Abstract
Introduction
Migraine’s astonishing prevalence and preserved genetic background contrast with the definition of a disease and the biological meaning of experiencing recurrent, severe headache attacks is still puzzling.
Methods
To provide a comprehensive explanation of the migraine evolutionary meaning, we review (i) the putative role of the autonomic nervous system in migraine attacks, (ii) the inter-ictal autonomic, functional, and metabolic signature of migraine patients, (iii) the bio-behavioral perspective of pain, and (iv) the allostatic perception of migraine chronification.
Results
Migraineurs have inter-ictal cortical hyperexcitability and metabolic dysfunction that predisposes to brain energetic imbalance. Multiple precipitating factors may lead to brain energy consumption over the migraine attack generation threshold. In response, the brain engenders adaptive, evolutionary conserved, autonomic-behavior responses through the antidromic activation of the trigeminovascular system. The sickness behavior and severe pain experienced during migraine attacks result in avoiding mental and physical activity, allowing brain energy restoration. Chronic exposure to stressors may result in an allostatic overload, leading to maladaptive chronic activation of these responses. In this bio-behavioral perspective, the chronification of migraine should be envisioned as a pathological process, whereas the migraine itself should not.
Conclusion
Migraine has an evolutionary (Darwinian) meaning.
Keywords
Introduction
Migraine is a complex neurovascular brain disorder characterized by recurrent, severe headache attacks, usually of unilateral and pulsating quality, variably accompanied by nausea, vomiting, photophobia, and phonophobia (1). Distinctive phases characterize the migraine attack, including the prodrome, aura, pain, and postdrome phases. The underlying biological underpinnings of migraine are still incompletely unveiled and involve a complex interplay of environmental, biochemical, epigenetics, and genetic factors (2). The autonomic nervous system arguably plays a neglected but pivotal role in this complex interplay. Different theories have tried to explain the multitude of molecules, neuronal pathways, and clinical manifestations exhibited during migraine attacks. Nonetheless, the astonishing prevalence of migraine, especially among fertility-age women, coupled with a preserved substantial genetic background (3,4), seems in contrast with the definition of migraine as a disease (5). Indeed, familial aggregation of this condition is frequently encountered and the significant maternal hereditary influence suggests a potential pivotal role of mitochondrial and epigenetics mechanisms (6,7). Therefore, a protective mechanism for migraine attacks has been proposed (1,3,8–10), yet, the biological meaning of experiencing recurrent, severe headache attacks is still puzzling (11,12).
To investigate the migraine evolutionary meaning, we reviewed (i) the putative role of the autonomic nervous system in migraine attacks, (ii) the inter-ictal autonomic, functional, and metabolic features of migraine patients, (iii) the bio-behavioral perspective of pain, and (iv) the allostatic perception of migraine chronification.
The role of the autonomic nervous system in migraine
The autonomic nervous system represents a complex neuronal network topographically distributed in the central nervous system, namely the Central Autonomic Network, and the peripheral nervous system (13). The autonomic nervous system collects internal and external information (afferents) to promote coordinated biological responses (efferents) in order to preserve the human body's homeostasis. For this purpose, the autonomic nervous system is densely interconnected with every human organ, modulates autonomic responses, and orchestrates behavioral, neuroendocrine, and neuro-immunological responses. Hence, the autonomic nervous system is crucial to every pathological and physiological human process, including pain. Accordingly, the autonomic nervous system has a complex role in headache disorders, contributing significantly to their development, clinical manifestations, and progression (14).
