Abstract
Objective
To review and discuss the literature relevant to the role of brainstem structure and function in headache.
Background
Primary headache disorders, such as migraine and cluster headache, are considered disorders of the brain. As well as head-related pain, these headache disorders are also associated with other neurological symptoms, such as those related to sensory, homeostatic, autonomic, cognitive and affective processing that can all occur before, during or even after headache has ceased. Many imaging studies demonstrate activation in brainstem areas that appear specifically associated with headache disorders, especially migraine, which may be related to the mechanisms of many of these symptoms. This is further supported by preclinical studies, which demonstrate that modulation of specific brainstem nuclei alters sensory processing relevant to these symptoms, including headache, cranial autonomic responses and homeostatic mechanisms.
Review focus
This review will specifically focus on the role of brainstem structures relevant to primary headaches, including medullary, pontine, and midbrain, and describe their functional role and how they relate to mechanisms of primary headaches, especially migraine.
Introduction
The brainstem consists of the medulla oblongata, the pons and the midbrain, moving rostrally through the brain, and at its most caudal it is contiguous with the spinal cord (Figure 1(a)–(b)). Its major function is the integration of information from the rest of the body through ascending connections with higher centres of the brain, such as the hypothalamus, amygdala, thalamus, and cortex (1). These are related to physiological survival regulation and reflex functions, including cardiovascular and respiratory control, alertness, awareness, consciousness, and sensitivity to pain (somatosensory function). These crucial connections are mediated via the corticospinal tract for motor signals, the posterior column-medial lemniscus pathway involved with fine touch, vibration sensation and proprioception, and the spinothalamic tract for pain, temperature, itch, and crude touch. In addition, there is the trigeminothalamic tract, specifically involved in relaying somatosensory signals from the head and face. A detailed description of all ascending/descending circuits associated with all brain structures relevant to primary headaches can be found elsewhere (2–6), and through other relevant reviews of this special issue. Ten of the 12 cranial nerves (III–XII) arise in the brainstem, including the trigeminal (V), facial (VII) and vagus (X) nerves (Figure 1(b)). These nerves supply the head, face and viscera. In relation to primary headaches, it is simplistic to suggest that the major function of the brainstem is in the processing of noxious somatosensory information relevant to the head and face. The complexity of their associated symptoms, particularly in migraine and trigeminal autonomic cephalalgias (TACs), such as triggers, which involve homeostatic stresses including stress, feeding and sleep disruption, and premonitory symptoms, including tiredness/alertness, irritability, as well as cranial autonomic symptoms, suggest an overall role in primary headache symptomatology processing and pathophysiology. While many primary headache disorders involved mechanisms related to the brainstem, particularly the medullary trigeminal nucleus, migraine is specifically associated with the brainstem through many imaging studies, which highlight selective brainstem activation, compared to other headache types such as the TACs. As the most prevalent and most well studied, this review will largely focus on the brainstem role in migraine, although where relevant we will reference other primary headache disorders.
Anatomical organisation of the brainstem and cranial nerves in the human brain. (a) The brainstem includes, moving rostrally, the medulla oblongata (emerging from the myelencephalon), the pons (emerging from the metencephalon) and the midbrain (emerging from the mesencephalon). At its most caudal, it is contiguous with the spinal cord connecting the trigeminal nucleus of the medulla. At its most rostral, it connects with diencephalic nuclei from the hypothalamus. (b) Ten of the 12 pairs of cranial nerves (III–XII) target or emerge from the brainstem. Nuclei of the oculomotor (III) and trochlear nerves (IV) are located in the midbrain. Nuclei of the trigeminal nerve (V), abducens (VI), facial (VII) and vestibulocochlear nerves (VIII) are located in the pons. Nuclei of the glossopharyngeal (IX), vagus (X), accessory (XI) and hypoglossal nerves (XII) are located in the medulla. Figure 1(b) is adapted from (1).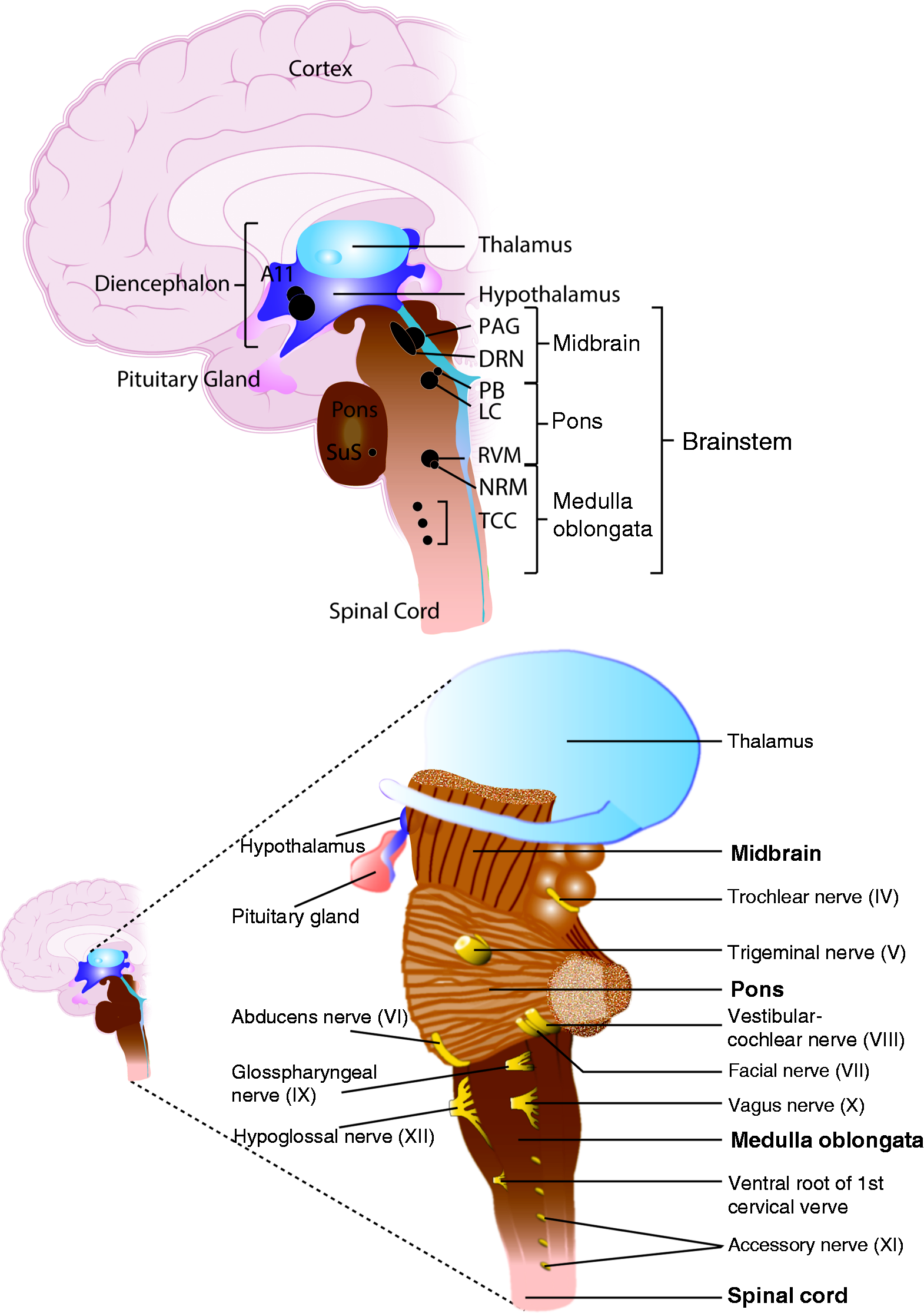
Medulla oblongata
The medulla oblongata, more commonly referred to as the “medulla”, is the most caudal portion of the brainstem and it connects the spinal cord with the rest of the brain via the pons (Figure 1(a)–(b)). It is here that there is decussation of approximately 90% of neurons to their contralateral side, ascending to cortico-thalamic structures. Structures within the medulla are primarily responsible for autonomic (involuntary; sympathetic and parasympathetic nervous systems) functions including vomiting and sneezing, as well as cardiac and respiratory centers. It therefore deals with the autonomic functions of breathing, heart rate and blood pressure. The medulla is also importantly involved in pain processing. Here is situated the rostral ventromedial medulla (RVM), including the nucleus raphe magnus (NRM). This forms part of the “endogenous pain modulatory pathway”, thought to provide descending modulation of nociceptive processing to the spinal and medullary dorsal horns (7–11). At the most caudal portion of the medulla is the trigeminal nucleus (Figures 2–3). These neurons are thought to be functionally contiguous with the upper cervical dorsal horn and relay all somatosensory information from the face and head, including intracranial structures such as the dura mater.
The trigeminal nerve
The trigeminal nerve is the fifth (V) and largest of the cranial nerves, and serves as the main sensory nerve of the head and face. It is also responsible for the motor nerves of the muscles of mastication (12–15). The trigeminal sensory root arises from the trigeminal ganglion, and is split into three major branches: The ophthalmic or first division (V1), the maxillary or second division (V2), and the mandibular or third division (V3). The V3 division is the only branch to have a sensory and motor component. Their intra- and extra-cranial innervation is summarised in Figure 2(a)–(b).
