Abstract
The aim of this study was to investigate central anti-nociceptive mechanisms of i.v. acetylsalicylic acid (ASA) and oral zolmitriptan (ZOL) in migraine patients and healthy subjects using the ‘nociceptive’ blink reflex (nBR). Twenty-eight migraine patients received ASA (n = 14, 1000 mg i.v) or ZOL (n = 14, 5 mg p.o) during the acute migraine attack and interictally. Thirty healthy subjects received either ASA or ZOL vs. placebo using a double blind cross over design. nBR was recorded in all patients and subjects before, 60 and 90 min after treatment. ASA and ZOL did not inhibit nBR responses in healthy subjects. Both ASA and ZOL suppressed nBR responses (ASA by 68%, ZOL by 78%) only during the acute attack but not interictally. The data suggest, that the anti-nociceptive effects of migraine drugs on the trigeminal nociceptive processing are different during and outside an acute migraine attack.
Introduction
Migraine is a common but nevertheless complex neurobiological disorder characterized by attacks of unilateral stabbing, pulsating headache (1). Migraine head pain arises within the trigeminal system (2). The pathophysiology of migraine headache as well the anti-nociceptive mechanisms of migraine drugs are still poorly understood.
We previously reported a novel technique for studying central trigeminal pain processing using the ‘nociceptive’ blink reflex (nBR) (3, 4). The nBR was elicited using a novel stimulating electrode, which by virtue of its concentric geometry produces high current densities by low current intensities. Thus, depolarization is limited to the superficial layer of the dermis containing nociceptive A-delta fibres but does not reach the deeper, A-beta fibre containing, layers. The nBR has been shown to be highly sensitive to changes in trigeminal nociception (5). Using the nBR a temporary facilitation of the trigeminal nociceptive system could be detected during acute migraine attacks but not interictally (6). The facilitation of the trigeminal nociceptive system has not been found during peripheral pain such as frontal sinusitis (7). Furthermore, nBR responses were suppressed following treatment with acetylsalicylic acid (ASA) and Zolmitriptan (ZOL) during the migraine attack in parallel to the headache relief (6). Therefore, the nBR has been thought to be a sensitive marker for the functional state of the trigeminal nociceptive system.
The aim of the current study was to evaluate whether nBR recordings can be used to assess central anti-nociceptive effects of potential migraine substances in humans. Therefore effects of two potent migraine drugs such as ASA and ZOL on the nBR were studied in migraine patients during and outside the acute attack and in healthy subjects. The data of the first 17 patients has been published previously (6).
Methods
The study was approved by the local ethics committee. Informed content was obtained from all subjects and patients prior the investigation.
Study design
Twenty-eight patients with migraine without aura (1) (21 women, 7 men; mean age 27 ± 5.5 years) were studied. Mean attack frequency was 2.8 ± 1.3 per month. Mean duration of suffering from migraine was 11.1 ± 5.4 years. None of the patients received preventive medication for the last 3 months.
Thirty age and gender matched healthy subjects (19 women, 11 men) with a mean age of 25.7 ± 4.8 years were investigated.
Fourteen patients received acetylsalicylic acid (ASA, lysine acetylsalicylate iv, Aspisol® 1000 mg, Bayer-Leverkusen, Leverkusen, Germany) during a strictly unilateral migraine attack within the first six hours after onset. All patients received ASA again during the interictal period. It was defined as migraine free at least 3 days before and after investigation.
Fourteen patients received zolmitriptan (ZOL, Zomig® 5 mg po, AstraZeneca, Plankstadt, Germany) during a strictly unilateral migraine attack within six hours after onset and interictally. Headache intensity was measured using the visual analogue scale (0 = no headache, 10 = very severe headache) before and 90 min after drug treatment. nBR recordings were performed in all patients before as well as 60 and 90 min after drug treatment.
Thirty healthy subjects were investigated. None of them had a personal or family history of migraine or cluster headache (1). Fifteen subjects received ASA or placebo using a double blind cross over study design. Fifteen subjects received ZOL or placebo using a similar double blind cross over design. nBR recordings were performed in all subjects before as well as 60 and 90 min after drug treatment.
