Abstract
Functional imaging of human trigemino-nociceptive processing provides meaningful insights into altered pain processing in head and face pain diseases. Although functional magnetic resonance imaging (fMRI) offers high temporal and spatial resolution, most studies available were done with radioligand-positron emission tomography, as fMRI requires non-magnetic stimulus equipment and fast on–off conditions. We developed a new approach for painful stimulation of the trigeminal nerve that can be implemented within an event-related design using fMRI and aimed to detect increased blood-oxygen-level-dependent (BOLD) signals as surrogate markers of trigeminal pain processing. Using an olfactometer, 20 healthy volunteers received intranasally standardized trigeminal nociceptive stimuli (ammonia gas) as well as olfactory (rose odour) and odourless control stimuli (air puffs). Imaging revealed robust BOLD responses to the trigeminal nociceptive stimulation in cortical and subcortical brain areas known to be involved in pain processing. Focusing on the trigeminal pain pathway, significant activations were observed bilaterally in brainstem areas at the trigeminal nerve entry zone, which are agreeable with the principal trigeminal nuclei. Furthermore, increased signal changes could be detected ipsilaterally at anatomical localization of the trigeminal ganglion and bilaterally in the rostral medulla, which probably represents the spinal trigeminal nuclei. However, brainstem areas involved in the endogenous pain control system that are close to this anatomical localization, such as raphe nuclei, have to be discussed. Our findings suggest that mapping trigeminal pain processing using fMRI with this non-invasive experimental design is feasible and capable of evoking specific activations in the trigeminal nociceptive system. This method will provide an ideal opportunity to study the trigeminal pain system in both health and pathological conditions such as idiopathic headache disorders.
Keywords
Introduction
Modern neuroimaging techniques such as positron emission tomography (PET) and functional magnetic resonance imaging (fMRI) have fundamentally improved our knowledge about the neuronal representation of pain (1–3). Previously published studies used different types of stimuli for inducing pain and revealed multiple cortical and subcortical areas activated by painful stimulation: the so-called pain matrix (4–6). Nevertheless, there is still limited information about the occurrence and location of brain activity in response to nociceptive stimulation of the trigeminal system in healthy pain-free volunteers and particularly in headache patients. Most studies to date investigating cerebral pain processing in headache patients have used either single-photon emission computed tomography (7–9) or PET (10–13). However, fMRI has some crucial advantages over PET. First, PET relies on the injection of radioactive tracers, whereas fMRI is entirely non-invasive. Further, fMRI has a higher spatial resolution and, especially with an event-related design, superior temporal resolution can be achieved. On the other hand, the magnetic field in MR leads to specific restrictions: material that is used in the high-magnetic-field room has to be non-ferromagnetic. In addition to the technical restrictions, several factors make stimulation of areas innervated by the trigeminal nerve more challenging than stimulation of other body parts: Trigeminal nociceptive stimulation has to be placed close to the subject's face, a particularly sensitive body part. Previous studies used thermal heat to initiate painful sensations on the skin of the forehead, maxilla or mandible (14–16), accepting the fact that tactile sensations become intermingled with nociceptive input. An alternative method to avoid tactile sensations during painful stimulation is the use of laser (17,18), where the head coil makes it difficult to place the thermode as well as the laser pointer on the subject's face and bears the danger of accidentally injuring the eyes. Finally, these procedures are highly susceptible to artefacts during fMRI measurements.
Other researchers have applied capsaicin superficially to the face within the receptive field of the trigeminal nerve investigating pain sensations, hyperalgesia or allodynia phenomena (16,19,20). However, capsaicin leads to sensations over a large area lasting for several minutes and needs several minutes to wean off, restricting the analysis to blocked designs and PET analysis (12). Event-related designs have important advantages over blocked designs: short stimuli can be presented repetitively without having undesirable peripheral habituation effects (21,22). Chemosensory trigeminal activation using intranasal CO2 application has been used in fMRI investigations (23–25); however, because of a possible general blood flow increase after CO2 application (26), the interpretation of fMRI data using CO2 is not without ambiguity.
