Abstract
Background
Recent studies on migraineurs and our own animal experiments have revealed that pituitary adenylate cyclase-activating polypeptide-38 (PACAP-38) has an important role in activation of the trigeminovascular system. The aim of this study was to determine the PACAP-38-like immunoreactivity (LI) in the plasma of healthy subjects, and parallel with the calcitonin gene-related peptide (CGRP)-LI in migraine patients in the ictal and interictal periods.
Methods
A total of 87 migraineurs and 40 healthy control volunteers were enrolled in the examination. Blood samples were collected from the cubital veins in both periods in 21 patients, and in either the ictal or the interictal period in the remaining 66 patients, and were analysed by radioimmunoassay.
Results
A significantly lower PACAP-38-LI was measured in the interictal plasma of the migraineurs as compared with the healthy control group (p < 0.011). In contrast, elevated peptide levels were detected in the ictal period relative to the attack-free period in the 21 migraineurs (pPACAP-38 < 0.001; pCGRP < 0.035) and PACAP-38-LI in the overall population of migraineurs (p < 0.009). A negative correlation was observed between the interictal PACAP-38-LI and the disease duration.
Conclusion
This is the first study that has provided evidence of a clear association between migraine phases (ictal and interictal) and plasma PACAP-38-LI alterations.
Introduction
It is possible to analyse the mechanisms of pain in animal models, but there are no really clinically relevant systems to appropriately mimic the human specificity of headache diseases. Human investigations are therefore particularly important to identify the key mediators responsible for the development and progression of migraine.
Several hypotheses have been proposed to explain the pathological mechanisms of migraine. One of the most widely accepted theories relates to activation of the trigeminovascular system (TS), which involves vascular and neuronal elements (1). The TS consists of the primary sensory pseudounipolar neurons in the trigeminal ganglion (TRIG), its terminals and the meningeal vasculature. The peripheral afferents innervate the supratentorial dura mater, the dural vasculature and the large and small pial arteries on the surface of the brain. The central fibres project to the area of the second-order neurons in the brainstem, the caudal trigeminal nucleus (TNC). The third-order neurons are located in the thalamus. The activation mechanism involves the release of neuropeptides from the terminals (e.g. calcitonin gene-related peptide (CGRP), vasoactive intestinal peptide (VIP) and substance P (SP) (2), and functional changes such as neurogenic inflammation (protein extravasation and vasodilation), mast cell degranulation and sensitisation (3).
Among the numerous “migraine-related” processes and substances, the vasoactive pituitary adenylate cyclase-activating polypeptide (PACAP) has been potentially implicated in the pathophysiology of migraine (4,5). PACAP belongs to the VIP/secretin/glucagon neuropeptide superfamily, and is considered to be a “brain-gut peptide” by virtue of its widespread expression and functions in the human body. PACAP exists in two forms: PACAP-27 and PACAP-38; the latter is expressed predominantly in mammals. PACAP acts at one specific (PAC1) and two non-specific receptors, which are shared between PACAP and VIP (VPAC1 and VPAC2). This can be explained by the 68% structural homology between PACAP and VIP (6–8).
This implies that PACAP-38 also has a vasodilating effect (9), and a broad range of data suggest that it is an integrator of nociceptive and sensitisation processes, besides being involved in neurogenic inflammation (10–13).
PACAP is presented in the primary sensory neurons of the TRIG (14), the parasympathetic otic and the sphenopalatine ganglia (15,16). Moreover, PACAP-38 is found in the cell bodies and nerve fibres of the human TNC and the upper regions of the cervical spinal cord, which suggests that PACAP may be closely related to the TS (2,17). We earlier furnished evidence for this hypothesis with animal experimental results. PACAP-deficient mice displayed reduced light-aversive behaviour (photophobia), while decreased meningeal blood flow and c-fos expression in the TRIG and TNC were detected relative to wild-type mice after nitroglycerol (NTG)-induced TS activation (18). Further, elevated levels of PACAP-27 and PACAP-38 were measured in the plasma and the TNC 90 and 180 minutes after the respective stimulation in two animal models of TS activation (19). These results suggest that PACAP may be an important mediator, and therefore a diagnostic marker of TS activation.
The available human data point to the involvement of PACAP in the mechanisms of migraine. Schytz et al. demonstrated that the intravenous administration of PACAP-38 causes headache in healthy subjects, and migraine-like attacks in migraine patients without aura, six hours on average after starting the infusion (4). However, no clinical data are available on endogenous alterations in PACAP levels in relation to migraine.
