Abstract
Background
Human and animal models suggest that central sensitisation plays a role in medication-overuse headache (MOH). We aimed to study pain perception in MOH patients before and a year after withdrawal.
Methods
We examined pain perception in 35 MOH patients before and two, six and 12 months after detoxification. For baseline comparison, we tested 40 healthy controls. We measured cephalic and extra-cephalic pressure-pain thresholds and supra-threshold pressure-pain scores and extra-cephalic pain thresholds, supra-threshold pain scores and temporal summation for electrical stimulation.
Results
Of the 35 patients, 21 patients completed the entire study and remained cured of MOH. Statistically significant differences between patients and healthy controls were found in cephalic pressure-pain thresholds (137.3 kPa vs. 170 kPa, p < 0.05), extra-cephalic pressure pain thresholds (213.3 vs. 274.3 kPa, p < 0.05), in cephalic supra-threshold pressure-pain scores measured on a 100 mm visual analogue scale (61 vs. 27 mm, p < 0.05) and extra-cephalic supra-threshold pain scores for electrical stimulation (19.0 vs. 10.0 mm, p < 0.05). Cephalic supra-threshold pain scores decreased statistically significantly from 50.3 mm at baseline to 28.0 mm at the 12-month follow-up. In contrast to controls, temporal summation was not found in MOH patients before withdrawal, but after detoxification temporal summation normalised.
Conclusion
The central nervous system is sensitised in patients with MOH. For the first time we demonstrate that the pain perception continues to normalise up to a year after detoxification. This emphasises the importance of detoxification and follow-up to prevent relapse.
Keywords
Introduction
Medication-overuse headache (MOH) is a chronic, secondary headache. The mechanisms behind the development of MOH are largely unknown, but previous studies including imaging, blood analyses, experimentally induced pain and sensory-evoked potentials have indicated a modification of the pain network (1–8). Furthermore, animal studies have shown changes in behaviour and in the expression of different pain-related substances such as substance P, calcitonin gene-related peptide (CGRP), nitric oxide and serotonin in animals treated continuously with drugs commonly overused by MOH patients such as opioids, triptans and simple analgesics (9–18).
Central sensitisation has previously been shown in chronic tension-type headache by demonstrating generally lower pain thresholds both in cephalic and extra-cephalic regions (19–24) and has also been suggested to play a role in chronic migraine (24–26).
Studies on MOH patients have shown increased pain-related reflexes before detoxification compared with healthy volunteers and both found that the reflex decreased after detoxification (1,5,6), but pain mechanisms in MOH have to our knowledge never been investigated in long-term studies. We hypothesised that MOH patients are centrally sensitised while they overuse medication and that the sensitisation will gradually decrease after withdrawal, and aimed to compare pain perception in patients with MOH and healthy volunteers and to investigate alterations in pain perception in MOH patients after detoxification.
Methods
Patients
Patients from the Danish Headache Centre (DHC) were recruited consecutively. We aimed to include 40 patients. The criteria for inclusion were: age above 18 years and MOH fulfilling the criteria defined by the International Headache Society in 2006 (14): headache present on ≥15 days/month, regular overuse for >three months of ergotamine, triptans, opioids or combination analgesics on ≥10 days/month or simple analgesics or any combination of ergotamine, triptans, combination analgesics or opioids on ≥15 days/month and headache has developed or markedly worsened during the overuse. Further, patients should have received treatment for MOH by a specialist in neurology, either as a structured detoxification programme or, as a minimum, advice on stopping medication overuse but should have failed to withdraw from medication overuse. Patients were considered ineligible if they had chronic post-traumatic headache, had coexisting pain conditions that could influence the pain assessment, failed to complete a baseline headache diary or were considered unable to undergo outpatient detoxification by the examining physician, either because they were overusing substances that presented too great a medical risk for outpatient withdrawal or they had co-morbidities expected to complicate outpatient withdrawal.
