Abstract
Background
Increasing evidence suggests that vasoactive neuropeptides such as pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38), substance P, calcitonin gene-related peptide, and vasoactive intestinal peptide are involved in the pathophysiology of migraine in adults, but their role in pediatric migraineurs remains unclear. We prospectively investigated plasma levels of these vasoactive neuropeptides in pediatric migraine patients without aura and compared the results with those of age-matched healthy controls.
Methods
Thirty-eight children aged 6–18 years with migraine without aura and 20 age-matched control subjects were included in the study. Neuropeptides in plasma samples from the controls, and in either the ictal or interictal periods in pediatric migraine without aura, were measured using ELISA.
Results
PACAP-38 and vasoactive intestinal peptide levels in both ictal and interictal plasma were higher in the patients with pediatric migraine without aura than in the controls (p < 0.001), although calcitonin gene-related peptide and substance P levels remained unchanged. Otherwise, no significant difference was determined between ictal and interictal periods in terms of all neuropeptide levels.
Conclusions
This study demonstrates increased plasma PACAP-38 and vasoactive intestinal peptide levels, but not calcitonin gene-related peptide and substance P levels, in pediatric patients with migraine during both attack and attack-free periods. The study findings suggest that PACAP-38 and vasoactive intestinal peptide may be implicated in the pathophysiology of migraine, particularly in pediatric migraineurs.
Introduction
Migraine is one of the most common and disabling neurovascular disorders. It has a complex pathophysiology; however, a large body of accumulating evidence suggests that sterile neurogenic inflammation plays a key role in the pathogenesis of migraine (1–3). This neurogenic inflammation is characterized by plasma protein extravasation, vasodilation, and mast cell degranulation in the dura mater (3). Preclinical observations indicate that stimulation of trigeminal sensory fibers causes neurogenic inflammation in the intracranial meninges (dura mater) through release of vasoactive neuropeptides, particularly calcitonin gene-related peptide (CGRP) and substance P (SP), from these afferent terminals through an axon reflex, and via dural mast cell degranulation (1–4). While SP leads to plasma protein extravasation secondary to capillary leakage, CGRP causes vasodilation in the dura mater (1–3), while both induce dural mast cell degranulation (5). When degranulated, mast cells release a wide range of pro-inflammatory and pro-nociceptive mediators to residents such as CGRP, SP, serotonin, prostaglandins, bradykinin, histamine and cytokines that further aggravate the neurogenic inflammation (6). This neurogenic inflammation consequently leads to activation and sensitization of the trigeminovascular system, resulting in the initiation of neurovascular type headaches including migraine.
In addition to CGRP and SP, there is growing evidence suggesting that the other vasoactive neuropeptides, vasoactive intestinal peptide (VIP) and pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38), are also involved in the pathophysiology of migraine (7–9). However, clinical and animal studies have reported little information about their relevance in migraine (8–11).
The cranial dura mater is regarded as the site of origin of migraine pain (12,13). It is largely innervated by sensory trigeminal fibers containing CGRP, SP and PACAP-38, by parasympathetic fibers containing PACAP-38 and VIP, and also by sympathetic fibers containing neuropeptide Y and ATP (11,14). It is generally agreed that one major component of migraine pain stems from chemical or mechanical irritation of the meninges, resulting in the release of these vasoactive neuropeptides (15). All these neuropeptides have potent vasodilating effects. However, CGRP and PACAP-38 exert pro-nociceptive and sensitizing effects on the peripheral and central sensory trigeminal fibers (8,16). Such effects make these neuropeptides a subject of particular interest in the investigation of the pathogenesis of migraine.
The vast majority of migraine studies focus on adults, although epidemiological studies have reported a quite high prevalence of pediatric migraine in the general population, at 3% in younger children and ∼20% in adolescents (17). It also remains unclear whether adult and pediatric migraine patients share common pathophysiological mechanisms. However, diagnosis of migraine is based on the clinical features described by patients, and pediatric patients are unable to clearly describe migraine symptoms. In addition, a number of medications are used for symptomatic and prophylactic treatment of migraine in adult patients, but their efficacy and safety in juvenile migraine patients are not yet clear. There is therefore an urgent need for parameters capable of use in the diagnosis of pediatric migraine. The detection of potential migraine biomarkers in the peripheral blood will thus greatly facilitate the diagnosis of migraine and decisions regarding the treatment to be administered, especially in pediatric patients. There have been rare studies investigating the diagnostic value of plasma CGRP, SP and VIP in pediatric migraine (18–20), although to the best of our knowledge, there has been no investigation of plasma PACAP-38 in children. The present study therefore prospectively investigated plasma PACAP-38, CGRP, SP and VIP levels in patients with pediatric migraine without aura (PMWA), and compared the results with those of age-matched healthy controls.
