Abstract
Background
Functional and structural disruptions to the pain matrix, which may involve changes in white matter (WM) pathways connecting the pain-processing system and hypothalamus, have been implicated in the pathophysiology of cluster headache (CH). However, previous studies have obtained inconclusive results regarding WM changes in CH, and WM variations between “in-bout” and “out-of-bout” periods of CH remain to be determined.
Methods
Multiple diffusivity indices obtained by diffusion tensor imaging (DTI) and post-hoc probabilistic tractography were used to elucidate CH pathophysiology.
Results
Compared to healthy participants, in-bout CH patients showed regionally higher absolute (radial and mean) diffusivities in the left medial frontal gyrus and frontal sub-gyrus and lower absolute (axial, radial and mean) diffusivities in the right parahippocampal gyrus of the limbic lobe. These changes during the in-bout period generally persisted in the out-of-bout period, except for the left cerebellar tonsil. Post-hoc probabilistic tractography showed highly consistent anatomical connections between these altered areas and the hypothalamus across participants.
Conclusions
Distinct WM changes were observed in episodic CH. Connections between the pain-modulation areas and hypothalamus may be involved in CH pathophysiology.
Keywords
Introduction
Cluster headache (CH) is a primary headache disorder that presents with periodic, severe unilateral periorbital pain, accompanied by parasympathetic or sympathetic symptoms. CH affects <1% of the population, mainly men (1,2). Episodic CH, which accounts for 80%–90% of CH cases, usually manifests in “bout periods” of daily high-frequency attacks lasting from one week to several months (the “in-bout” period), separated by month- to year-long remission (“out-of-bout” period) (1). The pathophysiology underlying CH is not completely understood. Previous functional imaging studies suggest that hypothalamic dysfunction is involved in CH pathophysiology (3–7). May et al. (8) demonstrated an increased posterior hypothalamic volume in CH patients, during both in-bout and out-of-bout periods, but subsequent structural magnetic resonance imaging (MRI) studies did not confirm this finding (9,10). Therefore, the involvement of structural changes in the hypothalamus in CH remains debatable.
Pain transmission in the central nervous system is highly complex and involves several brain structures/systems, traditionally known as the “pain neuromatrix,” which primarily includes regions involved in ascending pain processing (e.g. the sensory cortex, inferior parietal cortex and posterior insula) and those involved in descending pain modulation (e.g. the frontal cortex, cingulate cortex and anterior insula). Functional imaging studies have shown activation in regions associated with the pain neuromatrix during acute CH attacks (6,8,11). Moreover, Sprenger et al. (12) described changes in brain metabolism between in-bout (without acute attacks) and out-of-bout periods, with increased metabolism in the frontal pain-modulation areas and decreased metabolism in the cerebellopontine area. In addition, a structural MRI study (9), as well as our recent work (10), revealed reductions in the gray matter (GM) in regions associated with pain processing and modulation in CH patients during and between the in-bout and out-of-bout periods.
Collectively, these data support the notion of structural and functional alterations in pain-processing and -modulation regions in CH patients, with significant dynamic changes between the out-of-bout and in-bout (without acute attacks) periods. These alterations may also involve changes to white matter (WM) pathways. Studies of other chronically painful conditions (13,14) have found WM abnormalities in brain areas involved in sensory, modulatory and cognitive functions. Some of these WM abnormalities were associated with the clinical profiles for different conditions. Therefore, investigating the correlation between CH characteristics and measures of WM integrity may provide insights into whether CH drives (yet uncertain) changes in the WM microstructure.
Diffusion tensor imaging (DTI) is a novel MRI technique that enables noninvasive evaluation of microstructural alterations in brain WM (15). It uses a diffusion ellipsoid model to represent the three-dimensional characteristic of water molecular diffusion within the WM, and the ellipsoid can be decomposed into three mutually orthogonal eigenvectors and eigenvalues: the diffusion coefficient along the direction of maximal apparent diffusion (axial diffusivity or λ1), and the diffusion coefficients along two orthogonal directions embedded perpendicular to the maximal diffusion direction (λ2 and λ3). The most commonly used quantitative indices are mean diffusivity (MD, the mean of the three eigenvalues = [λ1 + λ2 + λ3]/3) and fractional anisotropy (FA, the standard deviation of the three eigenvalues), which reflect the amplitude and directionality of water diffusion within a measured region, respectively. MD and FA may be influenced by myelination, coherence, packing density, axonal diameter, neural tract structural integrity and fiber architecture complexity (16).
