Abstract
Background
The age of onset of cluster headache (CH) attacks most commonly is between 20 and 40 years old, although CH has been reported in all age groups. There is increasing evidence of CH with early or late onset and a different course of the disorder. The aim of the study was to analyze the influence of the age of onset on clinical features, disorder course, and therapy effectiveness in CH patients.
Methods
A retrospective and cross-sectional analysis was performed on 182 CH patients divided into three groups according to the age of onset. The first group consisted of patients with the first CH attack before 20 years of age, the second group was patients with age of onset between 20 and 40 years of age, and the third group was patients with age of onset after 40 years of age. Demographic data, features of CH periods and attacks, and the response to standardized treatment were compared among the groups.
Results
Patients with CH onset after 40 years of age reported a lower number of autonomic features and less frequently had conjunctival injection and nasal congestion/rhinorrhea phenomena during their attacks. Diagnostic delay was the longest in the patients with CH onset before 20 years of age.
Conclusion
The influence of the age of onset of CH is intriguing for further studies and could possibly extend the knowledge about CH pathophysiology. From a clinical point of view, the differences in CH presentation are insufficient to preclude a correct diagnosis and treatment because the same criteria could be applied regardless of patient age.
Introduction
According to the criteria provided by the International Headache Society, cluster headache (CH) is a primary headache disorder with attacks of severe or very severe, strictly unilateral, orbital, supraorbital, or temporal pain lasting 15 to 180 minutes and occurring from once every other day to eight times daily (1). The attacks are associated with at least one of the following cranial autonomic features, all of which are ipsilateral to the side of pain: conjunctival injection, lacrimation, nasal congestion, rhinorrhea, forehead or facial sweating and flushing, miosis, ptosis, eyelid edema, and a sensation of fullness in the ear. Autonomic symptoms are absent in about 3% of patients with CH. Unlike migraine, a sense of restlessness or even agitation is present during CH attacks. Two forms of the disorder, episodic and chronic, are recognized, delineated by a remission period lasting at least one month (1). Initial CH attacks most commonly occur in patients between 20 and 40 years old (1), although CH has been reported in all age groups (2,3). There is increasing evidence of the disorder in children, with reported cases in children as young as 2 years old (4). There are also reports of late-onset (LO) CH, with the oldest patient being 81 years old at the onset of CH (5). CH is a primary headache disorder with a clear male preponderance, although a trend toward a decreasing male preponderance was noted during the last few decades (6). Studies investigating female patients indicated two peaks of CH onset, the second one occurring after the age of 50 and characterized by longer CH periods and more frequently taking the chronic form (7).
The aim of this study was to analyze the influence of the age of onset of CH on clinical features, disorder course, and therapy effectiveness.
Patients and methods
This study was performed as a retrospective case series of CH patients diagnosed at the Headache Center, Neurology Clinic, Clinical Center of Serbia, between January 2000 and December 2012. The study was approved by the local ethics committee and was performed in strict accordance with Health Insurance Portability and Accountability Act regulations (Declaration of Helsinki). Informed consent for the use of patient data was obtained from all study subjects.
Our headache center is a tertiary referral center and serves in the diagnosis and treatment of patients with frequent and therapeutically resistant headaches. The patients are referred mostly from general practitioners, neurologists working in general hospitals, and other specialists from the Clinical Center of Serbia. There are four headache centers in Serbia, covering a population of approximately seven million inhabitants. Our headache center was founded in 2000 with 400 new patients per year; this number had increased to 800 by 2012.
Only patients with CH diagnosis established according to the criteria given in the second edition of the International Classification of Headache Disorders (ICHD) (8) were included in the study. Clinical data were collected during standardized face-to-face interviews and immediately stored in the headache database. We excluded 12 CH patients from the study: seven patients with a diagnosis of probable CH and five patients who were unable to recall the exact time of headache onset.
For the purpose of this study, patients were divided into three groups according to the age of CH onset. The first group consisted of patients whose first CH attack occurred before they were 20 years old (early onset, EO); the second group consisted of CH patients with age of onset between 20 and 40 years old (common onset, CO); and the third group was patients with CH age of onset after 40 years old (LO).