Earlier studies have already presented stimulation devices for chemosensory input that are compatible with the MR environment. However, these methods are designed specifically for odour input (27,28). The aim of the present study was to develop an experimental stimulation approach that can be implemented within an event-related fMRI pain paradigm to investigate, in addition to the olfactory system, trigemino-nociceptive pathways. To avoid tactile sensation on the skin and because all the material used in the high-magnetic-field room has to be non-ferromagnetic, we used an olfactometer (Fig. 1) for administering a chemical substance intranasally. Because of a possible general blood flow increase after CO2 application (26), we decided to use a low concentration of gaseous ammonia, administered intranasally (29). We validated our approach in a group of healthy subjects using fMRI, which offers unique opportunities to further explore and pharmacologically modulate the pathogenic mechanisms of the trigeminovascular system in different types of head and facial pain.
Set-up of the trigeminal nociceptive stimulation system in the functional magnetic resonance imaging environment. Gaseous substances are conveyed through an air-proofed tube system that ends in the subject's nose. Ammonia gas (2.5%) elicits irritation of the trigeminal nerve leading to painful sensation whereas rose odour excited the olfactory nerve. Air-puffs are implemented as a control condition. Only the Teflon tube enters the magnet-high-field room; the stimulation equipment, which is connected with the tube, is positioned in the anteroom.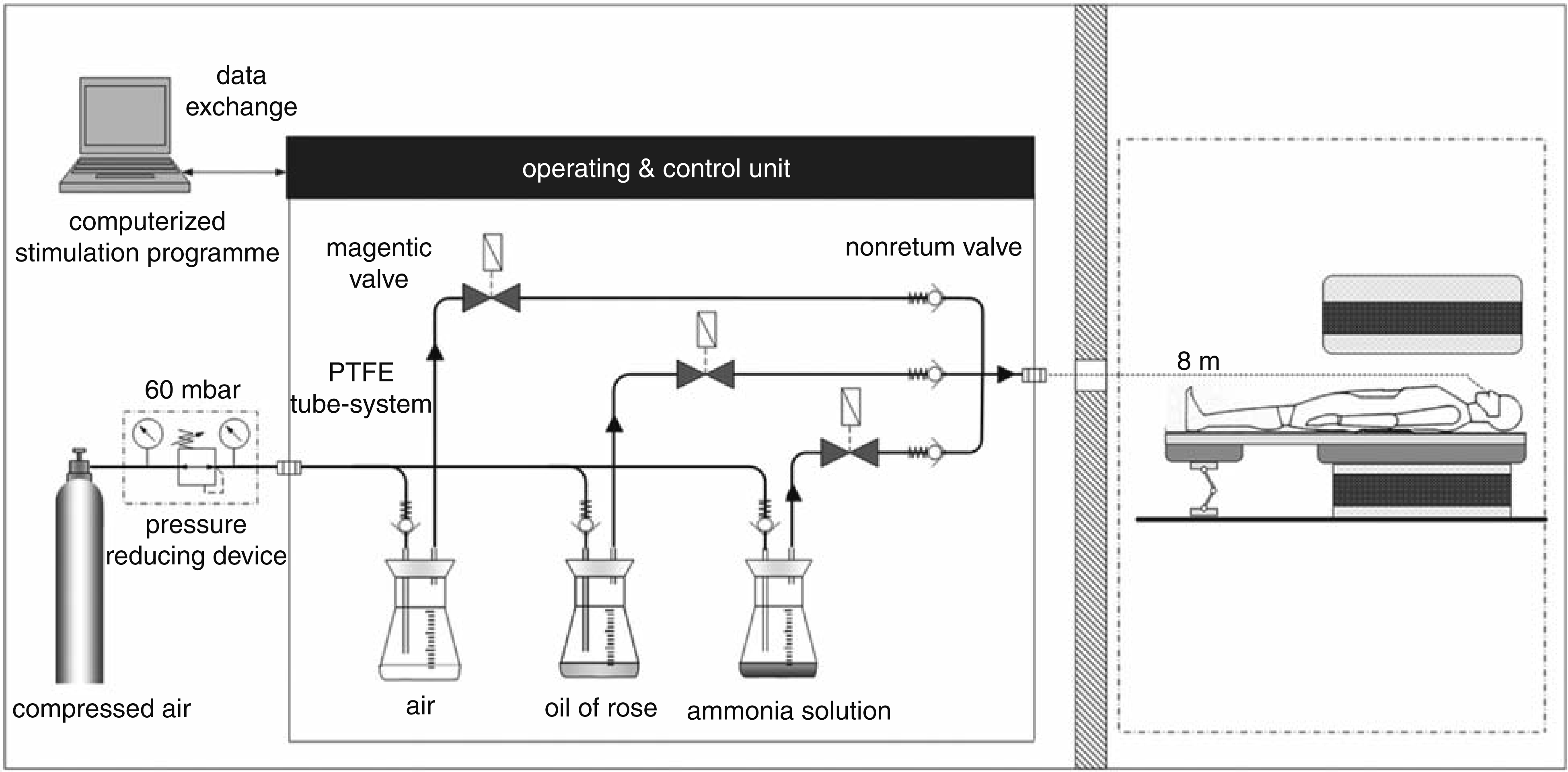
Methods and materials
Subjects
We studied 23 (n = 11 female) healthy subjects with a mean age of 28 years (range 20–39 years) who were recruited locally. None of them had a history of neurological or psychiatric diseases, particularly no history of pain or head pain syndromes, or was taking any medications. We also assessed any occurrence of minor pain events, including tooth and ear ache, headache or contusions up to 4 weeks prior to the study period.
All subjects gave their informed consent; the study was conducted in accord with the declaration of Helsinki and approved by the Ethics Committee in Hamburg, Germany. Before the beginning of the experiment, subjects were informed in detail about the purpose of the study and about the temporal sequence of the stimulation process. Subjects were also informed that they could withdraw from the experiment at any time.
