Abstract
Background
The trigeminal nociceptive system plays a pivotal role in the pathophysiology of tension-type headaches (TTH).
Objective
This study investigated and compared nociceptive-specific trigeminal pathways in patients with episodic and chronic TTH (ETTH and CTTH, respectively) using the nociceptive blink reflex (nBR) and nociceptive trigeminocervical reflex (nTCR).
Methods
We recorded nBR and nTCR in patients with ETTH and CTTH, and healthy controls using concentric electrodes and subsequently compared the threshold (i.e. sensory, pain) and parameters of reflex (i.e. the R2 component of the nBR and the late responses of the nTCR).
Results
Women with ETTH (n = 40) and CTTH (n = 32) and age-matched controls (n = 40) were recruited. CTTH patients displayed significantly lower amplitude and area under the curve (AUC) values of the R2 component for the nBR compared with those displayed by ETTH patients and controls (p < 0.05). Moreover, the amplitude and AUC of the R2 component was negatively correlated with the frequency of headaches, whereas the latency of the R2 component for the nBR was positively correlated with the frequency and duration of headaches in the TTH groups (p < 0.05). However, no significant differences in the late response parameters (i.e. latency, duration, amplitude, or AUC) were noted between the groups in terms of the nTCR.
Conclusions
R2 suppression associated with CTTH suggests decreased brainstem excitability. This may be the result of excessive descending inhibitory influences.
Keywords
Introduction
Tension-type headaches (TTH) are the most prevalent and costly form of headaches. Previous studies have focused on the pathogenesis of TTH; however, the mechanisms underlying TTH are currently unknown. Peripheral myofascial mechanisms and central dysregulation of pain-processing structures are implicated but their relative influence in the pathogenesis of TTH varies with the frequency of headache and among patients (1). Previous studies indicate central nervous system sensitization in patients with CTTH. Specifically, sensitization is present at the level of the spinal cord dorsal horn/trigeminal nucleus, as well as supraspinally, in patients with CTTH. Interestingly, central pain processing appears normal in patients with ETTH (2).
As no structural abnormalities are present with TTH, clinical neurophysiologic studies have been the most successful method for investigating TTH. In particular, a large number of studies have investigated the central trigeminal pathway to explore the role of pain control systems. Brainstem structures play a role not only in conveying nociceptive impulses but also in their modulation. Additionally, descending modulation of sensory transmission occurs largely in the brainstem. Therefore, an investigation of trigeminal pathways, including trigeminofacial and trigeminocervical reflexes, using the blink reflex (BR) and trigeminocervical reflex (TCR) may provide valuable insight into central control mechanisms in an effort to evaluate the role of the trigeminal system in TTH (3).
Previous studies that used blink recordings in TTH patients had disappointing results (4–8). Specifically, no significant differences were noted in the R1 or R2 latencies and R2 amplitudes in CTTH and controls (7,8). Normal R2 amplitudes and areas for both ictal and interictal phases have been reported in ETTH patients (4,5). Therefore, the nociceptive-specific BR (nBR) has been developed to facilitate the study of the mechanisms involved in trigeminal nociception using a special concentric planar stimulating electrode. As a result of the concentric design and small anode–cathode distance, a high current density was achieved, which allowed low current intensities to be used. Thus, depolarization was limited to the superficial layers of the dermis containing nociceptive fibers and did not reach deeper, non-nociceptive fiber-containing layers. The BR R2 integral that is elicited by the Aδ-fiber-specific concentric electrodes reflects neuronal excitability in the nociceptive trigeminal neurons of the reflex arc (9,10). Similar to the R2 component of the classic BR, the nociceptive R2 is bilateral and mediated via a polysynaptic neural network involving the spinal trigeminal nucleus, interneurons of the bulbo-pontine lateral reticular formation, and motor neurons of the facial nucleus innervating the orbicularis oculi muscles (11).
A dysregulated trigeminal nociceptive system using nBR is implicated in migraine during ictal and interictal phases (12,13). However, alterations in nBR with regard to central facilitation have not been observed in various headache disorders including medication-overuse headache (14), hypnic headache (15), and CTTH (16).
