Abstract
Background
Physiological studies have been determinant for the understanding of migraine pathophysiology and the screening of novel therapeutics. At present, there is no animal model that translates fully the clinical symptoms of migraine, and generally these studies are conducted on anesthetized animals.
Methodology
Pain as well as non-painful symptoms such as photophobia, need to have a conscious individual to be experienced; therefore, the new development and adaptation of behavioral assays assessing pain and other non-painful symptomatology in conscious animals represents a great opportunity for headache research and it is exciting that more and more researchers are using behavioral paradigms.
Summary
This review will describe the different behavioral models for the study of headache that are performed in non-anesthetized conscious animals. The pearls and challenges for measuring hypersensitivity in rodents such as the common tests for measuring mechanical allodynia and thermal hyperalgesia have been the landmark for the development of assays that measure hypersensitivity in the craniofacial region. Here we describe the different behavioral assays that measure hypersensitivity in the craniofacial region as well as the established behavioral models of trigeminovascular nociception and non-nociceptive migrainous symptoms.
Conscious behavioral research – historical context
The representation of the clinical symptoms needed to fulfill the diagnostic criteria that headache comprehends (1) is one of the biggest challenges that headache research faces in its quest for the creation of more suitable animal models for the study of headache. At present, there is no animal model that translates fully this multi-symptom disorder, and most of the established animal models for the study of migraine are based on the activation of nociceptive trigeminovascular pathways, resembling the physiological correlates of the pain perception when the animal is unconscious during anesthesia (2).
For the context of general pain, pain research has used for many years behavioral assays to study and assess pain in animals. Researchers are always seeking better and more accurate ways to assess behavioral nociceptive responses that translate, or intend to translate in a better way, to the human pain experience, yet headache research has concentrated predominantly on physiology and anatomy. However, as we have understood more about the physiology of the trigeminovascular system and with the onset of transgenic mice technologies, mechanisms to assess pain in rodents, and better and more novel ways to measure responses of pharmacological agents that reflect the human condition, behavioral assessment as a tool has become paramount. The assessment of nociceptive responses in the craniofacial region in rodents is challenging but necessary. Behavioral assays have been adapted or newly developed for the assessment of pain in the craniofacial region, and these assays represent a great tool for headache research as well.
While physiological studies were crucial in preclinical screening and understanding the mechanism of action of the 5-HT1B/1D agonists, “triptan,” and calcitonin gene-related peptide (CGRP) receptor antagonist class of drugs, and validating the use of some preventive drugs and the potential of novel compounds, these studies have their limitations. In general they are conducted on anesthetized animals, and it is accepted that most good anesthetics will dampen, to some extent, the central nervous system. Given that today it is believed that migraine and many primary headache disorders may be a consequence of dysfunction within the brain (3), resulting in a multi-symptom disorder, and perhaps best described as neurovascular disorders, having an animal model that has to some extent a compromised central nervous system may confound responses to nociceptive inputs, as well as responses to pharmacological agents.
One thing that we share with animals regarding pain research is that the pain experience is subjective and we identify pain based on behavior (4), and for pain as well as for other nonpain-related symptoms such as photophobia to be experienced requires a conscious individual. This review will describe the different behavioral models for the study of headache that are performed in non-anesthetized, conscious animals. The majority of studies conducted in the headache literature have focused on the mechanisms and measurement of hypersensitivity, which includes allodynia, the perception of pain in response to a stimulus that is normally considered innocuous in nature, and hyperalgesia, which is hypersensitivity to a known noxious stimulus. Finally, this review will address some of those behaviors that are not considered nociceptive specific, such a photophobia, nausea, and vomiting.
Of rats and mice in behavioral research
The rat (R. norvegicus) and mouse (M. musculus) belong to the family Muridae (5), and the divergence of both lineages happened approximately 12 millions of years ago based on fossil evidence (5,6). Therefore, it needs to be recognized that even though these two species of rodents share similarities, they are different in genetics, physiology, and behavior.
