Abstract
Chronic migraine accompanied by medication overuse is particularly difficult to treat. The number of treatment investigations is limited, few have included follow-up beyond 6 months and almost none has examined whether treatment leads to concurrent improvements in disability and functional impairment. This open-label study addresses these limitations. We have been prospectively following an initial cohort of 84 chronic migraine patients with medication overuse, who at the time of this evaluation had been reduced to 58, for an extended period to assess longer-term maintenance of effects, using measurement procedures identical to those in the original investigation. Thus, the specific aim was to determine the clinical status, with respect to pain indices and disability level, of chronic migraine patients with medication overuse who were treated and followed prospectively for 5 years. All patients completed a brief inpatient treatment programme, in which they were withdrawn from their offending medications and subsequently placed on more appropriate preventive antimigraine medications. Both end-point, wherein missing data points were estimated, and continuer analyses, wherein data analysis was limited to the 58 individuals with complete datasets, revealed significant improvement on all measures studied—headache days per month, analgesic consumption and Migraine Disability Assessment (MIDAS) total score. The percentage reduction from baseline to 5 years for the MIDAS total score was 76.0%, while the percentage of individuals revealing improvements of clinically significant magnitude (≥ 50%) on the MIDAS was 91.9%. MIDAS total scores were lower at 5 years than at some of the intervening follow-up intervals. Comparisons of those who completed the 5-year follow-up (n = 58) with those who did not revealed no differences at baseline. This finding, coupled with the nearly identical results for the end-point and continuers analyses, suggests that attrition did not have a bearing on outcome. None of the patients completing the 5-year follow-up had relapsed since the prior 3-year follow-up assessment. High levels of maintenance were revealed at 5 years, with disability scores showing some continued improvement over time. The implications of these findings and the limitations of the study are discussed.
Medication overuse is a significant problem among headache patients, especially so for migraineurs. For example, migraine is much more likely to be chronic (odds ratio 10.3), and thus more difficult to treat, when accompanied by analgesics overuse (1). A longitudinal study revealed that individuals who were taking excessive analgesics at the start of the study were much more likely to develop chronic migraine (relative risk 13.3) when reassessed 11 years later (2). These individuals are difficult to treat, relapse is typically high and little is known about long-term maintenance of effects (few studies have examined effects beyond 1 year) and what factors best predict outcome (3–6). Finally, minimal attention has been given to important outcome indicators beyond pain symptoms, such as impact and overall functioning. This is a significant shortcoming, as reduction in headache-related disability is one of the main treatment goals specified in the recent guidelines for the management of headaches (7).
We have been conducting a series of studies addressing many of these identified problems. Previously we published 1- and 3-year follow-up data for a group of migraine patients whose headaches were complicated by medication overuse (8,9). By repeatedly administering the Migraine Disability Assessment (MIDAS) (10), which measures the impact of headache on various life dimensions, we have been able to examine how this headache condition is influencing functioning as well as pain levels. These studies showed that treatment led to significant improvements in disability and functional impairments, as well as improvements in key headache indices. We have continued to follow the patients from this trial, incorporating measures identical to those earlier utilized. We now report on the clinical status of this group at five full years.
Methods
Patients
Type of medications taken at the start of the study by the 84 initial patients and the 58 patients who completed the 5-year follow-up evaluation

NSAIDs, non-steroidal anti-inflammatory drugs.
Outcome measures
Three primary measures were used to assess outcome, with two being extracted from 30-day headache diaries (12–14): (i) number of days per month during which any type of headache was present; and (ii) number of analgesic pills consumed per month. The remaining measure consisted of the total score derived from an Italian translation of the MIDAS that was developed and tested using a standardized methodology (15). The MIDAS contains five questions that inquire about headache impact over the prior 90 days (days of school or work missed, days of reduced productivity at work or school, days unable to complete household work, days of reduced productivity in household work and days of family, social or leisure activity missed). Responses to these five questions are summed to yield an overall or total score. MIDAS total scores can be grouped into four disability grade levels: I, little or no disability (0–5); II, mild disability (6–10); III, moderate disability (11–20); and IV, severe disability (21+). The MIDAS contains two additional questions that ask individuals to estimate headache frequency and intensity over the same time period. These latter two ‘retrospective’ measures are not reported here because of availability of ‘prospective’ diary-based measures. All measures were obtained prior to treatment and at 1, 2, 3 and 5 years following treatment completion.
Procedure
All 84 patients were provided a semi-standardized inpatient withdrawal treatment that consisted of the following (8,16): (i) intravenous hydration for 10 days; (ii) intramuscular indomethacin (maximum dose once per day) for patients experiencing severe rebound headaches during the first 3–4 days of withdrawal; and (iii) intravenous metoclopramide, intramuscular diazepam, parenteral neuroleptics or corticosteroids for those patients who revealed more intense withdrawal symptoms (vomiting, sleep disturbances, agitation). On day 6, patients were begun on one of several prophylactic antimigraine compounds recommended by published guidelines (7) (e.g. flunarizine 5–10 mg/day, pizotifen 2–3 mg/day, or propranolol 40–80 mg/day; amytriptyline 10–30 mg/day was added when associated mood disorders were present). Choice of medication was based on patient characteristics such as age, headache history and comorbid conditions (such as hypertension or depression), with the goal of minimizing the number of different compounds taken. Patients were encouraged to limit non-prophylactic medications to triptans and/or non-steroidal anti-inflammatory drugs, with no more than eight tablets being taken each month.