Materials and methods
Participants
Thirty-eight pediatric migraine patients without aura aged 6–18 years and 20 age-matched healthy controls who attended the Bolu Abant Izzet Baysal University Training and Research Hospital outpatient clinic, Turkey, between November 2018 and December 2019 were prospectively enrolled in this study. The diagnosis of pediatric migraine was based on the criteria of the International Classification of Headache Disorders 3rd edition, beta version (21). Comprehensive major symptoms of migraine were recorded, including unilaterality, throbbing headache, nausea, vomiting, photophobia, phonophobia and dizziness. The demographic and clinical characteristics of the patients and controls are shown in Tables 1 and 2. A detailed questionnaire was used to form a homogeneous migraine group in terms of features of their migraine diseases based on type of migraine, the duration of the migraine, and the attack frequency. Exclusion criteria were presence of drug intake, autoimmune and neuro-inflammatory diseases, mast cell-related diseases, depression, allergies, intracranial mass, congenital or genetic anomalies, and chronic disorders. The number of excluded patients and the reasons for exclusion are shown in Figure 1. The control group included 20 healthy children (seven boys and 13 girls) without headaches attending the Bolu Abant Izzet Baysal University Training and Research Hospital pediatric outpatient clinic for check-ups before engaging in athletic or school activities. The patients and healthy controls were all of Turkish origin and from the Western Black Sea region of Turkey. Sex, age, and physical and neurological examination findings were also recorded for the control group. The healthy controls were also checked for non-reported headaches.
Demographic data of 16 migraineurs in the ictal periods and 22 migraineurs in the interictal periods, respectively. The age, gender, type of migraine (without aura), disease duration (years) and attack frequency (per month) are shown for each patient.
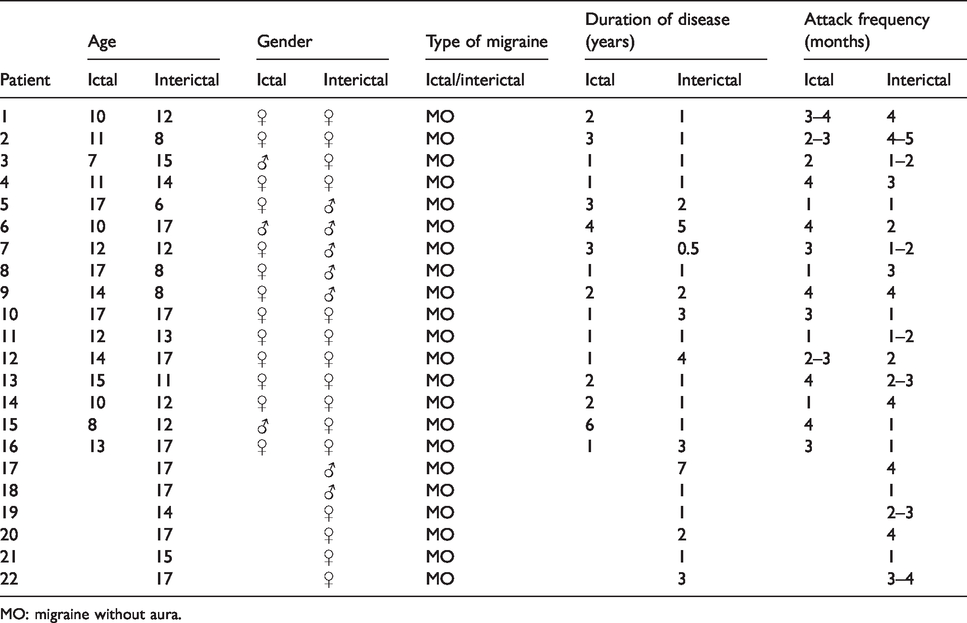
MO: migraine without aura.
Demographic data of 20 healthy control subjects. The age, gender, physical and neurological status are shown for each subject.