Based on the literature, we have hypothesised that the plasma concentration of PACAP-38 increases during migraine attacks. Our aims were therefore to reveal the potential relationships between the PACAP-38 levels of human plasma and the presence of migraine headache. Radioimmunoassay (RIA) measurements were carried out on peripheral blood plasma samples in order to show alterations of PACAP-38-like immunoreactivity (PACAP-38-LI) in migraine patients during the ictal and interictal periods in comparison with healthy control subjects. In addition, the clinical features of the disease, as well as plasma CGRP-like immunoreactivity (CGRP-LI) were also compared to the PACAP-38-LI to explore possible correlations.
Materials and methods
Participants
Mean data on healthy control volunteers and 87 migraineurs (age, gender, type of migraine (with or without aura), disease duration (years), attack frequency, allodynia and VAS score are shown.

MA: migraine with aura; MO: migraine without aura; allodynia: 0: absence of allodynia; 1: mild allodynia; 2: moderate allodynia; 3: serious allodynia; VAS: visual analogue score.
A detailed questionnaire was used to compile a homogeneous group of migraineurs as concerns the features of their migraine disease: the duration of the migraine, the attack frequency, allodynia (21) and the severity of pain during attacks as measured on a visual analogue scale (VAS). The relation of migraine attacks to the menstrual cycle (22) and to the presence of other non-migraine, chronic pain disorders (lumbago, low-back pain, knee- and hip-joint arthrosis) was also assessed. Depression was not clinically diagnosed in any of the cases. Healthy volunteers serving as controls were screened for non-reported/non-treated headaches. Subjects (patients and controls) who displayed any significant and serious non-migraine chronic disorders were excluded from the study.
Study design and procedures
Demographic data on 21 migraineurs, whose samples from both the ictal and the interictal periods were analysed. The age, gender, type of migraine (with or without aura), disease duration (years), attack frequency, allodynia, VAS score, PACAP-38-LI in plasma samples (fmol/ml) originating from interictal and ictal phases, the time of the previous attack before interictal blood sampling (days) and the duration of the present headache (hours) are shown for each patient.

MA: migraine with aura; MO: migraine without aura; allodynia: 0: absence of allodynia; 1: mild allodynia; 2: moderate allodynia; 3: serious allodynia; VAS: visual analogue score; PACAP-38-LI: pituitary adenylate cyclase-activating polypeptide-38 like immunoreactivity.
RIA measurements and data acquisition
Plasma concentrations of PACAP-38 were determined with specific and sensitive RIA techniques developed earlier in our laboratory (23). The PACAP-38 antiserum 88111-3 was raised in rabbits with synthetic peptides conjugated to bovine serum albumin (BSA) or thyroglobulin with glutaraldehyde or carbodiimide. The high specificity and C-terminal sensitivity of this antibody were confirmed by cross-reactivity studies, and no cross-reactivity was found with PACAP-27 or with other related neuropeptides in either case. Following centrifugation of the plasma samples (2000 rpm at 4°C for 10 minutes), the peptide was extracted from the plasma into three volumes of absolute alcohol.
After precipitation and a second centrifugation (2000 rpm at 4°C for 10 minutes), the samples were dried under a nitrogen flow and resuspended in 300 µl of assay buffer before RIA determination in order to achieve a 10 times higher concentration for the RIA procedure (10,23). The tracers were mono-125I-labelled peptides prepared in our laboratory. Synthetic peptides were used as RIA standards in concentrations ranging from 0 to 1000 fmol/ml. The assay was prepared in 1 ml 0.05 M (pH 7.4) phosphate buffer containing 0.1 M sodium chloride, 0.25% (w/v) BSA and 0.05% (w/v) sodium azide. The antiserum (100 µl, 1:10,000 dilution), the RIA tracer (100 µl, 5000 cpm/tube) and the standard or unknown samples (100 µl) were measured into polypropylene tubes with the assay buffer. After incubation for 48–72 hours at 4°C, the antibody-bound peptide was separated from the free peptide by the addition of 100 µl of separating solution (10 g charcoal, 1 g dextran and 0.5 g commercial fat-free milk powder in 100 ml of distilled water). Following centrifugation (3000 rpm at 4°C for 15 minutes), the contents of the tubes were gently decanted and the radioactivity of the precipitates was measured in a gamma counter (Gamma, type NZ310). The PACAP-38 concentrations of the unknown samples were read from calibration curves.
