Abstract
Background
Several centers have reported efficacious cluster headache suppression with deep brain stimulation (DBS) of the hypothalamic region using a variety of targets. While the connectivity of some of these targets has individually been studied, commonalities across these targets, especially with respect to network-level connectivity, have not previously been explored.
Methods
We examined the anatomic connectivity of the four distinct DBS targets reported in the literature using probabilistic diffusion tensor tractography in normal subjects.
Results
Despite being described as hypothalamic, the DBS targets localized in the midbrain tegmentum posterior to the hypothalamus. Common tracts across DBS targets and subjects included projections to the ipsilateral hypothalamus, reticular formation, and cerebellum.
Discussion
Although DBS target coordinates are not located within the hypothalamus, a strong connection between DBS targets and the hypothalamus likely exists. Moreover, a common projection to the medial ipsilateral cerebellum was identified. Understanding the common connectivity of DBS-targeted regions may elucidate anatomic pathways that are involved in modulating cluster headache attacks and facilitate more precise patient-specific targeting of DBS.
Introduction
Reported DBS cases and stereotactic coordinates in chronic cluster headache.

DBS: brain stimulation.
Although most reports refer to a “hypothalamic” target, the optimal targets reported by each series vary considerably and are generally not located within the anatomically defined limits of the hypothalamus (Table 1). To date, commonalities across the reported targets have not been analyzed. Moreover, the anatomical circuitry underlying DBS’s efficacy in cluster headache is not well understood. In light of the increasingly accepted notion that DBS modulates neural networks rather than isolated targets, in this report we explore the commonalities in connectivity of the various reported efficacious DBS targets. We used reported efficacious DBS stereotactic coordinates to seed probabilistic magnetic resonance diffusion tractography in healthy subjects and examined the resultant pathways shared by these various targets. Understanding the common connectivity of DBS-targeted regions may elucidate anatomic pathways that are involved in modulating cluster headache attacks and facilitate more precise targeting of DBS on a patient-specific basis.
Materials and methods
Subjects and image acquisition
Seven healthy subjects (six males, one female) underwent imaging under a protocol approved by the University of California Los Angeles (UCLA) Institutional Review Board. Ages ranged from 31 to 55 years (mean 44 years). Three of the patients were left-handed. T1-weighted axial and T2-weighted coronal MR images were obtained in a 3-Tesla machine (Siemens, Trio, Germany) as previously reported (6). T1-weighted axial (gradient echo; repetition time (TR) = 11 ms, echo time (TE) = 2.81 ms, matrix = 256 × 256, whole brain, voxel size = 0.9 × 0.9 × 0.9 mm3) and T2-weighted coronal (turbo spin echo; TR = 5000 ms, TE = 62 ms, matrix 512 × 512, 18 slices centered on the hypothalamic region, voxel size = 0.4 × 0.4 × 3 mm3). A single-shot axial spin-echo echo-planar sequence was used for diffusion tensor imaging (DTI): b0 = 1.000 s/mm2, matrix = 128 × 128, voxel size = 2 × 2 × 2 mm3 and diffusion gradients in 20 noncollinear directions (6).