Craniofacial sensory innervation territories of branches of the trigeminal nerve and cytoarchitecture of the trigeminal brainstem nuclear complex (TBNC). The trigeminal nerve is split into three major branches, the ophthalmic or first division (V1), the maxillary or second division (V2), and the mandibular or third division (V3). Intracranially (a), the V1 branch innervates the ethmoid and sphenoid sinuses, as well as the dura mater of the tentorium cerebelli and falx cerebri, along with portions of the dural vasculature, including middle meningeal artery (MMA), superior, sigmoid and occipital sinuses. The V2 branch innervates part of the nasal cavity, the tongue, maxillary and paranasal sinuses, the dura of the middle cranial fossa, and portions of the dural MMA. The V3 branch innervates the oral cavity, the gums of the mandible, the mucous membrane of the anterior two thirds of the tongue, the muscles of mastication, the external surface of the ear drum, and a small portion of the dural MMA. In the extracranial region (b), the V1 branch innervates the superior region of the face, including skin, eye and nose, the V2 branch innervates the cutaneous structures of the middle portion of the face, including the lower eyelid and cheeks. The V3 branch innervates the cutaneous structures overlying the mandible, the auricular, the lower lip, and the external surface of the eardrum. (c) Central afferent projections of the trigeminal nerve terminate on second-order neurons of the trigeminal brainstem nuclear complex (TBNC), which extends from the rostral pons to the C2 upper spinal cord. It is subdivided into four subnuclei, the principal or main sensory trigeminal nucleus (Pr5), and three spinal trigeminal nuclei (Sp5), oralis (Sp5O), interpolaris (Sp5I) and caudalis (Sp5C; trigeminal nucleus caudalis). GON: greater occipital nerve; C: cervical. Here we present the somatotopical organization in humans, but it is relatively conserved across species, for the rat organization see (245). Figures (a) and (b) are adapted from (20). Figure (c) is adapted from (246).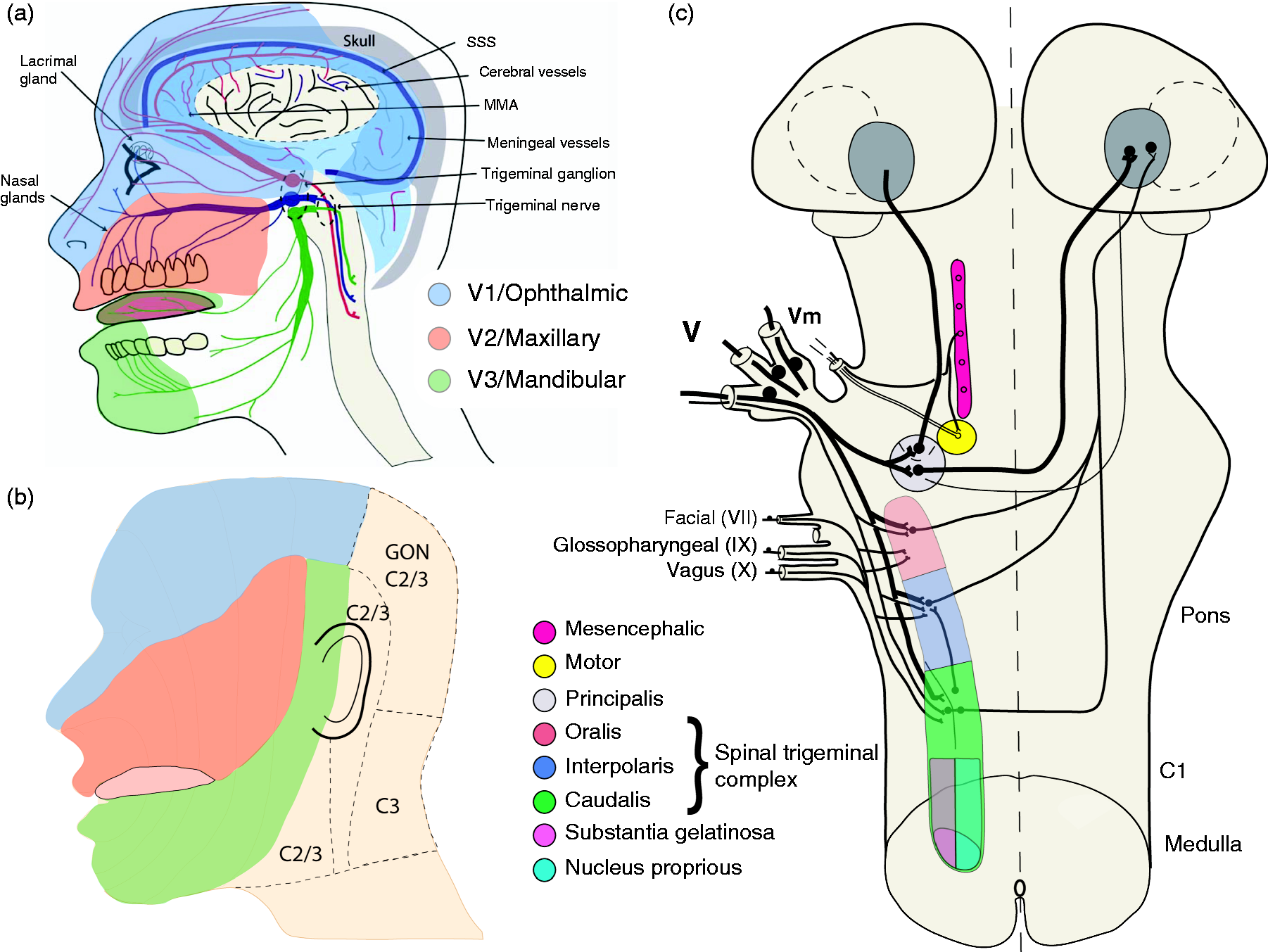
Trigeminal brainstem nuclear complex (TBNC)
In relation to primary headaches, the severe and throbbing head pain particularly associated with migraine and TACs, localized in the frontal, temporal, parietal, occipital and high cervical region, is thought to be the consequence of activation of the ophthalmic trigeminal projection to the intracranial dura mater. In TACs, the pain also predominates in the peri-orbital region, in and around the eye. A rich plexus of nociceptive nerve fibres that originate in the trigeminal ganglion (Tg) innervate the pial, arachnoid and dural blood vessels, including the superior sagittal sinus and middle meningeal artery (MMA), as well as large cerebral arteries (16–18). Noxious manipulation of these structures, particularly the dura mater, results in severe headache pain very similar to the pain in migraine and TACs, as well as other symptoms associated with migraine, including nausea and photophobia (16–18). Interestingly, stimulation of sites away from these blood vessels is much less nociceptive, with correspondingly milder symptoms of headache. The nociceptive innervation of the intracranial vasculature and meninges includes non-myelinated (C-fibres) and thinly myelinated (Aδ-fibres) axonal projections. This is mainly through the ophthalmic (V1) division of the trigeminal nerve, but also, to a lesser extent, through the maxillary (V2) and mandibular divisions (V3). There is also neuronal innervation of the dura mater from the cervical dorsal root ganglia (19). Central afferent projections from the Tg of these dural nociceptive primary afferent Aδ- and C-fibres enter the caudal medulla of the brainstem, terminating on second-order neurons of the trigeminal brainstem nuclear complex (TBNC), via the trigeminal tract. The TBNC is a long column of cells in the dorsolateral brainstem, which extends from the rostral pons to the C2 spinal level. This is subdivided into four subnuclei (Figure 2(c)): The principal or main sensory trigeminal nucleus (Pr5), and three spinal trigeminal nuclei (Sp5), oralis (Sp5O), interpolaris (Sp5I) and caudalis (Sp5C) (20). Sp5 is topographically organized in long rostro-caudal columns of cells, with midline structures (nose) represented medially and more lateral skin represented laterally, although each trigeminal branch conveys signals to all nuclei of Sp5 (Figure 3(a)–(b)). Indeed, animal tracing and electrophysiological studies indicate a dorso-ventral manner of somatotopic organization throughout the TBNC. Allowing for the natural curvature of the trigeminal nucleus, in general, the dorsal-most portion is mainly the V3 division, the V2 division is mainly central, and the V1 division is mainly the ventral-most portion of the dorsal horn (Figure 3(b)).
Somatotopical organization of the spinal trigeminal nucleus. The spinal trigeminal nucleus (Sp5) is topographically organized in rostro-caudal columns of cells (a)–(b). Sp5O (oralis) receives information relating to the mouth, lips, tongue, teeth and front of jaws, Sp5I (interpolaris) receives cutaneous and visceral signals from the middle of the face including nose, and Sp5C (caudalis) receives somatosensory signals from the most lateral territory of the face. The trigeminal brainstem nuclear complex is also somatotopically organized (b). In general, the dorsal-most portion of the trigeminal nucleus reflects mainly innervations from the V3 (mandibular) region of the trigeminal nerve. The V2 (maxillary) innervation reflects mainly the middle portion of the face, including the nose and just below the eye. The V1 (ophthalmic) division of the trigeminal nerve predominantly innervates the area above the eye, including the intracranial dura mater, and is found in the most ventral region of the dorsal horn. These boundaries are not clearly separated, and there is overlap between the specific divisions of the trigeminal nerve and the regions of the face, head and intracranial structures that they do innervate. The trigeminal nucleus caudalis (Sp5C; (c)–(d)) has a similar laminae organization and properties to the spinal dorsal horn. These layers include a marginal zone (lamina I, 1Sp), a substantia gelatinosa layer (lamina II, 2Sp), and a magnocellular layer, equivalent to nucleus proprius (lamina III-IV, 3-4Sp; (Figure 3(c))). (d) summarizes the fibre types and their somatosensory properties that terminate in the specific laminae of the medullary dorsal horn (Sp5C–C2). (c) is a hand drawing of the proposed lamina delineation of the mouse medullary and spinal dorsal horn adapted from the Allen Mouse Brain Atlas (http://atlas.brain-map.org/atlas), Allen Mouse Spinal Cord Atlas (http://mousespinal.brain-map.org), and Paxinos and Watson mouse atlas (247). NTS: nucleus tractis solitarius; Spt5: spinal trigeminal tract.