Healthy volunteers
Forty age- and sex-matched healthy volunteers were recruited by advertising on the website forsoegsperson.dk. Eligible subjects were aged between 18 and 65 years and had no more than one headache day/month. Criteria for exclusion were a history of migraine; headache at the time of examination or less than 12 hours before; intake of analgesics on more than five days per month or less than 12 hours before examination; serious, on-going psychiatric or somatic illness; pregnancy or breastfeeding.
All participants, patients as well as healthy volunteers, gave written, informed consent to participate in the study, which was conducted in accordance with the Declaration of Helsinki and approved by the regional Ethics Committee.
Study design
At a baseline visit before entering the detoxification programme, patients were interviewed and examined by a trained physician and underwent a general and neurological examination. Before the visit, patients had been asked to keep a diagnostic headache diary on a daily basis for at least four weeks (27). Patients who met inclusion criteria were then assigned to treatment. According to the time of inclusion, patients were assigned to one of two structured detoxification programmes. A more detailed description of the programmes is given in a prior publication (28).
The detoxification started at day 1 with abrupt cessation of the overused drugs. Patients in programme A were allowed symptomatic medication up to two days/week as prescribed by the physician at the baseline visit whereas patients in programme B were advised not to take symptomatic medications for the first two months. Symptomatic medications included simple analgesics, non-steroidal anti-inflammatory drugs (NSAIDs), triptans, or combination analgesics, all except the previously overused drugs. For patients in programme A, prophylactic treatment was prescribed by the physician at the baseline visit according to the patients’ primary headache, efficacy and side-effects of previous treatments, co-morbid disorders, and preferences and was started from day 1. For patients in programme B, prophylactic medication was postponed to the two-month follow-up.
After the two-month visit, patients in both treatment programmes were seen regularly by the physician approximately every fourth month until dismissal from the headache centre.
Headache diary
All patients were requested to keep a headache diary in the baseline period—i.e. from day −28 to −1, in the first two months after starting detoxification, and again for four weeks six and 12 months after detoxification. The diary assessed headache duration and intensity rated on a 1–3 scale, and headache quality as well as medication use (27).
Quantitative sensory testing (QST)
All measurements were conducted with the subject lying supine on a bed. All measurements were performed on the non-dominant side of the subject.
Sensitivity to pressure
Pressure-pain threshold (PPT) was recorded with an electronic pressure algometre (Somedic AB, Stockholm, Sweden). This was also used to apply the pressure when measuring supra-threshold pressure-pain score (STPP). We used a circular stimulation probe (0.5 cm2) and a pressure-loading rate of 10 kPa/s. The algometre is described in detail elsewhere (29). PPT was measured extra-cephalically on the dorsum of the middle phalanx of the second finger and cephalically at the temporal region on a standard point 3 cm rostral to the middle of a line between the lateral orbital edge and the external auditory meatus. The threshold was calculated as a mean of three measurements with an interstimulus interval of at least 30 seconds. To measure STPP, the PPT was multiplied by 1.5. This pressure was applied on the same points, three times at each point with an interstimulus interval of at least 30 seconds, and pain was scored on a 100 mm visual analogue scale (VAS) and STTP was calculated as a mean of these three values.
Sensitivity to electrical stimulation
Sensitivity to electrical stimulation was measured in the anterior tibial muscle, at a point located one-third of the distance from the lower tip of the kneecap (apex patellae) to the lateral malleolus. Small needle electrodes (13R24; Dantec Measurement Technology A/S, Copenhagen, Denmark) were inserted 15 mm into the muscle with the cathode approximately 5 mm proximal and anode approximately 5 mm distal to the standard point. We used a Keypoint Workstation (Alpine Biomed Corp., USA) to give the stimulations. All electrical stimuli lasted 1 ms. Pain threshold for a single electrical stimulus (PTSES) was recorded using the method of limits (30). Pain threshold was defined as the weakest stimulus perceived as painful. We calculated PTSES from the mean of three measurements of the pain threshold with an interstimulus interval of at least 30 seconds. Supra-threshold single electrical stimulus pain score (STSES) was measured by applying a single electrical stimulus with 1.5 times the strength of PTSES. The subject then scored the pain on a 100 mm VAS. This procedure was repeated three times and the mean was calculated to find the STSES.
