Abstract
Medication overuse could interfere with the activity of critical brain regions involved in the supraspinal control of pain signals at the trigeminal and spinal level, leading to a sensitisation phenomenon responsible for chronic pain. We hypothesised that medication-overuse headache (MOH) patients might display abnormal processing of pain stimuli at the spinal level and defective functioning of the diffuse noxious inhibitory controls. We tested 31 MOH patients before (bWT) and after (aWT) standard inpatient withdrawal treatment, 28 episodic migraine (EM) patients and 23 healthy control subjects. We measured the threshold, the area and the temporal summation threshold (TST) of the nociceptive withdrawal reflex before, during and after activation of the diffuse noxious inhibitory controls by means of the cold pressor test. A significantly lower TST was found in both the MOH (bWT and aWT) and the EM patients compared with the controls, and in the MOH patients bWT compared with both the MOH patients aWT and the EM patients. In the MOH bWT patients the cold pressor test induced a TST increase significantly lower than that found in the MOH aWT, EM and control groups. Abnormal spinal cord pain processing and a decrease of the antinociceptive activity of the supraspinal structures in MOH patients can be hypothesised. These abnormalities could, in part, be related to the medication overuse, given that the withdrawal treatment was related to an improvement in the neurophysiological findings.
Keywords
Introduction
The 2nd Edition of the International Headache Society's International Classification of Headache Disorders (ICHD-II, IHS 2004) (1) introduced the term medication-overuse headache (MOH: code 8.2) to indicate a condition in which an excessive intake of symptomatic drugs has played a role in the transformation of an episodic headache into a chronic form. Characteristically this condition shows a low response to prophylactic treatments and a marked improvement after drug withdrawal (1). Despite the wide prevalence of MOH, its pathophysiology is still unclear.
In humans, electrophysiological studies, based on psychophysical methods and cortical evoked potentials, showed abnormal responses to electrical (2,3) and laser (4) nociceptive stimulation at cephalic as well as at extracephalic sites in patients with chronic forms of primary headaches, including MOH. In addition, neuroimaging studies demonstrated, in chronic headaches, morphological abnormalities in brain areas contributing to pain processing (5,6) and, more recently, in MOH patients, functional metabolic abnormalities in cortical areas of the pain network such as the anterior cingulated and orbitofrontal cortex (7). These data suggest the existence of generalised abnormal processing of painful stimuli in patients with chronic forms of primary headache, including MOH. Moreover, in MOH patients the generalised facilitation of the pain responses, as well as the metabolic changes in cortical pain processing areas, normalised after withdrawal treatment (3,7), support a role for medication overuse in headache chronification.
It can be hypothesised that the generalised (cephalic and extracephalic) facilitation of the pain responses observed in MOH patients might be attributed to a dysfunction of antinociceptive supraspinal structures (3) (i.e. periaqueductal grey, subnucleus reticularis dorsalis, and rostral ventromedial medulla), which are involved in the descending pain modulatory network and diffusely project throughout the central nervous system to regulate nociceptive processing (8–10) and are known to play a role in the pathophysiology of primary headaches (5,11–14). Because, in both humans and animals, these antinociceptive supraspinal structures exert a strong inhibitory effect on the nociceptive spinal cord neurons (8–10,15,16), it would be interesting to evaluate whether MOH patients display abnormal processing of pain stimuli at the spinal level as well as defective functioning of the supraspinal descending control of pain.
To the best of our knowledge, in MOH patients neither abnormal pain processing at the spinal level nor defective functioning of the supraspinal descending control systems of pain has been demonstrated.
A useful tool to evaluate spinal pain processing and the influence of the supraspinal antinociceptive descending pathways on the spinal cord is the study of the wind-up phenomenon. This is a form of activity-dependent plasticity of trigeminal and spinal wide dynamic range neurons, consisting of a progressive, frequency-dependent facilitation of the neuronal response following the repeated stimulation of primary afferent nociceptive C-fibres, which results in prolonged increases in the neuronal excitability (17,18). Despite the controversial role of wind-up in the induction and maintenance of central sensitisation (18–21), it may be regarded as a neural mechanism physiologically devoted to encoding and detecting nociceptive inputs at the spinal as well as at the trigeminal level (22) and, in this sense, its enhancement could reflect the presence of abnormal pain processing at trigeminal as well as at spinal levels.
Temporal summation of painful stimuli in humans has been considered a clinical manifestation of the ‘wind-up’ phenomenon observed in animals (20,22,23) and it has been widely used to detect abnormalities in pain processing in chronic pain conditions, including fibromyalgia and headaches (24–26). The temporal summation of pain develops in parallel with the temporal summation of the nociceptive withdrawal reflex (NWR), which has been demonstrated to be a very sensitive tool for exploring physiological and pathophysiological mechanisms in spinal cord pain processing (15,16,21,27,28).