Stimulation approach
Our model was designed to achieve tolerable but effective trigeminal nociceptive stimulation. For nociceptive stimulation, we chose gaseous ammonia, which was applied to the intranasal receptive field innervated of the trigeminal nerve. Intranasally, this substance stimulates the nasal mucosa, leading to irritation of the first and second branch of the trigeminal nerve (V1 and V2), resulting in short-lasting, stinging or stabbing pain sensations (29,30). Control conditions consisted of either an odour (rose), which mainly excites the olfactory nerve, or an odourless stimulus (air puffs), applied intranasally.
The olfactometer consists of an air-proofed tube system made of polytetrafluoroethylene (Teflon). The technical construction of the equipment is presented in Fig. 1. One tube connects to a hermetically sealed glass bottle, which keeps the air pressure at a constant 6 kPa. A permanent air pressure is applied on the tube system. Three bottles were used in our experiment; one was filled with ammonia solution with a concentration of 4.3% weight per volume (10% solution mixed with distilled water). A second bottle contained synthesized rose oil and the third bottle contained air. These three substances (gaseous ammonia, rose and air) were conveyed through a thin tube (8 m long and with an inner diameter of 4 mm), which was placed in the subject's right nostril. Only the Teflon tube entered the magnet-high-field scanner room; the stimulation equipment, which was connected with the tube, was positioned in an anteroom.
A stimulation programme (presentation; http://www.neurobs.com; version 13.0) was used to trigger electrical inductors randomly, controlling magnetic valves between the bottles and the tube (closed during off-line setting). This programme controlled the opening and closing valves at fixed time points for 800 ms each. Stimuli (ammonia gas or odour) were embedded in a constant flow of odourless air and after each flow of substances an air flow neutralized the tube system. The stimulation PC triggered the inductor, which was connected with the empty glass bottle (air) in order to open the valve for 3 s.
