Abstract
Objective
The objective of this article is to test the hypothesis that cognitive reserve (CR) is related to migraine chronification, medication overuse and poor quality of life in migraineurs.
Design/methods
A cross-sectional study on patients with chronic migraine with medication overuse (CM-MOH), episodic migraine (EM), and controls, matched by sex, age and education, was carried out. CR was assessed by a specific questionnaire, and quality of life was measured by general and specific questionnaires (SF-36 and MSQoL). Migraine Disability Assessment Scale and Beck questionnaires for depression and anxiety were used. Medication dependence was evaluated by the medication-dependence questionnaire in headache (MDQ-H).
Results
Fifty-five individuals were enrolled: 18 CM-MOH patients (32.73%), 22 EM patients (40%) and 15 controls (27.27%). Fifty (90.91%) of them were females and aged 43.53 (7.54) years.
Univariate analysis showed a significant association between the study group and CR, and all items of the SF-36, anxiety and depression questionnaires, MSQoL and MDQ-H. The lower CR and CM-MOH group were related to a worse quality of life, more anxiety and depression and the highest medication dependence scores.
Multivariate analysis showed that higher CR scores were related to higher quality of life as measured by the physical and mental composite scores of the SF-36, and to lower anxiety (beta = −1.08, p = 0.001) and depression (beta = −0.56, p = 0.03) levels. Focusing on MSQoL, the increase in CR was predictive of a better quality of life (beta = 1.88, p < 0.0001). By all the models, the explained variance of the sample ranged from 39% (mental composite score) to 58% (MSQoL).
Conclusions
Low CR appears to be an independent factor associated with the deterioration of quality of life, the presence of anxiety and depression, and drug dependence and medication overuse in CM-MOH.
Introduction
Episodic migraine (EM) may transform into chronic migraine (CM) for unclear reasons. The latter is a disabling condition in which patients are difficult to treat, display a poor quality of life, and have frequent comorbidities, with an increased odds ratio for anxiety and depression.
A common clinical scenario is a patient with EM that transforms into CM in the setting of overusing one or more abortive drugs. Medication-overuse headache (MOH) is a subset of headache occurring on 15 days or more per month, four or more hours per day, for three or more months (1). The current classification of MOH no longer requires the improvement of headaches after medication withdrawal. The prevalence of MOH is around 1%–2% (2), and the annual incidence of new-onset MOH in patients with EM is 2.5% (3).
When patients meet the criteria for chronic migraine with medication overuse (CM-MOH), the International Headache Classification 2013 (beta version) indicates that both diagnoses should be applied (1).
Several factors, including a low socioeconomic status and household income (4), low level of education (5), high body mass index (6), female gender (7), intensity and severity of migraine attacks (8), use of opioids (9), allodynia (10), and childhood stressing experiences have been implicated in the development of MOH.
Emotions and different cognitive states, including attention and memory, modulate the perception of pain (11). Cognitive reserve (CR) has emerged as a novel concept defined as the difference in cognitive processes as a function of lifetime intellectual activities that explains differential susceptibility to functional impairment in the presence of pathology or other neurological insult (12). We hypothesize that those individuals with higher CR may be able to better modulate pain perception and therefore be less prone to CM-MOH, depression, anxiety, and therefore have a better quality of life.
In this study, we sought to establish if CR is associated with CM-MOH, depression and anxiety, and to the quality of life of these patients.
Methods
Participants
A cross-sectional study was carried out in patients meeting the criteria of the International Classification of Headache Disorders, third edition (1) for CM-MOH, and for EM, who were consecutively recruited over a six-month period from the Outpatient Clinic of General Neurology at our institution. These patients were referred by their primary care physicians for frequent headaches and therefore were not selected from a specific headache clinic. In short, the criteria for CM-MOH required that patients had headache occurring on 15 days or more per month, lasting four or more hours per day, for at least three months, which had the features of migraine headache on at least eight days per month. At the same time, they overused at least one class of migraine-abortive drug. Controls were recruited among individuals who consulted for nonspecific complaints, did not suffer from headaches and in whom neurologic examination was normal. The three groups were matched for age and education level.