The directional diffusivities—axial (Dax = λ1) and radial [Drad = (λ2 + λ3)/2]—may provide additional information on the potential underlying pathophysiology of WM changes (15). Alterations in Dax suggest axonal damage and/or Wallerian degeneration in the primary fiber orientation, whereas increased diffusivity perpendicular to the WM tract (Drad) implies demyelination or dysmyelination (17,18). The principal orientation of the diffusion (derived from DTI) can also noninvasively reveal WM pathways (19). Consideration of the different directional diffusivities is important because if the changes in diffusion along the axial direction (Dax) are proportional to those along the radial directions (Drad), then the FA (which is a function of the ratio) will remain unchanged (20).
Recent studies have utilized DTI to investigate potential WM microstructural changes in CH patients (9,21,22). However, their results are controversial. One study reported no diffusivity abnormalities of WM in CH patients compared to healthy controls (9), while two other studies found WM microstructural changes in regions widespread in the brain, mainly belonging to the pain matrix (21,22). The discrepancies in these findings may be attributable to the differences in the CH patients investigated: Absinta et al. (9) and Szabó et al. (21) recruited only out-of-bout CH patients, while Teepker et al. (22) recruited both in-bout and out-of-bout patients without assessing changes between the two groups.
In this study, we aimed to elucidate the potential involvement of the pain matrix and underlying WM microstructural changes in episodic CHs; to do this, we performed voxel-wise analyses of multiple diffusivity indices and probabilistic fiber tractography in both in-bout and out-of-bout patients, and compared these results with those for healthy volunteers. We sought to determine whether CH patients show: (1) altered WM microstructural integrity in the pain neuromatrix and hypothalamus, particularly in descending pain-modulation areas; (2) WM changes between in-bout and out-of-bout periods; (3) anatomical connections between brain areas with WM changes and the hypothalamus; and (4) correlations between WM changes and clinical variables.
Methods
Participants
Seventeen consecutive patients meeting the criteria for episodic CH were recruited from the Headache Clinic of Taipei Veterans General Hospital, Taiwan (2). Patients with vascular disease (including hypertension), diabetes mellitus and other major systemic or neurological/psychiatric conditions were excluded. All patients were first scanned during the in-bout period, with a second scan during the out-of-bout period. Average time between the scans was 9.1 months (range, six to 13 months). All patients were in a bout period for at least one week at the time of the first MRI, and were allowed to take triptans as acute pain treatment, along with verapamil or lithium as prophylactic medications. At the time of the MRIs, none of the patients had acute CH attacks.
Seventeen sex-, age- and handedness-matched healthy volunteers with no family history of CH or migraine, no previous diagnosis of other primary or secondary headache disorders except for infrequent episodic tension-type headaches (<1 day/month) and no other chronic pain conditions were recruited. All participants provided written informed consent to the institutional review board of Taipei Veterans General Hospital.
Image acquisition, preprocessing and tract-based spatial statistics (TBSS)
Imaging was performed with an eight-channel head coil on a 1.5 T whole-body MRI system (GE Medical Systems). Whole-brain DTI images were acquired using a single-shot spin-echo echo-planar imaging sequence in the axial-plane with the following parameters: repetition time/echo time = 17,000/67.8 ms; number of excitations = 6; matrix size = 128 × 128; field of view = 26 cm; slice thickness = 2.2 mm; 70 slices;
Statistical analyses of TBSS
The fsl RANDOMISE tool was used to conduct a permutation-based nonparametric inference in order to investigate the regional WM microstructural differences between CH patients and controls (analysis of covariance (ANCOVA) model with age and sex as covariates of no interest) and the longitudinal changes between the in-bout and out-of-bout periods of CH patients (paired
Post-hoc probabilistic tractography for the significant clusters in TBSS analysis
Probabilistic tractography was used to investigate the possible anatomical connections between the hypothalamus and WM areas with significant DTI index changes. For this method, clusters with significant changes in the diffusivity index were used as individual seed masks, and the hypothalamus ipsilateral to WM areas with significant DTI index changes was used as a waypoint mask. Tractography was performed in native DTI space by repeatedly sampling 5000 times at each voxel in a seed mask with a modified Euler integration approach. A curvature threshold of 0.2, step length of 0.5 mm, and a maximum number of steps of 2000 served as termination criteria for fiber tractography. The resultant individual native space connectivity distribution maps were then projected into Montreal Neurological Institute (MNI) space to produce population-based probability maps for anatomical connections between the ipsilateral hypothalamus and clusters with significant diffusivity index changes. Values reported in tractography maps in the figures reflect the proportion of subjects in whom a pathway was present (for details of the tractography reconstruction pipeline, see the eMethods in the Supplemental Material).