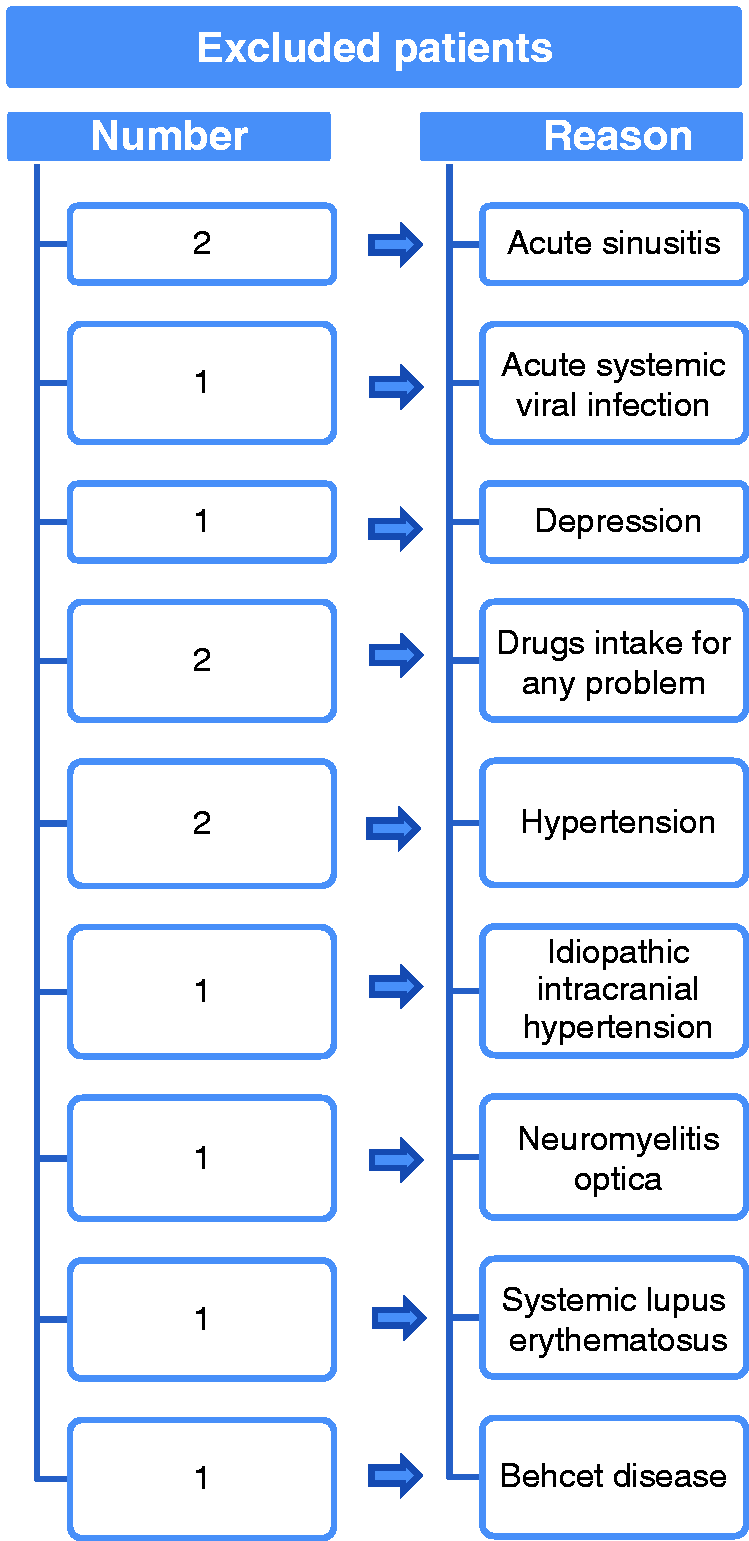
Flowchart indicating the number of excluded patients and reasons for the exclusion.
Study design and procedures
The study was approved by the Bolu Abant Izzet Baysal University Clinical Research Ethics Committee (licence number 2018/189). Signed, informed consent was received from all healthy and patient volunteers and their parents in compliance with the Declaration of Helsinki. No restrictions were imposed in terms of food and drink intake.
