Abstract
Austria is one of the countries, in which ergots are still the most commonly used acute anti- migraine drugs. Overuse and chronification is a clinical problem for ergots, but also for the recently developed triptans. In a retrospective study for the year 1999 we evaluated clinical data from all Austrian neurological hospitals including cost of withdrawal as well as the estimated cost for ergots and triptans on the pharmaceutical retail market. We identified a total of 96 patients that underwent withdrawal, all of whom because of ergot overuse, and some with considerable long-term side-effects. The cost of withdrawal (more than €300 000) together with direct cost of medication amounted to more than €1 million. In contrast, cost of medication for triptans was €2.8 million for the same year, without any cost for withdrawal. If only cost aspects were to be considered in the prescription of acute anti-migraine drugs, our data would suggest to choose ergots rather than triptans. However, as scientific evidence is clearly in favour of triptans, decision making for the prescribing clinicians is more complex and will primarily focus on optimizing patient care, but also depend on the respective socio-economic situation.
Introduction
For the past 70 years ergotamine has been widely used in the acute treatment of migraine. Its efficacy as an acute anti-migraine drug has been attributed to its serotonin(5-HT) 1 agonist activity and possibly to an effect on neurogenic inflammation (1, 2).
Since the introduction of the triptans in the early nineties as agents with a high and selective affinity to the receptor subtypes 5-HT1B and 5-HT1D, ergots were partly replaced in their role as acute anti-migraine drugs. In many countries, like in Austria, health care has increasingly become a matter of political controversy, as cost has steadily been increasing. In fact, because of their high cost, some of the recently developed drugs, such as triptans, are only partially reimbursed by health insurances in spite of their superior efficacy and favourable side-effect profile. On this background, there has been an ongoing discussion on the role of ergots in attack treatment in recent years, particularly on the background of their lower price, but with a less favourable side-effect profile compared to triptans.
In this paper, we evaluate acute anti-migraine therapy with ergots and triptans in Austria based on data from 1999 and take the cost of withdrawal following overuse into account. The evaluation is based on a retrospective analysis of the medical charts of all headache patients seen in our Pain Centre at the general hospital in Linz, which is the only specialized Hospital Department for Headache and Pain in Austria. The study was approved by the regulatory authorities. Furthermore, we obtained information from all neurological departments in Austria on in-patient withdrawal following overuse of ergots and triptans and we analysed data on the estimated cost of acute migraine medication on the pharmaceutical retail market in Austria.
Based on our study on the situation in Austria as an example of a country in which ergots are still predominantly used in acute anti-migraine therapy, we discuss the role of ergots and triptans including pharmacological, clinical and ethical aspects as well as the relative weight of cost issues.
Methods
In a retrospective chart analysis we determined the number of headache patients that had consulted the Pain Centre of the General Hospital Linz for the first time in 1999. According to IHS criteria of drug induced headache (3) we selected all patients overusing ergots or triptans, and extracted the underlying headache diagnosis (migraine, tension type headache, other headaches including all secondary headaches) as well as possible side-effects of medication overuse. In case of diagnostic overlaps between primary headaches the reported diagnoses are the primary headache diagnoses only. To prevent discrepancies all patients were personally examined and the diagnoses were determined from the first author. In a second step we identified those individuals who had been admitted for in-patient withdrawal to our Pain Centre and extracted information on the duration of hospitalization.
In order to evaluate the situation all of Austria, we obtained retrospective data from all 42 hospitals with neurological departments in Austria on the number of inpatients admitted for withdrawal because of overuse of ergots or triptans. The following steps were undertaken. One of the co-authors of this paper (AB) contacted the chairmen of all neurological departments by telephone and informed them of this study. All of them agreed to contribute. They determined a member of their medical staff who analysed admission statistics and provided us with information on all patients that were hospitalized for withdrawal due to drug overuse in the calendar year 1999. The number of patients, the underlying headache diagnosis, the duration of hospitalization and also side-effects attributed to medication overuse were obtained.
To supplement the above information, we obtained data on the amount of prescribed ergots and triptans (sold package units, one unit = one package) in 1999 for the diagnosis of headache in Austria from the Austrian Medical Statistical Survey (based on data of the Institute for Medical Statistics, IMS). These data refer to the entire Austrian pharmaceutical retail market, i.e. public pharmacies and dispensing medical practices. The indicated cost is based on the manufacturer's price. The products were classified in compliance with the international guidelines of the EPhMRA (European Pharmacy Market Research Association). The number of patients cannot be concluded as the data available is derived from statistics on prescriptions and consists only of the prescribed drugs and the diagnosis.