Statistical analyses of demographic and clinical characteristics
Statistical analyses of demographic and clinical characteristics were performed with SPSS (version 17 for Windows; SPSS) with the
Results
Demographics
Demographic and clinical characteristics of the patients with CH and healthy controls.

The variables are reported as means ± SD. The
CH: cluster headache; BDI: Beck Depression Inventory; HADS: Hospital Anxiety and Depression Scale.
Regional WM diffusivity index changes between CH patients and controls
Compared to controls, CH patients during the in-bout period had regional decreases in the MD, Dax and Drad for the contralateral (right side) parahippocampal gyrus of the limbic lobe (Figure 1, Table 2, Supplementary Figure 1). We observed a regional increase in the FA for the subgyral area of the ipsilateral frontal lobe and increased MD and Drad for the ipsilateral medial frontal gyrus during the in-bout period (Figure 2, Table 3, Supplementary Figure 2). Regional MD decreases were also detected in the contralateral subgyral area of the frontal lobe and the extranuclear area of the putamen (Figure 1, Table 2). These major WM changes in the parahippocampal gyrus, amygdala, insula, frontal sub-gyral area, extranuclear area of the putamen and medial frontal gyrus during the in-bout period also persisted during the out-of-bout period.
Regions showing significant reductions in diffusivity indices in CH patients compared with NCs. Different color maps show the cluster-level statistics with the FWE-corrected Regions showing significant diffusivity index elevations in CH patients compared with NCs. Different color maps show the cluster-level statistics with the FWE-corrected Anatomical regions with significant DTI index reductions in patients with CH compared with normal controls. All the diffusivity index values in this table were directly extracted from the significant clusters found in the voxel-wise TBSS analysis. Indicates that there was no corresponding cortical area region near the center of the 5 mm radius search range for the coordinates of the significant clusters. The unit of diffusivity indices is 103 × mm2/s. DTI: diffusion tensor imaging; CH: cluster headache; Dax: axial diffusivity; Drad: radial diffusivity; FWE: family-wise error; MD: mean diffusivity; MNI: Montreal Neurological Institute; NC: normal control; SD: standard deviation; TBSS: tract-based spatial statistic. Anatomical regions with significant DTI index elevations in patients with CH compared with normal controls. All diffusivity index values in this table were directly extracted from the significant clusters found with the voxel-wise TBSS analysis. tmax values represent the maximum t value of clusters that passed the cluster-based statistical criteria with DTI: diffusion tensor imaging; CH: cluster headache; Drad: radial diffusivity; FA: fractional anisotropy; FWE: family-wise error; MD: mean diffusivity; MNI: Montreal Neurological Institute; NC: normal control; SD: standard deviation; TBSS: tract-based spatial statistic.



Regional WM diffusivity index changes between in-bout and out-of-bout periods in CH patients
Compared to out-of-bout scans, the in-bout scans of CH patients showed only a significant regional increase in Dax in the ipsilateral cerebellar tonsil (Figure 3, Table 4, Supplementary Figure 3). No regional FA, MD, or Drad differences were found between the in-bout and out-of-bout periods in CH patients.