Statistical analysis
Data are expressed as median ± SD if not stated otherwise. The normality of the data was tested with the Shapiro-Wilk test. Group comparisons were carried out with Student's unpaired, paired t tests and the Wilcoxon-test with SPSS 17.0. Data were analysed with a multivariate test (repeated-measure analysis of variance (ANOVA)) in the case of menstruation cycle and chronic pain condition related to PACAP-38 level. Statistical significance was accepted at p < 0.05.
Results
Differences in plasma PACAP-38-LI between migraineurs and healthy controls
As concerns the total of 87 migraine patients (n = 87), a significantly lower PACAP-38-LI was determined in the interictal plasma of the migraineurs (n = 80; 24.60 ± 3.59 fmol/ml) than in that of the healthy volunteers (n = 40; 26.54 ± 4.43 fmol/ml; Student's unpaired t test, p < 0.011, t = 2.578) (Figure 1(a)). However, the plasma samples from the patients during their migraine attacks (n = 28) exhibited a significantly higher PACAP-38 concentration (27.39 ± 4.67 fmol/ml) as compared with the interictal samples (n = 59; 24.91 ± 3.73 fmol/ml; Student's unpaired t test, p < 0.009, t = −2.676) (the interictal data of those 21 migraineurs whose plasma samples were collected from both periods were excluded from this analysis to avoid the statistical problems caused by the paired samples) (Figure 1(b)). No difference was found when the ictal samples were compared with those of the controls (Student's t test for unpaired comparisons, p < 0.447, t = −0.765) (Figure 1(c)).
PACAP-38-LI (fmol/ml) was determined by RIA in the plasma of migraineur groups in comparison with those of healthy volunteers. Boxes indicate PACAP-38-LI (median ± SD, minimum and maximum values) of healthy control subjects (n = 40), and of migraineurs (n = 87) during the interictal (n = 80) and ictal periods (n = 28). Significant PACAP-38-LI decrease was observed between the control vs. the interictal group with Student's unpaired t test, p < 0.011 (a). To except the interictal data of the paired samples, the interictal (n = 59) vs. the ictal (n = 28) group comparison showed significantly higher PACAP-38-LI during migraine attacks with Student's unpaired t test, p < 0.009 (b). There were no significant PACAP-38-LI differences between the control and the ictal group with Student's t test for unpaired comparisons, p < 0.447 (c).