Exclusion criteria included a history of chronic painful conditions or any other neurological or psychiatric disorder except for depression or anxiety that required no medication. All participants had a normal brain magnetic resonance imaging. Three CM-MOH patients had several small subcortical white matter lesions.
During the initial visit, patients were requested to keep a headache diary recording the number of headache days, pain duration and severity, and number of medication units taken. Patients were also instructed as to how to manage a migraine attack with nonsteroidal anti-inflammatory drugs and triptans. Patients were informed about the convenience of stopping medication overuse.
During the second visit, three months later, the patients’ diaries were revised, allowing for their proper allocation and the need for preventive therapy. Migraine patients filled out a CR questionnaire (see below), a test for intelligence quotient (IQ), a generic and migraine-specific quality of life test (SF-36 and Migraine-Specific Quality of Life (MSQoL), respectively), the Migraine Disability Assessment (MIDAS), the Beck anxiety and depression inventories, and a medication-dependence questionnaire in headache patients (MDQ-H). Controls completed the test battery except for those tests specific for migraine (MSQoL, MIDAS and MDQ-H).
To avoid pain interference with their performance, patients were asked to rate their pain on the second visit. To this end, a verbal rating scale, where 0 equals no pain and 10 equals excruciating pain, was used. Migraine patients scoring > 4 were asked to fill out the questionnaires on another day, once their headache had subsided.
Both preventive and abortive therapy were coded for migraine patients.
The study was approved by the ethics committee of our institution and all participants signed a written consent.
Description of tests and questionnaires
CR
CR was assessed by a specific CR index questionnaire (13,14), which includes demographic data and eight items grouped into three sections: education, working activity and leisure time. The items are parental education, formal education, courses taken, job, languages spoken, reading habits, musical education, and frequency in practicing intellectually challenging activities (crossword, chess). According to the literature (13,14), all these activities are most relevant in forming CR. Each item is rated using a response scale with three to six categories. The higher the score, the better the CR, with a maximum score of 25. Quartiles were employed to determine the CR normative scoring levels.
IQ
IQ was estimated by the Word Accentuation Test (15), which requires the pronunciation of 30 low-frequency Spanish words whose accents have been removed. This task is equivalent to the National Adult Reading Test in a Spanish-speaking population.
General quality of life—SF-36 (16)
The SF-36 is a structured, self-reported questionnaire that includes 36 items measuring health status across eight domains. The scoring system generates subscale scores for physical functioning (PF), role limitations due to physical problems (RP), bodily pain (BP), general health perceptions (GH), vitality (VT), social functioning (SF), role limitations due to emotional problems (RE) and mental health (MH). Two summary scores derive from the SF-36: the physical component (PF, RP, BP and GH) and the mental component (VT, SF, RE and MH). The SF-36 scores range from 0 to 100, with a higher score indicating better health status. Scores for each individual were standardized for comparison with the Spanish population.
MSQoL (17)
The MSQoL is a self-administered questionnaire that consists of 20 items, each of which is rated using a response scale with four categories (1 = very much to 4 = not at all) grouped into three dimensions: avoidance (10 items), social relationships (six items) and feelings (four items). The scores were determined by adding the items for each domain. An overall score was also determined by adding the 20 items. Scoring ranged from 20 to 80. Higher scores indicate better quality of life.
MIDAS
This questionnaire assesses headache-related disability (18). Migraine patients answer five questions about the frequency (days) and duration of their headaches in the last three months, as well as how often these headaches limited their ability to participate in activities at work, at school, or at home. Scoring goes from 0 to five days, meaning no disability, to >21 days, representing severe disability.