In cooperation with the University of Technology in Hamburg (Institute of Measurement Technology), the concentration of the gaseous ammonia at the end of the tube (through which gas was applied) was repeatedly measured. Ammonia solution (25 ml), which was filled in the bottle, had a concentration of 4.3%, whereas gaseous ammonia at the end of the tube reached a concentration of 2.5%, which stayed constant over time, i.e. every ammonia stimulus had the same chemical concentration. Constant air pressure, technically precise electrical triggering of the magnetic valves and constant chemical concentration ensured reliable stimulus application during the experiment.
Training session
Prior to the fMRI experiment, subjects were trained in our model outside the scanner. All were asked to place the tube into their right nostril and were instructed to rate the intensity of trigeminally mediated pain sensations when receiving ammonia and the odour intensity when smelling rose. Subjects were trained to move the curser using a button box left (thumb) or right (index finger) to rate intensity on the numerical rating scale (NRS); a button press (middle finger) confirmed their rating. Only those subjects in whom the given ammonia concentration evoked a clearly painful sensation (which was pre-defined as ≥ 4 on the NRS) were included in the fMRI experiment and continued the entire protocol. During scan time, the tube was fixated with a band-aid on the subjects' upper lip and subjects were instructed to breathe orally during the experiment to avoid fluctuations in stimulus concentrations due to respiratory airflow.
Experimental paradigm
The experimental paradigm is shown in Fig. 2. At the beginning of each trial (15 trials per condition: ammonia, odour and air puffs) subjects underwent an attention task. A fixation cross were presented on a screen. This cross changed from red to yellow after a jittered time of 8–10 s. Subjects were instructed to press a button box as soon as the colour changed. The fixation cross then faded out and the screen went blank. Nine to 11 s later (jittered time interval) subjects were exposed to one of three stimuli (ammonia, odour, air). Each condition was presented 15 times per session in a randomized order. The triggered valve opened for 800 ms; immediately afterwards the valve of the empty bottle opened for 3 s to wash the tube free of any substances. The substances reached the subjects' nose 4 s after opening the respective valve. After a jittered time delay of 6–8 s, subjects were presented with a NRS on the screen anchored at 0 (no sensation) to 10 (highest imaginable intensity). All participants were instructed to evaluate pain and odour intensity on this scale. The choice of stimulation parameters was based on a psycho-physiological pilot experiment, in which we ensured that the stimulation stayed painful for the individual over the session. The use of online rating in our experiment is considerably important, as remembered pain ratings can differ substantially from concurrent pain ratings (31).
Experimental design: the stimulation model is separated into three parts: attention task, trigeminal stimulation, and rating procedure. Prior to each of the 45 trials (each stimulus is presented 15 times) a reaction task is implemented. Subjects are instructed to press a button immediately after the fixation cross changes its colour from red to yellow. After a jittered time delay, subjects undergo the trigeminal stimulation paradigm in which stimuli (2.5% ammonia, rose odour, air) are administered randomly. After each stimulus, subjects rate the intensity at a numerical rating scale and a randomized interstimulus interval follows. BOLD, blood-oxygen-level-dependent.
Behavioural data analysis
Behavioural data analysis was performed using Statistical Program for Social Sciences (
Image acquisition
Echo-planar images were collected on a 3-T scanner (Siemens-Trio; Siemens, Munich, Germany) using a standard 12-channel head coil. Functional scans used the following parameters: 42 axial slices (slice thickness 3 mm) were acquired, voxel size = 3 mm3, time to echo = 30 ms, repetition time = 2620 ms, flip angle 80°, field of view 192 mm2. Additionally, high-resolution T1-weighted structural images (1 × 1 × 1 mm3 voxel size) were acquired using a MPRAGE sequence.