Another method that has been used to evaluate nociceptive brainstem mechanisms is TCR. TCR is a nociceptive reflex that provides information about activation changes in the area between the trigeminocaudal nucleus and the dorsal cervical spinal horn. Previous studies have suggested that the TCR is more sensitive for revealing brainstem lesions than is the R2 component of the BR (17). TCRs, including an early and late response, can be obtained from several muscles (i.e. sternocleidomastoid muscle [SCM], semispinalis muscle) by electrical stimulation of the trigeminal nerve (i.e. supraorbital and infraorbital branches). The early responses are mediated by non-nociceptive afferents and functionally resemble the R1 component of the BR, whereas the late responses are related to head-retraction movements (i.e. protective responses to nociceptive stimuli applied to the face) (3).
Very few studies have investigated the TCR in TTH. Patients with CTTH and migraines without aura display a shortened TCR latency on the painful side compared with the non-painful side (18). A significantly decreased amplitude and increased latency of TCR has also been shown in patients with CTTH compared with controls (19). A single study was performed that used the nociceptive trigemino-cervical reflex (nTCR) in healthy controls. That study noted that nTCR monitoring was a sensitive tool for detecting changes in nociceptive transmission at the level of the trigeminocervical complex when the study was extended to patients affected by primary headaches (20). Investigating the trigeminal pathway at the level of the brainstem using standard electrodes has not yet been successful in the study of TTH. Therefore, nociceptive fiber-mediated brainstem reflexes using nBR and nTCR may be more sensitive for detecting changes in nociceptive-specific trigeminal transmission in patients with TTH. We hypothesized that the R2 component of nBR and the late response of nTCR may be altered to varying degrees in patients with CTTH. Investigation of nociceptive-specific trigeminal pathways at the brainstem level could give important information for understanding the pathophysiology of TTH. Therefore, we investigated and compared the properties of nociceptive trigeminal pathways by recording the nBR and nTCR in patients with ETTH and CTTH.
Methods
Subjects
Patients with TTH who received care at a tertiary hospital headache clinic between August and November 2011 were recruited for this study. Headache diagnoses were made by a board-certified neurologist using criteria from the International Headache Society (21). Subjects with ETTH were defined as those with headaches from 1 to 15 days per month (frequent ETTH). Subjects with infrequent ETTH were not included in the ETTH group. As infrequent ETTH has very little impact and is considered a normal phenomenon rather than a disease, it had an insignificant effect in our study. Patients with CTTH were defined as those having headaches at least 15 days per month for more than 3 consecutive months. To exclude other primary headaches, patients were required to have at least a 1-year history of headaches prior to enrollment in the present study. In order to be included in the TTH group, patients had to describe all characteristics typical of TTH following the common criteria of the International Headache Society: bilateral location, pressing and tightening pain, mild or moderate intensity, and no aggravation of headache during routine physical activity. Additionally, patients with migraine symptoms such as nausea and vomiting were excluded to distinguish them from having mixed headaches (i.e. coexistence of TTH and migraine). Patients were excluded who took analgesics or muscle relaxants within 24 hours of the study or overused medications as defined by the International Headache Society. The control group consisted of age-matched volunteers recruited as relatives or friends of patients. Controls were free of headaches for at least 3 months prior to the study, experienced no more than an occasional mild headache (<5 times per year), and had never sought medical treatment for headaches. Exclusion criteria included any other systemic disease, such as diabetes mellitus and polyneuropathy, the presence of neurologic disorders (i.e. stroke, multiple sclerosis), a history of Bell’s palsy, serious somatic or psychiatric illness including depression and anxiety disorder, daily medication to prevent headaches and/or antidepressant medication, and pregnancy or use of oral contraceptives. Only females were included in the present study to eliminate sex effects.
