Abstract
Keywords
Introduction
Tension-type headache (TTH) is the most costly and common form of headache. The second edition of the International Headache Classification of Headache Disorders further subdivides TTH into episodic (ETTH) and chronic TTH (CTTH), mainly on the basis of headache frequency. CTTH differs from the episodic forms not only in frequency but also with respect to pathophysiology, level of disability (greater), lack of response to most treatment strategies, rate of medication overuse (greater), and personal and socioeconomic costs (higher) (1).
The specific cause and pathophysiology of TTH are unknown, but they are most likely multifactorial (2–8), and vary depending on the form and individual. Whether the pain in TTH originates from myofascial tissues or from central mechanisms in the brain is still a matter of debate. The current consensus is that peripheral pain mechanisms probably play a role in ETTH, whereas central dysnociception is predominant in CTTH (9). In most patients, TTH develops from the episodic form to the chronic form (10). Prolonged peripheral nociceptive stimuli from pericranial myofascial tissues seem to be responsible for the conversion of episodic to chronic TTH (11,12). Therefore, identifying the source of peripheral nociception in patients with headaches may provide an insight into the interaction between peripheral and central changes in TTH.
Peripheral factors have traditionally been considered to be of major importance in TTH, and many studies have indicated that the most prominent clinical findings show that patients suffering from TTH have an increased tenderness in response to palpation of pericranial myofascial tissues (13–17). TTH is considered the prototype of headaches in which myofascial pain plays an important role (13). The role of myofascial factors in TTH, however, does not negate the importance of other physical factors. Therefore, a better understanding of musculoskeletal abnormalities of the craniocervical region can provide more information about the pathophysiology of TTH and may facilitate the development of new treatment programs using physical therapy.
Musculoskeletal impairment in the craniocervical region has traditionally been linked to various types of headache (18,19). Simons et al. described headache as the referred pain from myofascial trigger points (MTrPs) in the head and neck muscles (20). Increased pericranial tenderness has been found in patients with both ETTH and CTTH (17,21). In addition, migraineurs show increased tenderness in pericranial muscles, increased muscle sensitization to palpation, reduced pain threshold and an increased number of trigger points (19,22–24).
Posture is also known to be related to headache. The most frequently noted postural abnormality related to headache is an excessive forward head position or forward head posture (FHP). Greater FHP is considered a feature of cervicogenic headache or post-concussion headache, and it has also been reported in migraine, ETTH and CTTH (24–28). These findings suggest possible impairment of the craniocervical musculoskeletal system in individuals with headache, but the cause-and-effect relationship is not yet clear.
The function of the neck–shoulder region in headaches has been a matter of discussion for several decades. Reduced range of motion (ROM) in the neck has been considered a major feature of cervicogenic headache and studies have reported restricted neck mobility in migraine, ETTH and CTTH (24,25,27–29). However, another study involving assessment of neck mobility found no significant differences between patients with migraine or TTH and control subjects (29).
The present study was designed to investigate the presence of myofascial, postural and mechanical abnormalities in patients with ETTH and CTTH, and to compare these findings with the features of a headache-free control group.
This study was performed to measure several aspects of pericranial and cervical musculoskeletal function in patients with ETTH and CTTH, including MTrPs, postural (FHP) and mechanical (cervical active range of motion, AROM) abnormalities. In addition, we assessed the relationships among MTrPs, FHP, cervical AROM and several clinical parameters of headache related to the temporal profile and intensity of headache.
Methods
Subjects
For the ETTH and CTTH groups, we recruited patients with TTH who had visited the headache clinic of a tertiary care hospital between March and June 2008. The diagnoses of these headaches were made by the board-certified attending neurologist using the International Headache Society criteria (30). Subjects with ETTH were defined as those with headaches from 1 to 15 days per month (frequent ETTH). Subjects with infrequent ETTH were not included in the ETTH group. As infrequent ETTH has very little impact, it is considered a normal phenomenon and not a disease. Patients with CTTH were those who had headaches for at least 15 days per month for more than three successive months. To be included in the headache group, subjects had to have at least a one-year history of headache and typical symptoms of TTH to distinguish from mixed headaches (i.e. coexistence of TTH and migraine). Patients who were administered analgesics or muscle relaxants within 24 hours prior to the investigation or medication overuse headache as defined by the International Headache Society were excluded (30). Patients with neurological deficits or with a history of trauma or previous neck injury were also excluded.
The control group consisted of age- and sex-matched volunteers recruited from among hospital staff or the relatives or friends of patients. Participants in the control group were free of headache for at least three months prior to the study and experienced no more than occasional mild headache (occurring less than five times per year), for which they had never sought medical treatment. Furthermore, they had no history of cervical pain or injury for which they had sought treatment and no history of chronic pain.
All of the participants were examined physically and neurologically by an experienced neurologist. Computed tomography or MRI of the brain were performed in all headache patients. We also evaluated cervical-spine X-rays of all headache patients to exclude clear symptomatic causes such as fractures, congenital abnormalities, bone tumors, myelopathy, abnormalities of the craniocervical junction or rheumatoid arthritis with atlantoaxial subluxation (8). These radiological examinations of the brain and cervical spine indicated no pathological findings in any subject. They were asked to complete a questionnaire about their headache symptoms, including frequency, duration and intensity of headache in the last four weeks. Headache frequency (days/week) was calculated by dividing the number of days with headache by four weeks. Headache duration (hours/day) was calculated by dividing the sum of the total hours of headache by the number of days with headache and headache intensity (Visual Analogue Scale [VAS]: 0–10), which was then calculated from the mean of the VAS of the days with headaches. All subjects were examined on days when headache intensity was less than three points on the VAS. All subjects gave their written informed consent to participation in the study, and the study was approved by the local ethics committee.