Blood samples were collected from migraineurs during attacks (ictal) in 16 patients and in attack-free periods (interictal) in 22 patients. Blood collected during attacks was obtained at any time within the first 8 h of the headache episode. The non-attack (interictal) group consisted of patients who were headache free for at least 24 h before and after blood sampling. Data collected included age, sex, type of migraine, duration of disease, and attack frequency. A single blood sample was collected from each of the 20 controls. Five milliliters of blood was taken from the antecubital vein of each volunteer in a sitting position during rest. The samples were instantly transferred into ice-cold glass tubes containing the anticoagulant EDTA and protease inhibitor cocktail (cOmplete, Sigma-Aldrich), and centrifuged at 3000 rpm for 15 min at 4°C. The supernatants were stored at −80°C until being assayed for CGRP, PACAP-38, SP and VIP immunoreactivities using the ELISA method.
Measurements of plasma CGRP, PACAP-38, SP and VIP levels
CGRP, PACAP-38, SP and VIP contents in the samples were measured using enzyme-linked immunosorbent assay kits (BT Lab, Shanghai, China). The detection limits are ∼2 pg/ml for CGRP, ∼50 pg/ml for PACAP-38, and ∼5 pg/ml for SP and VIP. Assay protocols were carried out in line with the manufacturer’s instructions and in duplicate. Briefly, 50 µL of CGRP, PACAP-38, SP and VIP standard was added to their standard wells. Next, 40 µL of samples, and 10 µL of anti-CGRP antibody for CGRP assay and anti-PACAP-38, SP and VIP antibodies for PACAP-38, SP and VIP assays were then added to sample wells. Fifty microliters of streptavidin-HRP was added to each well, and the 96-well plates were incubated at 37°C for 60 min. The plates were subsequently washed five times with the wash buffer. Fifty microliters each of substrate solution A and substrate solution B was added to each well. The 96-well plates were incubated at 37°C for 10 min. Finally, 50 µL of stop solution was added to each well, and the optical density was immediately measured at 450 nm using a microplate reader (Epoch BioTek Instruments Inc., Winooski, VT, USA). Optical density curves were obtained using standards with defined CGRP, PACAP-38, SP and VIP concentrations.
Statistical analysis
The data obtained from the groups were expressed as mean ± SD. Statistical analysis was performed using SPSS for Windows (version 17.0, SPSS Inc., Chicago, IL, USA). The Shapiro-Wilk test was used to assess normality differences between or among groups, which were compared using the independent samples t-test, Mann-Whitney test, Kruskal-Wallis H test or ANOVA, followed by the Bonferroni post-hoc test. p < 0.05 was regarded as statistically significant.
Results
Plasma samples were collected from 38 children with migraine (10 boys) aged 6–18 years (mean age, 13.0 ± 3.3 years) and 20 healthy controls (seven boys, mean age 11.3 ± 2.9 years). The mean age of the migraineurs was not significantly different from that of the healthy controls (p = 0.069). Sixteen (42%) patients with migraine were blood sampled during headache attacks (ictal period) and 22 (58%) between attacks (interictal period). There was also no significant difference between the mean ages of the migraineurs in the ictal period and those in the interictal period (p = 0.33). The mean attack frequency per month of the migraineurs in the ictal period (2.7 ± 1.2) was not significantly different from that in the interictal period (2.4 ± 1.2, p = 0.647). Similarly, no difference in mean duration of disease (years) was observed between the migraineurs in the interictal period (2.1 ± 0.3) and the interictal period (1.9 ± 0.3, p = 0.44).
Plasma PACAP-38, VIP, CGRP and SP levels in the entire migraine patient group and healthy controls
Since the variables of PACAP-38 and VIP levels in control (n = 20) and migraine patient (ictal and interictal, n = 38) blood specimens exhibited normal distribution, these were compared using the independent samples t-test. Plasma PACAP-38 levels in the migraine group (1806 ± 724 pg/ml) were significantly higher than in the control group (830 ± 275 pg/ml, p = 0.001, Figure 2(a)). Plasma VIP levels in the migraine group (199 ± 75 pg/ml) were also significantly higher than in the control group (92 ± 38 pg/ml, p = 0.0001, Figure 2(b)). In contrast, since the variables of CGRP and SP levels in control (n = 20) and migraine patient (ictal and interictal, n = 38) blood specimens did not exhibit normal distribution, these were compared using the Mann-Whitney U test. There was no significant difference between the control and migraine groups in terms of either plasma CGRP or SP levels (control CGRP: 205 ± 41 pg/ml, migraine CGRP: 213 ± 59 pg/ml, p = 0.863, Figure 2(c); control SP: 139 ± 128 pg/ml, migraine SP: 158 ± 118 pg/ml, p = 0.262, Figure 2(d)).

