Abstract
Objective
The objective of this study was to identify brain regions having aberrant pain-induced activation in migraineurs, thereby gaining insight into particular aspects of pain processing that are atypical in migraineurs.
Methods
Functional magnetic resonance imaging assessed whole brain responses to painful heat in 24 adult episodic migraineurs who were at least 48 hours pain free and 27 healthy controls. Regions differentially activated in migraineurs compared to controls were identified. Activation intensities in these regions were correlated with headache frequency, number of migraine years, and time to next migraine attack.
Results
Migraineurs had greater pain-induced activation of lentiform nucleus, fusiform gyrus, subthalamic nucleus, hippocampus, middle cingulate cortex, premotor cortex, somatosensory cortex, and dorsolateral prefrontal cortex, and less activation in precentral gyrus and superior temporal gyrus. There were significant correlations between activation strength and headache frequency for middle cingulate (r = 0.627, p = 0.001), right dorsolateral prefrontal cortex (r = 0.568, p = 0.004), left fusiform gyrus (r = 0.487, p = 0.016), left precentral gyrus (r = 0.415, p = 0.044), and left hippocampus (r = 0.404, p = 0.050) and with number of migraine years for left fusiform gyrus (r = 0.425, p = 0.038). There were no significant correlations between activation strength and time to next migraine attack.
Conclusions
The majority of regions with enhanced pain-induced activation in headache-free migraineurs participate in cognitive aspects of pain perception such as attending to pain and pain memory. Enhanced cognitive pain processing by migraineurs might reflect cerebral hypersensitivity related to high expectations and hypervigilance for pain.
Introduction
Headache is typically the most recognized component of a migraine attack. However, migraine also consists of nausea, vomiting, photosensitivity, phonosensitivity, olfactory hypersensitivity, cutaneous allodynia, and symptom worsening with physical activity. Although symptoms are most prominent during individual migraine attacks, migraineurs commonly have less prominent migraine symptoms between attacks (i.e. when “interictal”). Interictal symptoms include hypersensitivities to visual, auditory, olfactory, and somatosensory stimuli (1–4). The interictal migraine brain is hyperexcitable in response to stimulation and migraineurs lack normal habituation to repetitive stimuli (5,6). Furthermore, interictal migraineurs maintain a state of hypervigilance regarding their next migraine, having high expectations for pain and feeling anxious about their next pain experience (7,8).
In this study, functional magnetic resonance imaging (fMRI) was used to contrast brain responses to moderately painful heat stimulation of the forearm skin in migraineurs who had been pain free for at least 48 hours to brain responses of nonmigraine healthy controls. We expected the hypersensitive migraine brain would show atypical activations in “pain matrix “regions (i.e. cortical and subcortical regions commonly activated by nociceptive stimulation). However, we used a whole-brain analysis approach to identify specific regions with atypical pain-induced activation in migraineurs. Identifying these regions and considering their specific roles in pain processing (e.g. sensory-discriminative, affective-motivational, cognitive) yields insights into the hypersensitive and hypervigilant migraine brain. Although there have been a few other migraine fMRI studies investigating brain responses to noxious cutaneous stimulation, most have taken a region-of-interest approach, an approach noted to have substantial limitations for studying migraine pathophysiology (9). This study adds to existing literature because it: 1) investigated whole brain responses to pain; 2) used stimulation temperatures that were individualized for each subject; 3) included more subjects compared to most migraine fMRI studies and thus had greater statistical power; and 4) allowed for fMRI findings to be correlated with markers of migraine burden, including headache frequency and number of years with migraine, and with time to next migraine attack.
Methods
Subjects
Twenty-seven migraineurs who met International Classification of Headache Disorders II episodic migraine criteria, who had not used migraine prophylactic medications for at least eight weeks, and who were not overusing migraine abortive medications, were enrolled (10). The control group consisted of 27 healthy individuals matched 1:1 to the migraine subjects, exactly matched for gender and to within five years for age. Control and migraine subjects were identified using the same methods, including the use of advertisements (e.g. posters) in our hospital, word-of-mouth referrals, and identification of subjects from a database of research volunteers maintained by Washington University School of Medicine. Subjects were 18–65 years old without contraindications to MRI or quantitative sensory testing (QST), without medical disorders that might affect QST or MRI results, without chronic pain or neurologic disorders other than migraine, and without acute pain. Controls had no headaches other than infrequent tension-type headache (<3 headache days/month).