Temporal summation
To measure supra-threshold repetitive electrical stimuli pain score (STRES), we applied a train of five stimuli with a frequency of 2 Hz. The subject scored the last of the five stimuli on the 100 mm VAS. The procedure was repeated three times and the mean was calculated to find the STRES. Temporal summation was defined as being present if the STRES was statistically significantly higher than the STSES (5,6,31,32).
Follow-up
QST measurements were repeated in MOH patients two, six and 12 months after detoxification was initiated.
Outcome measures
The primary endpoints were the differences in subjective pain ratings to cephalic STPP and to STSES between MOH patients and healthy controls and between MOH patients at baseline and 12 months. Supra-threshold pain scores have previously been described as having less inter-individual variation than pain thresholds (19).
Secondary endpoints were differences in ability to wind up between MOH patients and healthy controls and between MOH patients at baseline and two, six and 12 months. Further endpoints were differences in cephalic and extra-cephalic PPTs, in extra-cephalic STPP and in PTSES between MOH patients at baseline, two, six and 12 months and between healthy volunteers and MOH patients and differences in subjective pain ratings to cephalic STPP and to STSES between MOH patients at baseline and two and six months.
Statistics
The difference in supra-threshold pain scores was estimated to have a standard deviation of 35%. The clinical relevant difference was set to 25% between groups (33). A risk of 5% for type 1 errors and 20% for type 2 errors was accepted. The number of subjects needed in each group was estimated to be 31 (34). Addressing the risk of drop-out rate, we aimed to include at least 35 in each group.
Data are presented as medians and quartiles. Drop-outs were excluded from calculations of the primary outcome measures and were included only in the baseline data and for calculating drop-out rates. For data with a skewed distribution, the Mann-Whitney U-test was used. For dichotomous data, we used Fischer’s exact test for samples less than five and the χ2 test for larger samples. For pair-wise related samples, we used the Related Samples Wilcoxon’s Signed Ranks test.
To reduce the bias of missing values, we performed sensitivity analyses using the last observation carried forward method (LOCF). Statistical analyses were performed using the Predictive Analytics Software (PASW) version 18.0 (IBM, Armonk, NY, USA).