Interestingly, it has been demonstrated that the wind-up activity can be modulated by supraspinal structures such as the periaqueductal grey, subnucleus reticularis dorsalis, and rostral ventromedial medulla (8–10). In humans, the modulatory influences of the descending supraspinal control pathways on pain mechanisms can be usefully studied, evaluating the effect of heterotopic nociceptive stimuli, which activate the diffuse noxious inhibitory controls (DNICs), on the NWR, the temporal summation threshold (TST) of the NWR, and the related pain sensation (15,16,29).
Here we sought to determine whether MOH patients display abnormal processing of pain stimuli at the spinal level and whether this condition (MOH) is mediated, at least in part, by defective function of pain modulatory pathways. We measured the TST of the lower limb NWR and DNIC activity in a sample of MOH patients before and after drug withdrawal treatment. Moreover, we compared the MOH patients' responses with those of an additional group of subjects affected by an episodic form of migraine without aura.
Methods
The study had local ethics committee approval and all the participants gave their written consent.
Study population
Thirty-one patients suffering from chronic daily headache or almost daily headache, with a history of migraine without aura (coded as 1.1 in the ICHD-II) (1) and symptomatic medication overuse (coded as 8.2) (1), were enrolled as the MOH group; 28 patients with an episodic form of migraine without aura were enrolled as the episodic migraine (EM) group and 23 age-matched healthy individuals, without neurological disorders or a clinical (including family) history of neurological disorders, were the control group.
All the patients enrolled in the study filled in headache diaries (these were mailed to those on the waiting list for a consultation) every day for at least 3 months before undergoing their first neurophysiological examination and for 2 months after withdrawal treatment.
Exclusion criteria included: other primary or secondary headaches; chronic headache that did not improve (i.e. revert to the episodic pattern) 2 months after withdrawal treatment (MOH group); any serious systemic or neurological diseases or psychiatric disorders, including depression (Beck's depression inventory score > 17); fibromyalgia, complex regional pain syndrome or neuropathic pain (30–32); use of prophylactic medication for headaches; analgesic and/or antimigraine medication abuse (EM and controls); and use of opiates, antidepressants, benzodiazepines or hormones.
Nociceptive withdrawal reflex measurements
Nociceptive withdrawal reflex
The NWR from the right lower limb was investigated according to a validated method (15,16,27). In particular, female patients and controls were matched for cycle phases (follicular phase) in order to minimise the pain modulation across the menstrual cycle (16), and all the subjects were tested between 09.00 and 11.00 to minimise the effect of diurnal variation (16). Before formal measurements were started, the subjects underwent training to familiarise them with the pain threshold assessment procedure.
The subjects were seated comfortably in a quiet room at constant temperature (23 ± 2°C). Their lower limbs were positioned to ensure complete muscle relaxation (knee flexed at 130° and ankle at 90°).
The sural nerve was stimulated percutaneously via a pair of standard surface electrodes (Ag/AgCl) applied to degreased skin behind the right lateral malleolus. The transcutaneous electrical stimulus consisted of a constant current pulse train of five individual 1-ms pulses delivered at 200 Hz (equal to an inter-stimulus interval of 4 ms), randomly applied every 25–40 s. Electromyographic reflex responses were recorded from the capitis brevis of the biceps femoris via surface electrodes (Ag/AgCl). The filter bandpass setting was between 3 Hz and 3 kHz. The analysis time was 300 ms, with the sensitivity set at 100 µV. Each response was full-wave rectified and integrated in the 80–130 ms post-stimulus interval (16) (Medelec, Synergy, UK).
The staircase method was used to evaluate the NWR threshold (Th), defined as the stimulation intensity generating stable reflex responses with an amplitude exceeding 20 µV for more than 10 ms in the time interval 80–130 ms over five stimuli.
The stimulation intensity was fixed at 1.2xTh; five reflex responses were recorded and the mean NWR area under the curve (Area) was computed using a computerised method.
The subjects rated the psychophysical pain sensation for each stimulus on an 11-point numerical rating scale (NRS), graded from 0 = no pain to 10 = unbearable pain.
The first recording of each session was discarded in an attempt to reduce the influence of the startle reaction.
Temporal summation of the NWR
The sural nerve was stimulated using a constant current pulse train of five individual 1-ms pulses delivered at 200 Hz repeated five times at a frequency of 2 Hz, as previously described (16,27). The current intensity was increased (in 1 mA steps) from 2 mA until detection of temporal summation. A TST of the NWR was considered when a clear facilitation of the reflex response size (greater than 20 uV for 10 ms or more) in the 4th and 5th trace, compared to the 1st one, was detectable during the course of the five individual pulses train in the time interval 80–130 ms, and accepted when three consecutive recordings gave the same threshold.
The subjects rated the psychophysical pain sensation for the first and fifth stimulus on an 11-point NRS, graded from 0 = no pain to 10 = unbearable pain.