Historically, rats have been the most commonly used mammal model for the study of pain. They are bigger, easier to handle, manage handling well, and most of the existent pain behavioral assays have been developed using them. Mice have more affordable maintenance, they can be housed in groups of four or five in each cage, they are easier to breed and importantly, the use of mice in biomedical research has gained great popularity thanks to the development of transgenic mouse technology (7**). Therefore, to take full advantage of this technology, it is critical that the behavioral assays performed in rats be adapted to mice, but this is not an easy task. In addition, mice are very susceptible to stress produced by habituation, handling and housing that potentially could produce stress-induced analgesia (7**–9). It has been recommended to avoid repeated handling of mice because they can become more agitated, and some studies also have shown an increase in glucose levels when this happens (10). In our experience, handling the mice from the cage to the test room or test device in a noisy and accelerated manner can exacerbate agitation, but handling and petting the mice for a few minutes in the experimenter’s hand close to their cage and letting the animal walk around the hand and sniff around has shown to be quite the opposite.
To choose which species to work on depends on the question we want to answer as well as the experimental paradigm. Both species have their advantages and disadvantages and have been proved to be reliable and indispensable subjects for biomedical research.
Measurement of hypersensitivity in behavioral research
Activation of nociceptive systems can generate alterations in the somatosensory system, producing amplified responses and increasing the sensitivity to normally innocuous inputs (11). It is generally believed this is a consequence of sensitization of neural pathways and is called allodynia. Cutaneous allodynia has been described to be present in up to nearly 80% of migraineurs (12,13), and patients can develop it in the face, head, neck, and extending beyond the craniofacial region to extracephalic areas (14–16). Preventing the development of allodynia is of critical importance since its presence typically makes the migraine headache refractory to triptan therapy (14,17). In animal models of migraine, diverse methods are used to induce sensitization in the trigeminovascular system to recreate this allodynic state.
Allodynia can be measured by mechanical and thermal sensitivity tests. These tests have been described primarily to measure mechanical allodynia in extracephalic areas such as the hindpaw and the tail. The descriptions of these assays are essential not only because they are used for the evaluation of extracephalic allodynia in headache research, but because they are the foundation of the assays that evaluate allodynia in the craniofacial region.
A common way to measure mechanical allodynia is with the use of von Frey monofilaments (Figure 1). These are a series of calibrated nylon fibers of diverse thickness identified by different force values. These filaments exert several degrees of pressure when applied to the surface that is tested. The paw withdrawal assay using the von Frey test is the most common method to measure mechanical allodynia. In a traditional von Frey test, mechanical force is exerted via application of a particular monofilament to the surface of the rodent paw until buckling of the monofilament occurs in approximately two to four seconds. Plexiglas boxes with a wire mesh floor are the commonly used apparatus to allow their application. Specifically, the elevated wire mesh floor supports a rat or a mouse while the monofilaments are applied to the hind paw. Before testing, animals need to be acclimated to the new environment by placing the animals into the testing boxes in the behavior or testing room for several sessions. The test should be performed only when the animal is stationary and standing on all four paws after the animal’s habituation (this usually takes about 30 min) or as soon as the animal stops exploring.
Von Frey monofilaments. A. Von Frey monofilament bulging. B. Von Frey monofilament set.
Usually, a paw withdrawal upon probing or immediately upon release of the filament is considered a positive response (18). However, responses are not always clear-cut and their interpretation may vary between investigators. Most commonly, the filaments are applied to the central region of the plantar surface, but probing of the lateral or dorsal surface of the paw also has been performed. There are various paradigms to assess and define mechanical thresholds (18**–20).
This test is influenced by subjectivity, thus making it difficult to compare results obtained from different studies. It is important that the experimenter who performs the behavior test be blinded to the treatment groups. Moreover, bending forces of the monofilaments are influenced by the experimenter, ambient humidity and, to a lesser extent, temperature. To ensure constant pressure from a single monofilament, the tip of the monofilament needs to stay vertical and in full contact with the application surface, as variations in contact surface area will give rise to inconsistent pressure from a given von Frey filament. Finally, filaments may wear off with extensive use (21,22). The value of each monofilament needs to be calibrated frequently by measuring the magnitude in grams exerted by the monofilament when applied to an electronic balance. This needs to be performed because a slight fluctuation in the value of a filament may occur after each use. Moreover, repetitive testing with short intervals may bias mechanical threshold determinations (18**) and possibly induce sensitization.