Results
Participants
Baseline characteristics of patients with medication-overuse headache and healthy controls.
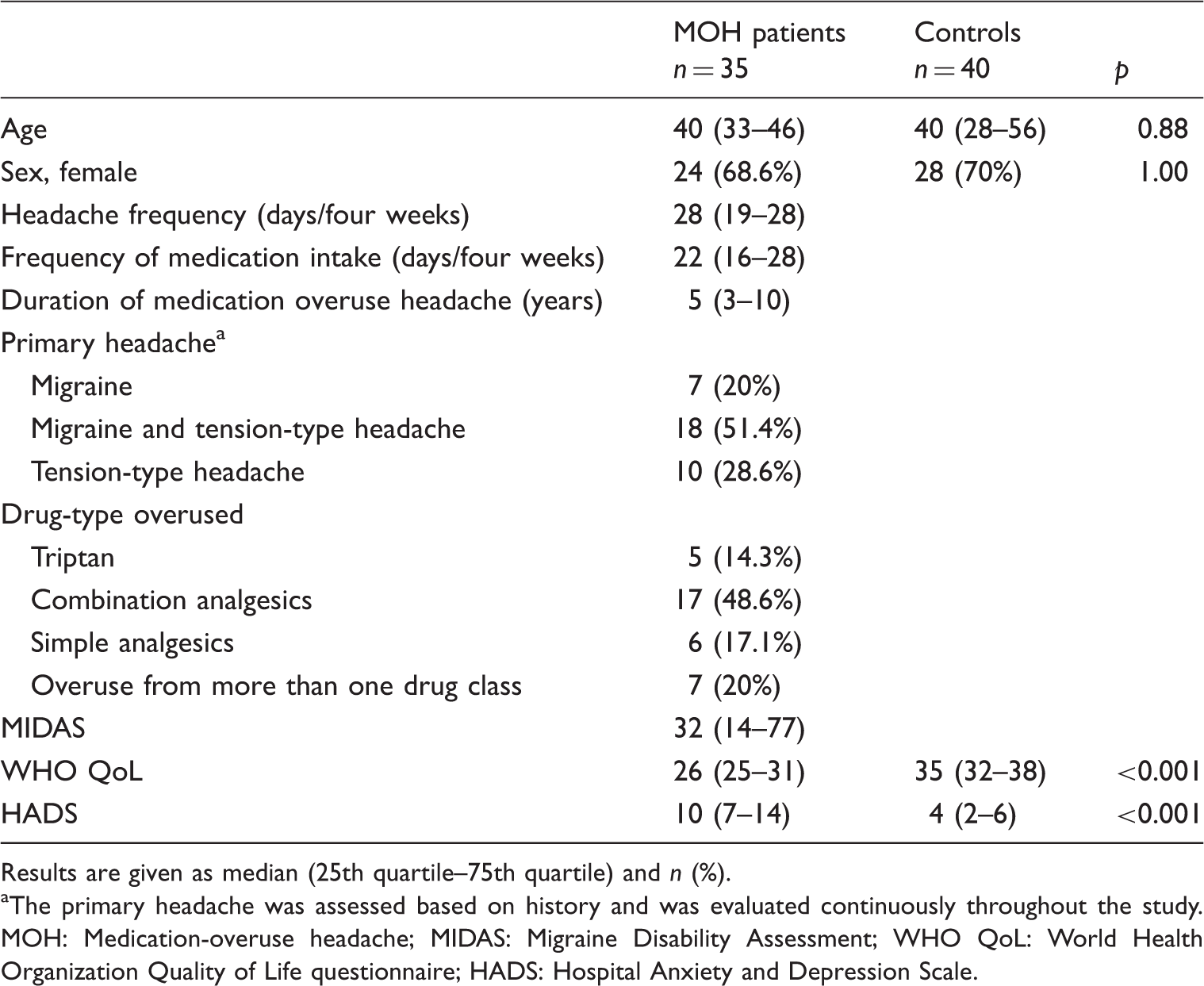
Results are given as median (25th quartile–75th quartile) and n (%). aThe primary headache was assessed based on history and was evaluated continuously throughout the study. MOH: Medication-overuse headache; MIDAS: Migraine Disability Assessment; WHO QoL: World Health Organization Quality of Life questionnaire; HADS: Hospital Anxiety and Depression Scale.
On the day of baseline QST, 21 patients had taken acute medication. We did not find any differences in baseline characteristics or in the results of the QST between those and the patients who did not take acute medication on the day of the baseline recording. We did not find any differences in QST results between patients who followed the two different treatment programmes at any time points. Five patients had fear of needles and thus declined the QST involving needle electrodes. One healthy volunteer experienced intolerable pain when the needle electrodes were placed and the electrical recordings for her were thus not performed.
Between baseline and month 12, headache frequency was reduced by 55.6%, from 24 to 10 days/four weeks (p < 0.001) with no difference between the two treatment groups (28,35). The number of days with medication intake was reduced from 21 to eight days/four weeks, a reduction of 63.6% (p < 0.001). The number of patients who remained cured of MOH after 12 months was 82.8%.
Between baseline and two months, three patients (8.6%) were lost to follow-up. In the remaining patients, headache frequency was reduced from 28 days/four weeks to 14.5 days/four weeks (p < 0.001) and medication use from 23 days/four weeks to 5.5 days/four weeks (p < 0.001).
Between two and six months, an additional five (15.6%) patients were lost to follow-up and four (12.5%) patients relapsed to MOH and were thus excluded from the analyses. The remaining 23 patients had reduced their headache frequency from 27 to 11 days/four weeks (p < 0.001 from baseline and 0.01 from two months) and the medication intake was now seven days/four weeks (p < 0.001 from baseline).
Between six and 12 months two patients (8.7%) relapsed into MOH and were excluded from the analyses.