Image processing and statistical analysis—fMRI
fMRI data and statistical analyses were processed using statistical parametric mapping (
In a second model a finite impulse response (FIR) basis function was defined to display activation time courses. The post-stimulus blood-oxygen-level-dependent (BOLD) response was averaged; a bin width of 2.6 s was used modelling a total of 10 bins from 0 to 26 s after stimulus onsets. This resulted in 10 regressors for each condition (ammonia, odour and air) and 30 regressors for all conditions. This basis set considered each time bin after stimulus onset individually to model the BOLD response at that time. Data were analysed for each subject individually in a first-level analysis before all 30 parameter estimates images were entered into a random effects analysis.
Results
Of 23 volunteers (nine female) only those subjects were included in the study who reported at least moderate pain (as defined by the NRS) to the trigeminal nociceptive stimuli in the pre-experimental testing phase. Two of them were ‘non-responders’ and one subject decided to drop out because of the unpleasantness of the used type of pain stimulus. Therefore, these subjects were excluded from our study, leaving complete datasets of 20 volunteers. Behavioural pain rating data (ammonia condition) showed a mean value of 5.78 on the rank scale (0–10) over all subjects (
Activation related to trigeminal nociceptive stimulation
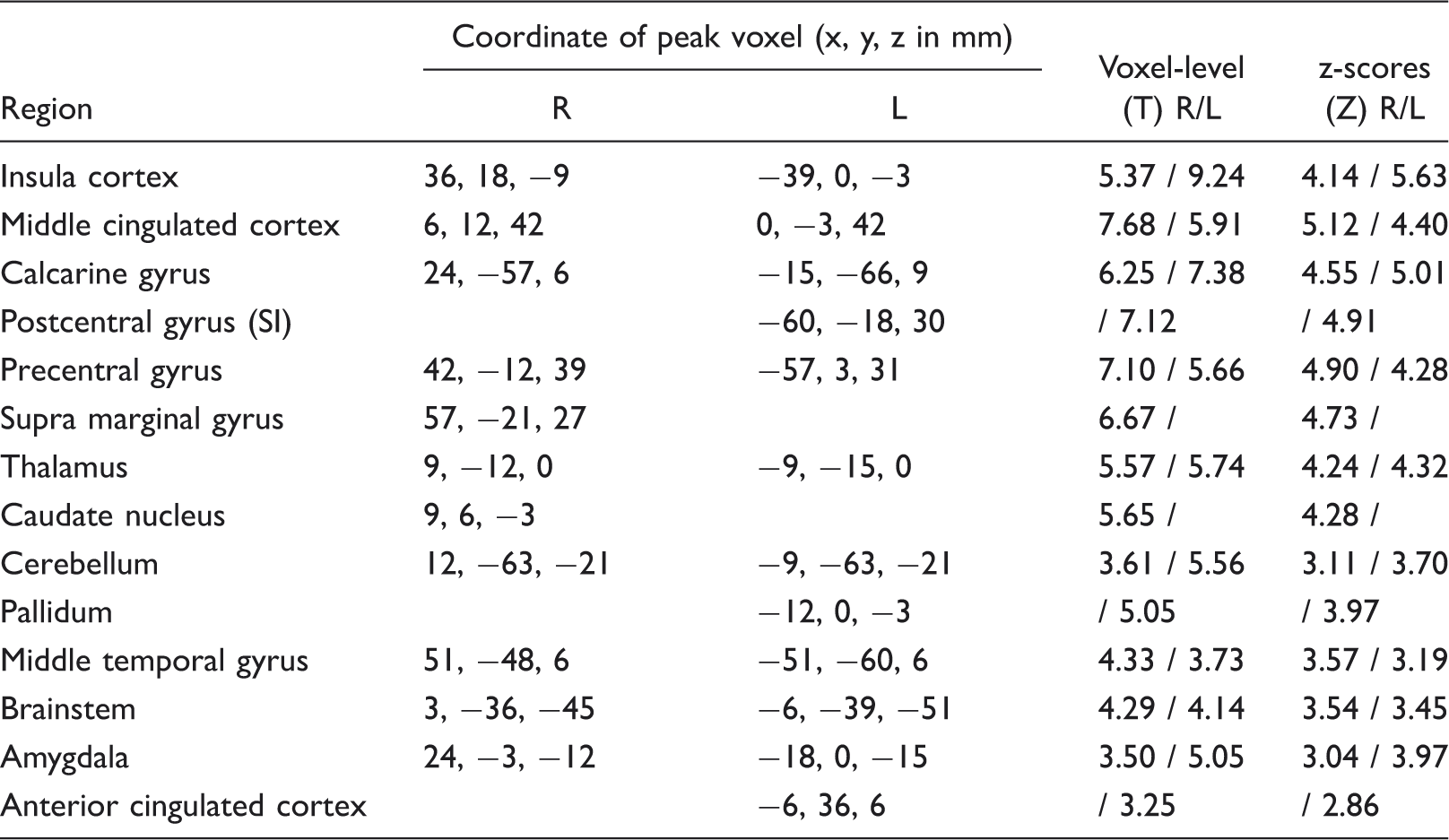
The voxel-by-voxel test resulting from data smoothed with a 10-mm3 kernel revealed statistically significant (FDR-corrected; P < 0.05) bilateral activation in the insula, thalamus, middle cigulate cortex, amygdala, precentral gyrus, calcarine, cerebellum, middle temporal gyrus and brainstem in the rostral medulla and lower pons. Increased responses to trigeminal nociceptive stimulation were also observed ipsilaterally in the caudate nucleus and the supra marginal gyrus, whereas contralateral activations were seen in left anterior cingulate cortex, postcentral gyrus and pallidum. Table 1 lists MNI coordinate locations of neuroimaging findings resulting from 10-mm3 FWHM kernel smoothed data. Brainstem activation patterns probably reflect increased BOLD signal of the spinal trigeminal nucleus (15,33). A t-score map of the linear contrast pain (ammonia > air puffs) resulting from 10-mm3 smoothed data is shown in Fig. 3a; brainstem activation is shown in Fig. 3b. To illustrate the time course of the BOLD signal during ammonia condition in the brainstem, the averaged post-stimulus response resulting from FIR basis set analysis is plotted in Fig. 4 (at the peak voxel of brainstem activation: 3, −36, −45).