The time and labor intensive nature of the traditional von Frey test has been reduced significantly by the availability of an automatic von Frey system by which the force of the probe increases continuously until a withdrawal response occurs (23). However, the system comes with probes that differ in tip diameter and stiffness, which are usually not mentioned in study reports. Reporting the tip specifications is strongly recommended, as pressure is defined as force divided by the contacting surface area; the same force will result in different pressure with different tip size.
The tail pinch assay is far less commonly used compared to the paw withdrawal test. This test is similar to the pricking pain test done during a neurological exam in a clinical setting. Animals are restrained in a restrainer with their tails out. A tail pincher that permits the application of increasing measurable pressure is applied to the animal’s tail. The measured parameter is the threshold (weight in grams) for the appearance of a given behavior. When the pressure increases, one can see the reflex withdrawal of the tail, and it is often accompanied by a vocal reaction. Compared to the paw withdrawal assay, there is a bigger individual variation in withdrawal thresholds. The tip of the tail is so delicate that it can be injured easily by the pinch applicator. Like the paw withdrawal assay, repetitive measurements with short intervals will sensitize the animals. The threshold also varies greatly by the position of the pincher on the animal’s tail. It is important to make sure that the applicator is applied to the same location of the tail during each measurement for consistent results.
The Hargreaves’ test used to measure cutaneous hyperalgesia to thermal stimulation in unrestrained animals was first described by Hargreaves et al. (24**). The test apparatus is composed of a moveable heat source, a glass pane onto which the animal enclosure will be placed, and a controller. Animals are placed into clear plastic or glass compartments. After acclimation, the heat source placed under the glass floor is positioned by the operator directly beneath the hind paw or the tail. When the animal feels pain and withdraws its paw or tail, the heat source switches off and the reaction time counter stops. The withdrawal latency to thermal stimulation is automatically determined.
The advantage of paw withdrawal over tail is that it allows independent assessment of either side of the body. Variation in the temperature of the glass that mice are sitting on will lead to inconsistent readings. If mice are placed on cold glass, their response to thermal stimulation will be delayed. It is recommended that a glass with built-in heating elements be used for consistent temperatures and measurement. In addition, the heating source needs to be calibrated from time to time to ensure the thermal stimulus delivers the same heat intensity. Variation in readings will also be affected by the location of where the beam projects to; the readings will be smaller if the beam is aimed at the toes, and will be larger if the beam is aimed at the heel. Extensive habituation similar to the von Frey test is required before data can be collected. Sleeping or grooming of animals will also render altered readings. Another major confounder of the test is that urine and feces tend to build up in the glass floor, and cannot be cleaned without disturbing the animals during the test.
Measurement of hypersensitivity in the craniofacial region
Measurement of allodynia in the craniofacial region in an awake animal is based on important electrophysiological studies (25–28) performed in anesthetized rats that modeled the dural craniovascular activation that is thought to occur in neurogenic inflammation (29). These studies have demonstrated that central sensitization of the trigeminovascular system induced an allodynic state, with increased neuronal sensitivity to innocuous mechanical stimulation of the dura mater as well as increasing the cutaneous receptive field that included not only the periorbital (ophthalmic) area but causes spreading beyond this to the maxillary and mandibular regions (27). The activation of dural nociceptors is attained by applying an inflammatory soup that consists of a mixture of bradykinin, serotonin, prostaglandin E2, and histamine (25,29). This inflammatory soup should be always of an acidic pH to induce sensitization of the meningeal nociceptors (25,30,31).
The measure of pain in such a small mammal can be challenging (7**) and much more when the source of pain is localized in the craniofacial region. The assessment of thermal and mechanical nociceptive thresholds that resemble allodynia and hyperalgesia in the craniofacial region may involve the measurement of the head withdrawal latency from the bending von Frey filament or heating source (32,33**). The head withdrawal may be accompanied by face-rubbing strokes directed to the area of testing and/or a quick withdrawal of the head or vocalization (33,34**–36). These tests are the modification of the hind paw withdrawal latency test used for the measurement of mechanical allodynia and hyperalgesia described above (37,38**).