Electrophysiological stimulation and recording setup
nBR were elicited using a concentric surface stimulating electrode, as described previously (4). Individual pain and tactile perception thresholds were detected using ascending and descending single pulses in 0.2 mA steps.
Stimulation parameters
Stimulation.
Blocks of 6 stimuli (monopolar square wave pulses, DS7A, Digitimer, UK), Stimulation intensity was 1.5 times the individual pain threshold, which was determined in subjects and patients during the first visit. Migraine patients were invited for the first evaluation outside the acute migraine attack. The stimulation intensity was kept constant for each subject or patient during the entire study. Median stimulation intensity in migraine patients was 1.4 mA range 0.8–2.0; median stimulation intensity in the group of healthy subjects was 1.35 mA, range 0.9–1.9. Stimulus duration was 0.3 ms, interstimulus interval 15–17 s (pseudo-randomized).
Recording.
Bilateral surface recording electrodes (silver chloride) placed infra-orbitally and at the root of the nose, bandwidth 1Hz-1 kHz; sampling rate 2.5 kHz, sweep length 150 ms (1401 micro, Signal Averager, Cambridge Electronic Design, Cambridge, UK).
Analysis.
Curves were analysed by an investigator blinded to the experimental conditions. The first recording sweep was discarded to avoid contamination by initial startle responses. Onset latencies of R2 responses were measured in individual sweeps. A mean value for each block was calculated.
For analysis of the ‘area under the curve’ (AUC) of the R2 response five subsequent curves were rectified, demeaned and averaged. The AUC of the R2 response was calculated between 27 and 87 ms using PC-based offline analysis with custom written software as described previously (4); Matlab, 5.3 MathWorks, Natick, Massachusetts (US).
Data analysis.
Differences in mean onset latencies for baseline and after treatment were compared. Because of heterogeneity of the raw AUCs, data were normalized and analysed as percentage deviations from the baseline values (baseline values considered as 100%).
Statistical analysis
Migraine patients
Two
Healthy subjects
Two
Two
Student's t-tests with Bonferoni's correction for multiple comparisons were used for posthoc analysis of significant factors and their interactions and group means. Factors and interactions with error probabilities P < 0.05 were considered significant.
Results
Clinical results
All patients suffered from strictly unilateral moderate to severe migraine headache (mean VAS score: 6.9 ± 1.2). All patients reported a significant reduction of the headache intensity by ASA (VAS baseline = 6.7 ± 1.1 vs. VAS 90 min after treatment = 0.9 ± 1.2) or ZOL (VAS baseline = 7.0 ± 1.4 vs. VAS 90 min after treatment = 1.0 ± 1.4; TIME d.f. = 1, F = 576.4, P < 0.0001). No significant differences were observed between the two drug effects (DRUG d.f. = 1, F = 0.067, P = 0.79).
Electrophysiological results
Migraine patients
A significant increase of nBR onset latencies (TIME∗ATTACK d.f. = 2, F = 8.6, P < 0.001) and a significant suppression of AUCs (TIME∗ATTACK d.f. = 2, F = 26.8, P < 0.001) were observed after application of ASA and ZOL during acute migraine attack but not interictally (Fig. 1a,b).

Effects of acetylsalicylic acid (ASA) and zolmitriptan (ZOL) on areas under the curves of the ‘nociceptive’ blink reflex. Data are shown as mean values and standard errors of the mean. (a) ASA in migraine patients during the acute attack (•) and interictally (▪). (b) ZOL in migraine patients during the acute attack (•) and interictally (▪). (c) ASA (○) vs. placebo in healthy subjects (□). (d) ZOL (○) vs. placebo in healthy subjects (□). BL = baseline, 60 min, 90min after treatment.
No significant differences were found comparing the effects of ASA and ZOL (Tables 1 and 2).
Effects of acetylsalicylic acid and zolmitriptan on onset latencies and areas under the curves (AUC) in migraine patients during the acute attack and interictally

P < 0.05.
