire outside attack for how they experience visual sensitivity (i) during attacks; (ii) in-between attacks (for those with ECH during a bout); and (iii) for those with ECH, out-of-a-bout (when free of attacks for at least one month). When answers were unclear or ambiguous, participants were contacted by phone for clarification.
For illustrative purposes, the L-VISS scores were compared with L-VISS scores from healthy controls (n = 86) and ictal and interictal scores from people with episodic migraine with aura (n = 67) or without aura (n = 66) that were obtained in a previous study (5). Healthy controls had no personal history of nor first-degree relatives with migraine or trigeminal autonomic cephalalgia.
Standard Protocol Approvals, Registrations and Patient Consent
The study was approved by the local ethics committee (METC of the Leiden University Medical Center). All participants provided written informed consent prior to inclusion.
Statistical analysis
Statistical analyses were performed using SPSS Statistics version 23. A linear mixed model was fitted on the L-VISS scores. The repeated-measures factor was set to compare ictal an interictal scores. Five fixed factors were included 1) diagnosis: episodic vs chronic cluster headache; 2) attack status: ictal or interictal; 3) aura status: cluster headache with vs without aura; 4) sex: male vs. female; 5) use of preventative treatment: yes vs. no; and 6) number of autonomic symptoms. Age was included as a covariate.
Another linear mixed model was performed in which the repeated measures factor was set to compare ictal, interictal and control scores. Two fixed factors were included 1) attack and disease status: controls vs ictal CH vs interictal CH and 2) sex: male vs. female. Age was included as a covariate.
A univariate general linear model (GLM) was performed to compare L-VISS scores between controls, participants with ECH out-of-bout and participants with CCH in-between attacks. Age and sex were included as covariates.
Finally, a separate analysis was performed for male and female participants. A linear mixed model was performed in which the repeated measures factor was set to compare ictal, interictal and control scores. Attack and disease status was included as a fixed factor: control vs ictal CH vs interictal CH. Age was included as a covariate.
Baseline subject characteristics and L-VISS scores are reported as mean and SD. Independent t-tests and χ2 tests were used for comparison of baseline characteristics when appropriate. For all analyses, a two-tailed p-value lower than 0.05 was deemed significant. A Bonferroni correction was used in post-hoc analyses to correct for multiple testing.
Data Availability
Anonymized data not published within this article will be made available by reasonable request from any qualified investigator.
Results
Participants
Of the invited patients from the initial study on visual aura in cluster headache 188/230 (71%) completed the screening questionnaire, after which n = 34 (18%) were excluded, mainly because of comorbid migraine (n = 23) or eye disorders (n = 7) (Figure 1) (11). No difference in age, sex or cluster headache subtype was observed between responders and non-responders. Of the remaining 154 participants, n = 95 had ECH (n = 11 with visual aura) of which n = 75 (79%; n = 11 with aura) completed the L-VISS, and n = 59 had CCH (n = 9 with visual aura) of which n = 55 (93%; n = 10 with aura) completed the L-VISS. Of these 73/75 (97%) with ECH and 48/55 (87%) with CCH completed the additional questions regarding whether they subjectively experienced visual hypersensitivity and whether this was bilateral or unilateral.

Response flowchart.
Data of 75 participants with ECH (of whom 11 (13%) with aura) and 55 participants with CCH (of whom 9 (16%) with aura) were analyzed and compared with n = 86 controls and n = 133 people with episodic migraine (n = 67 migraine with aura; n = 66 migraine without aura) from a previous study (Table 1) (5). Participants in the control group and migraine groups were younger and more often female in comparison to the cluster headache groups. Participants with ECH more frequently used preventive medication than participants with CCH.
Baseline characteristics.