Association between plasma PACAP-38-LI and duration of migraine
A negative correlation was found between the duration of the migraine and the interictal PACAP-38-LI (n = 87; linear regression, p < 0.044, R = 0.231) (Figure 2). Plasma PACAP-38-LI did not correlate with age, attack frequency, allodynia and VAS score (ANOVA, linear regression, p > 0.05) or differences were not found regarding gender, hormonal changes and pain (Student's unpaired t test, p > 0.05).
Interictal plasma PACAP-38-LI (fmol/ml) depends on the duration of the migraine disorder in the group of migraineurs (n = 87). A negative correlation was observed with linear regression on the graph, p < 0.044.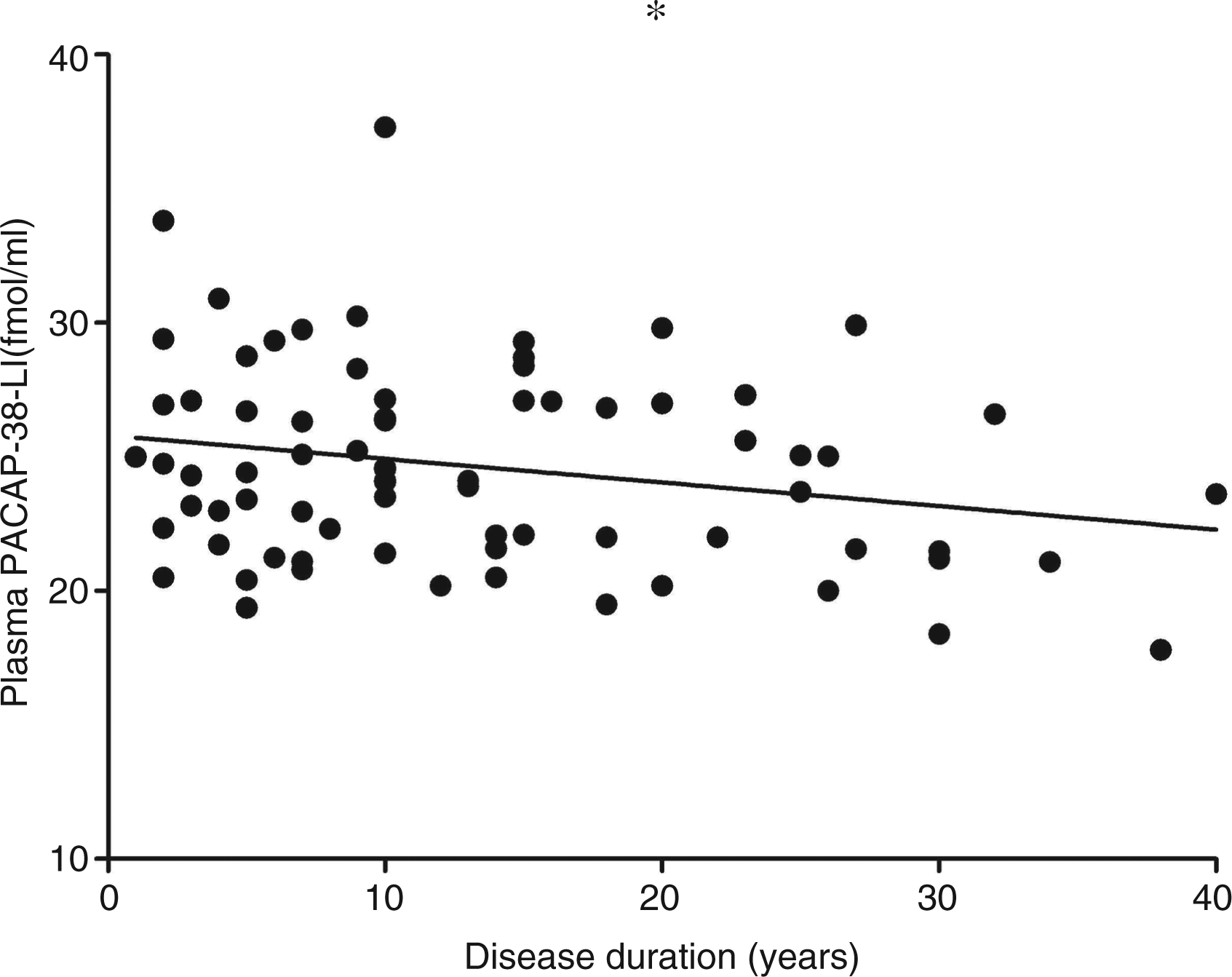
Changes in plasma PACAP-38-LI and CGRP-LI in 21 migraineurs
To gain insight into the changes of neuropeptide levels, we measured the plasma concentrations of PACAP-38 and CGRP in the same subject during a headache attack and interictally. The plasma PACAP-38-LI was significantly higher in the ictal period (28.04 ± 5.00 fmol/ml) than in the interictal period (23.74 ± 3.09 fmol/ml) (n = 21; Student's paired t test, p < 0.001, t = −4.134) (Figure 3(a)).
PACAP-38-LI (fmol/ml) was determined by RIA on plasma samples from migraineurs in both interictal and ictal periods. (a). Plots of individual data for each patient (n = 21). A significant difference was observed between the levels in the two phases with Student's paired t test, p < 0.001. CGRP-LI (fmol/ml) was determined by RIA on plasma samples from migraineurs in both interictal and ictal periods. (b). Plots of individual data for each patient (n = 18). A significant difference was observed between the levels in the two phases with Wilcoxon-test, p < 0.035. The thick grey lines represent the mean values.
The CGRP-LI was determined simultaneously with the PACAP-38-LI in both phases in 18 migraineurs. Significantly higher CGRP levels were observed in the plasma samples during the ictal period (53.74 ± 31.52 fmol/ml) as compared with the interictal period (39.74 ± 27.49 fmol/ml) (n = 18; Wilcoxon-test, p < 0.035) (Figure 3(b)).