Heterotopic noxious conditioning stimulation
In order to study the pain modulating system subserving DNICs, we investigated the effects of experimental heterotopic noxious conditioning stimulation, in the form of the cold pressor test (CPT), on the NWR. The CPT was chosen because previous studies in healthy human subjects showed that it induces a marked inhibition of both the Th and the TST of the NWR as well as of the related painful sensation (15,29). The patients immersed their whole hand up to the wrist in a circulating cold water bath (3–4°C) for not less than 4 min and not more than 5 min and rated the psychophysical pain sensation on an 11-point NRS (0 = no pain to 10 = unbearable pain). The water was constantly re-circulated to prevent laminar warming around the immersed hand. During testing, the neurophysiological measurements were taken, starting from the second minute after the start of the CPT.
Experimental procedure
All the MOH patients underwent a neurophysiological examination on admission, before the in-patient withdrawal treatment (bWT), and again 8–10 days after the start of the withdrawal treatment (aWT).
Neurophysiological measurements were recorded in all participants: (i) at baseline; (ii) during a non-painful (control) session (immersing the hand in water at 25°C); (iii) during a painful session (immersing the hand in water at 2–4°C); and (iv) at an after-effect session (4–8 min after taking the hand out of the water). The subjects were randomly assigned to the control or painful session and then crossed over to the other session. To avoid sensitisation of the skin receptors, the hand was dried after removal from the water and, except when evaluating the after-effects, a rest period of more than 20 min was allowed after each session.
The operator was blinded to the patient's diagnosis (control, EM or MOH) and withdrawal treatment status (bWT or aWT).
The intensity of the headache was evaluated on a four-point scale (0 = no headache; 1 = mild headache; 2 = moderate headache; 3 = severe headache).
In the MOH patients, the recordings were performed outside acute migraine attacks (headache intensity score 1 out of 3), and thus in between periods of exacerbation of migraine pain. All the subjects were tested at least 12 h after any drug intake. Prophylactic antimigraine treatment with valproic acid was started only after the second neurophysiological examination.
All the EM patients were examined in a headache-free interval, at least 3 days after and 3 days before a migraine attack (confirmed by telephone interview).
Statistical analysis
The groups were compared for gender, age, weight and blood pressure (measured at admission), in order to minimise physiological or pathological conditions liable to influence the various parameters of the NWR or the TST of the NWR (16).
We performed the Mann-Whitney rank sum test or Wilcoxon signed rank test for continuous variables. The discrete variables were evaluated using the χ2 test.
One-way
We used the Wilcoxon signed rank test for paired data to compare the bWT and aWT neurophysiological data (Th, Area and TST) and the NRS scores among the MOH patients, and to compare the number of headache days per month 1 month bWT and 1 and 2 months aWT.
In order to detect a temporal summation of pain, the Wilcoxon signed rank test for paired data was used to compare NRS scores for the fifth stimulus with the first stimulus recorded at the TST, both in patients and controls. In a further evaluation, separate one-way
The TST and Area of the NWR and the NRS scores, during the control session as well as during and after the CPT, were presented as percentages of baseline values.
Baseline values of all measurements were compared with the control session values in both patients and controls using the Mann-Whitney rank sum test or the Wilcoxon signed rank test for paired data, in order to verify that the experimental condition did not modify the baseline parameters.
In order to verify the effect of the CPT (painful session) on the neurophysiological and psychophysical parameters, a series of
Multivariate analysis of variance (
Spearman's correlation test was used to look for statistically significant correlations between MOH patient variables such as age, primary headache duration, duration of drug overuse, type and number of overused medications, number of headache days per months 1 month bWT and 1 and 2 months aWT, and neurophysiological values (Th, Area and TST). The same test was employed in the EM patients to detect a possible correlation between neurophysiological data (Th, Area and TST) and clinical variables, including age, age at onset, disease duration, and migraine attack frequency per month.
The results were expressed as mean values ±
All statistics were calculated using the SPSS (13.0) program for Windows (SPSS, Chicago, IL)
Results
Study population
No significant differences between the patients and controls were found in the physiological variables examined (gender, age, weight and blood pressure) (all P > 0.05).
Demography and clinical characteristics of the study population

MOH, medication overuse headache; bWT, before withdrawal treatment; EM, episodic migraine. (range).
In the MOH group, 19 (61.2%) patients presented triptan-overuse (coded as 8.2.2 in the ICHD-II) (1), 11 (35.4%) analgesic-overuse (8.2.3) and one (3.4 %) combination medication-overuse (8.2.5).
At follow-up 30 and 60 days aWT, we found a significant reduction (P = 0.001) in the number of headache days per month (7.0 ± 3.4 and 3.4 ± 2.0, respectively) compared with the bWT period.
In the EM group, migraine pain was unilateral or mainly unilateral in 11 patients, constantly bilateral in eight, and side-switching in nine.
No significant correlations emerged between neurophysiological (Th, Area and TST) and psychophysical (NRS scores) parameters and any of the clinical variables (see Table 1) in the study population (all P > 0.05).