Longitudinal white matter changes between in-bout and out-of-bout periods of CH patients. Compared with out-of-bout scans, in-bout scans were associated with regionally higher axial diffusivity in the left cerebellar tonsil. The violet color map shows the cluster-level statistics with the FWE-corrected Anatomical regions with significant axial diffusivity alterations between the in-bout and out-of-bout periods of patients with CH. The axial diffusivity values in this table were directly extracted from the significant cluster found with the voxel-wise TBSS analysis. CH: cluster headache; Dax: axial diffusivity; DTI: diffusion tensor imaging; FWE: family-wise error; MNI: Montreal Neurological Institute; WM: white matter; SD: standard deviation; TBSS: tract-based spatial statistic.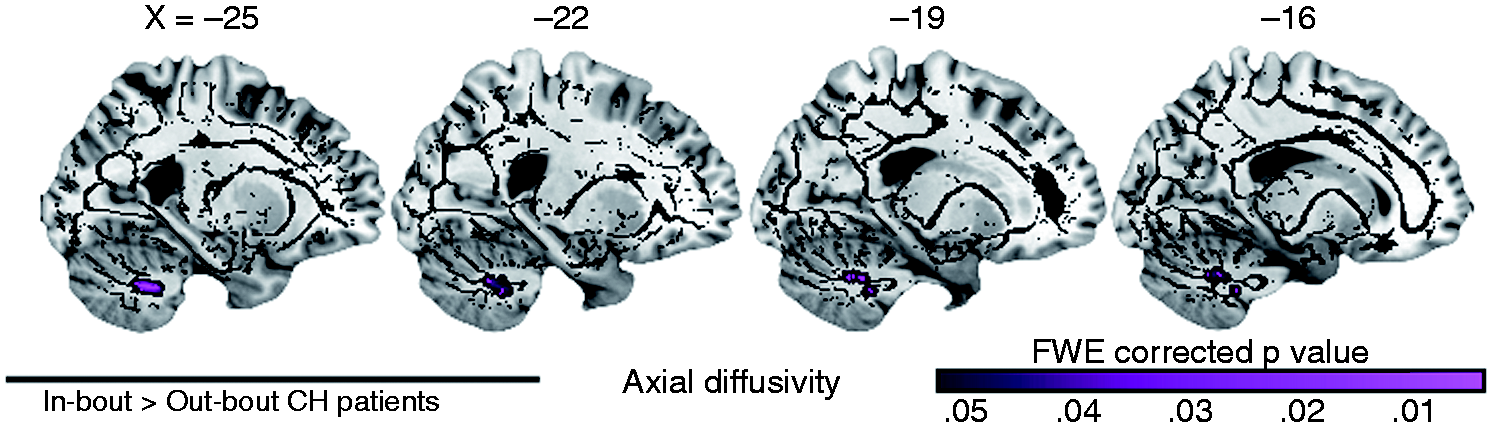

Consistent and direct anatomical connections between clusters with changes in diffusivity indices and the hypothalamus
Our population-based probabilistic tractography results demonstrated, across all study participants, the existence of highly consistent and direct anatomical connections between all the areas showing changes in diffusivity indices and the ipsilateral hypothalamus (Figure 4, Supplementary Figure 4).
Population-based probabilistic anatomical connection maps between the hypothalamus and clusters with significant between-/within-group effects. We performed probabilistic tractography using (a) a significant FA alteration cluster in the WM of the left frontal sub-gyrus, (b) a significant MD alteration cluster in the WM of the left medial frontal gyrus, (c) a significant axial diffusivity alteration cluster in the WM of the left cerebellar tonsil, (d) a significant MD alteration cluster in the WM of the right parahippocampal gyrus, (e) a significant MD alteration cluster in the WM of the right frontal sub-gyrus, and (f) a significant MD alteration cluster in the WM of the right extranuclear area of the putamen as seed masks and the ipsilateral hypothalamus as the waypoint mask to map any possible anatomical connections between the areas of interest. The values of these maps reflect the proportion of subjects in whom a pathway is present and overlaid on the MNI T1 template. BA: Brodmann area; FA: fractional anisotropy; MD: mean diffusivity; MNI: Montreal Neurological Institute; WM: white matter.
Correlations between clinical profiles of CH patients and regional DTI indices
None of the areas with significant changes in regional diffusivity indices demonstrated a significant correlation with the frequency of attacks per day, duration of illness, total number of bouts (lifetime), frequency of bouts per year, HADS score, or the BDI within the patient group.