Probabilistic tractography
Probabilistic diffusion tractography and image processing was performed using FSL tools (the Oxford Centre for Functional MRI of the Brain (FMRIB)’s Diffusion toolbox (FDT); http://www.fmrib.ox.ac.uk/fsl) in accordance with previously described methods (16). Briefly, before performing tractography, the eddy current correction tool within FSL was used to apply affine registrations to each volume in the diffusion dataset to register it with the initial reference B0 volume. Skull stripping was performed using the brain extraction tool (BET). Voxelwise estimates of fiber orientations and their uncertainty, accounting for the possibility of crossing fibers within each voxel, were calculated using BEDPOSTX, with two fibers modeled per voxel, a multiplicative factor (i.e. weight) of 1 for the prior on the additional modeled fibers, and 1000 iterations before sampling (17). Eight “seed” masks (four per hemisphere) were created in each subject’s high-resolution T1-weighted structural space. Coordinates used were those previously reported (x, y, z calculated from the midpoint of the anterior commissure-posterior commissure line) ±2, −3, −5; ±2, −6, −8 (Table 1); mean coordinates of effective contacts as previously reported (5): ±2.98, −3.53, −3.31; and 4 mm from the third ventricle wall, −2, −5 (9). Coordinates were transformed to diffusion space (using transformations defining the relationship between T1 and diffusion space as defined by the FMRIB’s Linear Image Registration Tool (FLIRT) using 6 parameter transformation and mutual information) and used as a 2 × 2 mm voxel “seed” to determine the probabilistic tractography of each seed with the rest of the brain using PROBTRACKX (using 5000 samples, a 0.2 curvature threshold, and loopcheck termination).
Analysis of tractography
To identify common tracts across seeds/targets and across subjects, we initially focused our analysis on the single subject level. For each subject, we generated two maps: a “single-subject common pathway map” (including voxels that contained projections from all four seeds/targets, generated by taking the product of the four within-subject projection maps and binarizing the result) and a “single-subject average projection map” (generated by averaging the projection maps of each of the four seeds/targets). Note that the single-subject average projection map is specifically generated to identify the average probability that a given voxel will be within the projection path of the targets. This is in distinction to a product map that would address the probability that all four targets within a subject simultaneously project to each voxel.
To generate an average projection map across all targets and across all subjects (“group average projection map”), all single-subject average projection maps were registered into MNI152 2 mm standard space (FLIRT, 12 parameter) and averaged. Given that this map is a group average of individual average of probabilities across multiple targets without normalization, the group average projection map does not represent an average but a weighted illustrative representation of the distribution of projections across all subjects and all targets. To identify common pathways across all subjects and all targets (“group average common projection