The total number of patients completing all four pain assessment sessions was 24. Of these, three patients had relapsed into MOH and were thus excluded from the calculations. The total number of patients excluded from the analyses at month 12 was thus 14 patients (40%), of whom three (8.6%) completed the pain assessments but relapsed into MOH and 11 withdrew their consent; eight (22.9%) of these remained cured of MOH and three (8.6%) relapsed to MOH. Data regarding headache and medication use were available for 33 patients (94.3%) at month 12.
We did not find any statistically significant differences in headache characteristics or medication use, neither at baseline nor at month 12, between the 24 patients who completed all four pain assessment sessions and the 11 patients who withdrew their consent.
Primary efficacy parameters
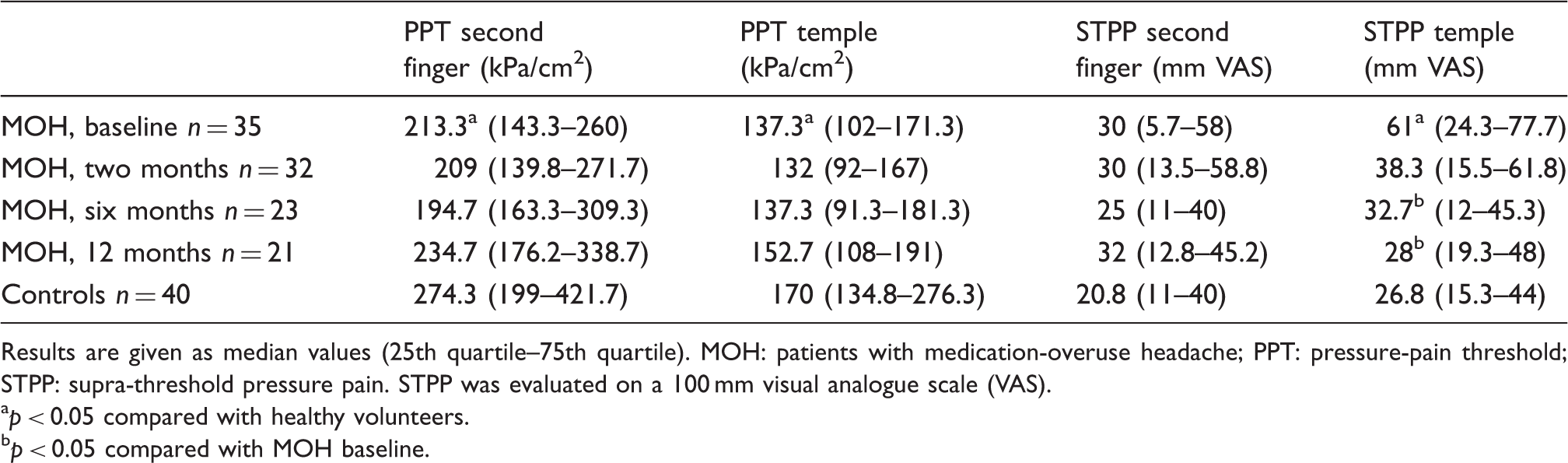
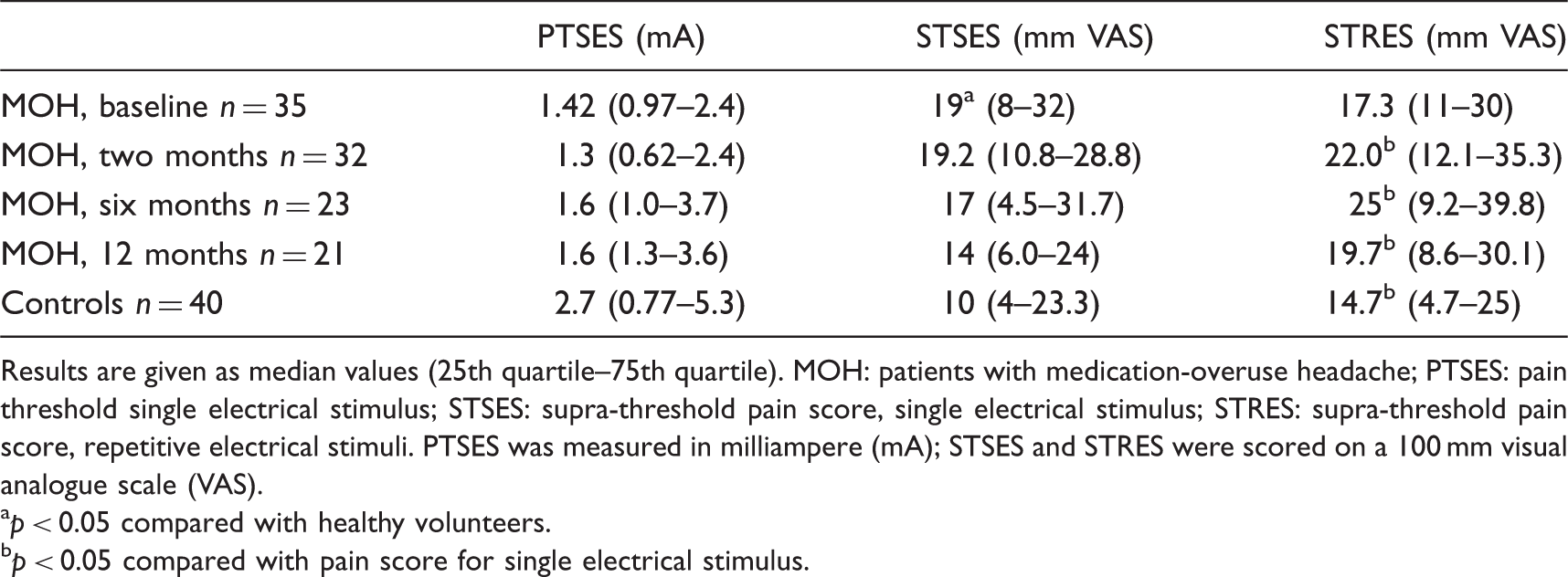
The cephalic STPP of 61 mm VAS in MOH patients before withdrawal was statistically significantly higher compared with the 27 mm VAS in the healthy volunteers (p = 0.001). After 12 months, cephalic STPP decreased markedly from 50.3 to 28.0 (p = 0.03) (Figure 1). Sensitivity analyses showed similar results for the STPP: a decline from 61 to 33.3 mm VAS (p = 0.003). Before withdrawal, the STSES was statistically significantly higher in patients than in healthy controls (p < 0.05) (Figure 2). Pressure-pain test results are listed in Table 2, and the results of the electrical stimulation are listed in Table 3.