Discussion
Here, we used voxel-wise analyses with multiple diffusivity indices and probabilistic fiber tractography to study the pathogenesis of WM changes in episodic CH patients during in-bout and out-of-bout periods. We found significant differences in WM microstructure with multiple (axial, radial and mean) diffusivities in the frontal, limbic areas and cerebellum during the in-bout and out-of-bout periods. Probabilistic tractography showed highly consistent direct anatomical connections between the altered areas and the ipsilateral hypothalamus. Our findings support the hypothesis that CH patients have altered WM microstructures that likely connect to the ipsilateral hypothalamus.
Changes in diffusivity indices in regional WM are associated with pain modulation
Our results indicate that the regionally higher MD in the ipsilateral medial frontal gyrus of the frontal lobe was driven by higher Drad, not Dax, suggesting that demyelination or dysmyelination may occur in this region of the pain neuromatrix in CH patients during in-bout periods (17,18). FA is generally assumed to be a marker of the integrity of WM tracts, and changes in FA can result from multiple causes, including alterations to myelination, axon size and density, path geometry and the presence of brain regions with complex fiber architecture. Thus, the pattern of changes in diffusivity indices reported here, indicating no change in MD, Drad and Dax, and higher FA in the ipsilateral frontal subgyral WM, may be related to complex fiber architecture (25). The higher FA in this area may be due to the selective loss of fibers in specific pathways in the fiber crossing area, which leads to an overall increase in WM coherence; similar phenomena have previously been reported (25).
The regionally reduced MD in the contralateral parahippocampal gyrus in our study was driven by lower Dax and Drad. A regionally reduced MD was also found in the contralateral sub-gyrus of the frontal lobe and the extranuclear area of the putamen. A reduced MD has been reported in patients after stroke (26) and is attributed to the swelling of cells and a subsequent neuronal depolarization (27). Tissue (cellular) swelling or altered intra-/extracellular volume ratios can be found following long episodes of neural activation (28). Changes in the extracellular matrix following structural tissue remodeling may also be responsible for changes in the diffusion properties of the tissue (29). Therefore, the cellular rearrangement of neural tissue could account for the reduction in MD. Here, most WM changes during the in-bout period persisted into the out-of-bout period. Furthermore, a higher Dax in the ipsilateral cerebellar tonsil in the in-bout scans was found when compared to out-of-bout scans (17,18), which suggests that axonal disintegration occurs between the periods, potentially causing the previously reported hypometabolism of the cerebellopontine area (12).
The frontal area, amygdala, hippocampus, parahippocampus and anterior cingulate cortex play a major role in modulating cognitive and affective dimensions of pain processing (30). It has been proposed that altered metabolism in the frontal pain-modulating circuits is related to CH pathophysiology (6). Our recent work (10) revealed GM reductions primarily in regions belonging to frontal pain-modulation areas. These changes in GM volume may reflect insufficient pain-modulating capacity in the frontal areas of CH patients. In the present study, we found significant differences in WM microstructure, with multiple (axial, radial and mean) diffusivities in the frontal, cerebellar and limbic areas primarily associated with pain modulation, in episodic CH patients during the in-bout and out-of-bout periods. These structural findings suggest that episodic CH is associated with distinct GM and WM changes, especially in the frontal pain-modulation areas, which may be highly involved in CH pathophysiology.
We observed a regional decrease in Dax for the ipsilateral corpus callosum (CC) during the out-of-bout period. Previous DTI studies in migraine patients showed a significant association between WM abnormalities in the CC and depression symptoms/scores (31,32). Considering that WM abnormalities of the CC were also reported in depression studies (33–35), the CC may be involved in the association of migraine with depressive symptoms (32). CH has also been associated with an increased risk for depression (36). Although we found significant changes in the regional diffusivity index of CC, we observed no significant correlation with the HADS or BDI scores. This result may be partially due to our relatively small number of subjects and the relatively lower HADS and BDI scores in the patient group. Further research involving more CH patients with different levels of depression severity is required to confirm this possible association.
Unlike other DTI studies that enrolled only out-of-bout CH patients (9,21) or investigated both in-bout and out-of-bout patients without addressing differences between the two groups (22), this study recruited episodic CH patients who underwent diffusion MRI scanning twice: once during the in-bout period and once during out-of-bout period. This difference may account for the discrepancies in the results between the studies. Unlike Teepker et al. (22) and Szabó et al. (21), who reported widespread WM microstructural changes in the brain, mainly in the pain matrix, and Absinta et al. (9), who reported no WM changes, we found WM microstructural changes primarily in the pain-modulation areas.