map”), in contrast to average pathways across all subjects and all targets, each single-subject common pathway map (which is a binarized single subject maps) was registered into MNI152 2 mm standard space (FLIRT, 12-parameter) and averaged, resulting in a map of the frequency with which “single subject common pathway maps” involved each voxel in standard space. Therefore, to identify voxels that were part of the common pathway in at least three subjects, the “group average common projection map” was thresholded at 0.29 and binarized for illustrative purposes. To illustrate a relative probability of connectivity of each of these voxels with the four seeds/targets in question, the resultant binarized “group common projection map” was multiplied on a voxel-by-voxel basis with the “group average projection map.” The final output represents the pathways across subjects common to all four seed/target regions with each voxel representing a hit by at least three subjects with color representing average probability of connectivity.
Results
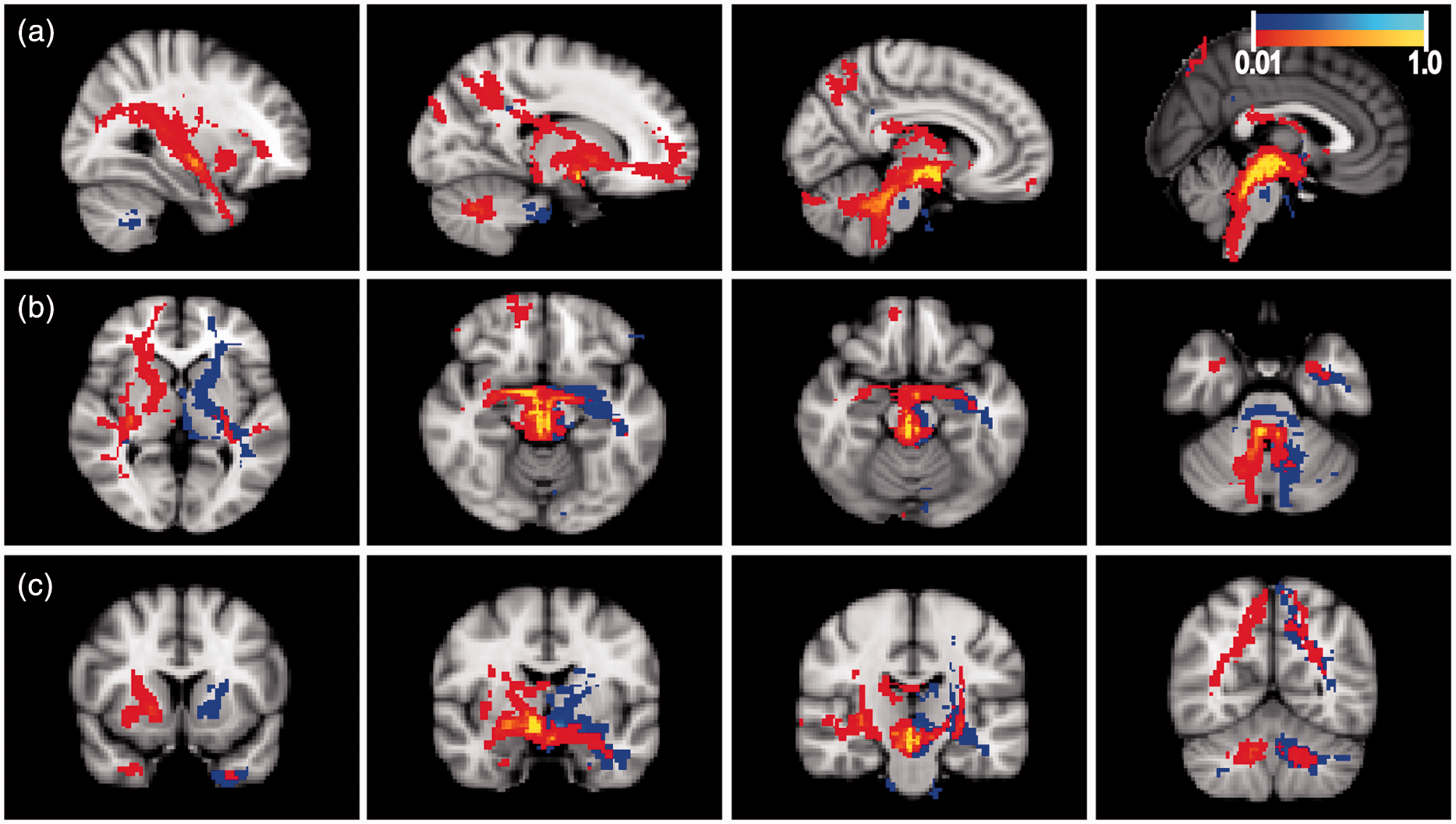
The stereotactic coordinates/targets used as seeds were localized in the gray matter of the midbrain tegmentum, posterior to the hypothalamus (Figure 1). The “group average projection map” (Figure 2), which illustrates the average projection of all targets across all subjects, showed diffuse projections to ipsilateral frontal lobe (orbitofrontal regions), temporal lobe, hypothalamus, reticular formation, and cerebellum, with the strongest projections on average to the latter three structures. The “group average common projection map” (Figure 3(a)) illustrates in standard space how often a voxel in standard space was included in “single subject common pathway maps” and as expected demonstrates a similar pattern to that seen in Figure 2, with most subjects demonstrating common pathways projecting to ipsilateral hypothalamus, reticular formation, and cerebellum. When the “group average common projection map” was thresholded to include only common pathways that were present in at least three subjects, the resultant maps reinforce the observation of common connectivity to the ipsilateral hypothalamus, ipsilateral reticular formation, and the ipsilateral cerebellar cortex via the superior cerebellar peduncle (Figure 3(b)). As can be seen in Figure 3(a), these projections to ipsilateral hypothalamus, ipsilateral reticular formation, and ipsilateral cerebellar cortex were robust and seen even when thresholding to identify those voxels present in at least 50% (four of seven) subjects (represented by bright yellow and bright blue voxels in Figure 3(a)). Similar patterns of common projections were also observed when group data were thresholded to identify those voxels involved in projection pathways in at least five of seven (71%) and six of seven (86%) of subjects. These data represent the most stringent analysis of pathways common to all four targets, with the elimination of pathways present in fewer than three subjects. Of note, in addition to the common pathways shown in the group analysis (Figure 3(b)), one patient had additional pathways to the bilateral parietal and frontal cortices, which is depicted in Figure 2 representing an average of all pathways from the four target seeds without thresholding.