Associations of changes in plasma PACAP-38-LI with menstruation cycle sensitivity and chronic pain conditions in 21 migraineurs
Changes in the plasma PACAP-38-LI proved to be influenced by two parameters: There was a significant PACAP-38-LI elevation in the ictal phase (31.01 ± 3.32) compared to the interictal phase (24.18 ± 2.52) in patients whose migraine headache was not sensitive to the menstruation cycle (Group 1: n = 11; Student's paired t test, p < 0.00002, t = −7.250). Meanwhile, there was no such significant increase during the ictal phase (24.78 ± 4.56) compared to the interictal phase (23.26 ± 3.70) in patients whose migraine headache was sensitive to the menstruation cycle (Group 2: n = 10; Student's paired t test, p < 0.344, t = −0.998) (Figure 4). A similar PACAP-38-LI increase was detected in the ictal phase (30.01 ± 3.69) vs. the interictal phase (24.15 ± 2.47) of patients, who did not have chronic pain-related conditions (Group 1: n = 15; Student's paired t test, p < 0.00005, t = −5.716). However, there was no difference in the ictal phase (23.13 ± 4.64) compared to the interictal phase (22.71 ± 4.40) in patients, who had other non-migraine, chronic pain disorders (Group 2: n = six; Student's paired t test, p < 0.833, t = −0.222) (Figure 5).
Plasma PACAP-38-LI (fmol/ml) in the interictal and ictal periods of two subpopulations of 21 migraineurs characterised on the basis of menstruation cycle dependence. Group 1: menstruation cycle-independent migraineurs (n = 11); group 2: migraine patients whose headache was sensitive to their menstruation (n = 10). Each box represents the median ± SD, minimum and maximum values of the results. A significant difference in PACAP-38-LI was observed between the interictal and ictal phases in group 1 with Student's paired t test, p < 0.00002. Plasma PACAP-38-LI (fmol/ml) in the interictal and ictal periods of two subpopulations of 21 migraineurs characterised on the basis of chronic pain conditions. Group 1: patients without chronic low back pain (n = 15); group 2: patients with simultaneous low back pain (n = 6). Each box represents the median ± SD, minimum and maximum values of the results. A significant difference in PACAP-38-LI was observed between the interictal and ictal phases in group 1 with Student's paired t test, p < 0.00005.

To reveal the relationship between ictal-interictal PACAP-38 levels and menstruation and chronic pain, a repeated-measure ANOVA was used. A multivariate test showed a significant main effect of PACAP-38 measurements in different phases of the disease (F(1,19) = 22.579, p < 0.0001) and a main effect of headache related to menstruation cycle (F(1,19) = 22.257, p < 0.0001). The interaction of the two factors was also significant (F(1,19) = 9.096, p < 0.007). A similar ANOVA was carried out for identifying if ictal/interictal PACAP-38 level changes are influenced by concomitant chronic pain conditions. While significant main effects were identified by analysis (ictal/interictal: F(1,19) = 11.392, p < 0.003, chronic pain condition: F(1,19) = 0.553, p < 0.446), the interaction was not significant in this case (p > 0.05).
No correlations were found between the level of peptides and age, gender, attack frequency, allodynia or VAS score.
Discussion
This study provided the first evidence that PACAP-38 concentration in the plasma of migraineurs is significantly lower in the interictal period as compared with that of healthy volunteers, but increases during migraine attacks relative to the attack-free period. These results suggest that PACAP-38 might be an important mediator of the pathophysiology of migraine. The difference in the plasma levels of PACAP-38 in migraineurs in the two periods indicate that this peptide is involved in the development of the attacks.
Effects of PACAP-38 related to migraine
PACAP-38 can be released from both the central and the peripheral terminals of the pseudounipolar neurons of the TRIG (24) in response to specific triggers. It enters the systemic circulation, and binds to its receptors (PAC1, VPAC1 and VPAC2) on the meningeal vessels and second-order sensory neurons. Zagami and colleagues published (25) that the stimulation of the superior sagittal sinus causes extracranial release of PACAP. PACAP is known to induce neurogenic inflammation, mast cell degranulation, neuronal activation and sensitisation (4,26). These effects can contribute to the development of severe headache and possibly to the aggravation of the attacks.
A magnetic resonance imaging (MRI) angiographic study revealed that PACAP-38 infusion-induced headache is related to significant dilation of the middle meningeal arteries (MMAs), in contrast with the middle cerebral arteries (27). The intracarotid infusion of PACAP-38 produces significant dilation of the dural artery in the rat, and this administration route has proved to be more effective than the intravenous mode (28). PACAP-38 also exerts potent vasodilating action on pressurised MMAs, which can be influenced by a PAC1 receptor antagonist. Multiple variants of the PAC1 receptor have been found in addition to the VPAC2 receptors in rat MMAs (29).