Anxiety and Depression evaluation
We employed the Beck Depression (BDI) and Anxiety Inventories (BAI) (19). These questionnaires consist of 21 self-administered items about how the patient has been feeling in the last week. Each question has a set of at least four possible choices ranging in intensity. Scoring between 11 and 18 indicates a mild depression, between 18 and 25 moderate depression, and over 30 severe depression. In the BAI, scoring 21 points or less represents a low anxiety level, between 22 and 35 moderate anxiety level, and over 36 severe anxiety.
MDQ-H
Medication dependence was assessed by the medication-dependence questionnaire in headache patients (MDQ-H) (20). MDQ-H consists of a 21-item, self-administered questionnaire inquiring about the number of units of treatment taken per week, the number of days of headache, and emotional distress related to the lack of medication, among others. For each item, subjects are asked to describe their medication consumption according to a seven-point Likert scale (1: never or not at all; 3: sometimes or a little; 5: often or quite a lot; and 7: always or completely). Total score was obtained by adding the scores for all the items. A high score means an important disturbance in the way the patient uses his or her medication.
All the questionnaires employed in this study have been validated in Spanish except for the MDQ-H (validated in French and English).
Statistical analysis
Descriptive statistics of socio-demographic variables and clinical data of the three groups of patients (CM-MOH, EM and control) were calculated using means and standard deviations for quantitative data, while categorical variables were expressed in terms of frequencies and percentages. We evaluated the relationship between the collected variables and study group using the Chi-square test (or Fisher’s Exact test when expected frequencies were lower than 5) for categorical variables, and nonparametric tests (Wilcoxon and Kruskal-Wallis) for continuous variables.
Mean and standard deviation of measured domains of the evaluated questionnaires (SF-36, MSQL, MIDAS, BDI and BAI, MDQ-H and IQ) were calculated. In order to gauge differences in the subscales across patients groups, Wilcoxon and Kruskal-Wallis tests were used. In addition, we conducted a specific post-hoc test to identify specific differences between groups, developing a relevant paired test for each pair and using Wilcoxon test and Bonferroni correction. The latter technique was also used for the assessment of the relationship of the aforementioned scales with the stratified CR levels according to quartiles (low: CR ≤ 11; low–medium: 11< CR≤ 15; medium–high: 15< CR≤ 18; high: CR> 18). When CR was considered continuous, Pearson’s correlation coefficient was calculated.
Finally, a multivariate analysis was undertaken to determine the role of CR on quality of life and BAI and BDI scales. To this end, the scores of the physical and mental components of the SF-36, and Beck anxiety and depression inventories were considered as outcomes whereas CR (as continuous) and patient group (controls as reference), age, gender and migraine therapy (abortive and preventive) were the independent ones. The MSQoL was also included in the analysis of migraine patients as the outcome with the aforementioned scores as independent variables. In each model, the variables significant at the 0.20 level in the univariate analysis were considered as potential independent variables. Several multiple linear regressions were undertaken in order to determine which variables represented final predictors with a significance level of 0.05 by using a backward selection process. To ascertain the percentage of explained variance of the outcomes by the final multivariate models, the R-square was computed.
All effects were deemed statistically significant at p < 0.05. All statistical analyses were performed using SAS System, version 9.2 (SAS Institute Inc, Carey, NC, USA).
Results
Fifty-five individuals were enrolled and divided into three groups that included 18 CM-MOH patients (32.73%), 22 EM patients (40%) and 15 controls (27.27%).
Descriptive analysis of socio-demographic and clinical data of the studied sample.

CM-MOH: chronic migraine with medication overuse; EM: episodic migraine; NA: not applicable for control patients; MIDAS: Migraine Disability Assessment.
Mean and standard deviations of the measured questionnaires (SF-36, MSQoL, Beck, MDQ-H and IQ) and cognitive reserve test according to the study group.