All participants were examined physically and neurologically by an experienced neurologist. Computed tomography or magnetic resonance imaging (MRI) of the brain was performed for all patients with headache. We also evaluated cervical spine x-rays of all patients with headache to exclude clear symptomatic causes (22). These radiological examinations indicated no pathological findings in any subject. The participants were asked to complete a questionnaire regarding their headache symptoms, including frequency, duration, and intensity of headaches during the last 4 weeks. Headache frequency (days/week) was calculated by dividing the number of days with headaches by 4 weeks. Headache duration (hours/day) was calculated by dividing the sum of the total hours of headaches by the number of days with headaches and headache intensity (numeric rating scale [NRS]: 0 = no pain to 10 = unbearable pain), which was calculated from the mean of the NRS for the days with headaches. Written informed consent was obtained from all subjects prior to enrollment in the study. The local ethics committee approved this study.
Experimental protocol
The experimental sessions were conducted at the same time of day (2–5 PM) to reduce variability related to circadian effects. Testing took place either in a lying position (nBR) or a sitting position (nTCR). Subjects were instructed to refrain from caffeine, nicotine, and alcohol for at least 24 hours prior to testing. Additionally, subjects were instructed to be examined on days in which headache intensity was <3 points on an NRS. Two planar concentric surface stimulating electrodes were used for nociceptive stimulation (Walter Graphtek, Lubeck, Germany, http://www.walter-graphtek.com), as previously described (9). The investigator was blind to the clinical diagnosis of the subjects. The painful sensation that was elicited by the electrical stimulation was rated by the subjects using a NRS.
Nociceptive blink reflex (nBR)
The subjects were in a lying position with their eyes closed during the nBR recordings. The concentric electrode was placed on the left lower forehead, approximately 10 mm above the supraorbital foramen, to stimulate the supraorbital nerve. Surface recording electrodes were placed on the infraorbital area (active) and at the base of the nose with a 2 Hz–1 kHz band-pass filter (sampling rate 2.5 kHz, 150 ms sweep length, 200 ms analysis time, and 100 µV sensitivity), which was used for recording. Sensory- and pain-perception thresholds were detected using ascending and descending single pulses of 0.1-mA steps, with a duration of 0.5 ms and an interstimulus interval of 15–20 s (randomized interval). The subjects were asked to identify their first sensation, which was subsequently defined as their individual sensory threshold (Is). Subjects were also asked to identify a pinprick-like pain stimulus, which was later defined as their pinprick pain sensation (Ip). A fixed stimulation intensity (SI) of 1.5 × Ip was used to evoke the nBR. Stimuli used to evoke the nBR were applied as trains that consisted of three pulses (each 0.5 ms in duration) with a 5 ms interpulse interval to further increase nociceptive specificity (23). Stimulation intensities >2 mA were excluded to minimize the risk of Aβ fiber co-activation (9). Repeated stimulation of the train pulse was applied at random intervals of 18–22 s to obtain at least 11 consecutive responses.
Nociceptive trigeminocervical reflex (nTCR)
Subjects were seated comfortably in a chair with their eyes closed during the nTCR procedure. Different head and neck positions were tried to minimize neck muscle activity. The concentric electrode was placed on the left lower forehead, approximately 10 mm above the supraorbital foramen to stimulate the supraorbital nerve. The surface recording electrodes were placed over the right side of the semispinalis capitis muscle near the C4 vertebrae on the midline and 2 cm below in a longitudinal direction over the muscles (24). The recording system was band-pass filtered at 10–2500 Hz and the sensitivity was set at 200–500 µV. The train of electrical stimuli was composed of three pulses, each pulse was 0.5 ms in duration (the inter-pulse interval was 5 ms) (20,23). Sensory (ST) and pain (PT) perception thresholds were defined as the minimum stimulation intensity that was perceived as tactile or painful, respectively. A stimulus with an intensity fixed at 1.5 × PT was used to record the baseline nTCR parameters. The six trials of each stimulation series were delivered randomly at 30–40 s.