Trigeminocervical complex (TCC)
The most caudal portion of the trigeminal nerve, the trigeminal nucleus caudalis (TNC; Sp5C), extends from the obex to the upper cervical spinal cord (C2). Indeed, stimulation of the dural vascular structures in animal models, including the superior sagittal and transverse sinuses, and MMA, results in activation of neurons in the TNC, C1 and C2 regions of the cervical spinal cord – together known as the trigeminocervical complex (TCC) (21–24). Furthermore, stimulation of the greater occipital nerve also causes neuronal activation in the same regions and enhances convergent inputs from the dural vasculature (25,26). These data suggest that the trigeminal nucleus extends beyond its caudalis boundary in a functional continuum, which includes the upper spinal dorsal horn. The TNC also has a similar laminar organization to the spinal dorsal horn (lamina I-V) (20) (Figure 3(c)), which disappears around the caudalis/interpolaris transition zone. This has led it to also be known as the medullary dorsal horn. Laminas V–VI have been described by some authors, although this appears to be less well defined. Nociceptive Aδ- and C-fibres predominantly terminate in the superficial laminae, I and IIo, as well as deeper laminae V–VI (24,27–31) of the TNC and cervical extension, whereas Aβ-fibres terminate predominantly in central laminae III–IV. Trigeminal primary afferent nerve fibres converge on three types of second order neurons in the medullary dorsal horn (32–34) (Figure 3(d)):
Low threshold (LT) neurons, which are activated by innocuous touch, vibration and pressure, receiving inputs predominantly from Aβ-fibres Nociceptive-specific neurons, which are silent at rest, become active in response to high intensity, noxious stimuli, including pain, temperature and itch, receiving inputs from Aδ- and C-fibres Wide dynamic range (WDR) neurons, which are activated by a range of noxious and innocuous stimuli (mechanical, chemical, thermal), and receive convergent inputs from first order extracranial cutaneous and intracranial visceral (viscerosomatic convergence) structures onto the same second-order neurons, receiving inputs from Aβ-, Aδ- and C-fibres
The major function of the trigeminal nucleus and its cervical extension is to relay all somatosensory information from craniovascular structures (face and head relevant to many headache forms), including intracranial and extracranial structures, to other areas of the brainstem and the diencephalon, via ascending connections involved in somatosensory processing. This includes direct projections to thalamic neurons, via the trigeminothalamic tract, as well as projections to hypothalamic nuclei via the trigeminohypothalamic tract (2,4,6). There are also projections to brainstem nuclei including the locus coeruleus (LC) and periaqueductal gray (PAG). Trigeminovascular neurons are also under the control of pain-modulatory circuits in the brainstem, including the PAG and rostral ventromedial medulla (both discussed in detail below), and therefore these neurons are considered as integrative relay neurons between peripheral and central nociceptive mechanisms. In the context of primary headaches, the convergence of neuronal inputs into the TCC (21–23) from intracranial and extracranial structures and cervical regions of different nerve fibre types, probably accounts for the distribution of pain perception and cutaneous allodynia in migraine and TACs over the frontal and temporal regions, plus the involvement of parietal, occipital, and higher cervical regions (35). Furthermore, while primary headaches such as migraine and the TACs are predominantly mediated via V1 activation, there is overlap between the specific divisions in the regions of the face, head and intracranial structures that they innervate. The result is considerable overlap of head pain into other trigeminal divisions beyond V1, especially in facial migraine (36), but also with the significant co-morbidity of several headache disorders with orofacial pain disorders, such as temporomandibular disorders (TMDs) (37–41). This overlap of innervation impacts the likely pathophysiology and occurrence of co-morbid pain disorders.
Rostral ventromedial medulla (RVM)
The rostral ventromedial medulla (RVM) is a cluster of neurons located within the medullary region that stretches as far as the pons, close to the midline floor. For anatomical localisation see Figures 1, 4, 6 and 8. Nuclei within the RVM make bidirectional connections with the ventral lateral PAG (vlPAG), and spinal and medullary dorsal horns. This pathway is thought to provide descending modulation of pain processing to the spinal and medullary dorsal horns. This was first evidenced by the high concentration of endogenous opioid signalling, via ON, OFF and Neutral cells, and is described as the “endogenous pain modulatory pathway” (8,42). Within the RVM, non-serotonergic ON cells facilitate pain and are inhibited by opioids, whereas OFF cells are tonically active, and they are activated by opioids (43,44). Neutral cells do not respond to peripheral noxious input, and whether they play a role in the transmission and modulation of central pain syndromes is not entirely clear. Functionally, ON and OFF cells in the RVM (including the nucleus raphe magnus) are also involved in the control of external stimuli, motor activity, and homeostatic processes (10,45), discussed in more detail in the NRM and PAG sections.
Vagus nerve
The vagus nerve is the Xth cranial nerve and arises in the medulla (Figure 1(b)). It is composed of 80% afferent fibres, projecting to the brain, and 20% efferent fibres, projecting to the rest of the body, each of which carries signals in both directions. These nerve fibres originate from cell bodies in the superior (jugular) and the larger, inferior (nodose) vagal ganglion (Figure 4(a)), and are made up of A-, B-, and C-fibres (46). The vagus nerve represents the major parasympathetic innervation of the autonomic nervous system. Efferent fibres project to the larynx, lungs, heart, stomach, liver, pancreas, and gut, and are involved in the primary function of the vagus nerve; regulating involuntary function including the control of heart rate, respiration and digestion. Visceral efferents participate in the pre-ganglionic parasympathetic nervous system and arise from the dorsal motor nucleus of the vagus (DMN) and nucleus ambiguous, innervating all thoracic and abdominal organs, and striate muscle. Fibres arising from DMN do not directly innervate peripheral organs, but on adjacent neurons in the parasympathetic ganglia close to these organs. Post-ganglionic parasympathetic neurons travel to cardiovascular, respiratory, and GI systems.
Summary of the neurophysiological connections of the medullary-pontine autonomic parasympathetic and sympathetic region. (a) The vagus nerve is the major parasympathetic innervation of the autonomic nervous system. Its efferents project to all areas of the body, including larynx, lungs, heart, stomach, liver and pancreas. Afferent nerve fibres originate in cell bodies in the superior (jugular) and inferior (nodose) vagal ganglion. From the jugular ganglion, they project to the nucleus tractus solitarius (NTS), where they directly or indirectly innervate many areas of the brainstem implicated in headache mechanisms, including locus coeruleus (LC), parabrachial nucleus (PB), preganglionic parasympathetic superior salivatory nucleus neurons (SuS), and raphe nuclei (DRN). From the nodose ganglion afferent fibres primarily project to the trigeminocervical complex (TCC), as well as the dorsal motor nucleus of the vagus (DMN) and nucleus ambiguus (NAm). (b) The SuS also receives a reflex connection from the TCC and projects to the cranial vasculature via the greater petrosal nerve (green), and its synapse with the sphenopalatine ganglion (SPG), as well as via the facial nerve (VIIth cranial nerve; blue). Together these form the cranial parasympathetic autonomic projection. There is also a cranial sympathetic autonomic projection (orange) to the intracranial vasculature, and facial region, predominantly via the superior cervical ganglion (SCG). Activation of these pathways contributes to cranial autonomic symptoms in primary headaches, especially trigeminal autonomic cephalalgias, but also migraine via the release of neurotransmitters (acetylcholine, ACh; vasoactive intestinal peptide, VIP; nitric oxide, NO; and pituitary adenylate cyclase-activating peptide, PACAP). The SuS also receives descending projections from a host of hypothalamic nuclei, the PB, as well as limbic (amygdala, hippocampus, thalamus) and cortical regions, and indirectly from the periaqueductal gray (PAG). Together they control SuS activation. Many of the hypothalamic and brainstem (PAG, RVM, parabrachial nucleus) nuclei are also involved in trigeminovascular nociceptive processing, of which the SuS is thought to contribute. RVM: rostral ventromedial medulla; TG: trigeminal ganglion; PH: posterior; SON: supra-optic; VMH: ventromedial, PON: pre-optic; LH: lateral; DMH: dorsal medial; PVN: paraventricular hypothalamic nuclei.