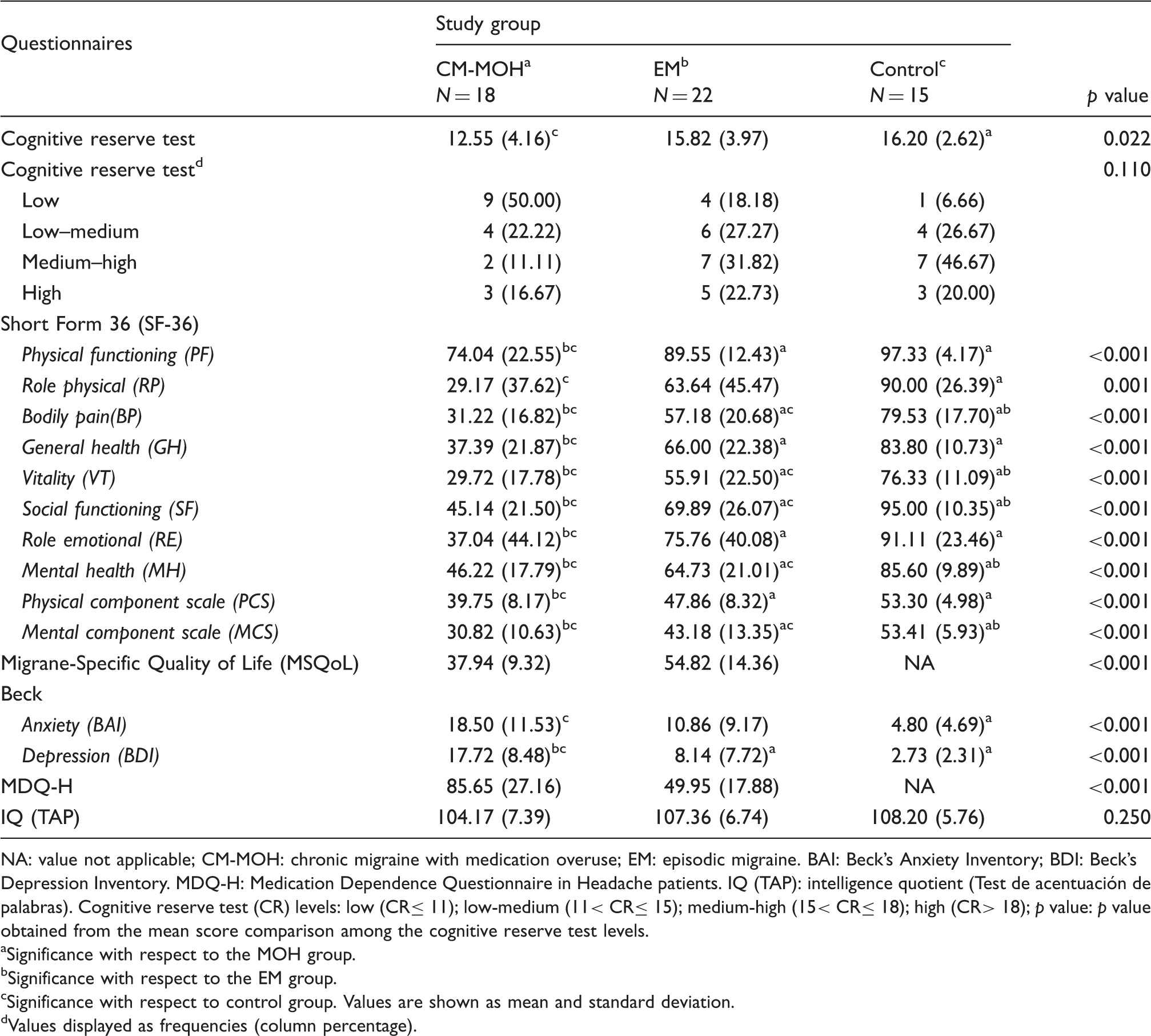
NA: value not applicable; CM-MOH: chronic migraine with medication overuse; EM: episodic migraine. BAI: Beck’s Anxiety Inventory; BDI: Beck’s Depression Inventory. MDQ-H: Medication Dependence Questionnaire in Headache patients. IQ (TAP): intelligence quotient (Test de acentuación de palabras). Cognitive reserve test (CR) levels: low (CR≤ 11); low-medium (11< CR≤ 15); medium-high (15< CR≤ 18); high (CR> 18); p value: p value obtained from the mean score comparison among the cognitive reserve test levels.