In the upper part (a) functional magnetic resonance imaging results from the contrast ‘ammonia (pain) > air puffs’ are demonstrated. The sagittal slice (X = 0) on the left side shows the localization of selected transverse slices (1–6) at different coordinates that are presented on the right side. Six pictures in transversal view of the brain (descending order: Z = 60, Z = −36, Z = 20, Z = 15, Z = 0, Z = –45) show detected blood-oxygen-level-dependent signals in areas known to be involved in pain processing including thalamus, insula, somatosensory cortex, middle and anterior cingulum (uncorrected; P < 0.001). The lower part (b) focuses on active brainstem parts during trigemino-nociceptive stimulation. Details of activation patterns in the lower pons/rostral medulla are presented at three different views at peak voxel coordinates in brainstem cluster (X = 3, Y = −36, Z = −45) (visualization threshold P < 0.001; uncorrected). The plot is derived from a finite impulse response (FIR) basis set. Parameter estimates demonstrate the averaged time course of the post-stimulus blood-oxygen-level-dependent (BOLD) signal in the peak voxel of brainstem activation (3, −36, −45) (red line; FIR). The black line shows the haemodynamic response function (HRF). The x-axis presents the post-stimulus time in bins from 0 to 26 s, whereas the y-axis is labelled as ‘parameter estimates u.a.’; these parameter estimates are directly proportional to the BOLD signal. Activity related to nociceptive painful stimulation (ammonia > air puffs; 10 mm3 smoothing)

Using a smaller FWHM kernel (6 mm3), significant activations correlating with nociceptive input were found bilaterally at the nerve entry zone into the pons at the anatomical localization of the trigeminal principal nuclei (right: 18, −27, −36; T = 3.96; Z = 3.34; left: −12, −27, −33; T = 3.87; Z = 3.28) as well as in brain areas corresponding to the ipsilateral localization of the TG (24, 3, −36; T = 2.62; Z = 2.39) (Fig. 5). Furthermore, smoothing the data with a smaller kernel elicited more differentiated BOLD signal changes in the pons (3, −30, −27; T = 3.96; Z = 3.34) and midbrain, probably representing the anatomical localization of the substantia nigra (9, −18, −21; T = 5.30; Z = 4.10) and the nucleus ruber (right: 12, −21, −15; T = 3.87; Z = 3.28; left: −12, −21, −15; T = 2.88; Z = 2.59). Uncorrected threshold of P < 0.005 was used for the 6-mm3 smoothing analysis.