The majority of afferent vagus nerve fibres, bringing information from the rest of the body, project bilaterally to the nucleus tractis solitarius (NTS) in the medulla. The remaining fibres project ipsilaterally to the SpV, area postrema, the DMN and nucleus ambiguus (47). Visceral afferent fibres from the thorax, heart and abdomen carry information whose cell bodies are located in the nodose ganglion and transmitted to the caudal NTS. Somatic afferents transmit sensory information from the lower part of the pharynx, larynx, trachea, bronchi, oesophagus, the ear and ear canal, and dura mater lining the posterior cranial fossa, via the jugular ganglion, terminating in SpV, where they project to somatosensory thalamic neurons. From the NTS, vagal afferents project to the LC, raphe nuclei, preganglionic parasympathetic neurons, the thalamus, the parabrachial nucleus, the PAG, the amygdala and hippocampus, and the cerebral cortex, traversing many synapses in the process (46,48,49). Through these many projections, the vagus nerve directly influences visceral sensory pathways, somatosensory, higher autonomic, extrapyramidal motor and limbic function, all of which are relevant to headache symptomatology and implicate it in its pathophysiology and treatment mechanisms.
Pons
The pons (or bridge) sits between the medulla and midbrain, and essentially forms an important bridge between these two regions to allow these areas to communicate with each other. It provides an important connection between the forebrain and cerebellum related to motor function, and is also involved with sleep, control of motor function such as respiration, swallowing, bladder control, as well as facial movement, taste and posture. The pons contains cranial nerve nuclei, including the pontine and motor nuclei of the trigeminal nerve (V), and the facial nerve (VII) (Figure 1(b)). These are involved in sensory processes related to touch and pain, facial sensation and expression, and secretion of saliva and tears. Nuclei within the pontine region are also involved in the control of touch and pain sensation, specifically cranial sensation, through descending projections to the medullary dorsal horn (trigeminal), via the LC, nucleus raphe magnus (NRM), and parabrachial nucleus. Structures within the pons are also concerned with autonomic function, specifically the parasympathetic nervous system, via the preganglionic superior salivatory nucleus (SuS) and its craniovascular projection, as well as the parabrachial nucleus.
Superior salivatory nucleus (SuS)
The SuS is part of the trigeminal autonomic reflex (Figure 4(b)), receiving a reflex connection from the TNC (50), connecting these two important somatosensory and autonomic pathways. The SuS contains the cell bodies of neurons that are part of the cranial parasympathetic autonomic vasodilator pathway (51). These neurons project predominantly through the greater petrosal branch of the facial (VIIth) nerve, via the sphenopalatine (pterygopalatine; SPG) ganglion (52) to the cranial vasculature, including the dura mater, and to the nasal and oral mucosa, and lacrimal glands. The SuS also receives inputs from the vagus nerve afferents, via the NTS, from the heart, thorax and abdomen (Figure 4(a)).
Preclinical studies demonstrate that activation of this SuS-parasympathetic projection to the cranial vasculature mediates vasodilation of cerebral blood vessels (53–55) and secretory responses from glands. Indeed, direct stimulation of the SuS in rats produces blood flow changes in the lacrimal gland, indicative of cranial autonomic features (56,57). These changes are specifically mediated by activation of the cranial parasympathetic pathway, as a specific SPG blocker attenuates these responses, as does the cluster headache treatment, inhaled oxygen (56,57). Activation of the cranial SuS-parasympathetic pathway is therefore thought to directly contribute to cranial autonomic symptoms found in primary headaches, including TACs such as cluster headache and paroxysmal hemicranias, as well as in migraine (57,58). The main neurotransmitters of this projection include acetylcholine (ACh), vasoactive intestinal peptide (VIP), pituitary adenylate cyclase-activating peptide (PACAP), and nitric oxide (NO). ACh is likely involved in the secretory responses, whereas VIP, NO and PACAP likely mediate vasodilatory and, potentially, sensory responses. In primary headaches, it is likely that both VIP and PACAP contribute to the cranial autonomic symptoms. In experimental clinical studies in migraineurs, both VIP and PACAP are known to cause severe cranial autonomic symptoms (59–61), as well as extracranial vasodilation. Also, VIP (62,63) and PACAP (64,65) levels in the extracranial and peripheral vasculature are increased during migraine and cluster headache. However, VIP levels are only increased during migraine when accompanied with severe cranial autonomic symptoms, suggestive of a definitive role. Taken together, it is clear that activation of the cranial parasympathetic pathway, via VIP and PACAP, is important in mediating cranial autonomic symptoms.
The cranial parasympathetic vasodilator pathway also influences the dural microenvironment, and as a consequence central trigeminovascular neurons and head pain in migraine and TACs. The dural blood vessels are richly innervated by parasympathetic nerve fibres (66) (Figure 4(b)), and activation of this pathway causes the release of ACh, VIP, PACAP and NO from dural vascular terminals of post-ganglionic sphenopalatine neurons. It is believed that activation can lead to a sensory neuro-inflammatory response, with dilation of intracranial vessels, plasma protein extravasation and local dural release of inflammatory mediators (67). Studies in rodents also demonstrate that direct stimulation of the SuS activates two populations of neurons in the TCC (57,68), one specifically via activation of the parasympathetic outflow to the cranial vasculature. It is thought that this in turn indirectly activates trigeminal afferents from the dura mater to the TCC. This neuronal response is attenuated by inhaled oxygen and an autonomic ganglion blocker (57), suggestive of direct activation of the cranial parasympathetic projection. Interestingly, only a modest dural vasodilation accompanies this neuronal response, but it is now thought that vasodilation during TACs and migraine is an epiphenomenon, and less relevant to the pathophysiology of the headache symptoms.
The SuS also has a network of bidirectional connections with areas of the hypothalamus, including lateral (51,69), paraventricular, dorsomedial and pre-optic hypothalamic nuclei (51,70,71), as well as the A5 (lateral tegmental area) and the parabrachial nucleus, and limbic and cortical areas (51). These are regions involved in homeostatic functions including feeding, sleep and stress, which alongside the central autonomic system are thought to be ultimately under the direct control of the hypothalamus. Many of these regions also send direct projections to the TCC, and therefore are thought to contribute to the triggering mechanisms involved in migraine and TACs, as well as to associated neurological symptoms (72). The SuS sits within a brainstem network that is therefore ideally placed to integrate and relay nociceptive and autonomic information to and from the trigeminovascular system, as well as being under descending control of the hypothalamus, therefore integral in the pathophysiology of headache and cranial autonomic symptoms in primary headaches.
Locus coeruleus (LC)
The locus coeruleus (LC) (Latin for Noradrenergic projection neurons, including locus coeruleus, located in the pons and medulla. (a) Noradrenergic (A groups) and adrenergic (C groups) neurons are located in the pons and medulla. A1/C1 cell groups in the nucleus ambiguus, and A2/C2 groups in the nucleus tractus solitarius both project to the hypothalamus. The A5 (lateral tegmental area), A6 (locus coeruleus; LC) and A7 (reticular formation) cell groups in the pons project to the spinal cord and modulate autonomic reflexes and pain sensation (a)–(b). The LC is the major synthesiser of noradrenaline in the brain. Its axons project to many forebrain structures, including the cerebral cortex, hippocampus, amygdala, cerebellum and thalamus, and it has an important role in arousal and attention. (a) is a representation of projections in the rat brain, (b) is a representation of projections in the human brain.
Moreover, the LC has a complex and specific role in the control of behaviour. In animal studies, it has been shown that changes in behavioural state are preceded by changes in LC activity. LC neurons fire in either tonic or phasic modes. During tonic activity, neurons discharge in a sustained and highly regular pattern, with higher rates during wakefulness, lowest during slow-wave sleep and with almost no firing during REM sleep (79). Highest rates are also found in response to stimuli that induce arousal and exploratory behaviour (80). On the other hand, phasic activity is driven by complex sensory input, task-relevant stimuli, and is critical for vigilance and for the efficient processing of salient information (75).
The LC has a key role in regulating the sleep-wake cycle and levels of arousal (81,82) through neural networks with hypothalamic nuclei. In connection with this, it is relevant to highlight the importance of arousal dysregulation in migraine (83). Sleep and circadian disruptions are reported to increase migraine attack frequency and chronification (84), fatigue is prevalent throughout the multiple attack phases (85,86), and sleep is frequently reported as a common abortive strategy (84). Furthermore, migraine attack onset is most frequent in the early morning when arousal networks, including the LC and the hypothalamic nuclei, are most active (87,88). The prominent role of the LC in regulating the sleep-wake cycle positions it as one of the potential mechanistic links between migraine pathophysiology and its sleep-wake disruption (83).
In support of this hypothesis, several neuroimaging studies in migraine patients have shown an altered hypothalamic functional connectivity with the dorsal rostral pontine region, a region including the LC among other nuclei, which could partially explain autonomic symptoms occurring during migraine attacks (89–91). In preclinical studies, the LC is activated by known clinical migraine triggers including nociceptive trigeminal activation (92) and systemic nitroglycerin injection (93), and electrical or pharmacological manipulation of the LC modulates dural-evoked nociceptive trigeminovascular activation (94). Additionally, modulation of the LC influences the susceptibility to cortical spreading depression (CSD), the experimental correlate of migraine aura. Direct electrical stimulation of the LC induces cerebral hypoperfusion, maximally in the occipital cortex (95–97), and indirect stimulation through vagus nerve activation (98,99) decreases CSD susceptibility (100).