Institutional review board approval was obtained for this study and all subjects provided informed consent.
Study timing and flow
All testing was completed during one visit. Subjects were studied when ≥48 hours pain free and ≥48 hours free from migraine medications, nonsteroidal anti-inflammatory drugs (NSAIDs), opiates, and other pain medications. Subjects completed questionnaires, then had QST, and then MRI. All subjects prospectively maintained a headache diary for seven days following their MRI (Day #1 = day of MRI), providing data on the timing until each subject’s next headache.
Clinical data
Migraineurs completed structured interviews for determining headache characteristics and the Migraine Disability Assessment (MIDAS) to determine severity of migraine-related disability. All subjects completed the State-Trait Anxiety Inventory and Beck Depression Inventory. Two-sample t-tests or chi-square tests were used to compare clinical data from migraineurs and controls.
Quantitative sensory testing
QST for determination of thermal pain thresholds was completed using a Medoc Pathway with a 30 mm × 30 mm thermode. Left ventral medial forearm pain thresholds were determined using the method of limits (thermode temperature change = 1℃/second). Testing was performed three times and the mean of the trials was considered the pain threshold.
Stimulation temperatures for fMRI
Stimulation temperatures causing moderate intensity pain (i.e. pain rating between 4 and 7 using an 11-point scale; 0 = no pain, 10 = most severe pain) were determined for each subject. Subjects were stimulated for 7.5 seconds at the left medial forearm (ramp and hold method) with a temperature equal to their pain threshold plus 1℃. Subjects rated the pain intensity from stimulation. If pain intensity was <4, the temperature was increased by 0.5℃ increments until it was rated between 4 and 7. Conversely, if pain intensity was >7, the stimulation temperature was decreased in 0.5℃ increments.
MRI paradigm
Images were obtained with a Siemens MAGNETOM Trio 3T scanner (Erlangen, Germany) with total imaging matrix technology using a 12-channel head matrix coil and Food and Drug Administration (FDA)-approved sequences. Structural scans included a high-resolution T1-weighted sagittal magnetization-prepared rapid gradient echo (echo time (TE) = 3.16 ms, repetition time (TR) = 2.4 s, flip angle = 8 degrees, 176 slices, 1 × 1 × 1 mm voxels, field of view (FOV) 256 × 256 mm2) and an axial T2-weighted turbo spin echo (TE = 88 ms, TR = 6280 ms, flip angle = 120 degrees, 36 slices, 1 × 1 × 4 mm voxels, FOV 256 × 256 mm2). Functional imaging used a blood oxygenation level-dependent (BOLD) contrast-sensitive asymmetric echo-planar sequence (TR = 2500 ms, TE = 27 ms, flip angle = 90 degrees, 36 slices, 4 × 4 × 4 mm voxels, FOV 256 × 256 mm2). During fMRI, subjects were intermittently exposed to an auditory stimulus followed by no further stimulation or followed by painful heat stimulation to the left ventral medial forearm. Interstimulus intervals varied from 44 to 46 seconds. A long interstimulus interval was used in order to avoid habituating or sensitizing effects. The experimental design is illustrated in Figure 1. Cutaneous stimuli were applied with the 30 mm × 30 mm thermode using ramp (10℃/second) and hold (7.5 seconds) methodology. Each stimulus was given three times in a randomized fashion within each of three fMRI runs.
Magnetic resonance imaging (MRI) paradigm. During each of three functional MRI (fMRI) runs, subjects were exposed to auditory cues followed either by no stimulation (three times) or moderately painful heat stimulation (three times) applied to the skin of the left forearm. The order of stimuli was randomized. Thus, data from each subject were derived from a total of nine auditory cues followed by painful stimuli and nine auditory cues followed by no stimuli.