Significance with respect to the MOH group.
Significance with respect to the EM group.
Significance with respect to control group. Values are shown as mean and standard deviation.
Values displayed as frequencies (column percentage).
Regarding the quality of life measures, mean scores and standard deviations of the different questionnaires according to patient group are also shown in Table 2. Statistically significant differences were found for all scores, except for the IQ (p = 0.250) scale. CM-MOH patients displayed significantly lower quality of life and CR values as compared to controls and EM patients, as well as higher anxiety and depression scores. Regarding the specific MSQoL, EM patients had higher scores as compared to CM-MOH patients (controls did not fill out this questionnaire). MIDAS was also worse in CM-MOH patients as compared to the EM group.
Mean and standard deviations of the measured questionnaires (SF-36, MSQOL, BECK, MDQ-H and IQ) and cognitive reserve test according to cognitive reserve test levels.

Values are displayed as means and standard deviation. BAI: Beck’s Anxiety Inventory; BDI: Beck’s Depression Inventory. MDQ-H: Medication Dependence Questionnaire in Headache patients. IQ (TAP): Intelligence Quotient (Test de acentuación de palabras). Cognitive reserve test (CR) levels: low (CR≤11); low-medium (11 <CR≤15); medium-high (15<CR≤18); high (CR>18); p value: p value obtained from the mean score comparison among the cognitive reserve test levels.
Significance with respect low CR group.
Significance with respect low-medium CR group.
Significance with respect medium-high CR group.
Significance with respect high CR group.
Pearson’s correlation coefficient was calculated to assess the association between the cognitive reserve test (as continuous) and the scales of the questionnaires.
The influence of the cognitive reserve in the anxiety, depression and quality of life, measured with different questionnaires and adjusted by patient group.

Generalized linear models were used. CM-MOH: chronic migraine with medication overuse; EM: episodic migraine. R2 (%): coefficient of determination, in percentage. Beta (s.e.): beta coefficient (standard error). p value: p value of the beta coefficient. Reference: reference group, in this case, controls are considered.
Generalized linear model was developed only for the CM-MOH and EM patient sample, since the MSQoL is a migraine-specific questionnaire and controls did not respond the survey. NA: not applicable for control patients.
Discussion
This study shows for the first time a clear association between CR and CM-MOH. The statistical models indicated that the combination of CR and patient group explained a 40% of the variance after proper adjustment for other variables, consistent with a strong association. Additionally, the explained variance by the CR itself ranged from 18% to 47%.
CR may contribute to these results by several mechanisms. CR explains the discrepancy between a person’s underlying level of brain pathology and the resulting cognitive deficits expected from that pathology. Such a discrepancy relates to the inter-individual variability in the efficiency of compensatory brain circuits that protect against brain insults (12). Although the neural substrate of the CR is unclear, an important component is the locus coeruleus (21), also a main component of the pain modulatory pathways involving projections from the midbrain periaqueductal gray matter to brainstem nuclei, and to the dorsal horn of the spinal cord (11). On the other hand, MOH patients have pronociceptive facilitation as well as weakened descending pain inhibition. It seems plausible, therefore, that those individuals with higher CR may display better pain-control mechanisms, explaining at least in part its negative association with CM.
Furthermore, chronic pain induces anatomic changes in several brain regions related to pain perception and chronification (11). The three cortical regions that consistently show decreases in gray matter are the anterior cingulate cortex, prefrontal cortex and insulae, all of them also involved in central pain modulation. Their dysfunction in MOH patients may contribute to poorer pain control and to addiction behavior in those individuals with a lower CR (22,23). Changes in pain sensitivity as a result of the aforementioned anatomical changes play a role in migraine chronification (24), which is consistent with the fact that allodynia, a marker of central sensitization, is an important factor in this regard (25).