Besides containing NA, LC neurons also co-express other distinct neuropeptides in a specific anteroposterior and dorsoventral distribution, providing heterogeneity to their function (101). Given the prominent role of calcitonin gene-related peptide (CGRP) in migraine pathophysiology (102), it is important to highlight that the highest CGRP immunoreactivity in the brainstem is found in the LC with approximately 80% of LC cell bodies being positive for CGRP in humans (103,104). Other neuropeptides known to be involved in migraine are also expressed in the LC, including PACAP, which is found in 40% of LC cells (103), substance P, found in LC fibres (103), and neuropeptide Y, found in lower density and only shown in rodents (105,106). LC neurons also express a variety of receptors that are modulated by other brain structures implicated in headache pathophysiology. These include neurokinin-1 tachykinin (NK1) receptor (107); NPY receptor type 1, 2, 4 and 5 (108); 5-HT1F receptor (109); µ-opioid receptor (110); and orexin receptor type-1 (111), which can be modulated through the dense projections received from the lateral hypothalamus (112), and could be involved in the dysfunction of arousal in migraine.
Raphe nuclei
The raphe nuclei constitute a collection of cell groups distributed in the midline region of the tegmentum (Figure 6). The term raphe refers to a seam or ridge along the midsagittal plane of the body where both left and right-side structures fuse or join. The raphe nuclei contain heterogeneous populations of neurons, with a majority of serotonergic neurons that provide the major source of serotonin synthesis in the brain. They are allocated into two groups: the rostral and the caudal groups, which are separated by a narrow serotonin-free transversal segment located at the middle of the pons (113). The rostral group, located within the pontine-midbrain region, includes the dorsal raphe (see midbrain section), the median raphe and the caudal linear nuclei, with major projections to the forebrain (Figure 6(a)–(b)). The caudal group, located within the medullary pontine region, includes the raphe magnus, raphe obscurus and raphe pallidus nuclei and parts of the adjacent lateral reticular formation, and mainly projects to the brainstem and spinal cord (Figure 6(a)–(b)).
Serotoninergic-raphe projection neurons located in the midbrain, pons and medulla. (a) The raphe nuclei are the major source of serotonin synthesis in the brain. The serotoninergic-raphe (B cell groups) neurons are located in the midbrain, pons and medulla of the brainstem, and project throughout the brain. Within the medulla B cell groups (B1, nucleus raphe pallidus; B2, nucleus raphe obscurus; B3 nucleus raphe magnus (NRM); B4, region of vestibular nuclei (MVe) and abducens nucleus) project throughout the medulla and spinal cord, and are involved in modulating afferent pain signals, thermoregulation, cardiovascular control, and breathing. (a)–(b) Within the pons and midbrain B cell groups (B5, nucleus raphe pontis; B6 forms the caudal portion of the nucleus raphe dorsalis (DRN), with B7 the rostral potion of the DRN adjacent to the peraqueductal gray (PAG); D8 median raphe nucleus (MRN); and B9 is the region of the pontine tegmentum) are involved in arousal, mood and cognition. These neurons also project throughout forebrain stuctures, including the cerebral cortex, hippocampus, hypothalamus, thalamus, and cerebellum. (a) is representation of projections in the rat brain, (b) is representation of projections in the human brain.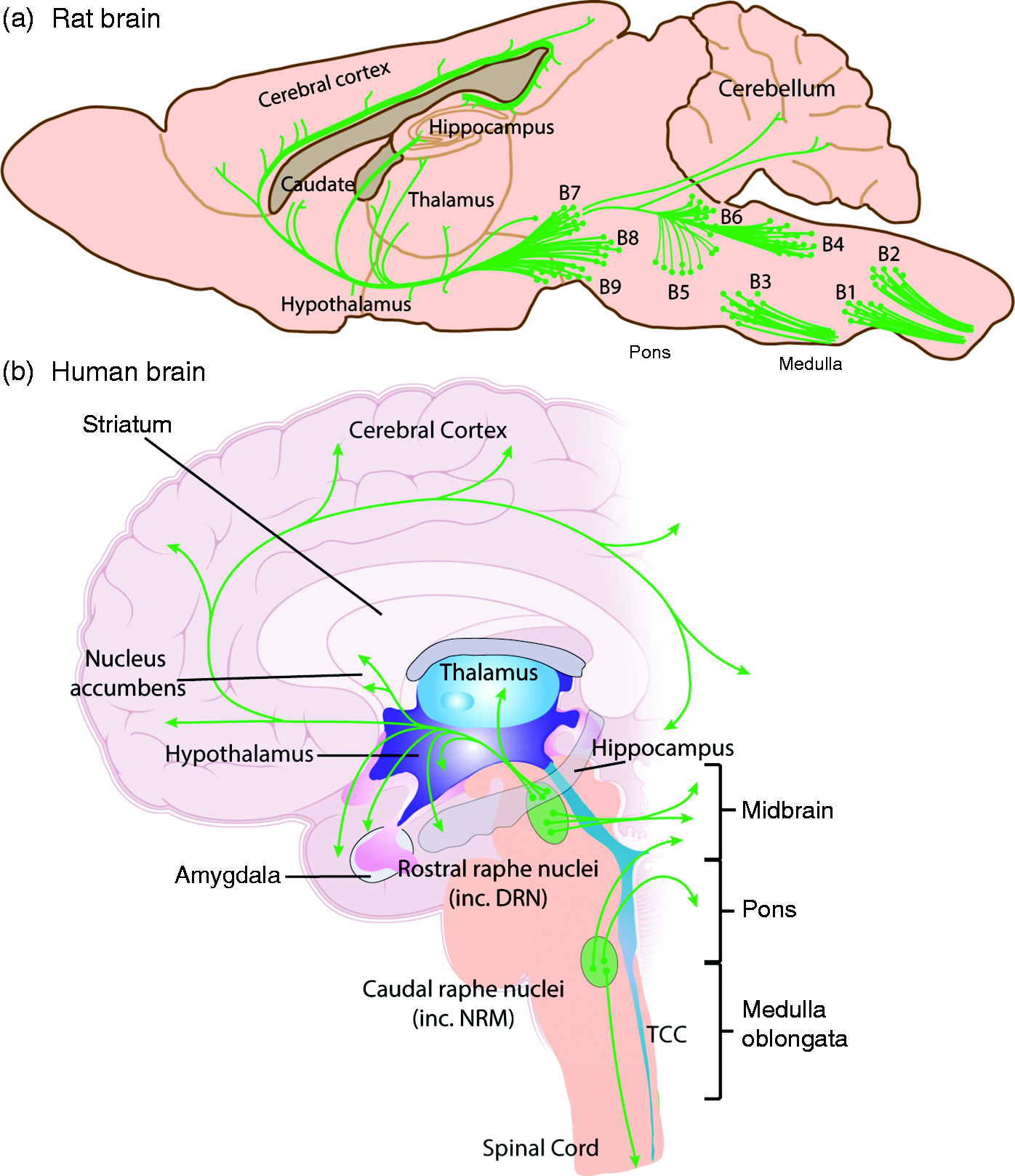
Nucleus raphe magnus (NRM)
The nucleus raphe magnus (NRM) is located at the junction between the pons and the medulla, at the level of the facial nucleus, and it is restricted to the midline (Figure 6(a)–(b)). It is the largest serotonergic nucleus of the raphe nuclei caudal group (113). This caudal group sends major projections to the caudal brainstem and spinal cord (114). The NRM specifically sends projections to the dorsal horn of the spinal cord, with dense innervation of laminae I and II of the TNC (115), the lateral hypothalamus, parafascicular nucleus, vlPAG, noradrenergic nuclei including LC, A5 and A7, and NTS (116). The NRM also receives afferent projections from the TNC, several cortical areas, lateral habenular nucleus, dorsolateral PAG (dlPAG), caudal pontine reticular nucleus and lateral paragigantocellular reticular nucleus (117). NRM neurons are largely serotonergic and differ in morphology depending on their localization. Bipolar serotonergic neurons are mainly found in the midline, and multipolar neurons are found in the paramedian and lateral reticular formation (114). Besides serotonin, NRM neurons can also co-express substance P (118) , neuropeptide Y (119), VIP (103), thyrotropin-releasing hormones (120) and other neuropeptides including encephalin, somatostatin and cholecystokinin (121).