PACAP and VIP receptors in the human meningeal and coronary arteries were recently characterised with pharmacological modulators, and these peptides were found to have lower potency and efficacy in meningeal vasculature than in coronary arteries. This study concluded that processes of PACAP-38-induced migraine-like headache may not involve meningeal vasodilation rather than sensitisation of peripheral and central sensory trigeminal fibres (9). The reported effects of a PACAP infusion regarding neurogenic inflammation and mast cell degranulation in relation to migraine are controversial (26). It was concluded from a study of the mast cell degranulating effects of different truncated PACAP and VIP fragments on rat peritoneal and dural mast cells that phospholipase C-mediated mast cell degranulation is implicated in PACAP-induced migraine (30).
Possible mechanisms related to the elevated PACAP-38-LI during migraine attacks
There are some anatomical and physiological factors which might be involved in the migraine-like headache-inducing effect of PACAP-38.
(a) PACAP-38 is a sensory (14), sympathetic (15) and parasympathetic (16) neuropeptide (31), which is released from the nerve endings (2,14,17,32) at the dural or other cranial compartments. It can modulate vessels and nerve fibres through its receptors leading to elevated intracellular cyclic adenosine monophosphate (cAMP) levels (33). There are several lines of evidence that the increased cAMP levels cause sensitisation and activation of the trigeminal neurons (34) and meningeal nociceptors (35,36), therefore inducing delayed headache (37).
The headache-inducing action of PACAP-38 was first described in 2009 by Schytz and colleagues, who demonstrated that PACAP-38 has a simple headache-evoking effect in healthy volunteers but provokes severe migraine-like attacks in susceptible subjects. A decreased mean blood flow velocity in the middle cerebral artery and an increase in diameter of the superficial temporal artery were observed 20 minutes following the infusion (4). It is possible that the PACAP-38 has a positive-feedback, self-amplifying effect, which can contribute to the maintenance and aggravation of headache.
Based on similar features and receptors of PACAP and VIP, as well as the supposed complex mechanism of migraine, it is likely that migraine cannot be related only to PACAP-38. Its causative role is equivocal, but its involvement is unquestioned.
This peptide can contribute to the evolution of migraine attacks in cooperation with other regulatory neuropeptides, molecules and enzymes, like VIP, CGRP, 5-HT, SP and nitric oxide (NO). When the headache starts in response to specific triggers (vasodilation, plasma protein extravasation, mast cell degranulation, neuropeptide release, neurogenic inflammation), the presence and aggravation of pain may lead to the release of PACAP-38 from the nerve terminals as a self-triggering process. This can be an explanation for the finding that the increase of plasma PACAP-38-LI in the ictal phases of migraineurs compared to the interictal one is moderate but significant.
(b) Numerous studies have approached the effects of PACAP-38 involved in headache from vasodilating aspects. There are controversial results regarding the relaxing actions of PACAP-38 on meningeal or cerebral arteries (9,29,38,39). The vasodilating properties to a small extent or indirectly are likely to contribute to the development of headache, e. g. PACAP induces vascular effects mediated via activation of perivascular nerves (40). The vascular effects cannot be excluded, but based on the literature it seems that these may be less relevant in migraine (9). PACAP-38 is more potent in sensitising trigeminal peripheral sensory fibres directly and also through mast cell degranulation (4,26,30). These mechanisms can be related to the phenomenon of throbbing headache, which can correlate with the increased PACAP-38-LI during the ictal phase of migraineurs found in our study. Subsequently, it would be plausible that the activation of the second-order trigeminal neurons by PACAP-38 can result in a direct central sensitisation (4).
Complementary mechanisms behind reduced PACAP-38-LI during interictal phase of migraine patients
The lower concentration of PACAP-38 observed in the interictal period, and especially the correlation of the interictal plasma PACAP-38-LI with disease duration, might be explained indirectly in terms of brain energy deficit (an impairment of the cerebral and striated muscle energy metabolism (41,42), elevated lactate levels (43,44), abnormalities of mitochondrial compartments (45) and imbalanced Mg2+ concentration in the neurons (46,47)).
It may additionally be hypothesised that the low interictal peptide concentration might be associated with degenerative changes affecting the PACAP-releasing circuitries (cortical atrophy (48), iron deposition in the periaqueductal grey matter (49) and increased levels of markers indicative of neuronal and glial damage (50)).