Results
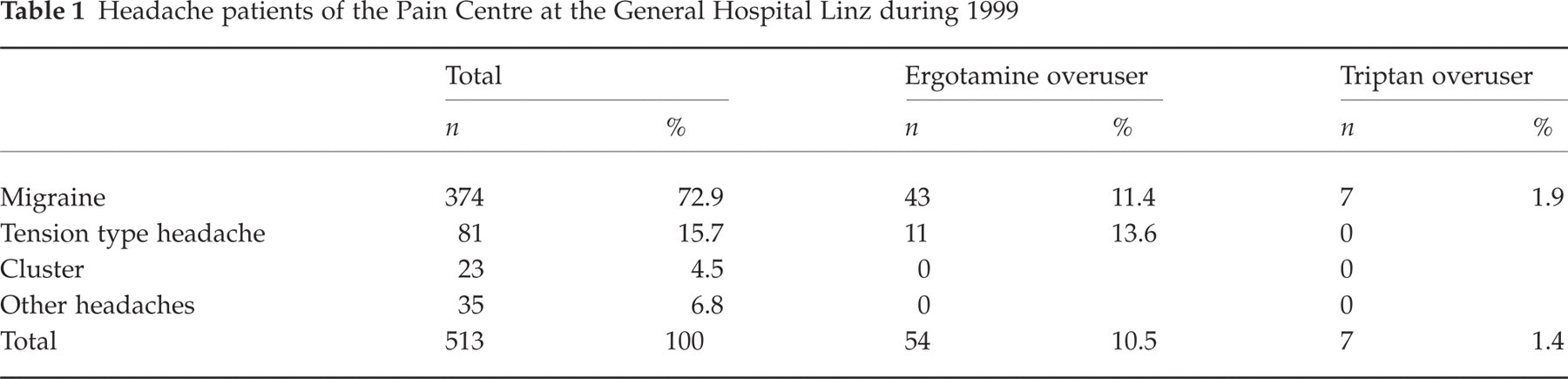
In 1999 a population of 513 new headache outpatients visited the Pain Centre of the General Hospital in Linz (Table 1). In this group we identified 374 (72.9%) patients suffering from migraine with and/or without aura. Eighty-one individuals (15.7%) were diagnosed with chronic tension-type headache, 23 (4.5%) with cluster headache and 35 (6.8%) with other headaches and facial pain syndromes. 43 out of 374 migraine sufferers (11.4%) were primarily identified as ergotamine overusers, 7 (1.9%) as triptan overusers (6 used sumatriptan, 1 zolmitriptan). The average age of the ergot overusers was 43.7 years (range: 29–68), of the triptan overusers 35 years (range: 25–48). 35 of the ergot overusers were female (81.4%) and 8 male (18.6%), all 7 triptan overusers were female. Among the 81 patients with tension-type headache 11 ergotamine overusers (13.6%) were identified. In the group of cluster headache and ‘other headaches’ no ergotamine and/or triptan overusers were found. All 54 ergotamine overusers (10.5% of all new headache patients in 1999) reported daily headache. Furthermore, 38 patients (70.4%) complained of other symptoms and signs that were attributed to ergot overuse (Table 2): nausea was reported by 24 patients (63.1%), dizziness and vertigo by 14 patients (36.8%), and fatigue by 9 patients (23.6%); three patients (7.8%) suffered from peripheral vascular insufficiency of the lower limbs and one patient (2.6%) had an anorectal ulcer all of which were attributed to ergot overuse. Triptan overuse was not associated with similar adverse effects in our small group of patients. It has to be pointed out, that none of the 374 patients with drug overuse had had an inpatient withdrawal before 1999. From the 61 overusers (54 ergots and 7 triptans) in our Pain Centre, 47 were admitted for inpatient withdrawal, all of who were overusing ergots. The average duration of hospitalization was 14,9 days.
Headache patients of the Pain Centre at the General Hospital Linz during 1999
Side-effects of ergotamine and triptans overuse

∗ergot-induced peripheral vascular insufficiency of the lower limb.
In the other 42 neurological departments 49 patients were hospitalized for ergotamine overuse, no patients overusing triptans were withdrawn. The mean duration of hospitalization was 8.2 days.
Taken together, a total of 96 patients with drug-overuse were withdrawn in Austrian neurological departments in 1999 with a mean duration of hospitalization of 11 days. One day of hospitalization at a neurological department in Austria in 1999 cost approximately ATS 4000 (1290) on average, without costs of examination and pharmacotherapy. Thus, for 1999, the cost for the in-patient withdrawal exceeded 1306 970.
In 1999, the entire pharmaceutical retail market amounted to 161 053 000 package units – 637 000 units were prescribed for the indication of headache (0.4% market share). Out of these, 430 000 were ergots (67%), with a cost of 1701 074. Together with the amount spent for inpatient withdrawal, the total amount was 11008 044.
For triptans, the cost for the 67 000 package units that were sold in 1999 amounted to a calculated cost of 12833 368. For triptans, there was no cost for inpatient detoxification.
Discussion
Austria is one of the countries where ergots are still by far the most commonly used drugs in migraine specific attack treatment. While ergots are free to prescribe and very low-priced, the reimbursement of triptans is monitored and restricted by the chief physician employed by the compulsory health care insurances. For ergots, the situation is different. Because of their cheap price, the free prescription modality in clinical practice and the absence of monitoring, overuse of ergots is to be expected and indeed a major problem.