Until recently, the main function of the RVM-NRM nuclei was thought to primarily be in the modulation of nociceptive inputs, forming part of the ‘endogenous pain modulatory pathway’. It provides descending control of nociceptive processing to the spinal and medullary dorsal horns via non-serotonergic ON and OFF cells. ON cells facilitate pain, and are most active during waking, whereas OFF cells are tonically active, their firing inhibits nociceptive inputs, and they are most active during sleep (43,44). There also exists a reciprocity in the connectivity between the PAG and the NRM that may be involved in the modulation of the PAG-NRM-spinal cord pathway (116). In fact, the PAG (see PAG section) has excitatory connections with the NRM, indicating that the anti-nociceptive effects of PAG stimulation may be mediated by the NRM (122). Stimulation of the NRM in animals has been shown to be anti-nociceptive in behavioural experiments (123), which has been linked to the inhibition of nociceptive dorsal horn neurons through the action of serotonin, endogenous opiates and/or NA. Interestingly, several reports also implicate the NRM in modulation of trigeminovascular pain pathways. In animals studies, the NRM is activated by dural nociceptive stimulation (124–127), and ON and OFF cells respond to various craniofacial inputs that are modulated by antimigraine treatments; intravenous naratriptan was shown to increase spontaneous activity of OFF cells and to decrease ON cell activity (125). Moreover, electrical or chemical manipulation of the NRM modulates trigeminovascular nociceptive activation in the TCC. Specifically, electrical stimulation of the NRM inhibits both dural and periorbital-evoked nociceptive neuronal activation in the TCC of cats (126). Whereas in rats, microinjection of GABA or muscimol (GABAA receptor agonist) in the NRM increases dural-evoked nociceptive neuronal activation in the TCC, while bicuculline (GABAA receptor antagonist) decreases it and neither baclofen (GABAB receptor agonist) nor 2-hydroxysaclofen (GABAB receptor antagonist) has a significant effect on the neuronal activation (128). On the same line, microinjection of orexin A and B in the NRM increases trigeminovascular nociceptive activation in the TCC, an effect that is mainly driven by OX2 receptors (129). Lastly, using experimental conditions to simulate potential migraine triggers in cats, using CSD and bright light stress, both were shown to inhibit spontaneous neuronal discharge firing in the NRM (126). In the same study, CSD antagonized the inhibitory effect of NRM stimulation on dural-evoked, but not peri-orbital-evoked, trigeminovascular nociceptive activation in the TCC.
Despite this plethora of data, the functional role of the NRM is now thought to go beyond simply modulating somatosensory mechanisms. Via a network of connections with other brain nuclei, including the PAG and hypothalamus, the RVM-NRM is also thought to respond in external innocuous stimuli (130), motor activity (131,132), and is involved in mechanisms related to homeostasis (44,133,134). ON cells are state-dependently active during waking hours, but not during feeding and micturition, or during sleeping (44,130,133,134). OFF cells are active during sleep, and only active during waking prior to micturition, as well as during feeding. Therefore, OFF cell firing during important homeostatic functions such as sleep, feeding and urination seems to suppress arousal, to prevent against responding to innocuous and even acute noxious stimuli. During waking, ON cell firing seems to facilitate alertness to sensory stimuli. In combination with the PAG, RVM-NRM neurons are now thought of as brain nuclei involved in modulating sensory, autonomic and motor processes in the spinal cord across normal conditions, and their network of connections to forebrain structures suggest that they are primed in readiness to prioritize responses to nociceptive inputs. If then one takes the bigger picture of migraine pathophysiology in particular, triggers such as sleep and food deprivation are homeostatic processes that now seem, in part, to be regulated by RVM-NRM and PAG pathways. Likewise, symptoms of migraine that accompany pain, including excessive urination, hunger and eating (or altered feeding), the need for sleep and the tendency to avoid physical activities, are all processes that are a result of OFF cell discharge. It is therefore possible that if altered responses of RVM-NRM ON and OFF cells are involved in migraine pathophysiology, they would result in homeostatic changes, similar to those described, as well as impacting somatosensory nociceptive processing.
Parabrachial nucleus
The parabrachial nucleus is located in the pons (Figure 4) and plays a significant role in many physiological processes such as arousal (135,136), thermoregulation (137) and the regulation of blood sugar (138). Beyond this, it is involved in the modulation of pain, including trigeminal pain (139–141). The TCC, in particular laminae I and II, connect to the medial and lateral parts of the parabrachial nucleus (139,140,142–145). Rostrally, the parabrachial nucleus connects to the RVM to access the descending pain modulatory system (141), as well as to higher subcortical structures including the thalamus and hypothalamus for pain processing and other subcortical structures such as the amygdala for integration with the central autonomic network (139,143,144,146–148). Some of these processes of the parabrachial nucleus and its interactions with other structures, such as the thalamus, are mediated by CGRP (149,150) and other neuropeptides, including PACAP (151), relevant to primary headaches (152). Given the high importance of these neuropeptides in the pathobiology of migraine and the described neuroanatomical connections within the relevant pain pathways, it may be hypothesized that the parabrachial nucleus plays a role in migraine. Following this hypothesis, results from preclinical
However, little is known so far on the role of the parabrachial nucleus in primary headache disorders. The neuronal activation observed, and the neuroanatomical and functional connections, could well be part of a modulatory network that may regulate the incoming pain signal. On the other hand, the parabrachial nucleus is a major relay center that converges nociceptive, visceral and thermoreceptive information to the forebrain and the central autonomic network. Through the parabrachial nucleus, trigeminal nociceptive inputs reach the subcortical areas of the autonomic network including the amygdala, hypothalamus, thalamus, the PAG and raphe nuclei (148). Activation of this network induces autonomic and behavioural responses as well as pain modulatory effects. Some of these effects such as alterations of arousal (tiredness), emotional responses, food intake (craving) and an increase in blood pressure can be observed during the premonitory, pain or postdromal phases of acute migraine attacks (85,86). Interestingly, this network, to some extent, uses endogenous CGRP as one of its main neurotransmitters (156). In this system, CGRP has been shown to act directly through the neuronal effects elicited after binding its receptor, as well as to interact with other receptor systems. In this context, CGRP acts on NMDA receptors and potentiates NMDA receptor-mediated synaptic transmission (157). Moreover, an interaction with GABAergic neurons is also described (158), highlighting the complexity of this modulatory system. In addition, the central autonomic network involves parasympathetic neurons that elicit the facial autonomic symptoms (e.g. lacrimation) during attacks of migraine or cluster headache. Therefore, it remains to be elucidated to what extent the parabrachial nucleus and the neuronal activations observed in animal experiments are linked to pain processing, or activation and transmission of neuronal signals of the central autonomic network.
Midbrain
The midbrain is the smallest brainstem component, sitting rostral to the pons. It is involved in linking components of the motor system, such as the cerebellum, basal ganglia and cerebral hemisphere, involved in the control of skeletal muscle. Indeed, the substantia nigra provides important inputs to the basal ganglia, which regulates voluntary movements; of particular interest in Parkinson’s disease through damage to dopaminergic neurons. Several regions are involved in the direct control of eye movement, and a relay for auditory and visual information. Many structures, including the PAG and dorsal raphe also provide important connections between pontine and medullary structures involved in modulating somatosensory inputs, as well as ascending connections involved in pain processing, such as hypothalamic, thalamic and cortical structures. These midbrain structures are believed to be important in the pathophysiology of head pain, as well as integrating inputs from other regions related to associated headache symptoms, such as altered arousal, reward and feeding states.
Dorsal raphe nucleus (DRN)
The DRN, located in the rostral pontine and caudal midbrain tegmentum, within the ventral part of the PAG, contains the largest group of serotonergic neurons within the raphe nuclei complex (Figure 6 (a)–(b)) (113,159).
The DRN is a complex and heterogeneous nucleus that has a marked topographical organization with distinct subdivisions receiving and sending specific projections to several areas of the brain (160). Mainly, the DRN receives afferents from the hypothalamus (arcuate nucleus, lateral, perifornical and preoptic areas), amygdala, lateral habenula, PAG, cerebral cortex and substantia nigra (159). It sends a vast majority of serotonergic projections to the caudate putamen and substantia nigra, and to a lesser extent, to the subthalamic nucleus, substantia innominata, motor cortex and, interestingly, to the TNC (161–164). The DRN also sends dopaminergic projections to the nucleus accumbens, lateral septum and medial prefrontal cortex (165). While the DRN contains predominantly serotonergic neurons (166), accounting for 70% of all DRN cells in humans (167), it also contains glutamatergic, GABAergic and dopaminergic neuronal cell types (168). These neurons also co-express other neuropeptides including nitric oxide (169), neuropeptide Y, substance P and VIP (170), among others, which have been previously linked to migraine pathophysiology.
The DRN modulates pain networks through direct and indirect descending projections to spinal dorsal horn neurons and through ascending projections that regulate the activity of thalamic pain sensitive neurons (171). It is also involved in regulating waking and rapid eye movement (REM) sleep (160) through hypothalamic and LC connections. Serotonergic neurons display a regular firing pattern during waking that is reduced during non-REM sleep and silent or almost silent during REM sleep (172–174). This suggests a possible mechanistic role between sleep-wake disruptions and headache, as discussed above. The DRN may also be involved in CSD, as DRN stimulation modulates cortical (175,176) and carotid blood flow (177,178), and chronic degeneration of serotonergic neurons of the DRN increases CSD velocity and amplitude in rats (179).
The DRN is described as having a role in mood disorders, which are highly co-morbid with headache and migraine (180). This is especially the case with depression (181); the DRN of depressed patients presents a 31% decrease in overall neuron number (182), and tryptophan hydroxylase (the enzyme involved in serotonin synthesis) immunoreactivity and mRNA levels in the DRN are higher in depressed suicide victims compared to controls (183). Furthermore, the dorsomedial part of the DRN is innervated by forebrain structures involved in the regulation of anxiety states and is activated by a number of stress- and anxiety-related stimuli (184).