The following data were collected in all patients: gender, form of CH, frequency of periods per year, and mean duration of periods, analyzed for CH patients with episodic form, diagnostic delay, features of CH attacks, autonomic features, and treatment efficacy, e.g. effectiveness of high-flow oxygen and prednisone 1 mg per kg during the first five days with subsequent gradual tapering and/or verapamil 240–560 mg daily.
The analyzed features of CH attacks were: daily attack frequency and duration, pain quality, presence of at least one autonomic feature, feeling of restlessness during attack, presence of at least one associated symptom typical for migraine attack (photophobia, phonophobia, nausea or vomiting), presence of other features during attack that are not the part of diagnostic criteria for CH (painful pulse of temporal artery, cough, hemifacial spasm, dry mouth, blurred vision, yawning, exhaustion, mydriasis, rise in blood pressure, or loss of consciousness), feeling of interictal tenderness in the periorbital region, presence of predominantly nocturnal attacks, presence of triggers (alcohol during CH period, certain foods, exercise, exposure to cold/hot external temperature).
The analyzed associated autonomic features were: presence of conjunctival injection, lacrimation, nasal congestion or rhinorrhea, eyelid edema or semiptosis, miosis, and forehead or facial sweating.
The collected data were compared among the three groups. An independent-samples t test was used for parametric data, and chi-square test, Kruskal-Wallis test, or Mann-Whitney U test were used for nonparametric data. The significance level for the analysis was set beforehand at 5% (p < 0.05).
Results
The whole examined group consisted of 182 CH patients, 61 (33.5%) females and 121 (66.5%) males. The mean age at the time of the first medical consultation for CH was 40.1 ± 12.6 years (range 18–81). Diagnostic delay was 7.8 ± 8.0 years in the whole group; 68.7% of patients had a diagnostic delay longer than two years.
EO of the disorder was reported by 29 (15.9%) patients, CO by 104 (57.1%) patients, and LO by 49 (26.9%) patients (Figure 1). The earliest CH onset was at the age of 10, and the latest one was at the age of 81.
Percentages of patients according to age at cluster headache (CH) onset.
Gender and form of CH in patients of various ages at disease onset.

aAnalyzed only for patients with episodic CH. CH: cluster headache; M: male; F: female; SD: standard deviation.
Features of CH attacks in patients of various ages at disease onset.

CH: cluster headache.
Autonomic features of CH attacks in patients of various ages at disease onset.
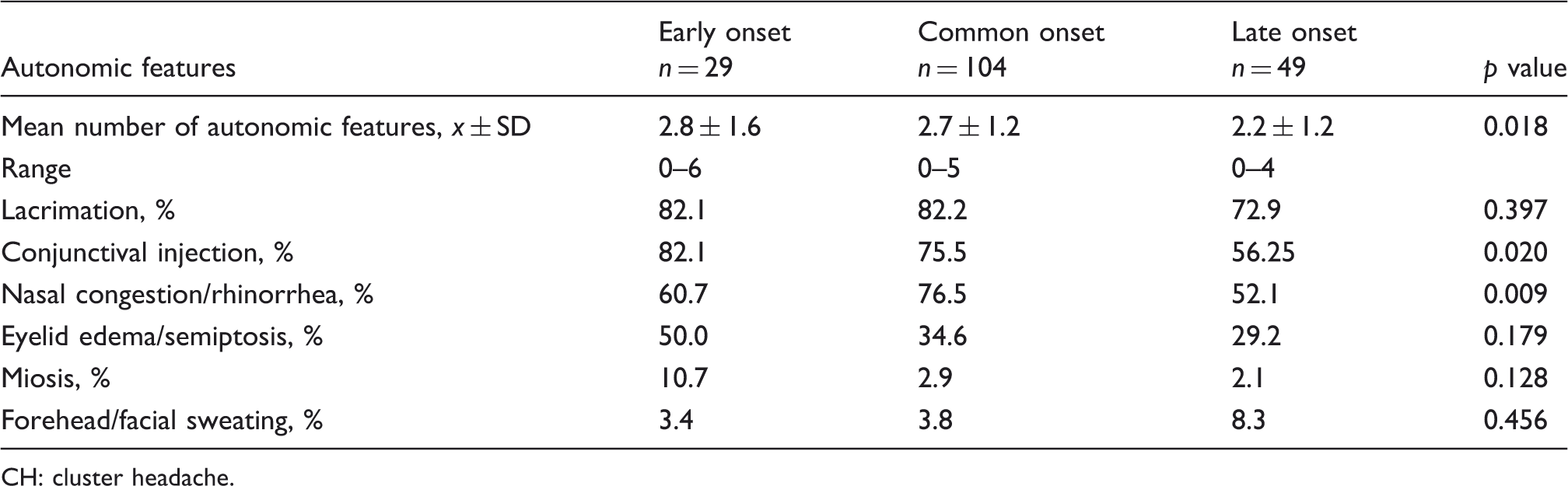
CH: cluster headache.