Placement of stereotactic deep brain stimulation (DBS) cluster headache “seed” targets on normal subject’s T1-weighted magnetic resonance image (MRI). Sagittal image (A) of right hemisphere showing target 1 (±2, −3, −5 from midcommisural point) and target 2 (±2, −6, −8), both located in the midbrain tegmentum. The third (±2.98, −3.53, −3.31) and fourth (4 mm from the third ventricle wall, −2, −5) targets are outside of the plane of the image. Target 1 is also depicted on axial (B) and coronal (C) sections in the left and right hemispheres. Group average projection map. Average single-subject projection maps of four stereotactic deep brain stimulation (DBS) cluster headache “seed” targets across normal subjects (n = 7) without thresholding overlaid on a standard structural brain image. Pathways projected to the ipsilateral frontal lobe (orbitofrontal cortex) (axial view/panel (b)), temporal lobe (sagittal view/panel (a), hypothalamus and reticular formations (panel (b) and coronal view/panel (c)), and the cerebellum (panels (a) and (c)). The strongest common projections were to the hypothalamus, reticular formation, and cerebellum (yellow color). Scale bar represents probability of a pathway passing through a voxel (probability of 1 is depicted in yellow for right hemisphere seeds and light blue for left hemisphere seeds). Average common pathways across subjects. Group average common projection map (panels (a)1–3) of four stereotactic deep brain stimulation (DBS) cluster headache “seed” targets across normal subjects (n = 7) illustrates in standard space the frequency by which each voxel was included in “single subject common pathway maps.” The pattern is similar to that of Figure 2, with most subjects demonstrating common pathways projecting to the ipsilateral hypothalamus, reticular formation, and cerebellum (yellow and light blue colors, panels (a)1–3). Note that solid yellow and light blue regions represent voxels involved in at least 50% of subjects. Average common pathways across subjects (panels (b)1–3) represents pathways present in all four DBS “seed” target projection maps and at least three subjects (thresholded data from panels (a)1–3). Common pathways projected to the ipsilateral hypothalamus, ipsilateral reticular formation, and the ipsilateral cerebellar cortex via the superior cerebellar peduncle (yellow and light blue colors, panels (a)1–3). Scale bar, panels (b)1–3 represents probability of a pathway passing through a voxel across targets and subjects (probability of 1 is depicted in yellow for right hemisphere seeds and light blue for left hemisphere seeds).

Discussion
The justification for the anatomic coordinates used to target DBS electrode placement (Table 1) is based on early PET and MRI studies with limited spatial resolution in patients experiencing cluster attacks (1,4,6,18). Whether the activation seen in these studies occurs in the posterior hypothalamus or the midbrain tegmentum has been debated (3). Using previously reported coordinates to create “seed” masks in our normal subjects, we confirm that these coordinates lie in the midbrain tegmentum gray matter adjacent to the third ventricle, posterior to the hypothalamus, a region that is hypothesized to be part of a pain network (5,13). Imprecise localization of electrodes based on poorly understood circuitry or individual anatomic variation may in part account for the 30%–40% of cluster headache patients (Table 1) for whom DBS is ineffective.
The mechanisms of neuromodulation and the anatomical underpinnings of effective DBS in cluster headache are not known. One important question is whether a local effect or distant neuromodulation underlies the effectiveness of electrical stimulation. We report three major regions of connectivity common to all seed coordinates evaluated: the ipsilateral hypothalamus, reticular formation, and cerebellar hemisphere. Given that it is currently not possible to differentiate directionality using DTI, we cannot determine whether these data represent one pathway projecting from the hypothalamus through the seed regions to the cerebellum, or vice versa, although the pattern of connectivity is suggestive of such a pathway.