The autonomic nervous system and the migraine attack
Migraine was previously considered a cerebrovascular disorder, yet, mounting recent evidence supports a much more complex pathophysiology. Indeed, activation and, possibly, sensitization of the trigeminovascular system secondary to a primary brain dysfunction seems responsible for headache generation (15). The trigeminovascular system is a neuronal network composed of upper cervical dorsal root and trigeminal ganglions (first-order neurons), the trigeminocervical complex (second-order neurons), and axonal projections ascending to the midbrain, thalamic and hypothalamic nuclei (third-order neurons) (Figure 1) (1). The structures sensitive to nociceptive stimuli of the head are the dural vessels, sinuses, and meninges innervated by trigeminal terminal ends. Activation of the trigeminovascular system results in the release of several neuropeptides from these terminal ends and trigeminal ganglion, including pituitary adenylate cyclase-activating polypeptide (PACAP), and calcitonin gene-related peptide (CGRP). These neuropeptides are critical actors of migraine pain, as supported by the highly therapeutic efficacy of their pharmacological inhibitors (16,17). The trigeminal fibers may have an orthodromic or antidromic activation (Figure 1) (18). Therefore, the trigeminovascular system may serve the autonomic nervous system as both an afferent and efferent pathway. In secondary headache disorders, the pathological inflammation of cerebrovascular structures results in the orthodromic activation of the trigeminovascular system. Conversely, in migraine, the antidromic release of vasoactive signaling molecules by the trigeminal fibers is postulated, suggesting that a central origin of migraine attacks is possible (18,19). In this context, in migraine patients, the autonomic nervous system might sense a homeostatic imbalance and use an existing defensive neuronal pathway, the trigeminovascular system, to alert the individual through pain. The first- and second-order neurons are likely responsible for the pathological pain mechanisms, while third-order neurons orchestrate the autonomic-emotional-behavioral responses to pain. Noteworthy, the central autonomic network exerts its control at every hierarchical level of the trigeminovascular system functions (20). Indeed, during a migraine attack, the cranial autonomic manifestations are governed by the sphenopalatine ganglion and the superior salivatory nucleus. Additionally, the lethargic and avoidant behavior experienced during migraine attacks is engendered by the trigeminovascular system third-order neurons. These latter neurons also exert control of cerebral blood flow, cortical hyperexcitability, and pain signaling (21). Furthermore, emotional and behavioral prodromes argue for a pivotal role of the autonomic nervous system in the migraine attack (1,3). Accordingly, activation of the hypothalamus, periaqueductal grey, and dorsolateral pons have been observed in the premonitory phase of migraine in functional imaging studies (22,23).