Cephalic supra-threshold pressure pain. Pain ratings on a 100 mm visual analogue scale (VAS) for supra-threshold pressure in the temporal region. MOH: medication-overuse headache. *p < 0.05 compared with healthy volunteers. ‡p < 0.05 compared with MOH patients at baseline. Pain ratings were significantly higher for patients with MOH before withdrawal compared with healthy controls but were normal 12 months after detoxification. Pain ratings decreased significantly in patients with MOH from baseline to six and 12 months’ follow-up. Extra-cephalic supra-threshold electrical stimulation. Pressure-pain thresholds and pain ratings for supra-threshold pressure in MOH patients before and two, six and 12 months after detoxification and in healthy controls. Results are given as median values (25th quartile–75th quartile). MOH: patients with medication-overuse headache; PPT: pressure-pain threshold; STPP: supra-threshold pressure pain. STPP was evaluated on a 100 mm visual analogue scale (VAS). ap < 0.05 compared with healthy volunteers. bp < 0.05 compared with MOH baseline. Pain thresholds for a single electrical stimulation and pain ratings for supra-threshold simulation with single and repetitive stimuli. Results are given as median values (25th quartile–75th quartile). MOH: patients with medication-overuse headache; PTSES: pain threshold single electrical stimulus; STSES: supra-threshold pain score, single electrical stimulus; STRES: supra-threshold pain score, repetitive electrical stimuli. PTSES was measured in milliampere (mA); STSES and STRES were scored on a 100 mm visual analogue scale (VAS). ap < 0.05 compared with healthy volunteers. bp < 0.05 compared with pain score for single electrical stimulus.