To have access to the craniofacial region, the animal is placed in a rodent restrainer or plastic tube. An effective restrainer should allow easy access for the rodent, and does not cause harm to the animal, while preventing or making it difficult for the animal to turn around. In mice, it is recommended that the restrainer be inverted at an incline of a 30-degree angle since mice prefer an upward facing position, which facilitates rapid entry into the testing position and prevents mice from turning around during the test (39**). As described above, it is very important that the animals get acclimated to the testing environment before the assay starts. This can be accomplished by placing the animal in the restrainer in the behavior or testing room for several sessions. The entrance of the animal to the restrainer should be without force, allowing the animal to recognize and adapt to it without stress. Rodents have an exploratory nature; the acclimation period should be short and sufficient to allow the animal to finalize its “exploring” and to find a stationary position to start testing. It is recommended that restraint time be less than 15 minutes, allowing five to 10 minutes for the animal to acclimate and no longer than five minutes for testing (39**).
The test consists of determining the pressure thresholds using the von Frey monofilaments applied to the periorbital region of the rodent on the right and left side of the face over the rostral portion of the eye or midline of the forehead (35**,36,39**,40) (Figure 2). The mechanical threshold is determined using an up-down method developed by Dixon, whereby a positive response is represented by withdrawal of the head, shaking of the head or rubbing of the area directing the use of a weaker stimuli (lower force) (see videos 1 and 2). A negative response represented by a non-withdrawal of the stimuli directs the use of a stronger stimulus (higher force) (18**,33,34**,35**) (see video 3). Moreover, as described above, repetitive testing with short intervals should be avoided since it can cause errors in the mechanical threshold readings (18**) and possibly induce sensitization.
Mouse forehead and periorbital regions. Image indicating forehead and periorbital regions in mice where the von Frey pressure stimulus is applied.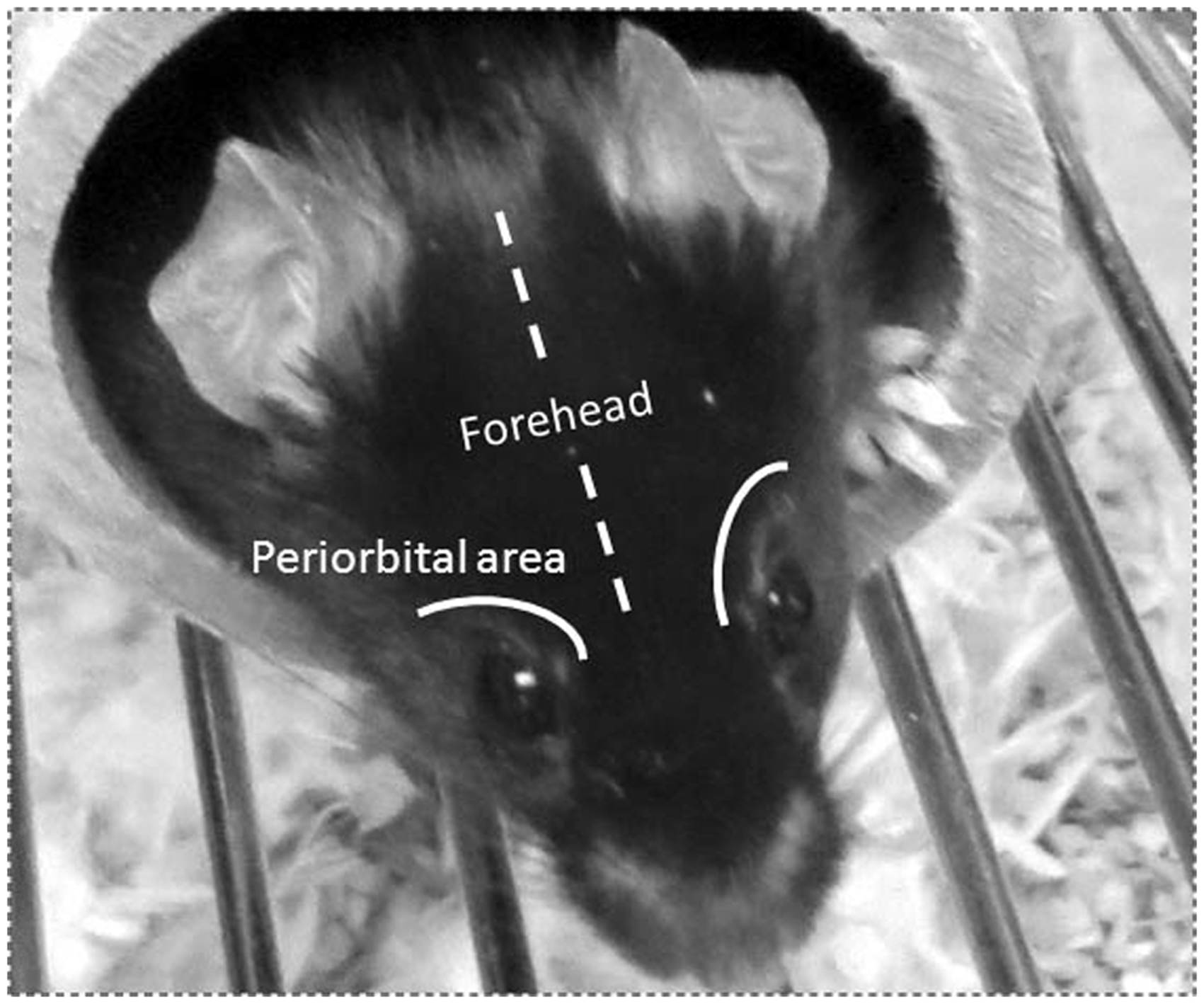
Another adaptation to measure allodynia in the craniofacial region is the use of an orofacial sensitivity assay that has been described and used in a model of chronic migraine (41**). This assay consists of training the rats to reach a bottle that contains a solution with sucrose. First the rats are placed in a behavioral area that has a hole connected to a plastic tube. The rats are trained to cross the plastic tube to reach the sucrose solution. When they perform this action, they need to be still and extend their face to reach the bottle, allowing access for a von Frey stimulation in the