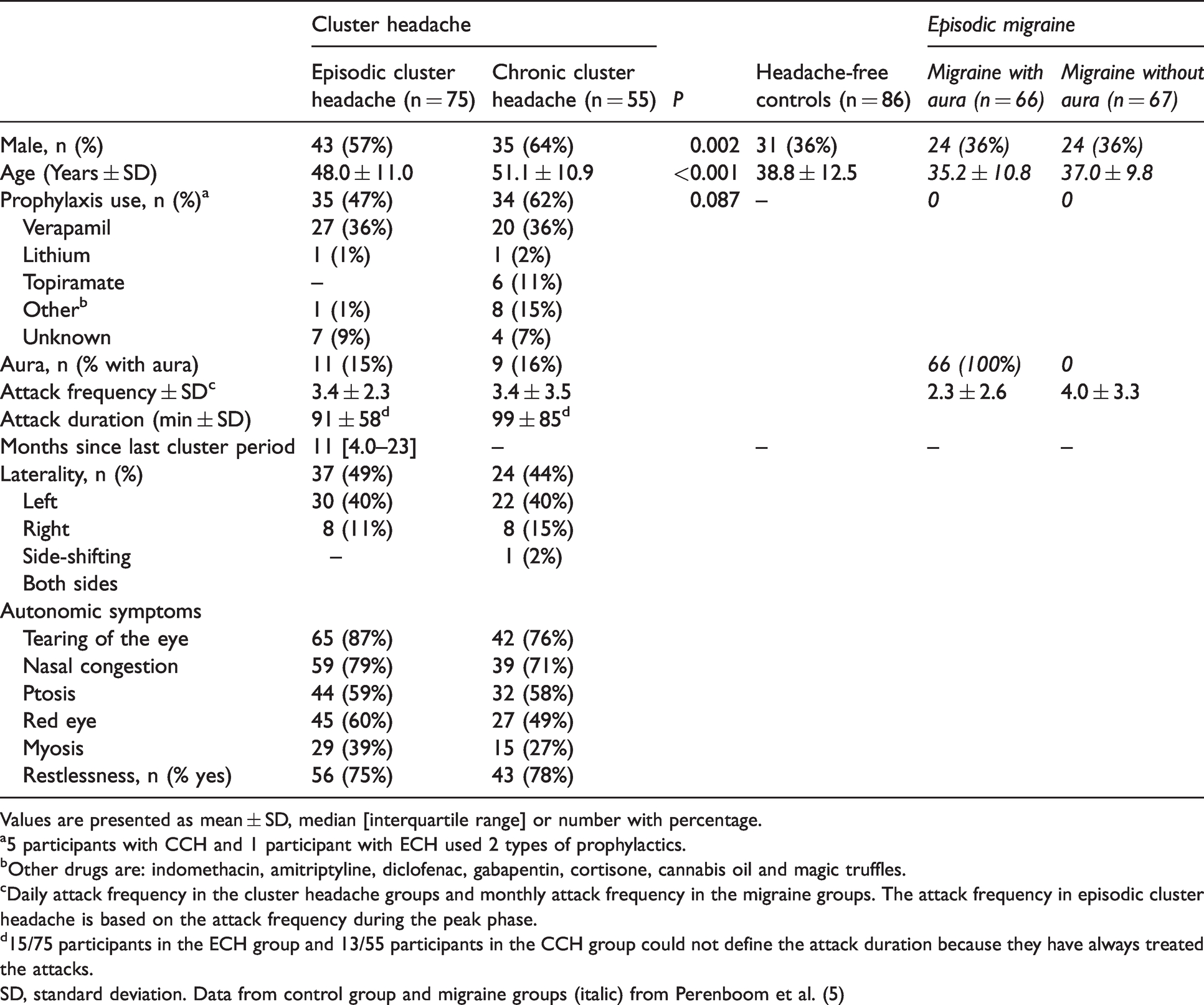
Values are presented as mean ± SD, median [interquartile range] or number with percentage.
a5 participants with CCH and 1 participant with ECH used 2 types of prophylactics.
bOther drugs are: indomethacin, amitriptyline, diclofenac, gabapentin, cortisone, cannabis oil and magic truffles.
cDaily attack frequency in the cluster headache groups and monthly attack frequency in the migraine groups. The attack frequency in episodic cluster headache is based on the attack frequency during the peak phase.
d15/75 participants in the ECH group and 13/55 participants in the CCH group could not define the attack duration because they have always treated the attacks.
SD, standard deviation. Data from control group and migraine groups (italic) from Perenboom et al. (5)
L-VISS scores
The L-VISS scores are summarized in Table 2 and Figure 2.
Mean LVISS score ± SD per group.