Nociceptive withdrawal reflex findings
Nociceptive withdrawal reflex
One-way
Mean values ±

*P < 0.01 vs controls; †P < 0.01 vs EM; ‡P < 0.01 vs MOH aWT.
Th, nociceptive withdrawal reflex threshold; Area, nociceptive withdrawal reflex area at 1.2xTh; TST, nociceptive withdrawal reflex temporal summation threshold; MOH, medication-overuse headache; bWT, before withdrawal treatment; aWT, after withdrawal treatment; EM, episodic migraine.
The mean Area was significantly greater in the MOH (both bWT and aWT) and EM patients compared with the controls (ES, d = −0.93, r = 0.42; d = −0.67, r = 0.32; d = −0.67, r = 0.31, respectively) and in the MOH patients bWT compared with aWT (Wilcoxon Z = −2.931, P = 0.003) (Table 2). No significant differences were found in the MOH patients (both bWT and aWT) compared with the EM patients (ES, d = −0.25, r = 0.12 and d = 0.06, r = 0.03, respectively) (Table 2).
No significant correlations emerged between neurophysiological parameters (Th, Area) and any of the clinical variables (see Table 1) (all P > 0.05).
Temporal summation of the nociceptive withdrawal reflex
One-way
The TST mean values were significantly reduced in both the MOH (bWT and aWT) and the EM patients compared with the controls (ES, d = 1.47, r = 0.59; d = 1.01, r = 0.45; d = 0.68, r = 0.32, respectively) (Table 2). A significant TST mean values difference was found between the MOH patients bWT vs aWT (Wilcoxon Z = −3.239, P = 0.001) (Figure 1), as well as between the MOH patients bWT and the EM patients (ES, d = 0.75, r = 0.35) (Table 2).
Nociceptive withdrawal reflex temporal summation threshold (full arrows) in a representative MOH patient before (above) and after (below) withdrawal treatment.
No significant differences were found between the MOH patients aWT and the EM patients (ES, d = 0.38, r = 0.18) (Table 2).
No significant correlation was found between the TST and any of the clinical variables (see Table 1) (all P > 0.05).
Psychophysical measurements
One-way
One-way
Mean values ±

P < 0.01 vs 1st s TST (Wilcoxon signed rank test for paired data).
P < 0.01 vs controls (
P < 0.01 vs MOH aWT (Wilcoxon signed rank test for paired data).
Differences between the 5th s and the 1st s were calculated for each subject before averaging.
NRS, numerical rating scale; Th, nociceptive withdrawal reflex threshold; TST, nociceptive withdrawal reflex temporal summation threshold; MOH, medication-overuse headache; bWT, before withdrawal treatment; aWT, after withdrawal treatment; 1st s, first stimulus of the TST; 5th s, fifth stimulus of the TST.
Significantly higher NRS scores for the fifth stimulus at the TST were found between the MOH patients bWT and the controls (ES, d = 0.27, r = 0.13). No significant differences were detected between MOH bWT and EM patients (ES, d = −0.11, r = 0.05). No significant differences were detected between MOH patients aWT and both EM (ES, d = −0.22, r = 0.11) and controls (ES, d = −0.40, r = 0.19) or between EM patients and controls (ES, d = −0.60, r = 0.29) (Table 3).
No differences were found between the MOH patients bWT and the MOH patients aWT in the NRS scores for either the first (Wilcoxon Z = −0.346, P = 0.730) or the fifth (Wilcoxon Z = −1.147, P = 0.251) stimulus at the TST.
For patients and controls, significant temporal summation of pain was observed in the NRS scores at the fifth compared with the first stimulus (Table 3).
In a further evaluation, we compared the differences between the NRS scores at the fifth stimulus and the NRS scores at the first stimulus in the patients and the controls. These differences were greater in the MOH patients than in the EM patients and controls, and we noted the emergence of a strong trend towards increased temporal summation of pain in the MOH patients. However, these values just failed to reach statistical significance (F(3,81) = 3.684, P = 0.030) (Table 3).
Effect of DNICs on the nociceptive withdrawal reflex and the psychophysical measurements
Multivariate analysis revealed significant differences between the MOH bWT, EM and control groups in mean TST values during the painful session (F(2,81) = 8.308, P = 0.001, η2 P = 0.209), while no differences were found in either the non-painful (control) (F(2,81) = 0.262, P = 0.770, η2 P = 0.003) or the after-effect sessions (F(2,81) = 1.266, P = 0.290, η2 P = 0.041). No differences were found in Area values across all three CPT sessions, non-painful (control) (F(2,81) = 0.436, P = 0.649, η2 P = 0.015), painful (F(2,81) = 4.014, P = 0.023, η2 P = 0.116) and after-effect (F(2,81) = 4.435, P = 0.016, η2 P = 0.133), respectively.