periorbital area, perimasseter area, and whisker pad. A positive response is indicated by head flinch or retraction in response to the monofilament stimuli (42). Additionally, a method of assessing nociceptive inputs arising from inflammatory and neuropathic pain in the trigeminal region of mice has described an innovative restraining method (43). This restraining method consists of tying the mouse tail with a wire covered by plastic and attached to a metal support with a magnet. The mouse is then covered with a plastic box that bears a heavy weight on top. The mouse has its head completely out and is accessible for testing. For the nociceptive assessment, von Frey monofilaments were used and air puffs were performed by directing an air current to the whisker pad (43).
There are other models that have attempted to measure allodynia in the facial region after nerve injury in rodents (20,44), and they could be used in headache research. These assays are performed without a restrainer device. In a study performed in rats, the examiner wore a leather glove and rats were habituated by petting them for 30 minutes until they leaned against the examiner’s hand and stood on their hind paws (20). This study can be conducted exclusively in rats since rats are bigger and their craniofacial region is easily accessible after they lean on the examiner’s hand. Mice are smaller and even after petting and cuddling into the examiner’s hand, they are still exploring and moving around, making accessibility to the craniofacial region difficult to test the same region every time. In other studies performed in mice, the threshold measurements were conducted while the animal was being held on the examiner’s hand (44). Mice are innately nervous creatures, so this assay would induce an incredible amount of stress not only for the mouse but for the experimenter as well, which potentially could induce misreadings.
Measurement of spontaneous nociceptive behaviors in the craniofacial region
The quantification of pain in rodent models, as has been described, has traditionally used withdrawal responses to an evoked mechanical or thermal stimulus induced by the experimenter for the measurement of hyperalgesia and allodynia (45). However, the responses of animals and humans to an evoked stimulus are only part of the pain experience and provide only an indirect measure of the subjective experience of pain. Indeed some researchers believe that spontaneous, non-evoked pain is a far more reliable assessment of the pain experience in patients (46). Certainly in primary headaches it is the spontaneous pain in the head and cervical region that define these disorders, while hypersensitivity to external stimuli is considered an additional symptom not present in all patients. Models that measure spontaneous nociceptive (nonevoked) behaviors have been developed and represent another method to further characterize the subjective pain experience.