Diagnostic delay and treatment efficacy for patients of various ages at CH onset.
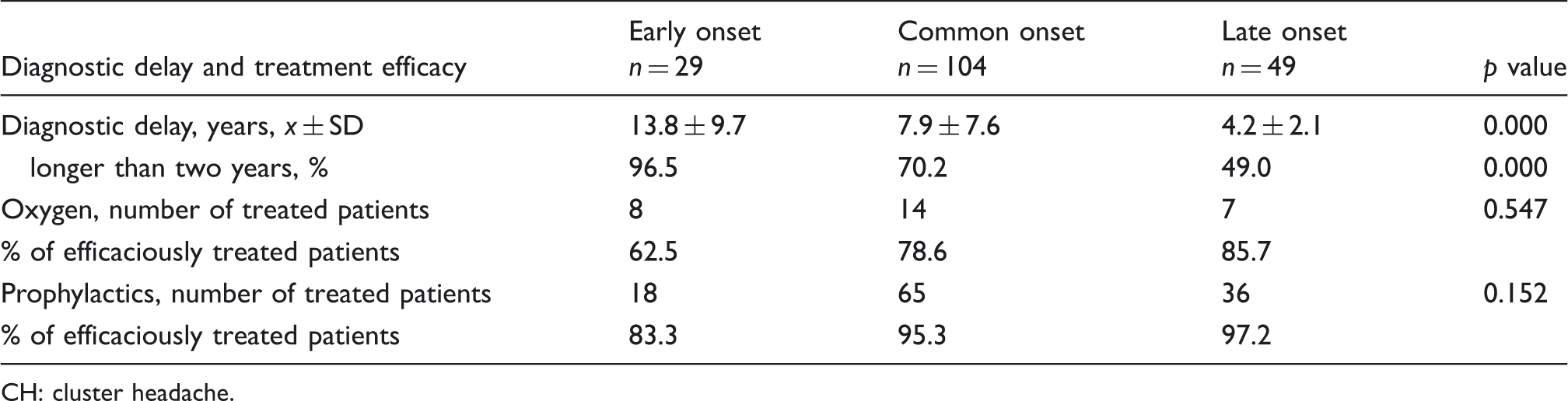
CH: cluster headache.
Discussion
The results of this study, performed on a large cohort of CH patients, demonstrated that the first attack of CH occurred between 20 and 40 years of age in nearly 60% of patients. There are, however, a significant number of patients with disease onset out of these boundaries. More than a quarter of our patients had LO, and a fifth of our patients had EO CH. A significant number of patients with the age of onset outside the common limits have been reported (6). Our youngest patient was 10 years old and the oldest was 81 at the time of disease onset; similar extremes have also been noted by other authors (5,9).