Significant activations in brainstem regions including data smoothed with a smaller 6-mm3 Gaussian kernel at the Z-coordinate –36. Active voxel could be detected in anatomical localization of the trigeminal ganglion (ipsilateral to the stimulation side) as well as bilaterally in the brainstem (more ipsilateral to the stimulation side) at the trigeminal nerve entry zone, possibly reflecting the principal trigeminal nuclei (prTNcl; P < 0.005; uncorrected). TG, trigeminal ganglion.
Functional imaging findings as regards olfactory processing
Activity related to olfactory stimulation (odour > air puffs)

Discussion
Mapping functional response of the trigeminal system during pain is a useful tool both for characterizing normal brain activity and for determining possible functional abnormalities in idiopathic headache syndromes such as migraine. The trigeminal system is, however, more challenging to study than other body parts, and special characteristics of the MRI environment restrict the experimental set-up. We present a MR-compatible stimulation device for trigemino-nociceptive stimulation without tactile sensation that can be successfully implemented in an event-related design during fMRI. Healthy, pain-free subjects were investigated in the present study to prove the validity of the experimental paradigm and to provide functional data for future comparisons with headache patients.
We aimed to detect BOLD signal changes in central brain areas involved in the trigeminal pain system including the peripheral ganglion, brainstem parts (trigeminal nuclei) and cortical regions (thalamus and somatosensory cortex).
Our results show robust activations during experimental trigemino-nociceptive stimulation in cortical and subcortical brain areas most commonly seen in the context of pain: these areas include the anterior cingulate cortex, the primary and secondary somatosensory cortex, the insula, thalamus, amygdala and frontal brain areas. Hence, our stimulation device enables us to detect brain responses as surrogate markers for trigeminal pain. Our results support findings reported in the literature with regard to pain processing in general (34). However, we aimed to identify specific trigeminal-related signal changes during the experimental condition. We therefore focused our interest on activations in the trigeminal pain pathway including the TG and spinal trigeminal nuclei.
Following trigeminal painful stimulation, significantly increased BOLD responses were detected in brainstem regions in the rostral medulla and lower pons, even with a relatively large smoothing kernel of 10 mm3. These activations were located bilaterally, although higher t-values were seen ipsilateral to the stimulation site. Sensory information (including pain) is conveyed to the central nervous system via the trigeminal sensory pathway converging on the TG, which contains the cell bodies of incoming sensory nerve fibres (35). The trigeminal nucleus is divided into three parts; pain and temperature fibres terminate in the spinal trigeminal nuclei, while touch is conveyed by sensory afferents to the principal sensory nucleus. From here, secondary fibres cross the midline and ascend in the trigeminothalamic tract to the contralateral thalamus (15,36,37). Precise identification of anatomical brainstem correlates is, however, difficult due to several technical limitations including restricted spatial resolution (voxel size in our study was 3 mm3), local field inhomogeneity-induced signal losses, image distortion and the lack of adequate template images. Furthermore, data processing procedures such as smoothing could also distort exact localization of anatomical and thus functional correlates (38,39). For this reason, we tested brainstem regions using a filter of 6 mm3 FWHM. Activation patterns were observed bilaterally in the brainstem at the nerve entry zone; pronounced ipsilaterality to the stimulation site probably reflecting the principal trigeminal nuclei.