Secondary efficacy parameters
In contrast to the healthy volunteers, temporal summation was not found in the MOH patients at baseline. But at the two-, six- and 12-month follow-up, the temporal summation was apparent as STRES was statistically significantly higher than the STSES (Figure 3), also when performing sensitivity analyses (Table 3).
Temporal summation. Pain ratings on a 100 mm visual analogue scale for supra-threshold electrical simulation in the anterior tibial muscle with a single stimulus and with repetitive stimuli. STSES: supra-threshold pain score, single electrical stimulus; STRES: supra-threshold pain score, repetitive electrical stimuli. Temporal summation was present if STRES was significantly higher than the STSES. *p < 0.05 between STSES and STRES indicating ability to wind-up. Before withdrawal, temporal summation was not seen in patients with MOH in contrast to controls. After withdrawal, temporal summation was found.
Both cephalic and extra-cephalic PPTs were statistically significantly lower in MOH patients than in healthy volunteers (p = 0.003 and 0.002, respectively).The PPTs did not change from baseline to after detoxification but tended to increase from baseline to 12 months. The extra-cephalic STPP was higher in MOH patients than in healthy volunteers, but not statistically significantly so. It remained stable throughout the follow-up.
After six months, the cephalic STPP was statistically significantly lower than at baseline (p = 0.006). This was also the case when performing sensitivity analyses (p = 0.002) (Table 2).
The PTSES was lower in patients than in healthy volunteers although not statistically significantly (Table 3).
We did not detect any changes between baseline and follow-up pain assessments in the patients who relapsed into MOH.
There were no statistically significant differences between any of the QST results of patients with a reduction in headache frequency of more than 50% compared with patients with less than 50% reduction or between patients with episodic headache and patients with chronic headache after two, six, or 12 months. The numbers of patients who relapsed to MOH at six and 12 months were too small to provide meaningful statistical data and hence we did not detect any statistically significant differences between relapsers and non-relapsers.
Discussion
This study presents detailed follow-up data on pain sensitivity in patients with MOH a year after detoxification, which, to our knowledge, has never been studied before. The first major finding of this study is the demonstration of central sensitisation in patients with MOH. We demonstrated that patients with MOH are sensitised by showing decreased pain thresholds and increased pain response for supra-threshold pain stimulation. This is in line with what has previously been found by others in patients with chronic tension-type headache and with MOH (5,6,8,21,24,26,31). Similar to the findings in chronic tension-type headache (19), the sensitivity for pressure was more increased cephalically than extra-cephalically, but extra-cephalic pain thresholds were also decreased compared with healthy volunteers. This indicates that central sensitisation is involved as the pain perception is altered in areas outside the area of the chronic pain (32,36).
The second important finding was the decrease in sensitisation after detoxification. The response to supra-threshold pain gradually approached that of the healthy volunteers. It has not previously been shown that the pain response in humans continues to decrease to a normal level several months after detoxification. This is probably reflecting the hypothesis that the neural plasticity involved in pain modulation where central neurons otherwise not related in the pain pathways are recruited during the overuse (36) take time to recover and revert to normal. Our important findings of continuous improvement in headache, where the headache frequency continues to diminish over time between two and 12 months after detoxification (28,35), support this theory.
The reduction in central sensitisation could be due to either the decrease in medication use or the decrease in headache frequency. Several lines of evidence speak for the first of these options. Zappaterra et al. found PPTs to be statistically significantly lower in patients with MOH compared with chronic headache without medication overuse (8), and Perrotta et al. have shown a decrease in pain response after withdrawal with only a minor reduction in headache frequency (5). In line with our results, animal studies have shown a marked reduction in pain thresholds that increase again after detoxification (10). Further, we did not find any correlation between the reduction in headache frequency and reduction in pain, which we would have expected if the pain reduction had been entirely related to the reduction in headache. It is therefore most likely that the reduction in medication intake is partly responsible for the reduction in central sensitisation.