Multivariate analysis revealed no differences between the MOH aWT, EM and control groups in either the TST or the Area values in the non-painful (control) (F(2,81) = 0.737, P = 0.483, η2 P = 0.015 and F(2,81) = 0.217, P = 0.806, η2 P = 0.013, respectively), painful (F(2,81) = 2.359, P = 0.104, η2 P = 0.067 and F(2,81) = 3.472, P = 0.038, η2 P = 0.094, respectively) and after-effect sessions (F(2,81) = 0.342, P = 0.712, η2 P = 0.013 and F(2,81) = 6.379, P = 0.03, η2 P = 0.173, respectively).
Multivariate analysis revealed significant differences between the MOH bWT, EM and control groups in mean NRS scores corresponding to the 1.2xTh (F(2,81) = 8.441, P = 0.001, η2 P = 0.129) and to the fifth stimulus at the TST during the painful session (F(2,81) = 7.901, P = 0.001, η2 P = 0.046), while no differences were found for these two measurements in either the non-painful (control) (F(2,81) = 0.129, P = 0.879, η2 P = 0.003 and F(2,81) = 0.234, P = 0.792, η2 P = 0.024, respectively) or the after-effect session (F(2,81) = 1.251, P = 0.294, η2 P = 0.015 and F(2,81) = 0.586, P = 0.560, η2 P = 0.004, respectively).
No significant differences were found in mean NRS scores corresponding to the first stimulus at the TST across all the CPT sessions (non-painful, F(2,81) = 0.470, P = 0.627, η2 P = 0.015; painful, F(2,81) = 2.032, P = 0.141, η2 P = 0.013; and after-effect, F(2,81) = 1.567, P = 0.218, η2 P = 0.053).
Multivariate analysis revealed no differences between the MOH aWT, EM and control groups in mean NRS scores corresponding to the 1.2xTh and to the first and fifth stimuli at the TST across all the CPT sessions (non-painful (control) (F(2,81) = 0.032, P = 0.969, η2 P = 0.003, F(2,81) = 0.437, P = 0.648, η2 P = 0.015 and F(2,81) = 0.527, P = 0.593, η2 P = 0.024, respectively); painful (F(2,81) = 5.361, P = 0.007, η2 P = 0.053, F(2,81) = 0.335, P = 0.717, η2 P = 0.013 and F(2,81) = 1.844, P = 0.168, η2 P = 0.046, respectively); and after-effect (F(2,81) = 1.420, P = 0.250, η2 P = 0.013, F(2,81) = 1.738, P = 0.185, η2 P = 0.053 and F(2,81) = 0.214, P = 0.808, η2 P = 0.004, respectively)]).
Non-painful (control) session
No significant differences in neurophysiological values (Th, Area and TST) and psychophysical measurements (NRS scores) were found between the baseline and control sessions either in the patient groups or in the controls, or within the whole patient sample (MOH patients bWT and aWT and EM patients) (all P > 0.05) (Tables 4 and 5, Figure 2).
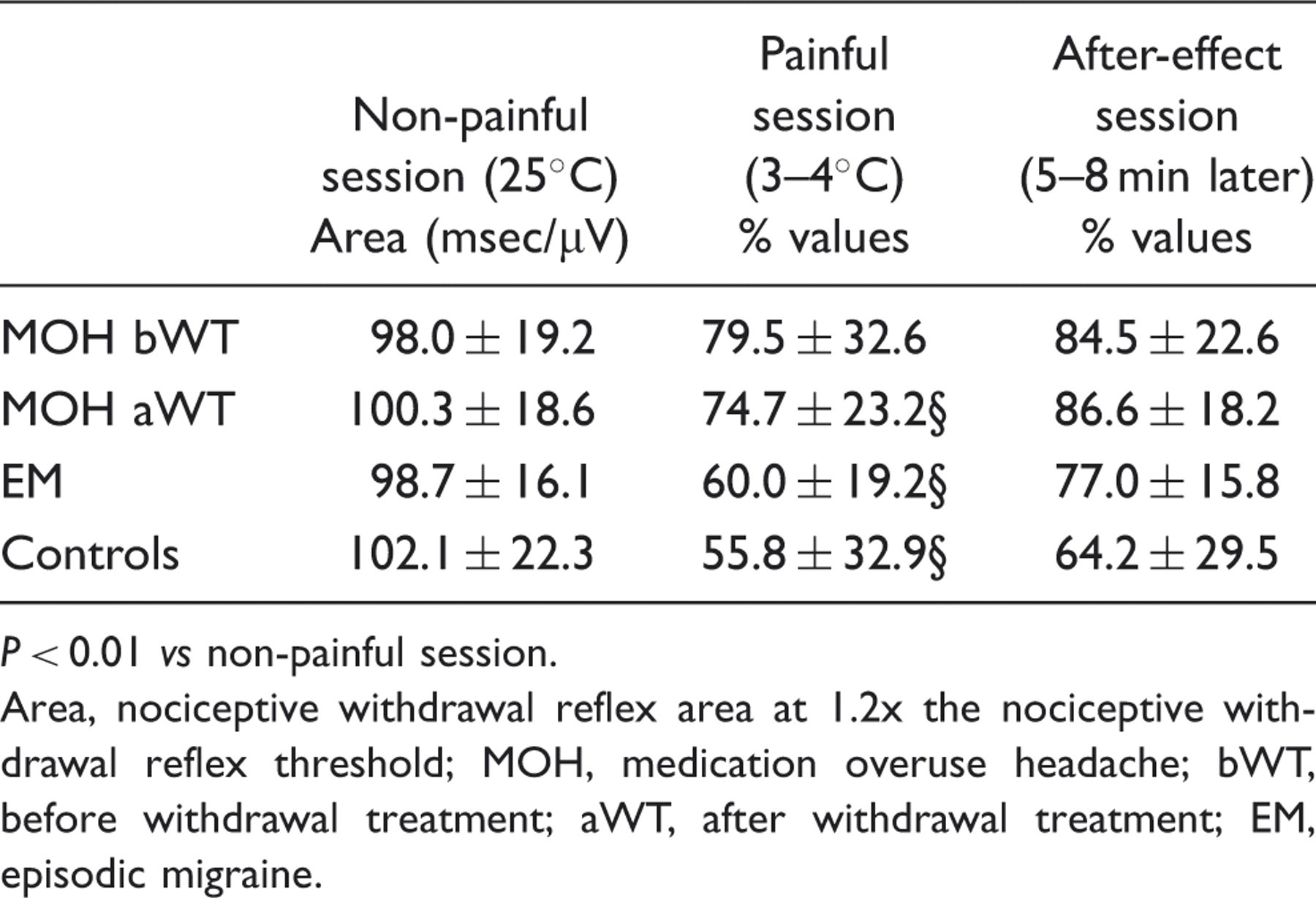
Temporal summation threshold of the nociceptive withdrawal reflex in MOH patients (bWT and aWT), in EM patients and in controls during non-painful, painful (cold pressor test) and after-effect sessions. Data are presented as percentage variations of the baseline values. Mean values ±