Analysis
An investigator who was blind to the diagnosis performed signal analyses. The recorded signals were amplified, full-wave rectified, and analyzed (Medelec Synergy, Oxford, UK). The first recording was discarded to reduce the influence of the startle response. For the nBR recording, the 10 trials for each stimulus series were rectified and averaged off-line. For the nBR recording, the following parameters were measured: (a) threshold: the stimulus intensity thresholds for non-specific and pinprick pain sensations were assessed (Is and Ip); (b) baseline parameters: the onset latency (ms), mean amplitude of the root mean square (RMS, µV), duration (ms), and area-under-the-curve (AUC, mV*ms) values from the ipsilateral and contralateral R2 were analyzed (iR2 and cR2). The onset latency was visually determined as the initial point from the baseline (ms), and the AUC was determined between 27 ms and 87 ms of the R2 component (mV*ms) (25).
For the nTCR recording, the EMG analysis was conducted 300 ms after the electrical stimulation. The first response was removed to avoid contamination by the initial startle response. At least five consecutive responses were recorded, full-wave rectified, and averaged. The following parameters were measured: (a) threshold: the stimulus intensity thresholds for tactile and painful sensations were assessed (ST and PT); (b) baseline parameters: the onset latency (ms), mean amplitude of the RMS (µV), duration (ms), and AUC (mV × ms) were analyzed. The onset and offset of the reflex corresponded to the point at which the amplitude of the EMG signal exceeded that of the background EMG activity by more than 30 µV and then returned to below this level according to the method of Serrao et al. (20).
Statistics
Statistical analyses were performed using SPSS 18.0 for Windows (SPSS, Inc., Chicago, IL, USA). All values are reported as means and standard deviations. A one-way analysis of variance was used to compare thresholds and parameters of nBR and nTCR between groups. Bonferroni post hoc correction for multiple comparisons was performed for the post hoc analyses. Pearson’s correlation analyses were used to explore the relationship between baseline parameters and clinical headache parameters (i.e. frequency, duration, intensity). Results were considered significant at a p < 0.05.
Results
Clinical characteristics
Females with ETTH (n = 40) and CTTH (n = 32) and age-matched healthy controls (n = 40) participated in the present study. No significant differences in age were noted (ETTH: 53.17 ± 10.49 years, CTTH: 55.93 ± 10.36 years, controls: 52.52 ± 11.41 years). The frequency of headaches according to the headache questionnaire (day/week) was 1.5 in patients with ETTH and 5.2 in patients with CTTH. The duration of headaches (hours/day) was 8.3 in patients with ETTH and 10.8 in patients with CTTH. The mean intensity per episode (NRS) was 4.3 in patients with ETTH and 4.8 in patients with CTTH. The duration of having the headache condition was 6.0 years in patients with ETTH and 9.7 years in patients with CTTH.
Sensory and pain thresholds
Mean sensory thresholds, pain thresholds, and perceived pain intensities in response to electrical stimulation.

All values are expressed as mean ± standard deviation.
p determined by analysis of variance.
ETTH: episodic tension-type headache; CTTH: chronic tension-type headache; nBR: nociceptive blink reflex; nTCR: nociceptive trigemino–cervical reflex; Is and Ip: sensory and pain threshold in nBR; ST and PT: sensory and pain threshold in nTCR; PIBR and PITCR: perceived stimulus intensity during the recording; SI: stimulation intensity in nBR.
Nociceptive blink reflex (nBR)
Nociceptive blink reflex parameters.

p < 0.05 between CTTH and ETTH.
p < 0.05 between CTTH and controls.
p determined with a Bonferroni post hoc test.
ETTH: episodic tension-type headache; CTTH: chronic tension-type headache; SD: standard deviation; nBR: nociceptive blink reflex; iR2: ipsilateral R2 component; cR2: contralateral R2 component; AUC: area under the curve.

Examples of the nociceptive blink reflex (nBR) and nociceptive trigemino-cervical reflex (nTCR) in healthy controls and patients with chronic tension-type headache (CTTH). Each curve represents the rectified recordings of a control subject or patients with CTTH. (a) nBR in a control subject; (b) nBR in a CTTH patient; (c) nTCR in a control subject; (d) nTCR in a CTTH subject.
Nociceptive trigeminocervical reflex (nTCR)
Nociceptive trigemino-cervical reflex parameters.

All values are expressed as mean ± standard deviation.