Image processing and fMRI data analysis
All preprocessing and general linear model (GLM) estimation of whole-brain activation patterns were performed using SPM8 software (Wellcome Department of Cognitive Neurology, Institute of Neurology, London, UK) interfaced with Matlab version 11.0 (Mathworks, Natick, MA, USA). Functional images were realigned to the mean volume in the series, motion-corrected, realigned to each individual’s structural images, normalized to standard stereotaxic space using Montreal Neurological Institute template (MNI 152), and smoothed using a 5 mm full width at half maximum (FWHM) Gaussian kernel. Data were checked for excessive motion and all included scans showed <3 mm movement in any direction. First-level, single-subject analysis was performed within SPM8 using a GLM approach. Brain regions associated with pain processing were identified by generating contrast maps representing activations associated with auditory cue followed by painful stimulation vs. activations associated with auditory cue followed by no stimulation. Each individual’s contrast map was then entered into a second level, mixed-effects analysis. A main-effects analysis identified brain regions engaged in migraineurs and controls, and a two-sample analysis identified regions differentially activated between subject cohorts. Main-effects analysis used the following parameters to determine significance: p < 0.05, cluster threshold corrected at 10 voxels, and family-wise error corrected for multiple comparisons. Group contrasts used the following parameters to determine significance: p < 0.005, cluster threshold corrected at 10 voxels (11). Regions of significant activation were anatomically localized by converting MNI coordinates into Talairach coordinates and labeled using Talairach atlas software (12,13).
Correlations between pain-induced activations with migraine burden and with time to next headache
Marsbar, a Matlab toolbox, extracted contrast parameters for further analyses within SPSS 21. Pearson’s correlation (two-tailed) analyses interrogated associations between headache frequency, years with migraine, and time to next headache with pain-induced activations in regions that differentially activated in migraineurs. Correlations were considered significant if surviving Benjamini-Hochberg multiple comparison correction allowing for a false-discovery rate (FDR) of 0.05. Correlations with p < 0.05, uncorrected, were also identified.
Results
Subjects and noxious stimulation parameters
Subject characteristics.

BDI: Beck Depression Inventory; MIDAS: Migraine Disability Assessment Scale.
Main effects of noxious heat (Figure 2)
As anticipated, numerous “pain matrix” regions demonstrated shared pain-induced activation in migraineurs and controls, including: anterior insula, middle insula, posterior insula, thalamus, anterior cingulate cortex, middle cingulate cortex, posterior cingulate cortex, midbrain, dorsal pons, medulla, cerebellum, somatosensory cortex (SSC), precentral gyrus, supplementary motor area, premotor cortex, lentiform nucleus, temporal pole, prefrontal cortex, basal ganglia, hippocampus, and parahippocampal gyrus.