Regardless of the age at CH onset, male preponderance was recorded in all three examined groups. This was particularly evident in the patients with the common age of onset, with the male to female ratio 5:1. In patients with EO and LO CH, the male:female ratio was lower, 2.4:1. The association between age of onset and male:female ratio was investigated in other studies (6,7). Results from the United States Cluster Headache Survey regarding gender differences demonstrated that there were two peaks of CH onset during the female lifetime, the first one occurring at an earlier age than in men and the second after 50 years of age (7). These results correspond well to the results obtained in our study, where these two peaks are expressed by lower M:F ratio in patients with EO and LO than in patients with CO.
The chronic form of CH was present in 11.5% of the examined patients. The presence of the chronic form of CH varies widely in different clinical studies. The lowest came from studies performed in Taiwan (10) and Japan (11), estimating the presence of chronic CH to be as low as 0%–3.5%, and the highest from Germany, where it was estimated to be 31% (12). As in our study, CH patients were recruited from headache centers in all the mentioned studies, and the different prevalence is probably a consequence of different referral modalities in these countries. In a large cohort of CH patients followed over 30 years, Ekbom et al. reported that the influence of the age of onset on the form of CH is related to gender (6). These results were confirmed by the results obtained from another large CH study with similarly long-term follow-up demonstrating that chronic CH was nearly five times more common in females with disease onset after 50 years of age (13). According to the results obtained from our patients, the form of the disorder was not influenced by the age of onset, in spite of the fact that the LO group had twice as many women as men. For the analysis, we applied 20 to 40 years of age as the common age of onset, in accordance with the Headache Classification (1). Several studies investigating LO CH mainly analyzed patients over 50 years of age (13,14). The observation of more often chronic CH and longer active periods of CH led to the conclusion that LO CH represents a negative prognostic factor (13). We could not confirm that observation, because neither the chronic form nor CH duration was different in our LO patients from the other patients.
The most intriguing result obtained by our study was the influence of age of onset on autonomic features. Although the presence of at least one accompanying autonomic feature was similar regardless of age at CH onset, LO patients had a lower number of these features during attacks, and fewer patients had conjunctival injection and nasal congestion/rhinorrhea phenomena, but also evident were all notified autonomic features with the exception of sweating (Table 3). Disease duration did not influence significantly the number of autonomic features. We decided to put together nasal obstruction and rhinorrhea, because patients often describe obstruction of the nostril on the painful side at the beginning and during the attack and clear and fluid nasal discharge when the attack subsides. Similar reasoning led us to combine eyelid edema and semiptosis. Even when observed by a neurologist, as was the case for the majority of our patients, it was hard to differentiate between these two features. According to the results obtained in our study, LO CH is followed by less prominent autonomic symptoms. Autonomic symptoms develop as the result of the activation of cranial parasympathetic fibers through the trigeminal autonomic reflex, functional connection between caudal trigeminal nuclei and superior salivary nuclei (15). Lacrimation, conjunctival injection, and rhinorrhea are signs of parasympathetic activation (16), while miosis, semiptosis, and forehead sweating are due to the dysfunction of sympathetic postganglionic fibers of the internal carotid nerve plexus (17). The role of the hypothalamus, suspected because of the seasonal and diurnal periodicity of CH attacks, is further confirmed by neuroimaging studies and mechanisms of action of deep brain stimulation to be modulatory on the nociceptive and autonomic pathways (18). The action of the hypothalamus is not necessary simultaneously on both pathways, which are shown by the cases of pain without autonomic features or rare cases of autonomic features without pain. Indeed, there were patients without autonomic features among our patients, as has been described in other published case series (6,7,13). Daily frequency, duration, and the typical sharp quality of pain were similar in our patients regardless of age at CH onset, so the influence of these traits of pain is unlikely. The possibility of overlap with migraine or other primary headache disorders could also be rejected because of the similar number of patients with migrainous features reported by less than a third of our patients, which is in line with data reported previously (12,19). The other associated symptoms, such as painful pulse of the temporal artery, cough, hemifacial spasm, dry mouth, blurred vision, yawning, exhaustion, mydriasis, rise in blood pressure, and loss of consciousness, as well as the feeling of interictal tenderness in painful regions, were reported by individual patients from all three groups. How the aged autonomic system responds to hypothalamic stimulation was not studied in CH and would not, certainly, be explained by this clinical study. Aging has a significant effect on the autonomic system, based on the knowledge of neurodegenerative diseases affecting mainly the autonomous nervous system, e.g. multiple systemic atrophy (20) or pure autonomic failure (21) that commonly have their onset in the elderly. While facial autonomic signs are prominent during CH attacks, they are not limited to the cranial autonomic system. A severe sympathovagal imbalance during spontaneous attacks evaluated by the heart rate variability was demonstrated in one study (22). We can only assume that the subtle degeneration of the autonomic system with aging is responsible for less pronounced autonomic, especially parasympathetic, responses to trigeminal stimulation. Sweating, the only symptom of sympathetic stimulation during CH attacks, was more frequently reported by patients with LO CH.