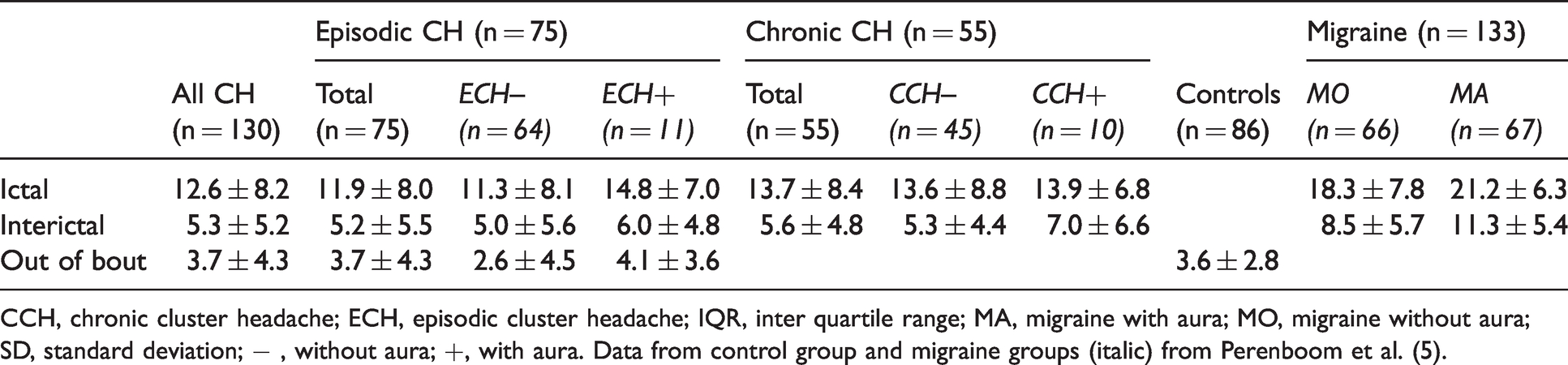
CCH, chronic cluster headache; ECH, episodic cluster headache; IQR, inter quartile range; MA, migraine with aura; MO, migraine without aura; SD, standard deviation; − , without aura; +, with aura. Data from control group and migraine groups (italic) from Perenboom et al. (5).

Individual mean raw L-VISS scores with group means and standard deviations. Mean L-VISS scores are higher ictally than interictally in participants with ECH (ictal: 11.9 ± 8.0, interictal: 5.2 ± 5.5) and CCH (ictal: 13.7 ± 8.4, interictal: 5.6 ± 4.8), and higher interictally in ECH when compared to ECH out-of-bout (5.2 ± 5.5 vs. 3.7 ± 4.3).
Mean ictal L-VISS scores were higher than interictal scores in ECH (ictal: 11.9 ± 8.0 vs. interictal: 5.2 ± 5.5) and CCH (ictal: 13.7 ± 8.4 vs. interictal: 5.6 ± 4.8). Attack status (p < 0.001) and number of autonomic symptoms (p = 0.001) significantly influenced L-VISS score, whereas cluster headache sub-type (p = 0.058), sex (p = 0.078), aura status (p = 0.544) and preventative treatment use (p = 0.122) did not (linear mixed model) (Figure 2, Table 2). Post-hoc pairwise comparison showed a higher L-VISS score in participants with n = 5 autonomic symptoms compared to a lower number of autonomic symptoms (all p < 0.05). The presence of any of the individual autonomic symptoms was not independently associated with a higher L-VISS score.
L-VISS scores differed between ictal CH (EM ± SE: 12.8 ± 0.72), interictal CH (EM ± SE: 5.5 ± 0.5) and controls (EM ± SE: 3.2 ± 0.3). Attack and disease status significantly influenced L-VISS score (p < 0.001). Post-hoc pairwise comparisons showed a difference between all groups (all between group comparisons: p < 0.001). (Figure 3). Furthermore, L-VISS score differed between interictal CCH, ECH out-of-bout, and controls (Univariate GLM (F[2,207] = 7.300, p = 0.001)). Post-hoc pairwise comparisons revealed higher estimated marginal mean interictal L-VISS scores in CCH (EM ± SE: 5.9 ± 0.5) when compared to out-of-bout ECH (EM ± SE: 3.8 ± 0.5, p = 0.009) and control scores (EM ± SE: 3.2 ± 0.4, p = 0.001). No difference was observed between ECH out-of-bout and control scores (mean difference [95% CI]: 0.620 [−0.964–2.204]). (Figure 4).

Individual mean raw L-VISS scores with standard deviation per group. A higher L-VISS score is shown ictally (mean ± SD: 12.6 ± 8.2; EM ± SE: 12.8 ± 0.7), than interictally (mean ± SD: 5.3 ± 5.2; EM ± SE: 5.5 ± 0.5), than in controls (mean ± SD: 3.6 ± 2.8; EM ± SE: 3.2 ± 0.3).