The third important finding was that temporal summation was not seen in MOH patients at baseline in contrast to the healthy volunteers, but could be demonstrated in patients at all time points after detoxification. Temporal summation is the psychophysical equivalent to wind-up, which is measured electrophysiologically (37). Wind-up may correlate to an experimental induction of central sensitisation (36,38,39). It can be induced in healthy subjects and has also been induced experimentally in different chronic pain conditions such as chronic headaches and fibromyalgia (19,24,36,40).
Previously, it has also been found in MOH patients with migraine as the primary headache (5,6). It does not seem to be enhanced in centrally sensitised subjects, neither with experimentally induced central sensitisation nor central sensitisation caused by disease (19,24,31,41).
Wind-up is dependent on the N-methyl-D-aspartic acid (NMDA) receptor, which is affected in central sensitisation and is known to play a role in both migraine and tension-type headache (36). Human and animal studies have shown alterations in the serotonergic system after prolonged exposure to analgesics leading to a decrease in the blood serotonin level (2,7). In animals, a low serotonin level can lead to phosphorylation of the NMDA receptor (42), and the stimulation needed to activate the phosphorylated NMDA receptor and produce action potentials is much lower than in the normal NMDA receptor (39). The cumulative depolarisation seen in repetitive stimulation might be non-existing as a single stimulation could depolarise the membrane maximally. The neurons could, so to speak, already be wound up so the single stimulation produces the same response as repetitive stimuli. It is possible that these or similar mechanisms explain the lack of temporal summation before withdrawal and why it is seen after detoxification in our MOH patients, but this remains speculative until we get an increased understanding of the mechanisms responsible for wind-up and temporal summation. The regaining of temporal summation is consistent with the reduction in the supra-threshold pain scores and an important finding indicating a normalisation in the NMDA receptor and hence a reduction in central sensitisation.
A strength of our study is the long follow-up period. Only few studies of the pain perception in MOH patients have been conducted and none with more than one follow-up visit only two to 10 weeks after detoxification (1,5,6).
On the testing days, we used repeated measurements to establish the pain thresholds and supra-threshold pain scores to be sure of consistent values. Pain perception in healthy volunteers was tested on only one occasion and not repeatedly over time as for the MOH patients. As previous studies have shown that pain perception remains stable in healthy volunteers over time, we did not find it necessary to repeat the measurements in those (43,44).
The number of MOH relapsers was fairly low and thus we were not able to detect the possible differences in pain perception between relapsers and non-relapsers.
A drawback of this study is the lack of a control population with untreated MOH. However, we did not find it ethically acceptable to withhold treatment for 12 months from these severely affected and from previously treatment-resistant patients with MOH.
The subjective measures of temporal summation could be a limitation of this study.
In conclusion, patients with MOH have altered pain perception indicating central sensitisation. This study shows that the alterations in pain perception are reversible and improve substantially after detoxification and over time approach the pain perception of healthy volunteers.
This study provides new insight in the pathophysiology of MOH and emphasises the importance of detoxification in the treatment of MOH.
Clinical implications
Patients with medication-overuse headache (MOH) have increased pain sensitivity for different modalities both in cephalic and extra-cephalic areas indicating central sensitisation. Medication overuse can alter the temporal summation but it can be regained after withdrawal. Pain sensitivity continues to normalise up to a year after withdrawal. Our results emphasise the importance of detoxification and close follow-up.
Footnotes
Acknowledgement
The authors thank Mrs Hanne Andresen for technical assistance during data collection.
Funding
The study was supported by grants from the Danish Headache Society. The funding source was not involved in any stage of the study.
Conflicts of interest
Signe B. Munksgaard has received a travel grant from MSD. Dr Bendtsen has received honoraria for lectures from MSD and Pfizer and serves on the scientific advisory board for Berlin-Chemie. Prof. Jensen has received honoraria for lectures and patient leaflets from MSD and Pfizer and serves on medical advisory boards for Allergan, LindeGas, ATI and Neurocore.