Noxious heat-induced shared activations. As anticipated, numerous regions of the “pain matrix” were commonly activated in response to moderate pain in migraine and control subjects, including anterior insula, middle insula, posterior insula, thalamus, anterior cingulate cortex, middle cingulate cortex, posterior cingulate cortex, midbrain, dorsal pons, medulla, cerebellum, somatosensory cortex, precentral gyrus, supplementary motor area, lentiform nucleus, temporal pole, prefrontal cortex, basal ganglia, hippocampus, and parahippocampal gyrus (p < 0.05, family-wise error corrected, cluster threshold corrected >10 voxels). Numbers next to each axial slice represent the Z-coordinate. Slices are shown with the right side of the brain on the right.
Differential activations in migraineurs vs. controls (Figure 3; Table 2)
Regions with differential pain-induced activation in migraine vs. control subjects.

Coordinates are in Montreal Neurological Institute (MNI) space. Lat: lateralization; L: left; R: right; BA: Brodmann’s Area.
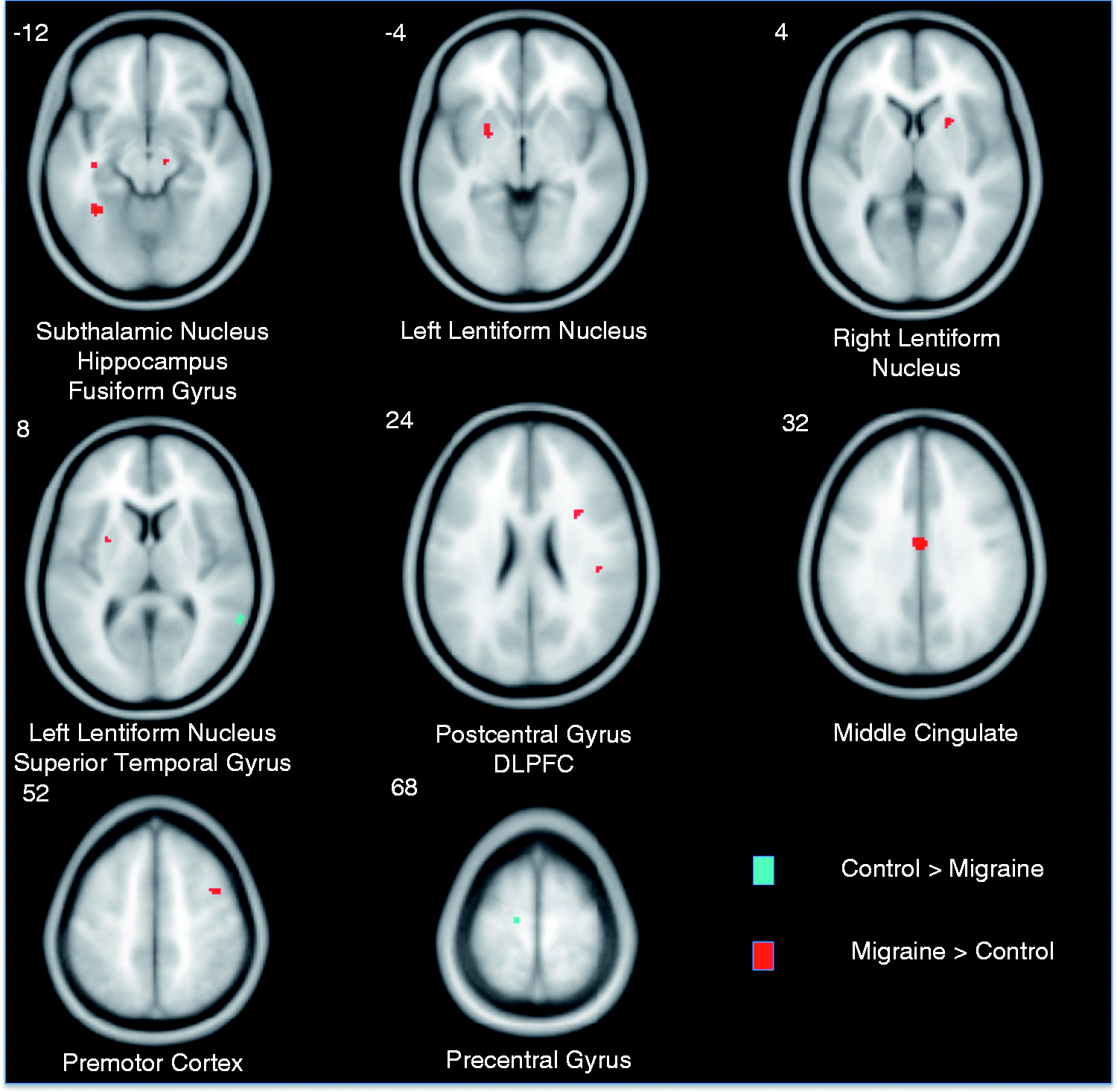
Noxious heat-induced activations differing between migraine and control subjects. Several regions differentially activated in migraine subjects compared to control subjects (p < 0.005, cluster corrected). Voxels colored red were more strongly activated in migraine subjects compared to control subjects. Voxels colored blue were more strongly activated in control subjects compared to migraine subjects. Coordinates, max z-stats, and number of voxels for each voxel cluster are reported in Table 2. Numbers next to each axial slice represent the Z-coordinate. Slices are shown with the right side of the brain on the right. DLPFC: dorsolateral prefrontal cortex.
Atypical activation correlates with migraine burden (Figure 4, Table 3)
There were positive correlations (p < 0.05, uncorrected) between headache frequency and pain-induced activation in middle cingulate (r = 0.627, p = 0.001), left fusiform gyrus (r = 0.487, p = 0.016), left hippocampus (r = 0.404, p = 0.050), right DLPFC (r = 0.568, p = 0.004), and left precentral gyrus (r = 0.415, p = 0.044). A positive correlation between years with migraine and activation of left fusiform gyrus was present (r = 0.425, p = 0.038). After FDR correction, two correlations with headache frequency remained significant: middle cingulate and right DLPFC. There were no significant correlations between pain-induced BOLD signal changes with depression or state-trait anxiety scores.
Correlations between pain-induced activation and headache frequency. Pain-induced activation of the middle cingulate, right dorsolateral prefrontal cortex, left fusiform gyrus, and left hippocampus significantly correlated (p < 0.05, uncorrected) with headache frequency (days per month with headache). Following Benjamini-Hochberg correction for multiple comparisons allowing for a false-discovery rate of 5%, correlations with the middle cingulate and right DLPFC were still significant. DLPFC: dorsolateral prefrontal cortex; BOLD: blood oxygenation level-dependent. Correlations between painful heat-induced activations and markers of migraine burden. Significant correlations are indicated by bold text. Region coordinates and size are indicated in Table 2. Lat: lateralized; r: correlation. aCorrelations with p ≤ 0.05, uncorrected. bCorrelations that survive Benjamini-Hochberg multiple comparisons correction allowing for a false discovery rate of 0.05.