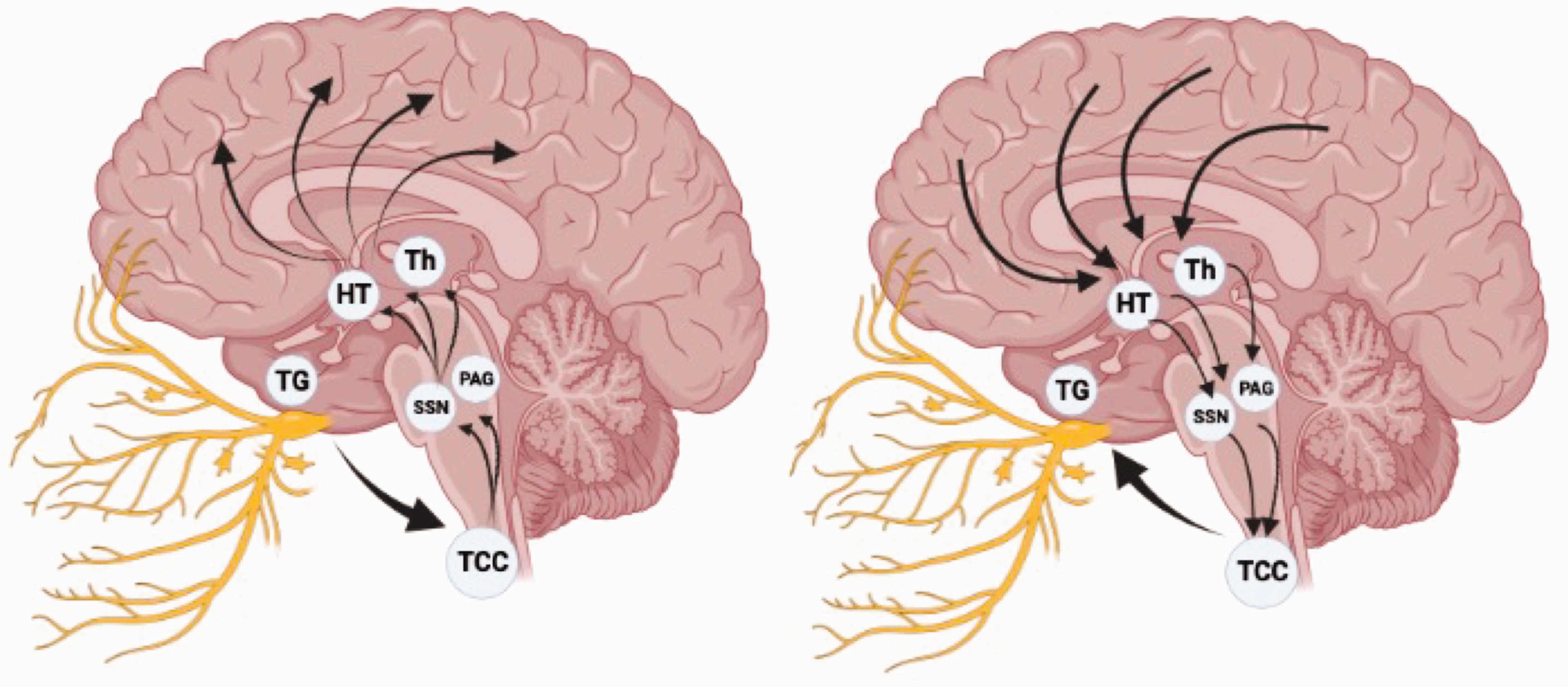
Simplified graphical view of the trigeminovascular system. In secondary headache disorders (on the left), the orthodromic activation of the trigeminal terminal ends results in ascending projections to the midbrain, hypothalamus, and thalamic nuclei. The same neuronal network is activated antidromically in migraine (on the right).
Autonomic manifestations during headache attacks
The painful attacks are often chaperoned by cranial autonomic manifestations related to a trigeminal parasympathetic outflow in primary headache disorders (14,24,25). These autonomic manifestations include facial sweating, rhinorrhea, nasal congestion, miosis, ptosis, periorbital swelling, lacrimation, and conjunctival injection. The predominance of unilateral autonomic manifestations is a pathognomonic feature of trigeminal autonomic cephalalgias (26).
Nonetheless, these autonomic symptoms may be present in migraine attacks but are usually bilateral and milder. Migraine patients who display significant unilateral autonomic symptoms usually experience more painful attacks and could represent a specific migraine endophenotype with potential treatment implications (27). These cranial autonomic manifestations are engendered by the trigeminal autonomic reflex, a protective physiological reflex activated by nociceptive stimuli in the trigeminal nerve territory (24). The trigeminal autonomic reflex is usually enhanced by painful stimulation of the eyes and sensitive facial skin areas, yet also the meninges and other intracranial structures may promote the reflex. The afferents information is conveyed and integrated into the trigeminocervical complex, thalamic nuclei, and several cortical areas, then transmitted to the superior salivatory nucleus and the sphenopalatine ganglion from where post-ganglionic autonomic fibers arise to innervate lacrimal glands, dural vessels, and other effector structures (24). Interestingly, the trigeminal autonomic reflex is activated antidromically in primary headaches, similarly to the trigeminovascular system. Therefore, the trigeminal autonomic reflex may also be considered an evolutionary conserved defensive mechanism borrowed for a different purpose in primary headache disorders. A direct relationship between the magnitude of the autonomic manifestations and pain exists, corroborating the protective role of the trigeminal autonomic reflex and its contributing role in the escalation of headache pain (28,29). Indeed, parasympathetic efferents project to intracranial arteries and meninges and an increase in parasympathetic tone activates meningeal nociceptors (30). Consistently, different preventive and acute headache therapies specifically target the trigeminal autonomic reflex.