Plasma pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38), vasoactive intestinal peptide (VIP), calcitonin gene-related peptide (CGRP) and substance P (SP) levels in the migraine and healthy control groups. Each box represents the median ± SD, minimum and maximum values of the results. Plasma levels of these peptides between migraine (ictal and interictal, n = 38) and control (n = 20) groups are compared by independent samples t-test or Mann-Whitney U test.
Plasma PACAP-38, VIP, CGRP and SP levels in healthy controls, and in the ictal and interictal periods of migraine patients
Since the variables of PACAP-38 and VIP levels in blood samples from the control (n = 20), and from migraine patients in the ictal period (n = 16) and in the interictal period (n = 22) exhibited normal distribution, these were compared using one-way ANOVA followed by the post-hoc Bonferroni test. PACAP-38 levels in both the ictal period (1638 ± 738 pg/ml) and the interictal period (1929 ± 706 pg/ml) were significantly higher than in the control group (830 ± 275 pg/ml, p = 0.001 for ictal vs. control, and p = 0.0001 for interictal vs. control, Figure 3(a)). However, PACAP-38 levels in the ictal period were not significantly different from those in the interictal period (p = 0.317, Figure 3(a)). Similarly to PACAP-38, VIP levels in both the ictal period (173 ± 71 pg/ml) and the interictal period (218 ± 74 pg/ml) were significantly higher than in the control group (92 ± 38 pg/ml, p = 0.001 for ictal vs. control, p = 0.0001 for interictal vs. control, Figure 3(b)). Moreover, VIP levels in the ictal period also did not differ significantly from those in the interictal period (p = 0.087, Figure 3(b)).

Plasma pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38), vasoactive intestinal peptide (VIP), calcitonin gene related peptide (CGRP) and substance P (SP) levels in the migraine and healthy control groups. Each box represents the median ± SD, minimum and maximum values of the results. Plasma levels of these peptides during attack (ictal, n = 16) or between attacks (interictal, n = 22) in the migraineurs and control (n = 20) groups are compared by one-way ANOVA or Kruskal-Wallis H test. p-values were analysed by post-hoc Bonferroni test.
In contrast, since the variables of CGRP and SP levels in blood samples from the control group (n = 20), and from migraine patients in the ictal period (n = 16) and in the interictal period (n = 22) did not exhibit normal distribution, these were compared using the Kruskal-Wallis H test. No significant difference was determined among the groups in terms of plasma CGRP (control: 205 ± 41 pg/ml, ictal period: 200 ± 59 pg/ml, and interictal period: 224 ± 59 pg/ml, p = 0.33, Figure 3(c)) or SP levels (control: 139 ± 128 pg/ml, ictal period: 130 ± 92 pg/ml, and interictal period: 178 ± 132 pg/ml, p = 0.277, Figure 3(d)).
Correlation between plasma PACAP-38 levels and attack frequency in migraine patients in the interictal period
Negative correlation was detected between attack frequency per month and plasma PACAP-38 levels of migraineurs in the interictal period (n = 22; Pearson correlation, p = 0.017, r = −0.503) (Figure 4). However, PACAP-38 did not correlate with age, gender or duration of disease (p > 0.05). Likewise, plasma PACAP-38 levels of migraineurs in the ictal period, and plasma levels of the other peptides in migraineurs in both the ictal and interictal periods did not correlate with age, gender, attack frequency or duration of disease (p > 0.05).