The possible role of the blood-brain barrier (BBB)
The ability of PACAP to pass through the intact BBB is limited; it needs specific transporters in specific brain areas (51,52). However, the enhanced BBB permeability in migraine may allow PACAP to penetrate into the cerebrospinal fluid and exert its central effects. From the opposite aspect, the PACAP released in the brain (6) can also penetrate through the BBB, and hence may be detected in the plasma. There are assumptions that the integrity of the BBB is disrupted in migraineurs (53,54). A transient brain dysfunction, vasogenic cerebral oedema and a damaged BBB can develop in lipopolysaccharide-induced brain injury in rats in response to the over-expression of matrix metalloproteinase 9 (55). This enzyme can degrade the basal membrane, resulting in structural impairment of the BBB, altered plasma levels of which have been observed in patients with migraine (56–59).
Correlation of PACAP-38-LI with the menstruation cycle and other chronic pain conditions in migraineurs
Migraine attacks are often closely related to female hormonal changes (22). Hence, it is interesting that PACAP-38-LI increases significantly during the ictal period only in those women whose headache is not related to the menstrual cycle. Although the influence of the menstruation cycle in the PACAP-38-LI changes cannot be excluded, there is evidence that the plasma concentration of PACAP-38 is relatively stable and independent of gender, age, food intake or female hormonal cycles in healthy subjects (60).
It is well established that PACAP-38 plays an important role in a variety of other pain conditions (61–63). In opposition, there was no increased PACAP-38-LI in the ictal phase in patients with other chronic pain conditions. Moreover, we did not find any statistical proof of the influence of the chronic pain and alterations of the PACAP-38 level.
Possible relations between the PACAP-38 and CGRP in migraineurs
More than 20 years ago, the release of CGRP was demonstrated in the extracerebral circulation of humans and cats in response to TS activation (64,65). CGRP-containing neurons have been detected in the human TRIG (14,66), and elevated plasma CGRP levels have been described in migraine (67,68) and other types of primary headache (69). Moreover, an intravenous infusion of human alpha-CGRP causes migraine headache (37,70), and there is a significant positive correlation between plasma levels of CGRP and headache severity scores in NTG-induced migraine attacks (71). However, one controversial study questions the importance of CGRP in migraine (72). In fact, CGRP-receptor antagonists are effective in the treatment of migraine disorder (73), but their side-effect profile, with special emphasis on hepatotoxicity, currently makes this drug development direction problematic (74–76). Our results draw attention to a possible influence between the PACAP and CGRP systems in migraine pathogenesis.
Conclusions
It is concluded that PACAP-38 might be implicated in the development of migraine headache. There is association between migraine periods (ictal and interictal) and alterations in PACAP-38 levels in the plasma. The quantitative changes in this peptide in the plasma are related to the disease duration, the menstruation cycle and the presence of other pain-related disorders. The causative role of PACAP-38 in migraine headache demands further studies. Our results indicate the need for investigations of the role of plasma PACAP-38 as a putative biomarker of migraine, which might provide new perspectives as concerns the identification of a new target in the therapy of migraine.
Clinical implications
The PACAP-38 has an important role in the mechanisms of migraine attack. A significantly lower PACAP-38 concentration in the interictal plasma of migraineurs as compared with healthy controls is associated with the duration of migraine disease. The elevated plasma PACAP-38 level in the ictal phase relative to the attack-free period is attributable to the releasing peptide from the PACAP-containing neurons, which can contribute to the development and/or the aggravation of migraine attacks.
Footnotes
Funding
This work was supported by Hungarian grants: Developing Competitiveness of Universities in the South Transdanubian Region (SROP-4.2.1.B-10/2/KONV-2010-0002, SROP-4.2.2.A-11/1/KONV-2012-0024 and SROP-4.2.2/B-10/1-2010-0029), the Neuroscience Research Group of the Hungarian Academy of Sciences and the University of Szeged, SROP-4.2.1.B-09/1/KONV-2010-0005, K-2012-TÁMOP-4.2.2.A-11/1/KONV.-0443534/130, OTKA (PD 104715) and FNUSA-ICRC (CZ.1.05/1.1.00/02.0123). B. Tuka is a Gedeon Richter Plc.-sponsored PhD candidate. Dr Kincses was supported by the Bolyai Scholarship Programme of the Hungarian Academy of Sciences.
Conflict of interest
None declared.