Interictal autonomic dysfunction in migraine
Different diagnostic techniques have been employed to investigate autonomic function in the inter-ictal phase of migraine patients, including cardiovascular reflexes, pupillary and cerebrovascular reactivity, as well as biochemical and pharmacological responses. Nonetheless, conflicting results have emerged with positive, negative, and unrevealing findings among studies (14,31). Hence, no inter-ictal dysautonomia is currently recognized in migraine.
Modulation of the autonomic nervous system as a treatment
Considering the trigeminal autonomic reflex's deep involvement in the pathophysiology of migraine and primary headache attacks, its modulation constitutes a compelling therapeutic approach. Accordingly, several pharmacological and non-pharmacological therapies that act on the trigeminal autonomic reflex, including oxygen, indomethacin, and neuromodulation of the vagal nerve, sphenopalatine ganglion, or hypothalamus, have revealed promising clinical results in migraine and primary headache disorders at large, especially in trigeminal autonomic cephalalgias (24). Indomethacin has a consistent efficacy in a subgroup of trigeminal autonomic cephalalgias, namely paroxysmal hemicrania and hemicrania continua, so much so that a complete clinical response also has diagnostic validity, yet it can be highly effective also in migraine (26,32). The biological mechanisms underlying its idiosyncratic therapeutic efficacy compared to the other analgesics have yet to be fully elucidated. Nonetheless, it likely depends on its ability to modulate the trigeminal autonomic reflex by inhibiting the nitrogen oxide-induced dilatation of dural vessels (24,33). High-flow oxygen therapy is a well-known effective acute treatment for cluster headache attacks (34) and, to a smaller extent, also for migraine attacks with prominent autonomic manifestations (35). Oxygen is thought to act on the parasympathetic outflow and its effectiveness in different primary headache disorders corroborates the hypothesis that it modulates the trigeminal autonomic reflex (36). Neuromodulation of different areas involved in this reflex has proven to be effective in refractory migraine (37). This discovery was initially a stroke of serendipity, as in comorbid epileptic and migraine patients, headache attacks were reduced after the adoption of non-invasive vagal nerve stimulation (nVNS) as an anti-seizure treatment (38). Therefore, neurostimulation was investigated specifically in migraine patients and proved effective (39,40). Subsequently, stimulation of the sphenopalatine ganglion and hypothalamus was also shown to be effective (41,42). Finally, biofeedback and cognitive-behavioral therapies that promote a reduction of stress have demonstrated efficacy in migraine, possibly by improving autonomic flexibility (25,43,44).
The inter-ictal migraineurs’ brain functional and metabolic signature
Overwhelming evidence suggests that migraine attacks arise in response to a mismatch between the brain’s workload and energy reserve (8,9). Accordingly, mitochondrial dysfunction coupled with glucose metabolism impairment in migraine patients has been consistently demonstrated (8,9,45–47). Additionally, cortical hyperexcitability and dysfunctional habituation of sensory processes are well-known inter-ictal migraine traits (12,48,49). These features are in line with the sustained sympathetic hyperactivity of migraineurs (50). Notably, experimental studies have also linked cerebral energy exhaustion with the activation of the trigeminovascular system and cortical spreading depression (9,51,52). Accordingly, the most common migraine attack triggers influence, directly or indirectly, cerebral energy metabolism, such as psychological stress, sleep deprivation, hypoglycemia, low arterial oxygen level, ovarian hormone changes, and physical exercise (9). Notably, metabolic enhancers, such as coenzyme Q10, riboflavin, and the ketogenic diet, are beneficial in migraine prophylaxis (1), while anti-seizure medications may be effective by diminishing the brain hyperexcitability (48). In summary, the combination of metabolic abnormalities together with altered cerebral responsivity determines the individual migraine attack threshold. This threshold level, in combination with predisposing environmental factors, likely determines the migraine burden (53).