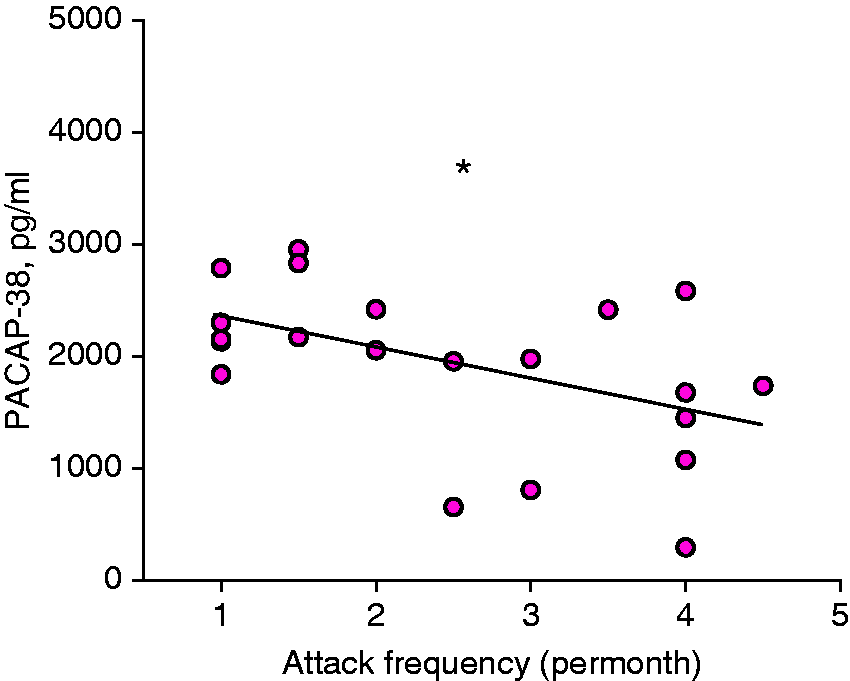
Plasma pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38) level depends on the frequency of migraine attacks in the group of migraineurs in interictal period (n = 22). A negative correlation was determined on the graph (Pearson correlation, r = −0.503, p = 0.017).
Discussion
Plasma PACAP-38 and VIP levels both in the ictal and interictal periods in children with migraine were higher than those in healthy children in this study. In contrast, CGRP and SP levels were unchanged in both the ictal and interictal periods compared to the healthy controls.
Although, with a few exceptions, the great majority of previous studies have reported increased plasma CGRP levels in adult migraineurs (22–24), reports concerning pediatric migraineurs are much more limited. One study reported higher plasma CGRP levels in children with migraine than in both non-headache patients and children with non-migraine headache aged 4–18 years (20). Another study reported elevated CGRP levels in plasma during attacks in young migraineurs with and without aura compared to the interictal period (19). In addition, higher plasma CGRP levels either during or between attacks have been reported in pediatric migraineurs than in both non-migraine headache patients and non-headache controls (18). However, we were unable to confirm those studies in terms of plasma CGRP fluctuations and observed no change in plasma CGRP levels during or between attacks in pediatric migraine patients compared to age-matched healthy controls. No significant difference in terms of plasma CGRP levels was also determined between migraine patients in the ictal period and the interictal period. Our CGRP findings are also not consistent with previous studies reporting increased CGRP levels in adult migraineurs. One possible reason for this may be different pathological mechanisms between adult and pediatric migraineurs. Further studies including pediatric migraineurs with and without aura are now clearly required to establish the exact pathophysiological mechanisms involved in these differences in plasma CGRP levels.
According to the neurogenic inflammation theory of migraine, SP has also been linked to dural neurogenic inflammation. SP has been reported to evoke plasma protein extravasation (1,3) and mast cell degranulation (5) in the cranial dura mater during activation of the trigeminovascular system, thus contributing to neurogenic inflammation. Despite this relevance to dural neurogenic inflammation, a recent systematic meta-analysis concluded that SP blockers failed to inhibit acute migraine pain in clinical trials (24).
However, previous studies have reported that plasma SP levels generally remain unaltered in adult and young migraine patients (19,22,24). One of those studies reported unaltered plasma SP levels in juvenile migraineurs (19), the others reported no change in adult migraineurs. We therefore set out to compare plasma SP levels in both the ictal and interictal periods with those in non-migraine control children. We found that plasma SP levels in juvenile migraineurs in both the ictal and interictal periods did not differ significantly from those in healthy controls, nor from each other. Our plasma SP findings are consistent with those previous studies. In addition, unchanged plasma SP levels in previous studies and in the current research may explain why SP inhibitors were ineffective in clinical trials (24). However, to the best of our knowledge, only one study in the literature has reported increased plasma SP levels in the interictal period in adult migraineurs both with and without aura compared to non-headache controls (23). However, our SP findings are of particular importance in terms of furthering our understanding of plasma SP levels in pediatric, rather than adult migraine. To the best of our knowledge, this is only the second study to investigate plasma SP levels in children with migraine.
Considering our CGRP and SP results together, plasma levels of both remained unchanged in both ictal and interictal migraineurs. Blood was collected from the ictal migraineurs at any time during the first 8 h of the headache episode. This is a limiting factor for our study, because these peptides may have been degraded during this time due to their short half-life in the peripheral blood circulation. The short half-life of CGRP and SP may also account for their unchanged plasma levels in the current study. Studies involving blood collected in less than the first 8 h of the headache episode are therefore also needed.