Assessment of spontaneous behaviors in rodents requires a freely moving animal as well as video recording and extensive analysis. In freely moving rodents, it is known that injury in the facial region results in alteration in the facial grooming patterns, and spontaneous behaviors become more persistent and directed to the area of injury (33**,47–50), whereas unilateral non-nociceptive facial stimuli do not produce this change in facial grooming (33**,34**,49). Determining reliable measurements of spontaneous behaviors in the head and facial region represents a significant future challenge for headache researchers, and probably lies in measuring alterations to normal behaviors that the animal unconsciously performs. Several recent studies have started to determine changes in the animals’ normal behavior directed to the head, and include changes to facial grooming and facial grimacing, which may be used in the future to assess pain similar to headache in experimental animals. As described above, grooming patterns are thought to be altered in response to pain in the facial region. A recent study describes alterations in facial grooming in response to an inflammatory injury in the orofacial region in freely moving animals (51**). In response to inflammatory pain in the mandibular region, mice increase short, vigorous, and repetitive rubbing or washing strokes to the head with their forepaw either unilaterally to the affected area or even bilaterally (see video 4). Mice also demonstrated repetitive rubbing of the inferior (lower lip skin) and lateral mandibular regions (cheek) against the bottom or side of the enclosure and scratching of the affected area with their hindpaw. These behavioral responses were blocked by morphine (51**). These responses are specifically directed to the lower region of the face where the pain is perceived, but they could be extrapolated for their use in future studies to other regions of the head such as periorbital, occipital, and cervical regions where pain related to a migrainous stimulus may be present. Another assay recently developed has coded the behavioral changes of facial expressions in response to noxious stimuli, called the mouse grimace scale (MGS) (52**). This scale involves several facial expressions recorded by a video camera while mice were experiencing pain. The coding comprehends facial expressions such as orbital tightening and cheek and nose bulging. In terms of migraine research, this scale was used to characterize further the phenotype of the Cacna1a knockout mice, which bear the human familial hemiplegic migraine, type 1 R192Q missense mutation that encodes the protein responsible for the gain-of-function effects on the neuronal voltage-gated Cav2.1 calcium channel (53). These mice were placed in Plexiglas cylinders and video recorded. They presented increased baseline MGS scores, with the authors speculating that confinement in the cylinder may have triggered a migraine. Interestingly, this MGS score was reversed by rizatriptan, implying there may be endogenous pain in these mice (52**). This also provides a good example of freely moving animals having an advantage over restrained animals, which may add a potential bias when measuring spontaneous pain and discomfort.
Behavioral models of trigeminovascular nociception
It is thought that primary headaches, including migraine, involve nociceptive activation of the trigeminovascular system (54). The trigeminovascular system includes the peripheral afferent projection from the pseudounipolar trigeminal ganglion to the craniovasculature, including the nociceptive-specific dura mater, and the central afferent projection to the trigeminal nucleus caudalis. In humans it is known that pain can be generated from stimulation of the cranial structures, and this pain is referred to the head (55,56). Many preclinical research methods into primary headaches have concentrated on nociceptive activation of the trigeminovascular system via stimulation of the dural vasculature in anesthetized animals, which are reviewed in detail in separate articles in this special issue. For the purpose of migraine research, the models described above have been adapted to measure the allodynic state in the craniofacial region after activation of the dural nociceptors by applying an inflammatory soup (25,29).
A rat model of episodic trigeminovascular nociceptive activation in conscious animals has been developed using activation of the dural meningeal nociceptors to induce dural inflammation, using a local inflammatory soup infusion. This assay proposes to model the hypersensitivity that patients with recurrent headache experience (12,35**). The study elegantly used the implantation of a cannula through the skull to infuse the inflammatory soup onto the dura mater at different time points/week. Tactile sensory testing was then performed in a plastic tube restraint, and the threshold was noted when the application of a von Frey monofilament in the periorbital region elicited a head retraction or a washing stroke directed to the face with the ipsilateral paw. Up to three days of repetitive infusion in rats produced a significant decrease in the periorbital threshold determined by tactile sensory testing; however, when the daily dural infusion was terminated the tactile responses returned to baseline. If the daily dural infusion was continued beyond three days, there was a continued decrease in bilateral periorbital pressure thresholds determined by tactile sensory testing (35**). Further, if these rats continue to receive the daily inflammatory soup infusions and are also injected with the nitric oxide (NO) donor, glyceryl trinitrate (GTN) (35**), known to be a trigger of migraine in migraineurs (57), it demonstrates prolonged bilateral periorbital pressure thresholds determined by tactile sensory testing for several hours (35**). The authors argue that this assay may be used to explain the mechanism for the development of chronic forms of headache from episodic disorders.