The posterior hypothalamus has been hypothesized to play a pivotal role in initiating cluster headaches owing to the periodicity of attacks, neuroendocrine alterations in patients with cluster headache (19), and purported hypothalamic PET and fMRI activation during cluster attacks (1,2,4). Conversely, a larger “pain matrix” in which the hypothalamus plays a role may be responsible for modulating cluster headache attacks, a hypothesis in agreement with diffuse activation during imaging of cluster attacks (1,2,4,13,20).
DTI probabilistic tractography in humans (15) has suggested a direct connection between the hypothalamus and cerebellum. Interestingly, PET (1,21) and MRI (4,20) studies have shown activation of both the ipsilateral hypothalamus and cerebellum in patients during cluster attacks, regions that are not activated in pain-free states. Furthermore, in patients with chronic cluster headache implanted with DBS electrodes, the cerebellum has enhanced 15-O-H2O PET activity during stimulator-on states compared to stimulator-off states (22) and chronic occipital nerve stimulation modulates cerebellar 18-FDG PET hyperactivation in cluster headache patients (21). Whether the cerebellum plays a direct role in modulating cluster headache is not known, although cerebellar activation has been suggested to play an active role in pain modulation (23) and may be part of a larger “pain matrix” (13).
Interestingly, our data align with data by Seijo et al. (13), which showed projections between DBS electrode target sites and the ipsilateral cerebellum in two patients with cluster headache using diffusion tensor tractography. Unlike Owen et al. (24), however, we do not show projections to parietal or frontal cortices as a pathway common to all seeded regions across patients. Although these pathways were present in unthresholded preliminary analyses (Figure 2), the more stringent treatment of diffusion data in this analysis eliminated pathways to these regions observed in one patient in our pooled analysis. This is consistent both with Owen et al.’s (24) and Seijo et al.’s (13) reports and figures that connections to the cerebellum and the reticular nucleus are much more robust. One should consider, however, that differences in studies may in part be due to limitations in the spatial resolution of DTI analysis and that DTI data with the spatial resolution used in this study (2 mm) may not completely account for differences in connectivity of all the targets studied.
By seeding DBS-target regions in diffusion tensor imaging, we show connectivity with the hypothalamus and cerebellum common to all DBS targets probed in normal subjects. Our data also suggest that tractography could be used to facilitate surgical planning, utilizing a pathway approach within patients to localize more precise anatomic targets. Despite grossly normal structural imaging in patients with cluster headache, subtle differences in brain structure (as suggested by May et al. (18)) or aberrant connectivity may underlie cluster headache attacks. Therefore, confirmation of the current findings in patients who have undergone effective DBS compared to patients for whom DBS is ineffective would elegantly address questions our current data raise regarding the pathways that are critical for cluster headaches and may suggest new targets for future surgical intervention.
Clinical implications
Reported deep brain stimulation (DBS) targets for treatment of cluster headache localize to the midbrain tegmentum rather than the hypothalamus. DBS targets demonstrate robust structural connections with the hypothalamus, ipsilateral reticular formation, and medial ipsilateral cerebellum. Frontal projections, as reported by others, are not common to all reported efficacious DBS targets. These common patterns of structural connectivity across DBS targets may elucidate anatomic pathways that are involved in modulating cluster headache attacks and facilitate more precise patient-specific targeting.
Footnotes
Funding
This work was supported by funds provided by National Institute of Biomedical Imaging and Bioengineering under Award Number K23EB014326 (N.P.) and the UCLA Department of Neurosurgery Visionary Ball.
Acknowledgment
We would like to thank the Jack Kent Cooke Foundation for its support of author C.D.C.
Conflict of interest
None declared.