In contrast, we observed elevated plasma PACAP-38 and VIP levels in both the ictal and interictal periods in children with migraine compared to healthy controls.
PACAP-38 is thought to play a key role in the pathophysiology of migraine. One preclinical study demonstrated that PACAP-38 led to delayed activation and sensitization of central trigeminovascular neurons via the PAC1 receptor (25). Reduced c-fos expression in the trigeminal nucleus caudalis and trigeminal ganglion, together with diminished meningeal blood flow following nitroglycerin treatment have also been shown in PACAP-knockout mice compared with wild-type controls (26). Additionally, a clinical study showed that PACAP-38 infusion led to headache in healthy volunteers (27) and migraine-like attacks in migraineurs without aura (8).
We also observed a negative correlation between interictal plasma PACAP-38 levels and attack frequency. This finding is in agreement with a previous study reporting negative correlation between the interictal plasma PACAP-38 levels and disease duration in adult migraineurs (28). Such a negative correlation might be interpreted indirectly in terms of brain energy deficit stemming from elevated lactate levels and imbalanced Mg2+ concentration in the neurons (28–30).
The trigeminal sensory nerves containing CGRP, SP and PACAP-38 innervate the cranial vasculature, in particular the intracranial dural vasculature and large cerebral arteries. The cranial meninges are regarded as the origin site of migraine pain due to dural neurogenic inflammation and headache involving the release of such neuropeptides as CGRP, SP and PACAP-38 (2,7). Both PACAP-38 and VIP are potent vasodilators (8,14) capable of degranulating the cranial dural mast cells (10).
Mast cells contain neurosensitizing, vasoactive and pro-inflammatory mediators such as CGRP, SP, histamine, interleukin (IL)-1β, IL-6, tumor necrosis factor, prostaglandin D2, bradykinin and nitric oxide (6). When these cells are activated by PACAP-38 or VIP, they may encourage dural neurogenic inflammation through the release of such mediators into the resident environment; in other words, the cranial meninges. One preclinical study showed that dural mast cell degranulation by the basic mast cell secretagogue compound 48/80 triggered nociceptive firing in trigeminal afferent nerve terminals innervating the meninges (31).
However, clinical studies investigating VIP levels in migraine patients have yielded evidence of the potential relevance of VIP to the pathophysiology of migraine. Higher VIP levels have been reported in peripheral venous blood in the interictal period of chronic migraine than in control subjects (32). Another study also reported higher plasma VIP levels in adult women with chronic migraine compared to healthy women (33). Our plasma VIP findings are consistent with these previous reports, although our results were observed in pediatric migraineurs. Another clinical study recently demonstrated that more prolonged VIP-mediated vasodilation provokes more headache than shorter vasodilation (34). Considered together, PACAP-38 and VIP may contribute to migraine pain through both vasodilation and dural mast cell degranulation.
PACAP-38 is a member of the VIP/secretin/glucagon neuropeptide family, and exhibits close structural and functional similarities to VIP. Both are capable of activating the VPAC1 and the VPAC2 receptors with approximately equal affinity (11,35). The concomitant increases in plasma PACAP-38 and VIP levels in the present study is consistent with their such structural and functional similarities. To the best of our knowledge, the present study is the first to show an increase in plasma PACAP-38 and VIP levels both during and between attacks in pediatric migraineurs without aura. Our plasma PACAP-38 and VIP findings may lead to a new horizon in terms of the diagnostic value of these peptides in pediatric migraine.
Limitations
Our study has a number of limitations. These include the low number of volunteers in the patient and control groups, and the fact that the patient group consisted only of migraineurs without aura, migraineurs with aura and chronic migraineurs being excluded. In addition, blood was not collected from a central venous vein such as the jugular vein, since this is problematic in pediatric patients.
Article highlights
Plasma PACAP-38 and VIP levels are increased in pediatric migraineurs without aura. Plasma PACAP-38 and VIP levels are increased in both the ictal and interictal periods in pediatric migraineurs without aura. Plasma CGRP and SP levels do not change in pediatric migraineurs without aura. Plasma CGRP and SP levels do not change in the ictal and interictal periods in pediatric migraineurs without aura.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Bolu Abant Izzet Baysal University Scientific Research Fund (Grant number 2019.08.23.1425).