CR has never been implicated as a protective factor in chronification of migraine but it seems to play a role. In fact, Attal et al. found that a limited cognitive flexibility predicted the development of chronic pain after surgery, suggesting that cognition is a protective factor against pain (26).
Cognitive and emotional factors have an important influence on pain perception. A negative expectation can reverse the analgesic effect of an opioid, whereas the expectation of pain relief is an important component of placebo analgesia. Clinical and experimental studies have shown that simple psychological manipulations, such as distraction, can have a powerful effect on our perception of pain (11).
The fact that individuals with poorer mental abilities are prone to develop chronic pain has been explained by their difficulties in interpreting bodily symptoms correctly and their heightened sensitivity to the workings of their body. Thus, people with lower intelligence and CR may be less able to apply appropriate coping strategies when faced with pain, increasing the likelihood of pain chronicity (27).
Intelligence has been associated with CR or resilience in normal population, and is considered as a complex capacity that is dependent on a global cognitive network rather than isolated brain areas (28). Thus, it is plausible that people with higher CR have more stable brain networks resulting in finer tuning of different functions including pain control.
Our study shows that CR is also inversely associated with the quality of life of these patients, since those patients with lower CR values scored significantly worse in the quality of life questionnaires, not only in emotional but also in physical components, including pain perception. This association was evident both with generic and migraine-specific quality of life questionnaires. Prior studies revealed a decreased score in all health-related domains of the SF-36 for patients with MOH compared with healthy individuals, particularly for bodily pain and physical functioning domains (29), but they did not analyze the influence of CR on the quality of life of these patients.
It is important to emphasize that there was not only a between-group association of CR and quality of life, but also a within-group association, i.e. among patients with CM-MOH those with lower CR scores also had a worse quality of life (generic and specific) perception. As an example, a CM-MOH patient with a CR score of 7 will score 25.78, 36.78 and 27.45 in the PCS, MCS and MSQoL, respectively. In contrast, a CM-MOH patient with a 24 score in CR will score 41.08, 46.13 and 59.41, in the PCS, MCS and MSQoL, respectively. On the other hand, the physical and mental composite scores of the SF-36 showed that those individuals with the highest CR scored similarly to the general Spanish population, again reinforcing the clear inverse relationship between quality of life and CR.
CR was inversely related to substance dependence according to the specific questionnaire employed in this study (MDQ-H). In fact, a large proportion of CM-MOH patients fit the criteria for substance dependence in the Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition. The presence of high values in the MDQ-H among patients with lower CR strongly supports the role of CR in promoting or facilitating drug overuse.
CR is also highly associated with anxiety and depression (30), two main comorbidities in headache patients, particularly those with CM. It remains to be seen if improving these conditions results in a quality of life improvement in migraine patients (31).
Finally, limitations should also be noted. The main weakness of our study was the sample size. The sample size obtained in our study is modest, since the recruitment period was only six months; it could be enlarged to increase the number of potential study participants. Moreover, as this is a cross-sectional study, where causal sequence is uncertain, it allows establishing an association between CR and CM-MOH, but does not permit us to formulate a causal relationship, either direct or reverse. Further follow-up studies are suggested to assess the causality. Eventually, an impact of preventive medications (i.e. topiramate) on CR is unlikely, since CR is a lifetime trait and does not include parameters (i.e. reaction time, planning) that can be interfered with by these drugs.
In conclusion, CR could represent a risk factor associated with migraine chronification, and may influence the quality of life of these patients and contribute to their potential for medication overuse. Future prospective studies should indicate if CR constitutes a predictor in the evolution of these patients.
Clinical implications
This article shows for the first time a clear association between cognitive reserve and chronic migraine with medication overuse. This association may allow for a prediction of the future in patients with headache, so as to forecast which patient will become a chronic migraineur and/or prone to medication overuse. Also, it reflects the importance of cognitive reserve in their quality of life and pain perception.
Footnotes
Funding
This work was supported by a grant of the Instituto de Salud Carlos III, FIS PI11/01243.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