In addition, and in accordance with the literature, a significantly increased BOLD signal ipsilateral to the stimulation was detected outside the brain at the anatomical localization of the TG (40,41) and in specific parts of the midbrain and pons—probably the nucleus ruber and substantia nigra. The latter structures are, along with the periaqueductal grey and locus coeruleus, thought to play a role in endogenous pain control (42–45). Taken together, our experimental design identifies specific structures involved in trigeminal pain processing in man. However, given that the brainstem is relatively small (15), further difficulties regarding the interpretation of our fMRI findings in small brainstem regions may derive from second level processing. As individual localization of brainstem nuclei probably differ from subject to subject and significant statistical activation patterns are based on average data of all subjects, small variations of individual localizations may lead to lower statistical power.
Consequently, in discussing BOLD responses in the upper medulla and pons, other brainstem areas have to be taken into account (46). The nucleus raphe magnus especially, which is close to the upper part of the spinal trigeminal nucleus (bilaterally in the lower pons), is a candidate whose possible role in trigeminal pain processing has be discussed (47,48). Raphe nuclei release serotonin in other brain areas and current studies suggest crucial involvement in endogenous analgesia (44). Increased BOLD response to the pain stimuli may also reflect its antinociceptive function; raphe nuclei send projection to the dorsal horn of the spinal cord to directly inhibit pain (49,50). However, one argument in favour of trigeminal nuclei over the spinal antinociceptive tracts is the fact that the activation was seen bilaterally and pronounced ipsilateral to the stimulation site, although arterial pulsation and subjects' head motions during the scanning period could limit the interpretation of side-specific activation patterns (16).
Analysing the activation following odorant stimuli we found an increase in BOLD signal in different brain areas, known to be responsible for olfactory perception. In recent years, neuroimaging studies have shed light on odour-induced brain responses in humans (51). Several brain structures could be identified which are summarized into the primary olfactory system (piriform and entorhinal cortex and amygdala) and secondary olfactory system (insula and orbitofrontal cortex) (52–54). However, imaging studies in humans show inconsistent activation in these structures (55). Our study revealed robust activation in the amygdala, whereas no activation could be observed in other parts of the primary olfactory system. Our knowledge about cortical representation of brain structures involved in odour perception derived originally from animal studies, which identified the primary olfactory cortex as a fundamental structure in odour perception. Rapid habituation phenomenon in the piriform cortex is known in the rat (56); a similar process could be responsible for weak or undetectable activity in these areas in humans. The present study has revealed further activity bilaterally in the insula. Peak voxel could be detected additionally in frontal brain structures such as the inferior frontal and middle frontal gyrus. However, activation patterns extend into surrounding areas; significantly activated voxel could also be found in the orbitofrontal cortex, which belongs (as well as the insula) to the secondary olfactory system (as described above). Finally, our stimulation paradigm leads to increased BOLD signal in brain areas that are not part of the classical olfactory system (i.e. pallidum, putamen, caudate nucleus, supramarginal, precentral and postcentral gyrus and cerebellum). These findings are in line with previous imaging studies and probably indicate that olfactory processing involves additionally higher-order associative processes, such as memory and somatosensory processes (51,52).
In summary, our findings demonstrate that the standardized application of trigeminal nociceptive input using an olfactometer is compatible with fMRI data acquisition and prompts reliable painful activation of the trigeminovascular system detectable with event-related fMRI. This device may be used to investigate the trigeminal pain pathway in acute migraine attacks and other types of chronic head and facial pain as well as mapping of pharmacologically induced modulations of these pathways.
Footnotes
Acknowledgements
He authors thank the Physics and Methods group at NeuroImage Nord in Hamburg, and further thank the staff of the Institute of Measurement Technology of the University of Technology in Hamburg (Germany) for technical support. This work was supported by an unrestricted grant from Allmirall, SA.