Migraine as a reflection of genetic Darwinian adaptive autonomic-behavioral responses
In migraine, pain is undoubtedly the most bothersome and disabling symptom, yet a wide range of other clinical manifestations exist, including autonomic, cognitive, and behavioral (26). These non-headache symptoms characterize the prodrome and postdrome phases and accompany the ictal phase. Additionally, pain can be absent in rare migraine attacks (e.g., migraine aura without headache) (26). To explain the evolutionary meaning of migraine attacks’ multifaceted phenomena, the underlying autonomic nervous system role must be integrated into a comprehensive behavioral perspective (54,55).
The behavioral meaning of pain
Pain can be perceived as either inescapable or escapable. These two pain modalities are conveyed with different neuronal networks and engender opposite autonomic-behavior responses (Figure 2) (54,56–58). Escapable pain is conveyed by skin A-delta fibers that project to the lateral and dorsolateral periaqueductal grey and the posterior hypothalamus, promoting a hyperactivation of the sympathetic system. This results in several autonomic-behavioral responses, including increased arterial pressure, heart rate, ventilation, catecholamines release, blood flow to skeletal muscles, restlessness, and aggressiveness (56,57,59). These clinical features represent integrated autonomic-behavioral responses that prepare the individuals to fight a stressor or flight from a threatening situation, namely a “fight-or-flight” response. Conversely, inescapable pain is conveyed by visceral C fibers that project to the ventrolateral periaqueductal grey, promoting inhibition of the sympathetic system. This results in decreased arterial pressure and heart rate, lethargy, and motor quiescence. These clinical features represent coordinated autonomic-behavioral responses that prepare the individuals to confront an inescapable stressor or situation, namely “sickness behavior” (56,57,59).

Schematic representation of the different neuronal networks underlying autonomic-behavioral responses of escapable and inescapable pain.
According to Charles Darwin's evolutionary theory, emotions (including pain perception) in animals and humans have an evolutionary meaning that promotes adaptive, homeostatic patterns of emotional behaviors (54,55,60–62). Therefore, pain perception is arguably a fundamental part of genetically predetermined autonomic-behavioral programs (54). Escapable and inescapable pain engenders different coordinated, innate, adaptive responses that serve homeostatic purposes (54).
The behavioral meaning of pain in migraine attacks
The autonomic-behavioral responses observed during migraine attacks remarkably overlap with those exhibited by all mammals in front of an inescapable threat, strongly substantiating the concept that it represents an adaptive evolutionary strategy (54). Indeed, a migraine patient avoids mental and physical activities, as well as sensory stimulations (1). As previously discussed, the sickness behavior exhibited during migraine attacks is believed to be triggered by a brain energy homeostatic imbalance signaled by the trigeminovascular system that aids the brain in restoring itself (9,63). This bio-behavioral framework identifies migraine pain as a visceral homeostatic emotion that serves a protective purpose, like hunger, drowsiness, and thirst (64). An alternative possible explanation to the behavioral manifestations observed during a migraine attack, such as photophobia and phonophobia, implies a failure of higher cortical functions to interpret and modulate sensory inputs. Nonetheless, the persistence of migraine as a tremendously frequent genetic trait worldwide supports its protective role and suggests that migraine individuals might have an evolutionary advantage (5,11,65). Accordingly, epigenetic mechanisms, especially those involved in the early stages of neurodevelopment, are likely pivotal in migraine and evolutionarily conserved behaviors (6,7,65). In conclusion, the migraine attack-related autonomic-behavioral response evolved for homeostatic purposes, yet, arguably, became maladaptive in migraine chronification (54).