Effects of acetylsalicylic acid and zolmitriptan vs. placebo on onset latencies and areas under the curves (AUC) in healthy subjects
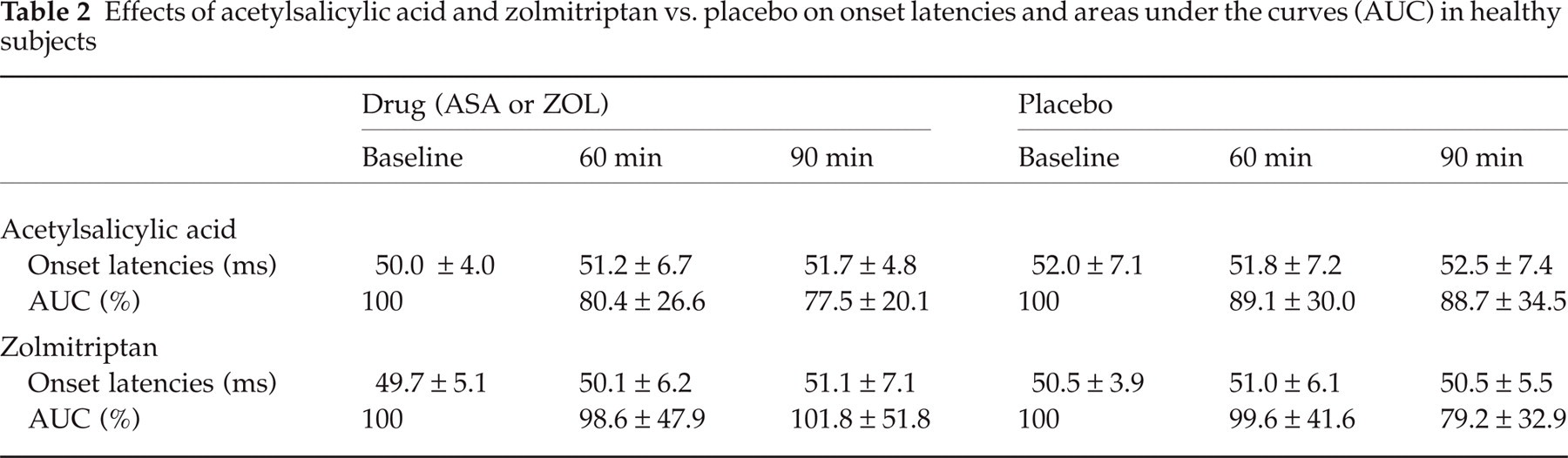
Healthy subjects
No differences were found between the ASA and placebo with regard to onset latencies (TIME∗DRUG d.f. = 2, F = 0.099, P = 0.91) or AUCs (TIME∗DRUG d.f. = 2, F = 1.3, P = 0.28; Fig. 1c).
No differences were found between the ZOL and placebo with regard to onset latencies (TIME∗DRUG d.f. = 2, F = 0.45, P = 0.64) or AUCs (TIME∗DRUG d.f. = 2, F = 0.46, P = 0.63; Fig. 1d).
Discussion
There are two important findings in this study
Firstly, a strong inhibitory effect of acetylsalicylic acid and zolmitriptan on trigeminal nociceptive transmission was observed during but not outside the acute migraine attack. Secondly, no differences were found between placebo and acetylsalicylic acid or zolmitriptan in healthy subjects.
We previously showed, that nBR can be used as a sensitive marker for the functional state of the trigeminal nociceptive system. nBR responses were facilitated during the acute migraine attack and normal between attacks (7, 6, 7). In parallel to headache relief, nBR responses were strongly suppressed by acetylsalicylic acid and zolmitriptan during acute migraine attack (6). Here, in a larger number of patients the same effect was seen. Interestingly, these anti-nociceptive effects have not been found in the same patients after administration of acetylsalicylic acid and zolmitriptan interictally. This difference indicates that – within the trigeminal system – the central anti-nociceptive effects of acetylsalicylic acid and zolmitriptan differ during and outside an acute migraine attack.