The clinical features of CH that are hallmarks of the disorder (1) were most frequently and equally present in patients with CO and EO CH and also in the majority of the patients with LO CH. The slight differences, demonstrated in LO patients regarding the presence of some autonomic features, were not responsible for prolonging the time for establishing the correct diagnosis. Diagnostic delay was long in all three examined groups, as shown in studies performed in other countries (23). The recent analysis of relevant published data on mismanagement of trigeminal autonomic cephalalgias concluded that CH was most frequently misdiagnosed as migraine, sinusitis, tooth/jaw disorder, or trigeminal neuralgia (24). The previous medical reports of our patients were insufficient and not consistently available, so we could not specify the type of misdiagnosis. Based on similar and typical CH presentations in the majority of our patients, the main reason for the delay could be only the limited knowledge of the specific characteristics of the disorder. According to the results obtained by our study, diagnostic delay was significantly longer if the disease onset occurred before the patient was 20 years of age. Compared to tension-type headache or migraine, the prevalence of CH is low and estimated to be 0.1%–0.3% during the lifetime (24,25). The one-year CH prevalence in adults of 0.05% (25) is higher than the estimated one-year prevalence in children of 0.03% (26). Trigeminal autonomic cephalalgias are poorly recognized even though the clinical features of childhood- and adolescence-onset CH are similar to those of adult onset (27,28). The diagnostic delay was the shortest for patients with CH onset over 40 years of age. A reasonable explanation could be that at this age the possibility of brain disorders is higher, so these patients are more readily sent to a neurologist than the younger patients are, and the neurological examination increases their chance of getting a proper diagnosis. We are not satisfied with the actual low diagnostic rate of CH in Serbia, regardless of the age of onset. This indicates the relatively small number of patients referred to our headache center in a 12-year period.
Regarding acute treatment, only the effectiveness of high-flow oxygen was analyzed, as the only available treatment. However, the oxygen treatment is not reimbursed in Serbia, resulting in a small proportion of examined patients (15.9%) treated with this method. Furthermore, triptans other than sumatriptan tablets are not available on the Serbian market.
There are two main limitations of the study. The latest edition of ICHD (1) introduced two additional autonomic symptoms, forehead and facial flushing and a sensation of fullness in the ear. As our case series of patients were diagnosed according to ICHD-II (8) criteria before December 2012, these autonomic features were not included, which influenced the patient selection and possibly the interpretation of the results. The second limitation derives from the natural course of primary headache disorders, where the disease duration possibly affects some headache characteristics. The presented study is retrospective in the sense that the analyzed data were previously collected. However, the data were collected for every patient in one time point, as in cross-sectional studies, so a further course of autonomic features was not covered. Follow-up studies are needed.
In conclusion, the influence of age at CH onset is intriguing for further studies and could possibly extend the knowledge about CH pathophysiology. From a clinical point of view, the differences in CH presentation are insufficient to preclude a correct diagnosis and treatment because the same criteria could be applied regardless of patient age.
Clinical implications
Late-onset cluster headache is followed by less prominent autonomic symptoms. Early-onset cluster headache is associated with a longer diagnostic delay.
Footnotes
Funding
This work was supported by a grant from
Conflict of interest
None declared.