In a similar study using a single application of the inflammatory soup to the dura mater of rats, observations were made of the time course of cephalic and extracephalic allodynia to explore the time-related cephalic and extracephalic allodynia that patients experience during a headache attack (36). Measures of sensory thresholds in the periorbital region as well as the hindpaws as a measure of extracephalic allodynia were made. The cephalic and extracephalic allodynia was maximal after three hours but was significantly reduced in the presence of sumatriptan, naproxen, and a CGRP receptor antagonist.
Furthermore, the same model was used to explore the mechanism of ethanol and its metabolites in hangover headache (40). Ethanol treatment decreased the sensitivity to periorbital pressure thresholds for the first two hours, but it was increased four to six hours later in sensitized rats with inflammatory soup. Acetate administration in sensitized rats induced hypersensitivity significantly, decreasing their sensory thresholds (40).
In addition, another study developed a preclinical model of chronic migraine similar to the previous reported models (35**,36) with the use of repetitive infusions of inflammatory soup with the help of a cannula. The allodynic state was measured in the periorbital and perimasseter area but with the use of the orofacial sensitivity assay described above. Interestingly, female rats showed a decrease in mechanical nociceptive thresholds in the periorbital area versus the male rats, in addition to a reduction in locomotor activity (41**).
NO donors, such as GTN, are well known research tools in primary headaches, known to reproduce a migrainous phenotype as classified by the International Headache Society (IHS) (1) in migraineurs; many hours after administration (58,59) and in animal studies, NO donors have been shown to induce allodynia (38**,60). GTN injection in mice induced mechanical and thermal allodynia that was reversed by sumatriptan injection (38**). The assessment of allodynia was measured only in extracephalic areas as indicated by hindpaw withdrawal; however, the decrease in nociceptive thresholds correlated with increased neural activation in the trigeminocervical complex as shown by increased Fos expression.
Behavioral models of non-nociceptive migrainous symptoms
While primary headache disorders are most commonly associated with pain in the head and cervical region, these disorders, particularly migraine, are also classified by their accompanying symptoms, which may not be specifically pain related, or may be symptoms that exacerbate the pain perception in patients during an attack rather than being pain-provoking themselves. Photophobia is part of the symptomatology present during migraine (61) and is described as pain worsening in response to light, or the light may indeed be painful in itself. Hence there is a tendency for migraineurs to often seek a darkened room during an attack. It has been shown that bright light can activate neurons in the trigeminal nucleus caudalis (62). Moreover, it has been described that a novel pathway from retino-thalamo-cortical neurons may be responsible for exacerbation of migraine by photic signals (63).
Recently it has been demonstrated that a specific aversion to light can be demonstrated in rodents. For the study of light-aversion behavior, a chamber with two compartments is needed. One compartment is dark and closed and the other is light and open. The study involves evaluating the preference of the animal to stay in either one of these two compartments. This preference (behavior) can be recorded, and the total time the animal spends in the light versus dark compartment is quantified (64).
Until now only one behavioral model has been proposed to study this migraine symptom. The Nestin/hRAMP1 mouse is a conditional transgenic model with increased expression of the human receptor activity-modifying protein 1 (hRAMP1) (65**) that forms part of the CGRP receptor complex. These mice have increased sensitivity to CGRP and express selectively the hRAMP in the central nervous system (65**). The authors tested the hypothesis that increased CGRP produces light-aversive behavior in mice that could be a surrogate of photophobia in humans (66). Nestin/hRAMP1 mice presented a strong light-aversive behavior that is enhanced with the administration of CGRP and blocked with olcegepant administration.