Results
The primary data analysis consisted of two doubly multivariate
End-point analyses
The MIDAS total scores from baseline through 5 years of follow-up for the end-point and continuers analyses. Mean values and standard deviations (within parentheses) for the three measures for the end-point analyses, with percentage improvement relative to pretreatment and percentage of sample revealing clinically significant improvement at 5 years MIDAS, Migraine Disability Assessment.
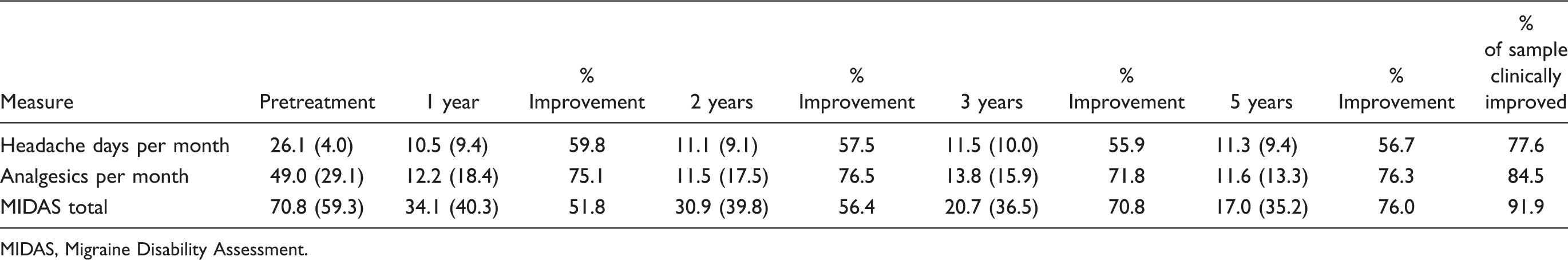
When comparing disability grade levels at 5 years with those at the start of treatment, 78% dropped to a lower grade level whereas 22% remained at the same grade level. No patient increased to a higher grade level. Thirty-eight individuals (65.5%) dropped from the highest disability grade (level IV) to the lowest disability grade (level I).
Continuers analyses
The
Without exception, post hoc pair-wise comparisons with Bonferroni adjustments showed that every data point subsequent to the pretreatment value was significantly less for every measure. The amount of improvement obtained at 5 years exceeded that at 1 and 2 years for MIDAS total scores (as found in the end-point analyses).
Continuers vs. non-completers
Gender distribution and mean pretreatment values for continuers (n = 58) vs. non-continuers (n = 26), along with statistical tests for differences as appropriate

MIDAS, Migraine Disability Assessment; NS, not significant.
Discussion
This long-term follow-up study shows improvements made following treatment have held very well for a full 5 years, no matter which analysis is used and which dataset is included. Furthermore, the amount of improvement observed at 5 years for the MIDAS total scores exceeded that observed in some of the intervening years. The fact that some patients reported further improvements with respect to functional impact is quite encouraging. Deserving of further comment are the findings of the high level of overall percentage improvement shown on the MIDAS, the high percentage of patients revealing improvements of ≥ 50%, and the large number of patients dropping to a lower disability grade level, many of whom dropped from the top to the lowest grade level. In fact, the level of disability shown as a whole by the group of patients is now comparable to that of a population of non-migraine headache sufferers (17). Jacobson and Truax (18) have suggested several criteria for judging clinically significant improvement. Two of the criteria they mention seem to be satisfied here: having a high percentage of individuals who show a given level of improvement, and achieving high end-state functioning. Although the magnitude of reductions in medication was considerable, on average the level of medication consumption continues at a less than desirable level (mean exceeds our monthly target of eight or fewer tablets).
These encouraging findings notwithstanding, we acknowledge a number of limitations. A major limitation continues to be the absence of a control or comparison condition. Comparative outcome investigations, incorporating appropriate control and/or comparison conditions [such as inpatient vs. out-patient, pharmacological alone vs. pharmacological augmented by behavioural and related procedures (16,19)], examining predictors of treatment response (such as type of medication and duration of use, comorbid conditions) (20,21), and attempting to identify specific categories of medication that, when overused, increase the likelihood of progression to chronic migraine, are warranted. Regrettably, we did not collect sufficient information to conduct such analyses with our present cohort. Preliminary analyses of relationships between age, gender and illness duration revealed no significant findings. It is of course not possible to determine the exact status of those patients lost during follow-up, and some may indeed have relapsed. Without an appropriate control/comparison condition we cannot determine the effects of regression to the mean or rule out other factors that may have influenced outcome (seeking treatment elsewhere, life circumstances, reporting biases, etc.). Finally, although the MIDAS is generally accepted as a psychometrically sound measure of disability, it would be preferable in the future to include additional measures of functional impact.
Footnotes
Statistical Analyses Performed by: S. Kass and F. Andrasik.
Competing interests
None to declare.