P < 0.01 vs non-painful session. Area, nociceptive withdrawal reflex area at 1.2x the nociceptive withdrawal reflex threshold; MOH, medication overuse headache; bWT, before withdrawal treatment; aWT, after withdrawal treatment; EM, episodic migraine. Means values ±
P < 0.01 vs controls; †P < 0.01 vs baseline. NRS, numerical rating scale; Tr, nociceptive withdrawal reflex threshold to single stimulus; TST, nociceptive withdrawal reflex temporal summation threshold; MOH, medication-overuse headache; bWT, before withdrawal treatment; aWT, after withdrawal treatment; EM, episodic migraine.

Painful (CPT) session
In the MOH patients bWT the CPT induced a non-significant change in the TST (F(2,60) = 2.916, P = 0.062, η2 P = 0.100) and Area (F(2,60) = 4.133, P = 0.021, η2 P = 0.133) compared with the control sessions (all P > 0.01). Conversely, the CPT in the MOH patients aWT induced a significant change in both the TST (F(2,60) = 9.444, P = 0.001, η2 P = 0.268) and the Area (F(2,60) = 11.169, P = 0.001, η2 P = 0.339) when compared with the control session (all P < 0.01), the values obtained resembling those of the EM patients and the controls (Table 4; Figure 2).
During the CPT, a lower TST increase was found between the MOH patients bWT and both the EM patients and the controls (Figure 2); this difference was highly significant.
The MOH patients aWT showed a significantly higher TST increase during the CPT compared with the MOH patients bWT (Wilcoxon, Z-2.875; P = 0.004), while no significant differences emerged between the MOH patients aWT and either the EM patients or the controls (Figure 2).
The Area values showed a lower reduction during the CPT in the MOH patients bWT and aWT compared with the controls; however, these differences did not reach statistical significance; similarly, no significant difference was found between the EM patients and the controls (Table 4).
No significant changes in the NRS scores corresponding to the 1.2xTh and TST (at the fifth stimulus) were found in the MOH patients bWT (F(2,60) = 2.258, P = 0.114, η2 P = 0.080; F(2,60) = 2.058, P = 0.137, η2 P = 0.073, respectively) and aWT (F(2,60) = 2.746, P = 0.083, η2 P = 0.198; F(2,60) = 2.924, P = 0.062, η2 P = 0.122, respectively) during the painful compared with the non-painful (control) session (all P > 0.01) (Table 5). Conversely, the EM patients and controls showed a significant reduction in the NRS scores corresponding to the 1.2xTh (F(2,54) = 24.259, P = 0.0001, η2 P = 0.714 and F(2,44) = 12.190, P = 0.0001, η2 P = 0.372, respectively) and TST (at the fifth stimulus) (F(2,54) = 9.291, P = 0.007, η2 P = 0.653 and F(2,44) = 13.958, P = 0.0001, η2 P = 0.407, respectively) during the CPT compared with the control session (all P < 0.01) (Table 5).