Raw mean individual LVISS scores with standard deviation per group. Higher interictal L-VISS scores are observed in CCH (EM ± SE: 5.9 ± 0.5) when compared to out-of-bout ECH (EM ± SE: 3.8 ± 0.5) and controls (EM ± SE: 3.2 ± 0.4, p = 0.001).
A paired samples t-test showed higher mean interictal L-VISS scores in ECH when compared to L-VISS scores in ECH out-of-bout (5.2 ± 5.5 vs. 3.7 ± 4.3, p < 0.001) (Figure 2).
Of the 121/130 (93%) participants with cluster headache who completed the supplementary questions, 110 (91%) reported subjective visual hypersensitivity; in 70/110 participants (64%) this was partly (n = 34; 31%) or always (n = 36; 33%) unilateral, and in 69/70 participants ipsilateral to the ictal pain and facial autonomic symptoms.
No correlation between age and L-VISS score was observed (Pearson Correlation: −0.056, p = 0.561). LVISS scores in males differed between ictal CH (EM ± SE: 11.3 ± 0.9), interictal CH (EM SE: 4.9 ± 0.6) and controls (EM ± SE: 2.9 ± 0.4). Furthermore, L-VISS scores in females differed between ictal CH (EM ± SE: 14.6 ± 1.1), interictal CH (EM ± SE: 6.0 ± 0.7) and controls (EM ± SE: 3.9 ± 0.4). Post-hoc pairwise comparisons showed higher L-VISS scores ictally than interictally than controls in males and females (male interictally vs. controls: p = 0.17, female interictally vs controls: p = 0.041, all other comparisons p < 0.001). Finally, no correlation between attack frequency and L-VISS score was observed ictally (R2 = 3.08E-5) or interictally (R2 = 1.72E-5).
Discussion
In this cross-sectional study, we found increased visual sensitivity in people with cluster headache, particularly during attacks, but also outside attacks and bouts. In two thirds of cases, the visual hypersensitivity was unilateral, mostly ipsilateral on the side of the ictal pain, similar to a previous small study in trigeminal autonomic cephalalgias (14). This observation should help distinguish cluster headache from migraine, where photophobia is typically bilateral and generally more severe (5,7,14). Unilateral mild to moderate photophobia, ipsilateral to the ictal pain side, can thus be considered a frequent part of the clinical spectrum of cluster headache and could be added to the diagnostic criteria to facilitate clinical differentiation from migraine.
Subjective visual hypersensitivity was reported by 91% of the participants with cluster headache, which is considerably higher than in previous studies (14-17). This might be because in the present study visual sensitivity was examined on a linear rather than a binary scale (1,2,4,8,14,15).
In ECH, interictal visual sensitivity was higher during a bout than visual sensitivity outside a bout. This would indicate a ‘lingering visual hypersensitivity’ that we observed in CCH as well. It is possible that this is because the interictal recovery time is too short in patients with a high attack frequency (17). Increased ventral occipital responsiveness to light has been postulated to explain ictal photophobia in migraine and presence of trigeminal pain increases this responsiveness (18,19). By analogy, since many patients with cluster headache have permanent mild trigeminal pain between attacks, increased occipital responsiveness to light could possibly explain visual hypersensitivity in cluster headache.
L-VISS scores are higher in participants with the most autonomic symptoms, suggesting a common underlying pathophysiological mechanism (e.g. a reflection of a more severe trigeminovascular activation).
L-VISS scores were slightly higher in female participants with cluster headache and in participants with cluster headache who also reported having aura (but no other migraine features) during at least some attacks. This could indicate the presence of a slightly more ‘migrainous’ phenotype in women with cluster headache (20,21). Interestingly, in two recent parallel genome-wide association studies of cluster headache, among other gene loci, the migraine risk FHL5-locus was identified as well and with a remarkably large effect size, indicating a partially overlapping genetic basis between these two types of paroxysmal headache (22,23).
Strengths of the current study are (i) a clinically well-defined study population of patients with ECH or CCH in whom the presence or absence of visual aura was specified and in whom comorbid migraine and severe eye disorders were excluded to avoid confounding factors. The importance of this exclusion was highlighted by a recent study reporting that the presence of aura in trigeminal autonomic cephalalgias is mediated by comorbid migraine with aura in most patients (24); and (ii) the use of a validated instrument to measure visual sensitivity in combination with additional questions as to whether the participants experienced visual hypersensitivity and if so on which side.