Atypical activations and time to next headache
Among the 24 migraineurs, eight had their next headache on the day of their MRI (i.e. Day 1), three experienced a headache on Day 2, six on Day 3, three on Day 4, two on Day 5, 0 on Day 6, and two on Day 7. There were no significant correlations between the extent of pain-induced activations in regions that differed in migraineurs vs. controls and the time to next headache.
Discussion
The main study finding is that episodic migraineurs have greater pain-induced activation in brain regions that participate in neurocognitive aspects of pain processing, suggesting that migraineurs have altered cognitive responses and cognitive perceptions of pain. Other regions with greater activation in migraine included those important for pain modulation and sensory-discriminative pain processing. Enhanced activation of these regions in migraineurs may be the result of stimulus-induced adaptations secondary to recurrent migraine attacks or could represent an underlying hyperactivity of these regions and thus predisposition to migraine symptoms.
Cognitive pain regions with increased activation in migraine
Neurocognitive aspects of pain processing affect human perception of pain. These neurocognitive processes include attention to pain, pain expectations and vigilance, and reappraisal, all of which are affected by prior pain experiences and pain memory (14). Numerous studies have demonstrated impacts of attending to pain, pain expectations, and one’s perceived ability to tolerate pain on pain intensity ratings (15–17). The person who expects severe pain and is hypervigilant about anticipated pain is likely to rate pain as more severe and to react with anxiety. Pain responses are reduced in patients who reframe their interpretation of pain in less emotional terms (i.e. pain reappraisal) and in those who perceive they have control over pain (18,19). Since attention, expectations, vigilance, and reappraisal of pain are affected by prior experiences, recurrent and chronic pain can modulate cognitive pain processing.
As expected in patients with chronic, recurring, unpredictable attacks of pain, migraineurs have enhanced pain expectation and negative appraisal of their ability to control pain (7,20). Migraineurs have interictal hypervigilance, watching out for symptoms of their next migraine attack and calculating the risk of exposing themselves to potential migraine triggers vs. avoiding those potential triggers (8). In this study, migraineurs had greater pain-induced activation of several cognitive pain-processing regions, including middle cingulate, fusiform gyrus, hippocampus, and premotor cortex. Although enhanced cognitive pain processing might be an adaptive response in patients with acute pain, we theorize that it is maladaptive in patients with chronic, recurring functional pain like migraine. Enhanced cognition of pain by the migraineur might relate to greater vigilance and attention to pain based on memory of prior pain experiences, expectations for future pain, and negative pain appraisal.
Middle cingulate cortex
The middle cingulate is involved in cognitive, affective, attention, orienting, and motor withdrawal aspects of pain (21,22). In this study, migraineurs had increased activation within the posterior subdivision of the middle cingulate, a region predominantly involved with executive function (23). Prior studies have found middle cingulate to differentially activate in migraineurs when exposed to olfactory stimuli and when exposed to painful heat (24,25). We hypothesize that enhanced activation of posterior middle cingulate represents a greater attention and orienting response to painful stimuli by migraineurs. Increased attention to pain in patients who have experienced many years of recurrent migraine pain is likely a learned response. Although one might expect that recurrent pain leads to less attention to pain over time, migraineurs do not habituate normally to recurrent stimuli and they have interictal hypervigilance to pain (25). Strong correlations between headache frequency and pain-induced middle cingulate activations found in this study suggest that recurrent migraines sensitize the middle cingulate to painful stimuli.
Fusiform gyrus
The fusiform gyrus is an integrative zone participating in the mental imagery process related to retrieval of pain perception (26). Atypical function and structure of the fusiform gyrus have been identified in chronic low back pain, fibromyalgia, and cluster headache patients (27–29). Greater left fusiform gyrus activation in migraineurs may be due to enhanced mental imagery of pain based on memory of recurrent migraine headaches.