Pain aggravated by or causing avoidance of physical activity is one of the diagnostic criteria in migraine (1). Mice have an innate exploratory behavior. Rearings are an action represented when a mouse stands completely erect on its hind legs and is an indicator of motor activity and exploratory behavior (66,67). Interestingly, CGRP-treated Nestin/hRAMP1 mice presented a reduction of locomotion and rearing activity when they were in the dark compartment as if they were intentionally avoiding any physical activity (65**,66), which is particularly relevant as mice are by nature nocturnal. The measurement of locomotion can be measured by the open field test. This test is an open area used to measure behavioral responses such as locomotor activity, hyperactivity, exploratory behaviors, and anxiety (68). In the chronic migraine model described above, in addition to measuring allodynia with the orofacial sensitivity assay, they also measured the difference in locomotion between sexes (41**). Female rats presented a decrease in distance traveled, low mobility, and increased spatial confinement in comparison to the male rats after dural infusion of inflammatory soup when tested in the open field. In addition, females presented a greater range of induction of the CGRP pathway than males. Interestingly, both infusions of either inflammatory soup or phosphate-buffered saline induced changes in CGRP-related genes but were unrelated to the behavioral changes. The authors suggested that the CGRP pathway may respond to changes in intracranial pressure and that the behavioral changes may occur after meningeal inflammation (41**).
Behavioral models of nausea and vomiting
Nausea and vomiting are very distressing symptoms presented during a migraine attack; however, rats and mice are incapable of vomiting and for these types of studies other species have been used (69). Some paradigms of condition taste aversion used in rats have shown a suitable alternative for the study of nausea and vomiting. In these studies, rats learn to avoid a taste paired with toxins that produces nausea (70,71). Moreover, in addition to ingestive behavior disturbances, an association between pain and loss of appetite has been documented in migraine (72). Rats presented loss of appetite after sensitization of the trigeminovascular system by dural stimulation with an inflammatory soup. This finding could implicate a loss of appetite provoked by the pain (73) or a possible result of the brainstem and diencephalic activation causing taste aversion. Certainly future studies would be to use taste aversion and appetite paradigms in the genetic migraine mouse models that more accurately represent the migraine genotype to determine if they also fully exhibit this migrainous phenotype.
Pearls and pitfalls of behavioral research in the craniofacial and extracephalic regions
For all behavioral tests mentioned in this review, there are rules that apply. Following these rules will optimize the assay as well as the data collection and recording, and ignoring them will likely produce unreliable data that are difficult to interpret. First and of fundamental importance, a stressed and anxious animal will not provide reliable results. It needs to be acknowledged that rodents are nervous by nature and can be easily disturbed; in addition they are calmer when they recognize the environment they are in. Therefore, the room in which the animals are to be tested needs to be as quiet as possible. Most of the time, due to the diverse distribution of the animal facilities, the test room may not be in the animal housing room. A good suggestion is to bring the animals to the test room several times a week before starting a new experiment. Second, the acclimation period is essential in behavioral research; therefore, before testing the animals, they need to be habituated in their testing boxes or restrainers and allowed to explore the area. It is recommended to pet them for a few minutes before placing them in their testing devices to avoid stress, and always be gentle. Third, allow enough time/days to record a reliable baseline. This depends on the experimental paradigm. Fourth, repetitive recordings during a short duration are not recommended because they will sensitize animals to the stimulus. Fifth, always apply the stimulus at the same spot on the animals’ paw, tail, face or periorbital region to avoid different measurements. Finally, the experimenter needs to be well trained in the assay to secure an optimal performance. Moreover, the experimenter needs to be comfortable working with animals. A stressed experimenter will not perform the assay cautiously and could induce stress in the animals. The experimenter needs to be the same person who acclimated the animals, and it is recommended they not wear cologne or perfume to avoid any distraction for the animal. In addition, since all behavioral tests are subjective, the experimenter who carries out the measurement should be blinded to the treatment groups.
Conclusion
Animal models for the study of migraine have primarily concentrated on representing trigeminovascular nociception in the anesthetized animal. However, the pain experience is subjective and, as for other nonpain-related symptoms in migraine, it requires a conscious individual. Translational behavioral models for the study of migraine featured in conscious animals intend to offer a surrogate in the resemblance to human migraine symptomatology. It is exciting that more and more scientists are taking into account the importance of behavioral tests in headache research. These models have been essential in advancing our understanding of migraine pathophysiology and for the development of novel therapeutics and will continue to do so in the future.
Footnotes
Acknowledgements
The authors would like to thank Dr. Vanessa Pardi for her technical support and Dr. Simon Akerman for his assistance in reviewing the draft of this review.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