Despite the low spatial resolution found in human imaging, several studies have hypothesised an important role for the DRN based on its general location within activated regions. Early PET studies reported an increase in cerebral blood flow in a region that includes the DRN during spontaneous migraine attacks. This activation persisted after effective treatment with the 5-HT1B/1D receptor agonist, sumatriptan (185). More recently, a functional MRI study has reported an altered hypothalamic functional connectivity within the dorsal rostral pons, a region that also includes the DRN among other nuclei, which could partially explain autonomic symptoms occurring during migraine attacks, as reported above (90). A PET study has provided indirect support for 5-HT1B receptors expressed in the DRN being temporarily downregulated during the migraine attack, probably in response to higher cerebral serotonin levels in the ictal phase (186).
Preclinically, the DRN is activated by trigeminal nociception induced by intracranial capsaicin (92), an experimental model of trigeminal nociception and CGRP release, but it is not activated by electrical stimulation of the trigeminal ganglion (127). The effects of antimigraine drugs in the DRN have been analysed in several studies. In one, using autoradiographic techniques in the cat, the highest density of specific binding sites of 3H-dihydroergotamine was found in the DRN and the medial raphe nucleus. Both nuclei were also labelled after intravenous administration of 3H-dihydroergotamine (187) and
Periaqueductal gray (PAG)
The PAG (Figure 7) represents one of the most significant elements of the endogenous descending modulatory system. Caudally, the PAG projects to the TCC and the spinal dorsal horn, predominantly through the RVM, rostrally it connects to supraspinal structures including the hypothalamus, thalamus, amygdala and prefrontal cortex (Figure 8). Given this neuroanatomical setting, the PAG is ideally placed to modulate incoming pain and other sensory and homeostatic signals, including the endogenous modulation of pain by cognitive influences, emotional factors and stress.
Functional subdivisions of the central periaqueductal gray (PAG). The PAG is thought to be divided into four functional subdivisions, organised as longitudinal columns, running parallel with the central aqueduct (248). These four columns are the dorsomedial (dmPAG), dorsolateral (dlPAG), lateral (lPAG), and ventrolateral (vlPAG). The dmPAG, lPAG and vlPAG each project directly to the lower brainstem, the dlPAG does not. These different columns have different functions; for instance, both the lPAG and vlPAG are involved in the control of heart rate and blood pressure, but with opposing effects, hypertensive vs. hypotensive, respectively. The PAG is also involved in the endogenous descending modulation of pain mechanisms, via rostral ventromedial medullary projections. Again the lPAG and vlPAG appear to have opposing effects, with activation of lPAG producing an aversive response, whereas the vlPAG is thought to be analgesic. Further, within these separate subdivisions there are also differences in their response to opioids and development of tolerance to analgesics, and the dual role of ascending/descending pathways, or descending only. This figure is adapted from (248). Brainstem descending control of nociceptive inputs in the medullary dorsal horn. Nociceptive trigeminovascular neurons, including those that innervate the dural vasculature and project to the trigeminocervical complex (TCC) region, are under the control of various endogenous descending pain modulatory mechanisms. These impact the neurotransmission of the nociceptive input, and therefore the perception of pain, or not, during primary headaches, as their signal is relayed and processed via ascending projections to hypothalamic (H), thalamic (Thal) and cortical neurons. The periaqueductal gray (PAG) is indirectly involved in the control of pain transmission in the medullary dorsal horn via projections to the rostral ventromedial medulla (RVM). This involves both inhibitory (green) and facilitatory (red) projections. The PAG receives projections from the anterior cingulate, S1 somatosensory and insula cortices, the hypothalamus, and amygdala, and thus is the main output of the limbic system and its involvement in the emotional and motivational control of pain. The PAG also projects to the locus coeruleus (LC), which has descending noradrenergic projections to provide control of TCC nociceptive neurons. Within the RVM there are also inhibitory and facilitatory projections to the TCC involved in the control of nociceptive transmission. In addition, there are separate serotoninergic projections (yellow) from the nucleus raphe magnus in the RVM involved in pain modulation. Opioid receptors present in the PAG and RVM further provide descending mechanisms for endogenous pain modulation of dural-nociceptive inputs to the TCC. Finally, direct descending projection neurons from the cortex and hypothalamus also provide descending modulatory control nociceptive inputs to the TCC. The figure represents the rat brain.
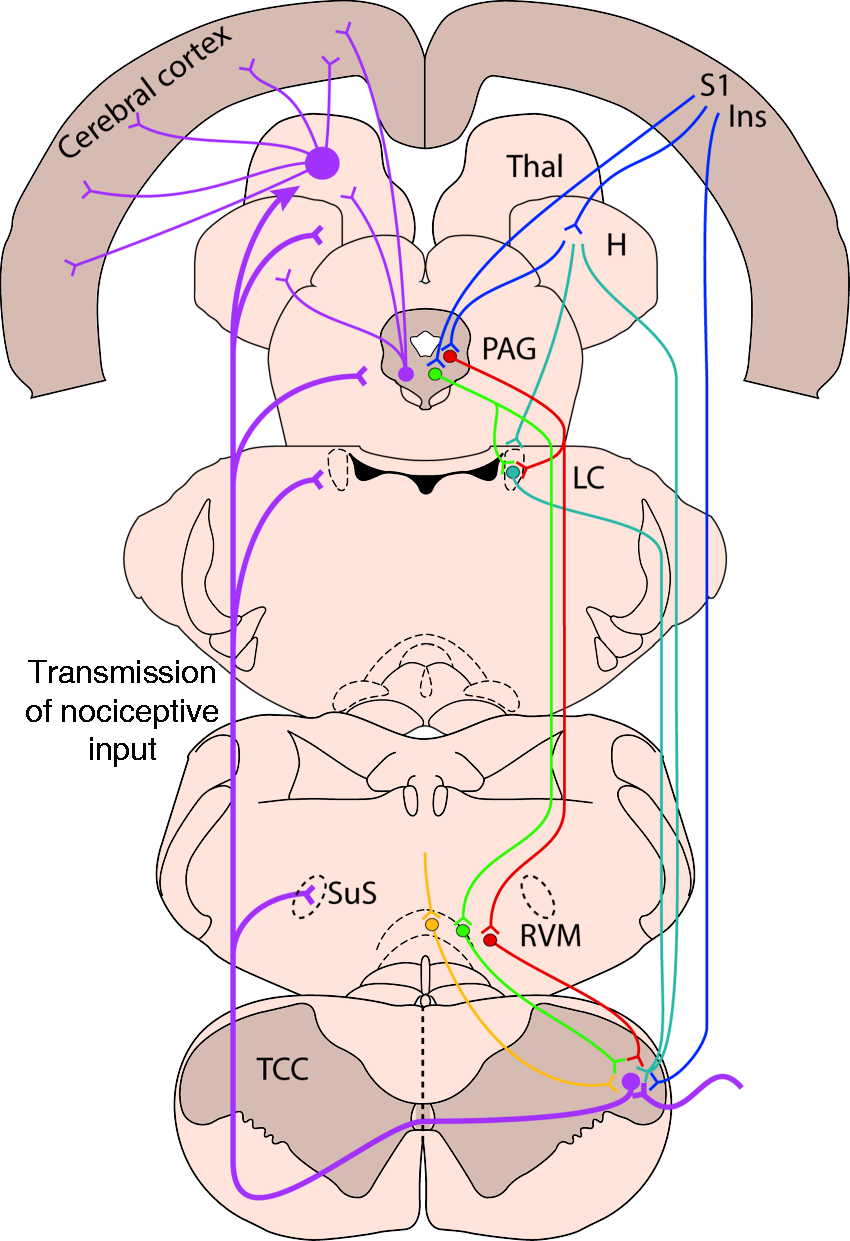
The descending modulatory system, and in particular the PAG-RVM pathway (Figure 8), is involved in all pain conditions, regardless whether the pain is of peripheral or central origin. As discussed in the RVM-NRM section, this descending modulatory system is also involved in mechanisms beyond pain, to include motor function, responses to innocuous stimuli, as well as related to homeostatic functions. The function of the endogenous ‘pain’ modulatory system was initially discovered in animal experiments in rats (190) and later confirmed in humans (191,192). Beyond that, these studies impressively demonstrate the potency of this system, allowing surgery or endurance of severe lesions without experiencing pain. Given the importance of this mechanism, it could be expected that it would play a role in headache disorders. Raskin et al. published a case series in which patients that underwent electrode implantation in the PAG started to suffer from headache with migrainous features, which in most cases was responsive to dihydroergotamine (193). The fact that PAG stimulation induced rather than inhibited headache underlines the fact that the action of the PAG-RVM pathway is not unidirectional. In addition, it can’t be excluded that the frequency of the electrical stimulation was decisive of the functional outcome of the PAG stimulation. A few years later, the hypothesis that the PAG is relevant in migraine was given significant support by the studies of Weiller et al. (185). Using PET imaging, they identified specific brainstem activation during spontaneous migraine headache. They hypothesised that the midbrain region that was activated during these spontaneous migraine attacks was very likely the vlPAG, and these active areas remained so, even after headache resolution with abortive treatment. Based on these findings, a number of
In the PAG endogenous modulation of pain signals is mediated to a large extent through opioid receptor-based mechanisms (9,197–200). In migraine, serotonin receptor agonists (5-HT1B/1D) are more effective than opioids, and for this and several other reasons the use of opioids is not recommended (201,202). These observations raised the possibility that in primary headache disorders other non-opioid receptor systems may be as important for pain transmission, and that this may also apply for pain transmission and modulation in the PAG. Given that triptans are highly effective in the treatment of migraine, Bartsch et al. investigated their effect in the PAG. The data showed that microinjection of the 5-HT1B/1D receptor agonist naratriptan into the vlPAG inhibited nociceptive neuronal activity in the TCC following electrical stimulation of the dura mater (196). A similar effect was observed with administration of sumatriptan into the RVM in two distinct models of visceral pain (203). Considering that triptans act, at least in part, through modulation of CGRP release, it would be conceivable that CGRP itself may also act, among other sites, within the PAG. This hypothesis was recently confirmed in an animal model of migraine in which microinjection of CGRP into the vlPAG facilitated nociceptive activity within the TCC, an effect that could be reversed with the CGRP receptor antagonists, CGRP8-37 and olcegepant (204). These results suggest that the pathobiology of migraine could potentially involve a dysfunction within this descending modulatory system. This hypothesis is fuelled by the orexins, which may also act at specific orexin receptors in the PAG. Orexins are synthetized exclusively in the hypothalamus and regulate arousal. Beyond that, and particularly important in migraine, orexins modulate trigeminal nociceptive processing (205–207). The site of action of this modulation is not limited to the hypothalamus, as orexinergic neurons project to the prefrontal cortex, thalamus and other subcortical areas, including the PAG (31,206,208–211). In this context, Holland et al. demonstrated that administration of orexin A into the vlPAG inhibits dural nociceptive responses in the TCC (206). The exact mechanisms of this action are not entirely clarified, but the fact that the orexin A-induced antinociceptive effect can be blocked by a 5-HT1B/1D receptor antagonist suggests an interaction with the triptan and other receptor systems. A similar observation was made with a CB1 receptor antagonist, which when microinjected into the vlPAG also had an inhibiting effect on dural-evoked nociceptive activity in the TCC that could be modulated through an action at 5-HT1B/1D receptors (212). Therefore it remains unclear if these receptor systems have direct influence on the PAG or if their effect is mediated via 5-HT1B/1D or perhaps even 5-HT1F receptors.