During the CPT, the MOH patients bWT showed a lower reduction in the NRS scores corresponding to both the 1.2xTh and the TST (at the fifth stimulus) compared with the controls (Table 5). No significant differences in NRS scores were found between the MOH bWT and the MOH aWT patients.
After-effect session
In the after-effect session no significant differences were found between the MOH patients (bWT and aWT), the EM patients and the controls in either the TST or the Area values.
After the CPT session, the NRS measurements at the 1.2xTh and TST of the NWR did not show statistically significant differences between the patients and the controls (Table 5).
Discussion
The main findings of this study were the following: (i) the stimulation intensity needed to evoke the TST of the NWR was markedly reduced in MOH patients, being significantly lower than that needed both in controls and in EM patients; (ii) the Th and the Area of the NWR evoked by a single stimulation were significantly abnormal in MOH patients; (iii) the psychophysical measurements (NRS scores) revealed enhanced pain perception following single and repeated stimulation and increased temporal summation of pain (albeit barely reaching statistical significance) in MOH patients, showing a pattern similar to that of the neurophysiological abnormalities; (iv) these abnormalities of both neurophysiological (NWR and TST of the NWR) and psychophysical (NRS scores) parameters tended to improve in MOH patients after withdrawal treatment; (v) the CPT activating the DNICs did not produce any significant effect on the neurophysiological (TST and Area) or psychophysical (NRS scores) parameters in the MOH patients bWT, although a significant normalisation was observed aWT; and (vi) the TST of the NWR was also found to be significantly lower in the EM patients compared with the controls.
We suggest that these results might be explained by the presence of widespread abnormal spinal cord pain processing in MOH patients, which may be, at least in part, underlain by defective functioning of the supraspinal control of pain.
Furthermore, the fact that in MOH patients both the neurophysiological and the psychophysical responses, as well as the activity of the DNICs, improved after the withdrawal treatment, allows us to hypothesise a link between medication overuse, defective functioning of the DNICs, neuronal hyperexcitability in pain pathways and chronification of migraine. However, because a facilitation of spinal cord pain processing was also detected in the EM patients, we hypothesised that the heightened sensitivity to incoming sensory signals (painful stimulation) observed in our MOH patients is already present, albeit to a lesser degree, in EM patients during the interictal period and that this could represent a predisposing factor making migraineurs, or a subgroup of them, more prone to developing chronification of headache after medication overuse.
The increased excitability of the spinal nociceptive system found in the MOH patients and reflected in the reduced TST of the NWR and the increased psychophysical temporal summation of pain, is in line with the results of previous studies that found a facilitation of the temporal summation of pain after electrical stimulation of extracephalic regions in patients with transformed migraine with medication overuse (2) and in patients with chronic tension-type headache (CTTH) (26). Furthermore, generalised pain sensitivity at the extracephalic level seems to be a common feature in patients with chronic pain, having also been demonstrated in patients with fibromyalgia and whiplash syndrome, who also showed a reduced TST of the NWR coupled with increased temporal summation of pain (24,28).
In our study, the abnormal excitability of the spinal nociceptive system in the MOH patients was further confirmed by the reduced NWR Th, the increased NWR Area and the increased related psychophysical pain sensation detected following single painful stimulation in this group compared with EM patients and control subjects. These data confirm previous reports of abnormal NWR parameters in primary chronic headaches (33–36) and are in line with more recent studies in which extracephalic facilitation of laser evoked potentials (4), of pain-related evoked potentials (3) and of the related pain sensation was found in patients with MOH.
Another noteworthy finding of this study was the reduced ability of the MOH patients to activate the DNICs following the CPT. This dysfunction was widely evident, being detected at neurophysiological (TST and Area of the NWR) as well as at psycophysical levels (temporal summation of pain) and it suggests a defective supraspinal control of pain in MOH patients. Interestingly, abnormal functioning of the DNICs has also been demonstrated in chronic pain conditions without medication overuse, including CTTH (36,37) and fibromyalgia (38).
Accordingly, the sensitisation of the pain pathways linked to a defective supraspinal control of pain could be seen as a common trait in patients with chronic forms of pain or who are prone to developing chronic pain. The role of the defective supraspinal control of pain and of dysfunction of the DNICs in the pathophysiology of MOH and of generalised sensitivity to pain can only be hypothesised.