There were several technical differences between the present research and previous DTI studies (9,21,22) First, the studies employed different field strengths for the MR scanner. Similar to Teepker et al. (22) and Szabó et al. (21), we used a 1.5 T scanner, whereas Absinta et al. (9) used a 3.0 T scanner. Second, unlike other DTI studies that investigated only the diffusivity indices of FA and/or MD (9,22), our study investigated multiple (axial, radial and mean) diffusivities simultaneously. Our approach provides a more complete picture of changes in the microstructural tissue organization in CH patients. Third, we performed probabilistic tractography to investigate the possible anatomical connections between the hypothalamus and WM areas with significant DTI index changes. This approach has not been used in previous studies. Finally, to obtain more accurate and reliable direction estimates for the DTI dataset in our study, each applied gradient direction was reoriented with a corresponding transformation matrix that described the rotation parameters of subject motion. This additional preprocessing step allowed us to obtain more accurate fiber orientation estimates than previous studies (37).
Tractography between the hypothalamus and regions with diffusivity changes
Though the hypothalamus plays a key role in CH pathophysiology (4,8), no involvement of the hypothalamus was found in our study. There are several potential explanations for this result, and possible methodological constraints should be considered. For instance, to achieve higher registration accuracy across subjects and to minimize the influence of partial volume effects, the TBSS approach focused only on major WM skeleton voxels, and statistical tests were performed on these voxels. Therefore, it is possible that the WM skeleton crossed only a part of the hypothalamus rather than the entire hypothalamus, and our study may have been unable to detect subtle differences in areas outside the WM skeleton voxels within the hypothalamus. Second, metabolic or structural changes in the hypothalamus mainly occur during spontaneous or provoked acute attacks of CH (4–6,8), and while all of our patients were scanned during an in-bout period, they were not experiencing acute attacks during the time of the MRI.
To further investigate the possible anatomical connections associated with CH pathophysiology between the altered WM areas and the hypothalamus, we located the margins of the hypothalamus and performed probabilistic tractography across the regions. We found highly consistent anatomical connections between all of the altered areas and the ipsilateral hypothalamus in all participants. The anatomical connections between the hypothalamus, cerebellar tonsil and frontal areas that we describe are similar to those of Owen et al. (38), who found highly consistent anatomical connections with the cerebellum and inferior medial frontal gyrus. The anatomical connections found between the hypothalamus and the frontal and limbic areas are also similar to those found via tracing experiments in non-human primates (39). Bidirectional posterior hypothalamic connections to the cerebellum have also been reported in animal studies (40) and are extensively involved in autonomic regulation and pain (40,41).
Although DTI with probabilistic fiber tractography can help in mapping the WM architecture and anatomical connection, it could not offer information related to “directionality”: that is, it could not indicate or differentiate whether the connective pathway was afferent or efferent in nature. Further research with an effective connectivity approach using functional MRI may be able to provide unique physiological insights regarding the causal relationships between brain areas (42,43).
Possible implications of the study results
It is still unknown why CH is characterized by a strictly unilateral headache that is associated with ipsilateral autonomic symptoms. During a cluster period, both sides of the hypothalamus may have the ability to induce cluster pain, but only one becomes active (44). We found higher MD in the ipsilateral medial frontal gyrus and reduced MD in the contralateral parahippocampal gyrus, both of which were highly connected to the ipsilateral hypothalamus. Descending projections to the primate hypothalamus arising from the limbic and prefrontal areas may directly influence the hypothalamus, initiating autonomic responses (45). We therefore propose that the ipsilateral hypothalamus becomes more active because of disinhibition from the medial frontal gyrus, and that the contralateral hypothalamus becomes relatively inactive because of inhibition from the contralateral parahippocampal gyrus. Compared to out-of-bout scans, we found a significant regional increase in Dax in the ipsilateral cerebellar tonsil in the in-bout scans. Other than the cerebellum, no other areas demonstrated significant dynamic differences in WM between the two periods. We also found a highly consistent direct anatomical connection between the altered area and the hypothalamus, suggesting that this connection was altered between the two periods, further reducing inhibition of the hypothalamus by the cerebellum. This latter mechanism may explain the shift between the bout periods. Further research with a more specific analysis method focusing on the cerebellum is required to determine the association.