The acute migraine attack is characterized by dysfunction of nociceptive and nonnociceptive brain structures at cortical (8, 9), diencephalic (10) and brain stem levels (11, 12). As the most likely substrate for migraine head pain the trigeminal nociceptive system is significantly involved into this global brain dysfunction (6). The fact, that nBR responses were suppressed only during acute migraine attack, but not interictally might reflect a modulatory effect of migraine drugs on the trigeminal nociceptive system. It is possible, that migraine drugs such as zolmitriptan and acetylsalicylic acid strongly suppress the facilitated trigeminal nociception but do not or only to some extent affect the trigeminal nociceptive system in the resting state. The trigeminal reflexes, however, are strongly influenced by supra-segmental control at cortical level and by cognitive factors (13, 14). Therefore, these higher cortical mechanisms, such as arousal or expectation of pain relief may partly account for the observed suppression of nBR responses.
Another explanation for different effects of migraine drugs during and outside the acute migraine attack could be an impaired function of the blood brain barrier controlling access of migraine drugs to the brain stem as suggested by clinical (15) and experimental animal data (16). In experimental animals intravenous zolmitriptan and acetylsalicylic acid, both, have been shown to be able to cross the blood brain barrier and affect central pain processing (17, 18). A direct inhibitory effect of zolmitriptan on second-order trigeminal neurons has been shown after microiontophoretic administration (19). In migraine patients and healthy subjects zolmitriptan modulated the intensity dependence of cortical auditory evoked potentials suggesting a change in serotonergic sensory control (20). Similarly, anti-nociceptive effects of acetylsalicylic acid have been demonstrated in animals (21). Thus, although acetylsalicylic acid and zolmitriptan, both penetrate blood brain barrier easily, it is possible, that an impaired blood brain barrier during migraine attack could increase their central anti-nociceptive effects.
In healthy subjects we could not find any differences between zolmitriptan or acetylsalicylic acid and placebo. This finding could be explained by an insufficient sensitivity of the nBR method. In a recent study using a similar stimulation and recording paradigm a significant inhibition of the contralateral nBR responses by a novel adenosine A1 receptor agonist GR79236 was seen. On the ipsilateral side the suppression did not reach statistical significance (22). Whether the contralateral nBR responses are more sensitive to pharmacological intervention than ipsilateral responses is not known. Another tentative explanation is that GR79236 is a more potent inhibitor of central trigeminal transmission than acetylsalicylic acid or zolmitriptan as observed in similar animal assays (23). Whether this is of relevance for the clinical efficacy in migraine remains to be addressed in clinical trials.
The lack of a detectable effect of acetylsalicylic acid and zolmitriptan on nBR in healthy volunteers suggests that this reflex response cannot be used as a pharmacological screening tool in the search for drugs with anti-migraine action in healthy subjects.
Several attempts have been made to use protective reflexes and their laboratory surrogates as human models in physiology and pharmacology. The corneal reflex has been suggested to be ‘nociceptive’ since the human cornea is predominantly innervated by nociceptive A-delta (24). In healthy subjects using the corneal reflex anti-nociceptive effects of several drugs including acetylsalicylic acid and piroxocam were demonstrated (25, 26). Both studies, however, had a relatively low sample sizes and were not placebo controlled.
The main draw back of the study is the lacking of placebo control in migraine patients treated with acetylsalicylic acid or zolmitriptan. It could be argued that the presence of the placebo control would reduce the net size of the drug effect. However, we found a very strong anti-nociceptive effect (reduction of R2 AUCs by 70–80%) of ASA and ZOL in migraine patient during acute attack. Interictally this effect was significantly lower, approximately 20–25%. A similar reduction was found also in healthy subjects either by ASA and ZOL or by placebo. This reduction is most likely caused by the unspecific habituation process as shown previously by Katsarava et al. (3). Therefore, although it is not known to which extent the placebo effect may be larger during acute migraine it is unlikely that it could account for the whole extent of the observed reduction.
Hence, the main finding of this study is that both potent migraine drugs, acetylsalicylic acid and zolmitriptan suppressed trigeminal nociceptive processing during acute migraine attacks but not interictally and not in healthy subjects. And finally, the data suggest, that the nBR technique is an useful tool to quantify the activation of the trigeminal nociceptive system during the acute migraine attack and to evaluate the central anti-nociceptive effects of migraine drugs during the state of facilitation.
Footnotes
Acknowledgements
The study was supported by a research grant of the Bayer AG, Leverkusen, Germany.