Homeostasis, allostasis, and allostatic load in migraine
In order to explain the transformation of migraine from an adaptive protective mechanism to a maladaptive response that leads to chronic migraine, the concepts of homeostasis, allostasis, and allostatic load need to be introduced (66). The term homeostasis refers to the need of the human body to constantly maintain a stable milieu inside the body. Adaptive responses are promoted to face external changes that may threaten homeostasis, mainly through negative feedback regulatory mechanisms. Allostasis refers to the ability to maintain homeostasis through these responses (67). The brain is the central organ of stress that orchestrates the autonomic, behavioral, and neuroendocrine responses to stressors (66,68). In migraine, a metabolic brain imbalance is perceived as a stressor by the brain itself (69,70). In this context, the brain promotes an allostatic response, namely the migraine attack, to restore the cortical energetic deficit (51,71). Indeed, the avoidant behavior toward any stimulation, such as noise, light, and smell, and any physical or mental activity, namely sickness behavior, arguably results in an energy-preserving/recovering state.
This conceptualization paves the way to consider the migraine attack as a protective response and, therefore, migraine itself not as a disease (4,6). Nonetheless, high-frequency episodic migraine and chronic migraine patients suffer from substantial disability and burden, thus a pathological process must be recognized. The presence of repeated and chronic exposure to stressors results in an allostatic load where the adaptive autonomic responses become maladaptive (72). Accordingly, the most critical risk factors for transforming episodic to chronic migraines are depression, stressful life events, and acute medication overuse (73). The formers result in chronic exposure to stressors, while the latter may partially contribute to the chronic inhibition of the protective, energy-restoring responses mediated by migraine attacks. In this allostatic perspective, the chronification of migraine is a pathological process mediated by an allostatic overload (66).
Conclusions: The evolutionary meaning of migraine
Migraineurs are constantly over-alerting to both external and internal stimuli. This might have determined an evolutionary advantage in the past since they are the first to sense a potential threat, such as an incoming predator or a monoxide carbon leak (5,54). This potential evolutionary advantage is also suggested by the high persistence of migraine as a genetic trait worldwide (55). Yet, these over-performing cortical activities are very energy-consuming; therefore, the price to pay is a predisposition to developing a brain metabolism imbalance. The migraine attack is, therefore, an allostatic response that serves a homeostatic purpose: to recover brain energy by avoiding any physically or mentally energy-consuming activity for a brief interval. For this purpose, the brain engenders genetically, predetermined, adaptive neuronal responses through the antidromic activation of the trigeminovascular system, promoting visceral pain and sickness behavior. In this bio-behavioral perspective, migraine attacks are not merely painful conditions, but they represent behavioral responses to brain metabolism homeostatic perturbations that include pain within their clinical features (6). Incorporating all of these concepts, low-frequency episodic migraine might not be envisioned as a disease (3,11). However, chronic exposure to brain energy imbalance results in an allostatic overload and a maladaptive chronic activation of these adaptive responses, namely high-frequency episodic migraine and chronic migraine. In this allostatic perspective, the chronification of migraine is considered the pathological process. Migraine patients constantly seek an explanation for their apparently “meaningless” pain. Informing them of the true meaning of migraine pain should be pursued as a therapeutic act. Indeed, it may promote more conscious choices toward life habits, non-pharmacological treatment, and acute medication intake. This comprehensive bio-behavioral view explains the evolutionary (Darwinian) meaning of headache.
Clinical implications
Migraine’s astonishing prevalence and preserved genetic background contrast with the definition of a disease. Migraineurs have inter-ictal cortical hyperexcitability, low sympathetic hyperactivity, and metabolic dysfunction that predisposes to brain energetic imbalance. Migraine attacks represent behavioral responses to brain metabolism homeostatic perturbations that include pain within their clinical features. The transformation from episodic to chronic migraine should be envisioned as a pathological process, whereas the migraine itself should not. Inform the patients of the true meaning of migraine pain should be pursued as a therapeutic act.
Footnotes
Acknowledgement
In loving memory of Prof. Pasquale Montagna & Prof. Elio Lugaresi, founders of the School of Neurology of Bologna and continuous source of inspiration for all the neurological community of Bologna.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The publication of this article was supported by the “Ricerca Corrente“ funding from the Italia Ministry of Health.