Limitations
There are several limitations to this study. First, our patients were not drug free during the in-bout period; all were treated with prophylactic medications (verapamil or lithium) and acute pain treatment (triptans). Because of the debilitating pain of CH, it was deemed unethical to ask patients to stop their medication(s) at the risk of increasing their headache frequency and intensity during the in-bout period. However, none of the patients had taken triptans for at least 24 hours before they were scanned. Although no brain structural changes have been reported for triptans, we acknowledge the potential influence of acute pain treatment on brain structure, which could contribute to our findings. With respect to lithium therapy, lithium-related differences in GM or WM tracts have been reported in bipolar patients (46,47). No structural changes have been reported for verapamil to date. Although only two of 17 of the CH patients in our study were on lithium at the time of scanning during the in-bout period, we can hardly exclude the potential impact of medications on our findings.
Second, a longitudinal control group was not included, and this prevented us from completely determining whether the pattern detected in CH patients of the follow-up evaluation is disease related or technique/scanner related. Previous studies have evaluated the intra- and/or inter-scanner variability of DTI measurements (48–51). Most such studies investigated reliability by performing repeated scans on healthy participants acquired within the same scan session or within short scan intervals. Inter-scanner variability is generally larger than intra-scanner variability. Recently, Takao and colleagues evaluated intra- and inter-scanner reliability using longitudinal data obtained on 224 normal subjects. They examined the effects of longitudinal drift in intra-scanner and inter-scanner variability on longitudinal changes in global and regional DTI measurements using longitudinal (one-year) data. The inter-scanner variability (bias) significantly affected the longitudinal results, whereas the longitudinal results of FA, Dax and Drad were relatively stable within the same scanner (52). All of our CH patients were scanned in the same MRI machine. The time interval between scans was less than one year (mean 9.1 months). Our longitudinal results between the in-bout and out-of-bout periods (regional increase in Dax in the cerebellar tonsil) were also considered significant under a relatively strict statistical level (FWE-corrected
Third, nicotine consumption may have had an effect on our results. Nicotine has known effects on the brain (53). Furthermore, smoking has a high prevalence rate among CH patients (1), and our study group consisted of 15 patients (88.2%) who were either ex- or current smokers. Thus, we cannot exclude the potential effect of nicotine on our findings. Finally, the signal-to-noise ratio (SNR) is an important factor in DTI index calculations and tensor orientation estimations (54). A recent review suggested that the SNR needs to be considered when setting the protocol for DTI-based studies (55). Although the current DTI datasets were suboptimal in terms of the number of diffusion-encoding directions, the SNR (∼ 45) was sufficiently high to produce reliable DTI-related measurements.
Conclusion
In this study, we have demonstrated significant differences in WM microstructure with multiple diffusivity measures of DTI in episodic CH patients during in-bout and out-of-bout periods. We have also provided evidence for an association between the hypothalamus and regional WM changes in the frontal and limbic areas and cerebellum, which may partially explain why CH manifests with strictly unilateral attacks and shifts between in-bout and out-of-bout periods. Further studies are required to determine the role of the WM changes and to unravel the complexity of these connections with the hypothalamus in relation to CH pathophysiology.
Clinical implications
Diffusion tensor imaging was used to study cluster headache (CH) patient white matter microstructure. Multiple (axial, radial, and mean) diffusivities were examined. Regional brain differences were found between the in- and out-of-bout periods. Probabilistic tractography was also performed. The altered regions and the ipsilateral hypothalamus were anatomically connected.
Footnotes
Funding
This work was supported in part by grants from the Taiwan National Science Council (NSC 100-2628-E-010-002-MY3, NSC 101-2321-B-010-023); NSC support for the Centre for Dynamical Biomarkers and Translational Medicine; Taipei Veterans General Hospital (102AC-D715, V96C1-081); Brain Research Center, National Yang-Ming University; National Central University, Taiwan (NSC100-2911-I-008-001); Tri-Service General Hospital (TSGH-C101-159); and a grant from the Ministry of Education, Aim for the Top University Plan of Taiwan.
Acknowledgments
The authors would like to acknowledge MRI support from the MRI Core Laboratory of National Yang-Ming University, Taiwan.
Conflict of interest
None declared.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