ETTH: episodic tension-type headache; CTTH: chronic tension-type headache; nTCR: nociceptive trigeminocervical reflex; AUC: area under the curve.
Correlations between clinical headache parameters and parameters of nBR and nTCR
Headache characteristic correlation coefficients from the headache questionnaire and nociceptive blink reflex parameters in patients with tension type headache.
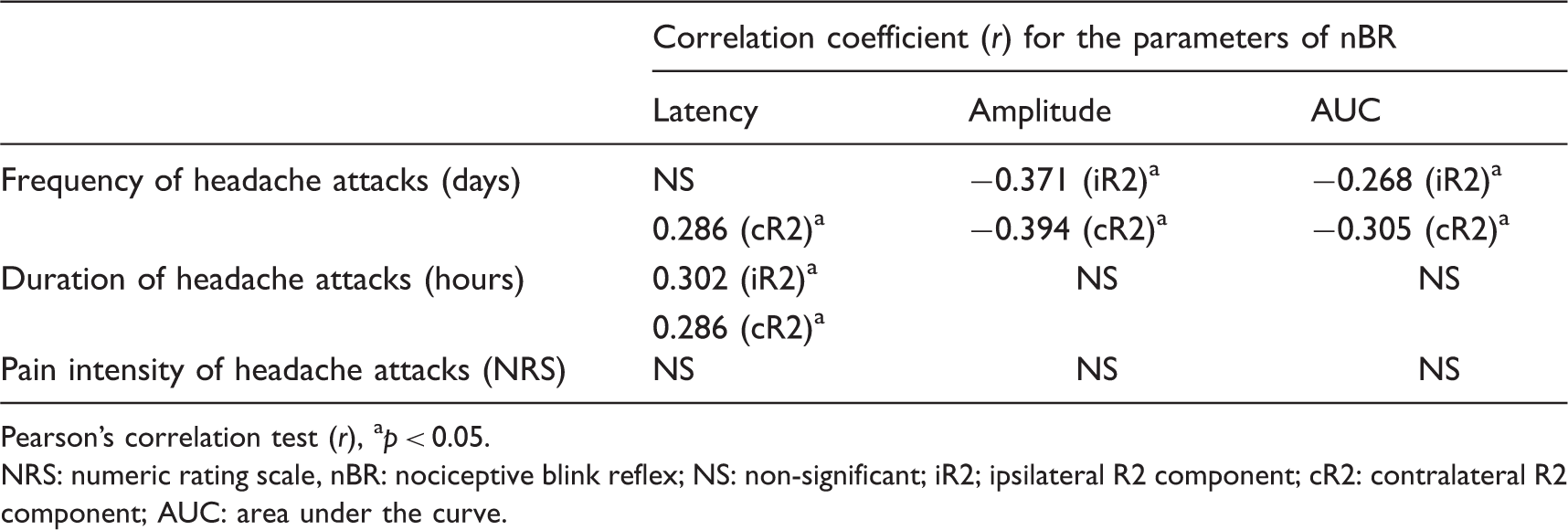
Pearson’s correlation test (r), ap < 0.05.
NRS: numeric rating scale, nBR: nociceptive blink reflex; NS: non-significant; iR2; ipsilateral R2 component; cR2: contralateral R2 component; AUC: area under the curve.
Discussion
This study observed significant R2 suppression with nBR in CTTH patients, but no significant differences were noted with nTCR in all groups. In patients with CTTH, significantly low amplitudes and the AUC of the R2 component were observed with the nBR, and the R2 latency was positively correlated with the frequency and duration of headache parameters. A decrease in onset latency and an increase in the reflex integral (AUC) suggest facilitation of a spinal or medullary reflex (26,27). The R2 integral of the BR elicited by the Aδ-fiber-specific concentric electrode reflects the neuronal excitability in the nociceptive trigeminal neurons in the reflex arc (9). Our findings suggest decreased lower brainstem excitability, as well as hypoexcitability of R2 interneurons and the circuit.