Hippocampus
In addition to its role in memory and learning, the hippocampus participates in determining pain-induced stress responses and mediates behavioral pain responses, processes likely affected by prior pain experiences (30). Atypical hippocampal activation and functional connectivity have been demonstrated in prior migraine studies (31). We theorize that greater pain-induced hippocampal activation in migraineurs is due to strong memory of pain from recurrent migraines. The positive correlation between pain-induced hippocampal activation and headache frequency found in our study supports this theory. In addition, enhanced hippocampal activation might produce a greater stress response to pain, thus enhancing the migraineur’s desire to avoid pain. Although a strong stress-response to pain is adaptive when experiencing acute and escapable pain, this enhanced response is likely maladaptive in the migraineur, leading to greater pain-induced anxiety and unpleasantness, and somatosensory amplification (24,32).
Premotor cortex
The premotor cortex participates in motor response inhibition and impulse control (27,33). Pain-induced activation of the premotor cortex correlates with pain intensity (34). Pain-induced activation of premotor cortex might inhibit motor reactions to pain. Prior studies have identified atypical premotor cortex function and structure in migraineurs (35,36). Since premotor cortex is involved in inhibiting motor responses to pain and since migraine pain is exacerbated by physical activity, enhanced pain-induced activation of premotor cortex in migraine might reflect the migraineur’s learned response to avoid movement during pain.
Other regions with increased activation in migraine
Other hyperreactive regions in migraineurs included DLPFC, basal ganglia, SSC, and subthalamic nucleus. These regions are primarily involved in pain modulation (DLPFC), pain integration (basal ganglia), and sensory-discriminative pain processing (SSC, subthalamic nucleus).
The DLPFC plays a pain-inhibiting role, perhaps via top-down inhibition of the midbrain-thalamic-cingulate pathway (37,38). DLPFC transcranial magnetic stimulation increases thermal pain thresholds and pain tolerance and reduces postoperative pain, neuropathic pain, fibromyalgia symptoms, and may reduce migraine symptoms (39–44). Enhanced pain-induced activation of DLPFC in migraineurs and the correlation between DLPFC activation and headache frequency found in our study might indicate that greater pain-induced DLPFC activation is a pain-inhibiting and compensatory response.
The basal ganglia participate in habit and stimulus-response learning via integration of inputs from cortical and thalamic regions participating in sensory-discriminative, cognitive, affective, and modulatory pain processing, and is involved in multisensory integration (45–47). Prior migraine studies demonstrate atypical pain-induced basal ganglia activation, caudate volume, and resting state functional connectivity of basal ganglia regions (46,48). We hypothesize that enhanced pain-induced activation of putamen and of regions providing input to basal ganglia (e.g. premotor, hippocampus) found in our study could result in or be a marker of atypical integration of different pain aspects (e.g. sensory discriminative and cognitive) and aberrant multisensory integration, features typical of migraine (49).
Primary SSC is likely involved in sensory-discriminative pain processing (50). Greater pain-induced activation of contralateral SSC in our study (estimated to be within cortex involved in facial/head sensation) is consistent with prior studies suggesting that interictal migraineurs have primary SSC hyperexcitability and abnormal sensory-discriminative pain processing (4,51). It is unknown if functional alterations of SSC in migraine are due to repetitive migraines or if they represent an underlying hyperexcitability that predisposes to migraine symptoms.
The subthalamic nucleus participates in pain and temperature sensation. Subthalamic nucleus stimulation is associated with reductions in clinical pain and reduced sensitivity to mechanical and thermal pain (52,53). Greater subthalamic nucleus activation in migraineurs might represent a pain-inhibiting response.
Regions with less activation in migraine
A region within precentral gyrus (within the supplementary motor area) had less pain-induced activation in migraine subjects compared to controls. The supplementary motor area (SMA), a region prominently involved in motor planning, is commonly activated in response to painful heat stimuli (54). Pain-induced SMA activation might indicate a preparation for moving the body away from the painful stimulus (55,56). Since physical movement worsens the pain of migraine, less activation of SMA in migraineurs might indicate the migraineur’s learned response to avoid movement during pain, an explanation that is also consistent with our study finding of enhanced activation of premotor cortex (a region implicated in motor response inhibition) in migraineurs.
A region in the superior temporal gyrus (in the area of the temporoparietal junction) also had less pain-induced activation in migraineurs. The temporoparietal junction activates in response to novel sensory stimuli including pain and is thought to play a role in salience of pain (57–59). Migraineurs’ experience with recurrent pain and expectation for pain might result in experimental pain lacking novelty, thus activating the temporoparietal junction to a lesser extent.