Compared to other European countries, like Italy, Sweden, Norway or the United Kingdom, Austria shows a different development in its prescription pattern (Fig. 1). While, in Italy, the number of sold units has been balanced between ergots and triptans over the last couple of years, there is a steady relative increase of sold triptans units in the other above-mentioned countries.

Sold units of ergots (□) and triptans (▪) in some European countries illustrating a different prescription behaviour in acute migraine treatment.
The exact reasons for this prescription pattern in Austria, which is different from other European countries, are unknown. One important aspect might be the influence on prescriptions by the health insurances (vide supra), part of which is that the first prescription of a triptan has to be done by a neurologist and is not permitted for general practitioners.
In our study, we retrospectively evaluate clinical characteristics of ergot and triptan overuse, the cost of withdrawal as well as cost of medication on the pharmaceutical retail market in Austria. In the year 1999, a total of 96 patients were hospitalized for withdrawal in Austria, all of them for abuse of ergots. The calculated cost of inpatient withdrawal amounted to more than 1300 000 (without additional cost for examination and drug therapy).
From the physician's point of view, a question can be asked, whether it is ethical to prescribe a type of medication that is cheaper, but potentially more harmful than a modern, but more expensive type of drug for which side-effects of comparable severity have not been reported.
The situation in Austria in 1999 without any in-patient withdrawal because of triptan overuse is certainly not representative for European countries. Recent publications from Germany, where triptans have been used far more frequently than in Austria, show that overuse of triptans and subsequent withdrawal is a well-known clinical problem (4). Typically, the onset of chronification seems to be earlier for overuse of triptans (mean of 1.7 years compared to 2.7 years for ergots). On the other hand, for an inpatient setting, Katsarava et al. (5) reported withdrawal symptoms to be shorter following triptan overuse (4.1 days vs. 6.7 days for ergot overuse) and also less severe and at the end of withdrawal (day 14) a larger proportion of triptan overusers was headache-free (85% of triptan overusers vs. 57% of ergot overusers). There is no published data on long-term prognosis of patients after withdrawal from triptans – even the recent paper by Fritsche et al. 2001 (6) has no significant number of patients in this group.
For the prescribing physician, the quality of evidence has become another ethical aspect in decision-making. For the acute treatment of migraine with ergots the number of controlled clinical trials is small (7), most of them based on nonrandomized historical controls, individual case reports and surveillance data (2). The efficacy data are sometimes contradictory and the effect size is also relatively small. On the other hand, triptan studies provide a much higher level of evidence based on a large number of randomized, controlled trials with large numbers of patients and including clinical safety data. Due to the limited interest of industry in ergots, this is not likely to change in the future. Furthermore, due to the high first pass effect, ergots have a low bioavailability and a low degree of receptor selectivity. The recent review by Tfelt-Hansen et al. (7) includes a European consensus of physicians on the use of ergots in the acute treatment of migraine. It was concluded that for most migraine sufferers a triptan is generally a better option from both efficacy and side-effect perspectives.
If only cost of medication and withdrawal are considered in the prescription of acute anti-migraine drugs, our data are clearly in favour of a prescription of ergots rather than triptans. However, on the basis that scientific evidence clearly favours triptans, decision making for the prescribing clinicians is more complex and will include the quality of patient care.
Our study has several limitations. The data provided by the other neurological departments is somewhat less reliable than the numbers obtained from our own Pain Centre and it is also less detailed. For this reason, it is likely that we missed some drug overuse headache patients that were treated in other than neurological departments. Therefore, the number of cases, and consequently also cost, is probably underestimated in this study. A further limitation is the retrospective nature of the study.
Sumatriptan was the first triptan introduced to the Austrian market in 1992, second generation triptans followed in 1995. The low number of triptan overusers in our study probably reflects the low numbers of triptan prescriptions in Austria as detailed above. Our study takes only data from the year 1999 into account. However, on the background of the practically unchanged drug prescription numbers over the last 5 years (Fig. 2) our data have most likely preserved their actuality.

Sold units of ergots (□) and triptans (▪) on the pharmaceutical retail market 1996–2001 in Austria (data of the Institute for Medical Statistics, IMS).
The major drawback of triptans in the current health economical situation is their high direct cost. This might change, to a certain extent, as soon as sumatriptan, the first triptan on the market, will be available in form of cheaper generics. While, for the prescribing physician, the advantages in clinical care have high priority, the financial burden for the societies cannot be neglected in the decision-making processes. The development of new drugs is one of the sources of increased cost in our health economies. To make constantly improving therapies available for their populations has been and will be one of the challenges for modern societies.
Footnotes
Acknowledgements
The authors thank all chairmen of the neurological departments of Austria and their coworkers for providing us with data. We also thank Prof Stephen Silberstein, Philadelphia (U.S.A.) and Dr Volker Limmroth, Essen (Germany) for valuable suggestions on this manuscript.