Previous BR studies have suggested a primary dysfunction in central nociceptive control systems in patients with TTH (3). Our data are consistent with results from previous studies that used different methods. Increased BR latencies associated with increased headache duration have been reported in TTH patients. The authors suggested that hypoactivity of trigeminal sensory neurons and facial motor neurons developed over time in patients with CTTH. Moreover, they suggested that brainstem serotonergic hypoactivity may be an explanation for the increased BR latencies (7). Other reports have demonstrated a diminished recovery curve of the BR R2 component in patients with TTH. These findings indicate reduced excitability of the brainstem interneurons in patients with TTH that may be related to a relative depletion in specific neurotransmitters, such as serotonin or noradrenaline (4).
Previous studies have reported that the nBR R2 component evoked by nociceptive stimuli from a concentric electrode was over six times greater during migraines than during headache-free intervals. This is consistent with sensitization of cutaneous nociceptive afferents or second-order neurons in the trigeminal nucleus caudalis (12). Moreover, trigeminal sensitization may persist subclinically during headache-free intervals (13). The facilitation of trigeminal nociception may be specific to migraines rather than a consequence of peripheral pain, such as frontal sinusitis. The authors indicated that the observed activation of brainstem nuclei was not simply a reaction to trigeminal pain but was a central migraine generator driving force for migraine attacks (28). Our findings are not consistent with electrophysiologic evidence of trigeminal sensitization in migraines. However, no differences between the trigeminal processing of patients with hypnic headaches and that of healthy controls were observed either at the brainstem level (nBR) or the supraspinal level (pain-related evoked potential, PREP). These patients did not show any hyperexcitability despite the chronic nature of this disease. Thus, disease chronicity does not seem to be the only predictor of electrophysiologically conceivable cerebral overexcitability (15). In medication-overuse headache, activation of somatic and trigeminal PREP was found, but not of nBR, indicating that sensitization of central nociceptive mechanisms mainly involved structures outside the trigeminal system and probably occurred at the supraspinal level (14).
In experimental pain models of CTTH, a significantly reduced integral of the R2 component from the first BR recording after an infusion of saline was shown in CTTH patients compared with healthy volunteers on the contralateral side. This difference, however, was only significant at one time point, and facilitation of the brainstem could not be induced in this study (29). A previous experiment hypothesized that painful stimulation of the temple ipsilateral to supraorbital nerve stimulation would facilitate electrically evoked pain and R2 due to convergence of nociceptive signals in the brainstem trigeminal nucleus. However, the R2 component of the BR was strongly suppressed during and after painful conditioning stimulation. Thus, inhibitory influences (e.g. diffuse noxious inhibitory controls, DNIC) appeared to mask ipsilateral segmental facilitation of R2 during ice-induced headache. The local facilitatory effect induced by the conditioning stimulus competed with the general inhibitory influence on R2 (30). Facilitation of brainstem reflexes has been described in humans after homotopic conditioning painful stimuli (31). However, heterotopic conditioning pain has been demonstrated to inhibit the BR, possibly via DNIC (25).
Previous reports revealed widespread bilateral pressure-pain hypersensitivity in adults with CTTH and children with frequent ETTH, suggesting that the sensitization process is not only restricted to the trigeminal second-order neurons but also to the extratrigeminal nociceptive neurons (32,33). This generalized hyperexcitability of the second-order (or higher) nociceptive neurons in the central nervous system leads to sensitization and plastic changes in higher centers and may provoke broader inhibitory influences, and this may mask R2 facilitation at the trigeminal brainstem level. Alternatively, early potentiation followed by excessive inhibition may be the underlying issue (possibly a long-term depression-like mechanism) (8).
Pain transmission in the central nervous system is influenced by a wide range of neurotransmitters, including serotonin, norepinephrine, dopamine, gamma-aminobutyric acid, and enkephalins/endorphins. The BR is directly related to dopamine levels in the central nervous system. An increased reflex excitability occurs with lower levels, such as in Parkinsonism, and is reversed by dopaminergic drugs (11). Arousal also alters the excitability of the R2 component of the BR. Analysis of R2 during sleep showed a marked reduction in stages II, III, and IV, and a substantial recovery during rapid-eye-movement sleep (34). Taken together, it appears that dopaminergic, serotonergic, and noradrenergic activity alters R2 excitability (4).