Prior migraine studies investigating painful heat-induced whole brain activation
A few studies have contrasted thermal pain-induced whole brain activations in headache-free migraineurs to controls (48,60,61). In a study of 16 migraineurs, painful heat induced greater activation of perigenual anterior cingulate cortex in migraineurs and less activation in bilateral secondary SSC (60,61). The authors hypothesized that their findings represented an “analgesic compensatory reorganization” of pain-processing regions that are “performing additional work to perceive pain at the same subjective intensity” of controls. Another study, including 11 migraineurs, found increased pain-induced activation within contralateral temporal pole and ipsilateral parahippocampal gyrus (48). Exploratory analyses (p < 0.05, uncorrected, cluster threshold seven voxels) identified several regions that differentially activated in migraineurs that we also found to be atypically activated, including: fusiform gyrus, basal ganglia, middle frontal gyrus (designated DLPFC in our study), precentral gyrus, and SSC.
Limitations and future directions
Results did not demonstrate differences in pain-induced activation of brainstem-descending modulatory regions like have been identified in a prior migraine study (62). Differential activations in brainstem regions might be identified in our study if a region-of-interest approach were used, if parameters for statistical significance were eased, or if we specifically studied migraineurs with cutaneous allodynia (63). As with all fMRI studies, the spatial resolution of fMRI is limited, resulting in difficulties differentiating small brainstem structures like subthalamic nucleus from nearby structures like substantia nigra (64). To increase confidence that the midbrain structure differentially activating in migraine was within subthalamic nucleus, we carefully visualized the region on T2-weighted scans, determined the region coordinates corresponding with the maximum z-stat, and used these coordinates to name the region according to the Talairach atlas. Since this is not a longitudinal study, it is impossible to determine if fMRI findings are secondary to recurrent migraines or if they represent an underlying brain predisposition to migraine symptoms. Unmeasured confounding variables could have influenced study results. However, we minimized the effect of potential confounders by matching migraine and control subjects according to age and sex, excluding subjects with medical disorders that might affect QST or MRI results, excluding subjects with chronic pain or neurologic disorders other than migraine, and excluding subjects with acute pain. Furthermore, there were no differences in anxiety scores or depression scores between migraineurs and controls and no significant correlations between pain-induced activations of regions that differentially activated in migraineurs vs. controls with anxiety or depression scores. However, we cannot entirely exclude the possibility that residual confounding due to small differences between migraine and control subject ages, anxiety, depression, and unmeasured variables, could have influenced study results. The average temperature that caused moderate pain in the headache-free migraine subjects was no different from the average temperature that caused moderate pain in healthy control subjects. Some published reports have suggested that headache-free migraineurs are more sensitive to thermal cutaneous stimulation compared to controls (4,65). Additional studies are needed to further elucidate this controversial issue. The majority of migraineurs in this study developed their next headache within several days of their MR imaging. Thus, whether these subjects were “interictal” is debatable, since it is possible that brain function is altered prior to onset of migraine symptoms. Development of headaches within several days of MR imaging in a subset of our subjects allowed for a fairly unique opportunity to investigate correlations between the extent of atypical pain-induced brain activations with the duration until next headache. The absence of significant correlations between the extent of activity in regions that differed when comparing migraineurs to controls with time to next headache suggests that time to next headache is not a significant contributor to the study results. Finally, since this study did not evaluate patients with nonmigraine pain, we cannot determine if fMRI findings are migraine specific or if they would be shared by patients with other types of longstanding and recurrent pain.
Conclusions
In this study, headache-free episodic migraineurs had enhanced pain-induced activation of several pain-processing regions. Although these regions participate in different aspects of pain processing, many are specifically involved with cognitive aspects of pain. We hypothesize that greater activation of these regions represents an experience-driven sensitization of the migraine brain to pain. Although acclimation to recurrent pain might be expected in the healthy brain, our study suggests that the migraine brain instead develops an enhanced cognitive response to pain. This enhanced response may reflect hypervigilance and anxiety about pain and a greater attentiveness to pain, features that are typical in migraine.
Clinical implications
Headache-free migraineurs have enhanced pain-induced activation of brain regions that participate in cognitive aspects of pain processing. Enhanced cognitive processing of pain by migraineurs might reflect an underlying cerebral hypersensitivity related to high expectations for pain and hypervigilance for pain.
Footnotes
Funding
This study was funded by a grant from the National Institutes of Health (K23NS070891) to TJS.
Conflict of interest
None declared.