In animal models it has been shown that several brainstem structures, including the periaqueductal grey, rostral ventromedial medulla, nucleus raphe magnus and nucleus reticularis giganto cellularis (8), can exert modulatory influences on spinal cord activity. The periaqueductal grey and dorsal raphe contribute strongly to endogenous analgesia through the rostral ventromedial medulla, which directly inhibits trigeminal and spinal dorsal horn pain transmission (8–10,12). On other hand, there is growing evidence that stimulation of nociceptors activates a spinobulbospinal loop, exerting an opposite descending facilitatory influence (rostral ventromedial medulla-mediated) on the nociceptive spinal neurons (9). Similarly, in humans, the DNICs represent a descending inhibitory system acting to modulate pain perception via structures located in the caudal medulla, including the subnucleus reticularis dorsalis, which exert their inhibitory action on wide dynamic range neurons via descending pathways in the dorsolateral funiculi (39), but it is known that DNIC circuits may also exert a facilitatory influence on dorsal horn nociresponsive neurons (39,40). All these descending inhibitory and excitatory inputs have been shown to contribute to the development and maintenance of the wind-up phenomenon and the central sensitisation of nociceptive spinal neurons (8,39). On these bases, an imbalance of excitatory and inhibitory mechanisms could variably contribute to the development and maintenance of chronic pain conditions and could, together with the inter-subject variability, contribute to the prevalence of facilitatory or inhibitory influences following acute noxious stimulation. This might explain our previous report, in which the CPT performed in a sample of migraine and CTTH patients gave rise to facilitation of the NWR responses (36), rather than the reduced or absent inhibition of the nociceptive responses observed in the present study. Further studies are thus needed to clarify the clinical and temporal factors that could drive these different phonotypical manifestations of the dysfunctional control of pain.
Changes within the above-mentioned descending pain modulatory network are known to play a role in the pathophysiology of chronic pain (10) and in functional pain disorders, including primary headaches (5,12–14). In MOH patients, descending influences from brain regions belonging to the pain network that elicit inhibition or facilitation of nociceptive transmission via brainstem structures are found to be hypometabolic and rapidly revert after withdrawal treatment (7). In addition, in rats, daily exposure to antimigraine medications, such as triptans and analgesics, caused a significant down-regulation of the serotonin receptors, 5-HT1B/D and 5-HT1B/2/3, respectively, in various subcortical regions, including the periaqueductal grey and the locus coeruleus (41,42). Thus, overuse of a triptan or an analgesic can significantly interfere with anatomical structures, in particular serotoninergic descending inhibitory pathways, such as the periaqueductal grey, locus coeruleus, rostral ventromedial medulla and subnucleus reticularis dorsalis, which are involved in the processing of nociceptive signals, leading to a functional change in the central antinociceptive system.
On the basis of the above data we propose that the abnormal nociceptive spinal cord excitability observed in our MOH patients may reflect an abnormal balancing of nociceptive and antinociceptive mechanisms in subjects with a high frequency of migraine attacks in which medication overuse is an additional aggravating factor.
Indeed, we observed a significant recovery (increase) of the TST values in MOH patients after withdrawal treatment, including the rate of DNIC inhibition, suggesting that the abnormal excitability of the spinal neurons may have been reversed by the medication withdrawal. However, the recovery of the TST values, after the withdrawal treatment, was only partial, these values remaining significantly lower than those of the control subjects.
Other important findings of this study were the abnormal neurophysiological (TST and Area) and psychophysical (NRS scores during the temporal summation of pain) parameters detected in the EM patients as compared with the healthy controls, possibly indicating abnormal spinal excitability to repeated nociceptive inputs between attacks. Because EM patients do not complain of allodynia or hyperalgesia while pain-free, we propose that their spinal cord is in a state of subclinical hypersensitivity. This hypersensitivity or suballodynic condition, as Weissmann-Foegel et al. (25) defined it, could be related to a dysfunction in central antinociceptive pathways. In this regard, it has been reported that P/Q-type and N-type Ca2+ channels, which are involved in spinal hyperalgesia, are expressed in structures such as the periaqueductal grey and the rostral ventromedial medulla (43). Migraine patients could have a genetic substrate that predisposes them to dysfunction of the supraspinal pain control system acting both on trigeminal and on spinal neurons. Such chronic hyperexcitability may contribute to their susceptibility to repeated migraine attacks. This last finding is in accordance with recent studies that have shown a generalised hypersensitivity involving the sensory pathways in migraine during the interictal phase at both the cephalic and extracephalic level (25,44–46).
We speculate that high frequency of migraine attacks and overexposure to medication contribute to a further impairment of antinociceptive control structures, leading to the progression from EM to a MOH form.
In the present study, we found that the NRS score changes paralleled, only in part, the neurophysiological ones. This indicates that pain perception, too, is abnormal in MOH and EM patients, confirming the finding of hypersensitivity to painful stimulation, but the psychophysical response, in this case, seems to indicate a lower sensitivity in detecting changes in spinal cord pain processing. However, these results should be considered with due caution, because this study was not designed specifically to evaluate the psychophysical aspect of the temporal summation of pain.
In conclusion, the facilitation of spinal cord pain processing, the defective function of the DNICs and the improvement of these parameters after the withdrawal treatment in MOH patients, strongly suggest a link between medication overuse and abnormal modulation of pain perception and, at least in part, chronification of migraine. The TST of the NWR, which is also able to detect subclinical alterations in EM patients during the pain-free period, seems to be a useful tool for exploring the integration and processing of pain at the spinal level.