Brainstem actions of headache therapies and the blood-brain barrier (BBB)
Within this review we have highlighted how many headache therapeutics, such as triptans and targeting CGRP and its receptors, can be used to modulate trigeminovascular nociceptive mechanisms within the brainstem. Some of these are from preclinical studies that allow direct administration into specific nuclei. While they are suggestive of a potential therapeutic mechanism of action, they are not definitive. At best they demonstrate that certain pharmacologies are very likely relevant to the pathophysiological mechanisms that underpin these headache disorders, with potential, rather than defined, mechanisms of action in therapeutics. The question of access to the brain for therapeutics is a long running issue, which has been amplified since the general acceptance that vascular mechanisms are unlikely to be as relevant to many primary headache disorders, especially migraine and the TACs. It is relevant that probably the most commonly prescribed headache therapeutic, sumatriptan, is actually the least brain penetrant of all the triptans (213). In preclinical studies, sumatriptan is unable to inhibit dural-evoked activation of central trigeminovascular neurons (214), or reverse dural-inflammatory mediated central sensitisation (215,216), unless the BBB is disrupted (217). The more brain penetrant naratriptan (218), zolmitriptan (214) and rizatriptan (219), are able to inhibit dural-nociceptive central trigeminovascular responses. Furthermore, zolmitriptan has been shown to bind to the medullary TNC and its C1 and C2 cervical extension (220). Yet, it is clear sumatriptan can act via an exclusively neural mechanism (221), and it is at least equally effective in aborting migraine headache as other triptans, and highly effective at aborting cluster headache. So how can this be rationalised?
Either triptans and other therapeutics do not need to gain access to the brain to be effective, or they are accessing the brain via another mechanism to mediate their therapeutic effects. The only area of the brainstem not protected by a blood-brain barrier (BBB) is the area postrema. It sends projections to brainstem nuclei, including the parabrachial nucleus, as well to several hypothalamic nuclei. It is possible therapeutics are accessing the brain this way, but at the moment there is no evidence to support this. Another hypothesis is that there is BBB disruption, particularly during migraine with aura, which contributes to the spread of noxious mediators in the dura mater, but also access of therapeutics to the brain (222). However, several recent studies now suggest that there is no disruption of the BBB during migraine with and without aura, and no difference in brain permeability during migraine compared to their interictal state (223,224). Dihydroergotamine is also an effective migraine abortive and is able to inhibit dural-evoked central trigeminovascular neurons (225). Yet in a human binding study there was little or no binding of 11 C-dihydroergotamine in the brain parenchyma during nitroglycerin-induced migraine (226), again suggesting an intact BBB.
Migraine and TAC preventive drugs, including topiramate, sodium valproate, lamotrigine, and amitriptyline, are widely accepted to cross the BBB, based on their primary use as anti-epileptics and anti-depressants. Preclinical studies support this, as they are effective in models of cortical spreading depression (227–229) and dural-nociceptive trigeminovascular activation (230–232), which require access to discrete areas of the brain. The development of non-vascular centrally acting novel therapeutics such as the -ditans, also suggest that access to the brain in medullary regions is an effective mechanistic approach (233,234). However, the development of molecules that target CGRP and its receptors raises a further issue. Olcegepant was proven to be highly effective in the abortive treatment of migraine (235), and several preclinical studies suggest it is also able to modulate central trigeminovascular neurons (57,236–238). The lipophilicity of olcegepant is not known but it is thought to have poor brain penetrance, based on a lack of effect on the cerebral vasculature (239). The recent development and reported success of CGRP-related antibodies (240–243) in migraine treatment, molecules considered too large to cross the BBB, suggest they are acting in the periphery to be effective. Despite the overwhelming data describing the important role of brainstem nuclei in mediating symptoms of primary headache, particularly the medullary trigeminovascular relay, we are left in the situation where we have to conclude that access to the brain to reach brainstem areas is not obligate for either acute or preventive treatment success in many primary headaches. While in the main it is not clear exactly where many drugs are acting to mediate their therapeutic effects, what is very likely, and perhaps proven by the success of many preclinical models in predicting therapeutic efficacy, is that modulating dural-responsive central trigeminovascular neurons and their ascending brainstem projections is likely to lead to therapeutic success. Much of the trigeminovascular system sits outside the BBB, including the dura mater, trigeminal ganglion, the central axonal trigeminal projections, as well as cranial autonomic projections, modulating brainstem structures including the medullary trigeminovascular relay. Therefore, via these peripheral networks, it is very likely that some migraine drugs mediate their therapeutic action to modulate brainstem networks, whereas other drugs, which do pass the BBB, mediate their effects more directly.
Summary
Brainstem nuclei make up an important part of the central pain pathway related to primary headaches. Their normal function is to integrate information related to physiological survival and reflex functions, arousal and sensitivity to pain, from the rest of the body to higher centres in the brain. The role of brainstem nuclei in headache mechanisms is to integrate inputs from the periphery, such as from environmental stimuli and intracranial nociceptive inputs, relayed through the medullary trigeminovascular system, with those mediated centrally from endogenous homeostatic stresses, such as altered arousal, feeding and life stress. Due to the complex integration and processing of information in brainstem nuclei with each other, and their roles in normal function, the net result of activation of this complex neural network can be headache, due to an altered perception of craniovascular inputs. But also, there is a host of associated symptoms based on the nuclei involved, such as a generalized increase in sensitivity to sensory inputs, mood change, fatigue, yawning, neck stiffness, gastrointestinal disturbance, autonomic features, and cognitive phenomena. All of these can precede, accompany or follow the headache (86,244). This activation might be mediated as a normal functional response to external stimuli and endogenously-mediated homeostatic stressors, or potentially through dysfunctional processing within the brainstem to what are considered normal stimuli and stresses. Together, the normal function of brainstem nuclei, and their potential dysfunction in processing neural information, is integral to mechanisms related to primary headaches.
Article highlights
The brainstem consists of the medulla oblongata, pons, and midbrain, and includes the trigeminal nucleus, raphe nuclei, locus coeruleus, parabrachial nucleus and periaqueductal gray. Normal brainstem function is to integrate information related to physiological survival (cardiovascular/respiratory/autonomic control), arousal and sensitivity to pain, from the rest of the body to higher centres in the brain. In a headache context, activation within brainstem nuclei can mediate cranial pain, and associated headache symptoms that reflect altered sensory, autonomic, affective, and cognitive processing. Activation in the brainstem, and primary headache symptoms, might be mediated as a normal functional response to external stimuli and endogenously mediated homeostatic stressors. Alternatively, primary headache symptoms are a consequence of dysfunctional processing within the brainstem to what are considered normal stimuli and stresses, but which are perceived as noxious, and become a homeostatic threat.
Footnotes
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MVP declares no conflict of interest. JH has received honoraria for consulting activities and/or serving on advisory boards as well as for speaking from Allergan, Autonomic Technologies Inc. (ATI), Chordate Medical AB, Novartis and Teva. MRR and SA report an unrestricted grant, honoraria and travel reimbursements from electroCore LLC, unrelated to the submitted work.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