In a mouse model of TTH, hypertonic saline infusion into the neck muscle induces long-lasting facilitation of the brainstem reflex (35). This reflex is an accepted model for investigating altered excitability in sensory brainstem neurons with convergent afferent input from different craniofacial tissues such as neck muscles. Intramuscular infusion of α, β-methylene adenosine 5-triphophate (α, β-meATP) provides noxious input from neck muscles that causes sustained reflex potentiation. Recent studies have demonstrated that administration of the nitric oxide synthase inhibitor L-NMMA prevents brainstem-reflex facilitation evoked by intramuscular α, β-meATP in a dose-dependent manner. Thus, nitric oxide synthase (NOS) is involved in the induction and maintenance of facilitation (36). Additionally, acetylsalicylic acid (ASA) inhibits α, β-meATP-induced facilitation of neck-muscle nociception in mice. Thus, a central nervous system antinociceptive mode of action for ASA via a cyclooxygenase-related mechanism and/or NOS may account for this phenomenon (37). The increased NOS activity shown in platelets of patients with CTTH is also related to serotonin depletion and to increased glutamate content (38). A previous study found that sublingual nitroglycerine induces changes in nBR and visual evoked potentials in healthy subjects, which are comparable to those shown to be abnormal in migraine. Nitroglycerine could modify evoked cortical responses indirectly via an increase in serotonergic transmission (39). However, these results do not agree with those reported by Kowacs et al. Those authors found no significant change in the nBR in healthy subjects after an intravenous infusion of glyceryl trinitrate (40). Recently, it was discovered that inhibition of nitric-oxide (NO) production can counter both the cortical hyperexcitability and facilitation of trigeminal nociception in the serotonin-depleted rat model (41). Considering the results of these experimental studies, suppression of R2 in patients with CTTH may be related to a dysfunction of certain neurotransmitters. In particular, dysfunction of the brainstem NO and monoaminergic systems, which reflects a relationship between NO and serotonin hypoactivity, may explain the reduced excitability of the nBR.
Furthermore, changes in BR-circuit excitability may also be due to damage of the neural structures along the BR circuit. Lesions may occur in the trigeminal or facial nerves, as well as in the brainstem (42). Schmidt-Wilcke et al. demonstrated a significant decrease in gray matter in the brainstem of patients with CTTH using MRI and voxel-based morphometrics. The reduced gray matter areas were regions involved in pain processing, including the cingulate gyrus and the dorsal rostral pons, near the periaqueductal gray. The authors suggested that the results may be a consequence of central sensitization, generated by prolonged nociceptive input from pericranial myofacial tissues (43). Atrophy in CTTH patients may directly influence or have an impact via supraspinal modulation on the R2-component suppression associated with the nBR.
This study has several limitations. Highly selected patients in specialized headache clinics were recruited. The size of the sample was small, and the study used a cross-sectional design that provided limited information. In order to further understand the mechanisms underlying TTH, longitudinal electrophysiologic follow-up studies are warranted. Future studies that include other primary headaches are necessary to encourage the use of nBR and nTCR as part of a neurophysiologic assessment of primary headaches. Females may show alterations in blink rates, tear composition, and/or other ocular factors due to hormonal fluctuations associated with the menstrual cycle, pregnancy, or pseudo-pregnancy produced by the use of oral contraceptives (44). The present study did not classify subjects according to menstrual cycle or menopausal status. Only females were included in the present study because of potential effects of age and sex on the BR (45). Additionally, pregnant females and women who use oral contraceptive were excluded. Therefore, menstruation status as a confounding factor may have similarly affected control subjects and patients with headache.
In conclusion, this study provided electrophysiologic evidence that interneuronal brainstem pathways can be altered in patients with CTTH. Therefore, the nBR may be useful in evaluating alterations in brainstem neuronal networks in patients with TTH, as well as other types of primary headaches.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflict of interest
None declared.