A major limitation of the present study is that we compared L-VISS scores in people with CH with those from healthy controls and people with migraine obtained in a previous study. As a result, the three groups were not matched for age and sex. However, we believe the conclusions still hold. Firstly, both studies were conducted by the same study group and were using the same detection tools (5). Secondly, we did not observe any correlation between age and L-VISS score, which is in line with previous results (9). Thirdly, we observed lower L-VISS scores in men than in women in the healthy control and CH groups. Since there were fewer women than men in the cluster headache group, sex matched comparison groups would have probably resulted in even more robust differences. Finally, to limit possible confounding effects, we adjusted for the differences in sex and age in the statistical analyses. Furthermore, we performed a separate analysis between (i) males with CH and healthy males and (ii) females with CH and healthy females with similar results. Notwithstanding these arguments, direct comparisons with the migraine and control groups should be interpreted with caution.
Another potential risk of comparing study groups measured at different times of year is that the results may differ due to seasonal variation. However, because L-VISS measures visual sensitivity retrospectively (what it was like during attacks and what it was like outside attacks), different measurement times are unlikely to have much influence. Some participants (n = 17) were chronically using preventives, potentially preventing active bouts, we cannot exclude that some of them were actually having (effectively treated) CCH rather than ECH. We do not think this has materially affected the results since L-VISS scores of participants with CCH are generally higher, thereby potentially increasing actual between-group differences.
Unilateral visual sensitivity was reported by 64% of participants in the cluster headache group. Unfortunately, we do not have information on possible laterality of visual sensitivity in the migraine group. However, in a previous study 4% of episodic and 13% of chronic migraine patients reported unilateral photophobia (14). Furthermore, data on bout or disease duration were not available. However, we do not expect a correlation between these variables and visual sensitivity.
Due to the retrospective nature of the questionnaire, recall bias cannot be excluded. Participants might have focused on the most extreme or most recent attacks rather than on the entire bout period. However this seems unlikely to explain the observed differences between ictal, interictal and out-of-bout scores as well as the occurrence of visual hypersensitivity per se. Furthermore, participants are capable of accurately describing specific symptoms other than pain, which is highlighted in a study assessing the course of CH attacks (25). It is possible that the visual sensitivity differs between attacks for each patient. To properly address this issue, a prospective registration that assesses intrapatient attack characteristics (attack intensity, number of autonomic symptoms and attack duration) and visual sensitivity could be considered. The risk of selection bias is also low since we included patients that were previously selected for another unrelated study.
Finally, participants were recruited from the Leiden University Medical Center headache clinic, a tertiary referral center. This might have led to a more severely affected and less responsive population in which, relatively, many patients had stopped using prophylactic medication because of lack of response. It is possible that our results cannot be directly translated to the ‘general’ cluster headache population. However, most of the participants were self-recruited through the web-based LUCA questionnaire, which is less likely to be influenced by referral bias since patients apply for the study themselves.
In summary, cluster headache is associated with ictal and interictal visual hypersensitivity, which in most cases is unilateral and ipsilateral on the side of the pain during attacks. It might be considered to add this feature to the diagnostic criteria of cluster headache to facilitate clinical differentiation from migraine.
Article Highlights
Cluster headache is associated with ictal and interictal visual hypersensitivity In most cases, this visual sensitivity is unilateral and ipsilateral on the side of the pain during attacks Visual sensitivity ipsilateral to the pain could be added to the diagnostic criteria of cluster headache to facilitate clinical differentiation from migraine
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RBB reports support for conference visits from Allergan; VC reports no relevant conflict of interest; PGGD reports no relevant conflict of interest; IFC reports no relevant conflict of interest; MJLP reports no relevant conflict of interest; JAC reports consultancy and lecture fees from Novartis, Teva, Allergan, Lilly and UCB; RM reports no relevant conflict of interest; GMT reports consultancy support from Novartis, Allergan, Lilly, and Teva, and independent support from Dutch Organization for Scientific Research, the Dutch Heart & Brain Foundations, IRRF and Dioraphte; MDF reports no relevant conflict of interest; RF reports consultancy and lecture fees from Novartis, Teva, Allergan, Lilly and TEVA, and independent support from the Dutch Brain Foundation, Leiden University Fund and Innovation Fund Dutch Healthcare Providers.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