Myofascial evaluation: Identification of myofascial trigger points
For myofascial evaluation, the location and number of MTrPs were determined. The rehabilitation doctor, who had no information about the patients (including the results of diagnosis), performed MTrP identification in the bilateral upper trapezius, sternocleidomastoid, temporalis and suboccipital muscles in headache and control subjects. We used the methods of Gerwin et al. (31) to identify MTrPs.
The existence of MTrPs was investigated using snapping palpation (first to locate a taut band of muscle, placing the fingertip at right angles, and then moving the thumb tip back and forth to roll the underlying fibers) to induce a local twitch response and flat palpation (placing the padded aspect of the thumb at a right angle on the muscle fibers and applying pressure against the underlying tissue or bone) to induce local pain and referred pain. The presence of MTrPs was determined according to the proposed diagnostic criteria of MTrPs (20): the presence of a palpable taut band, a tender spot within a taut band, a local twitch response by snapping palpation of the taut band and referred pain evoked by flat palpation of the tender spot that reproduces pain on the typical headache site. A MTrP was considered active if the referred pain evoked by its compression reproduced the same headache in the patient, whereas MTrPs were considered latent if the evoked referred pain failed to reproduce any familiar headache symptoms (20,31). The numbers of active and latent MTrPs on each muscle were calculated.
Posture abnormality evaluation: Measurement of flexor head posture
FHP was used for evaluating posture abnormalities. The physical measures of the cervical FHP were conducted by an examiner blinded to the headache or non-headache status of each subject. Photographs were used to obtain a series of measurements of postural alignment of the head and shoulders in sagittal planes with the patient in a comfortable sitting position on a straight-backed chair. A camera was mounted on a tripod, the center of the lens was 1 meter from the subject, and the base of the camera was matched with the height of the subject’s shoulder. A plumb line was suspended from the ceiling, and horizontal lines were drawn at the wall to provide vertical and horizontal references. Bony landmarks with red squares of 1 cm2 were attached at the left tragus and the C7 spinous process, which would be visible in photographs. Subjects were seated in a comfortable posture on a backed chair and the instructions were given to place their weight evenly on both buttocks and feet, maintain the hips and knees at an angle of 90° and place the hands on their laps with their eyes looking forward. They were asked not to sit straight up in the “best posture” because the purpose of the photograph was to capture their habitual or usual sitting postures.
Measurements of FHP in the sagittal plane were made from left-profile photographs of the subjects. The photographs were digitized in one dimension, and skin markers, anatomic landmarks and reference lines on the image were used to calculate the degree of FHP.
The sagittal C7-tragus angle was determined (Figure 1); a line was drawn between the midpoint of the tragus of the left ear and the C7 spinous process, and the angle of this line to the horizontal was calculated in degrees. This measurement was performed using PiView STAR picture archiving and communication systems (Infinitt, Seoul, Korea). This sagittal C7-tragus angle described the position of the head relative to C7. As the head was positioned further forward, the value increased. This procedure was described in a previous study (32), and this method has already been confirmed to have high reliability, with a high intraclass correlation coefficient.
Measurement of the sagittal C7-tragus angle.
Mechanical abnormality evaluation: Measurement of the active range of neck motion
For evaluating mechanical abnormalities, measurement of neck mobility was performed in triplanar cervical motion. AROM was measured with a Dualer IQ electronic dual inclinometer system (JTECH Medical, Salt Lake City, UT, USA) according to the manufacturer’s instructions and the technique recommended in the American Medical Association Guides to the Evaluation of Permanent Impairment for the two-inclinometer method (33).
We recruited healthy control subjects to assess the reliability of the Dualer IQ electronic dual inclinometer system. In addition, the range of neck motion was measured in the directions of flexion (forward), extension (backward), lateral flexion (right, left) and axial rotation (right, left) of the cervical spine in six ways. These measurements were performed over two trials in each of six movement planes by one of the authors (JHS) and a medical resident of the participating institution. One week later, one of the authors (JHS) measured the cervical mobility of the subjects repeatedly to check intra-tester reliability. Then we assessed the inter- and intra-tester reliability of the digital inclinometer findings. The results of reliability measurements for subjects showed high intra-tester reproducibility in all planes (intra-tester reliability: forward 0.924, backward 0.959, right lateral flexion 0.866, left lateral flexion 0.793, right rotation 0.854, left rotation 0.982; inter-tester reliability: forward 0.843, backward 0.875, right lateral flexion 0.684, left lateral flexion 0.712, right rotation 0.752, left rotation 0.681). Next, an examiner blinded to the condition of the patients measured the neck mobility of headache and control subjects. The test was conducted twice in each way and the average value was recorded.
Statistical analysis
SPSS statistical software (version 12.0; SPSS, Chicago, IL, USA) was used for all analyses. The numbers of active and latent MTrPs and mean values of the C7-tragus angle and all cervical AROM were calculated. Differences in the number of MTrPs and values of the sagittal C7-tragus angle and AROM of cervical mobility between the study groups were assessed with the Kruskal–Wallis analysis of variance (ANOVA) test with the Mann-Whitney U-test. The chi-square test was used to compare the presence of latent and active MTrPs. Pearson’s correlation test (
Results
Subject demographics
Demographic data and headache features
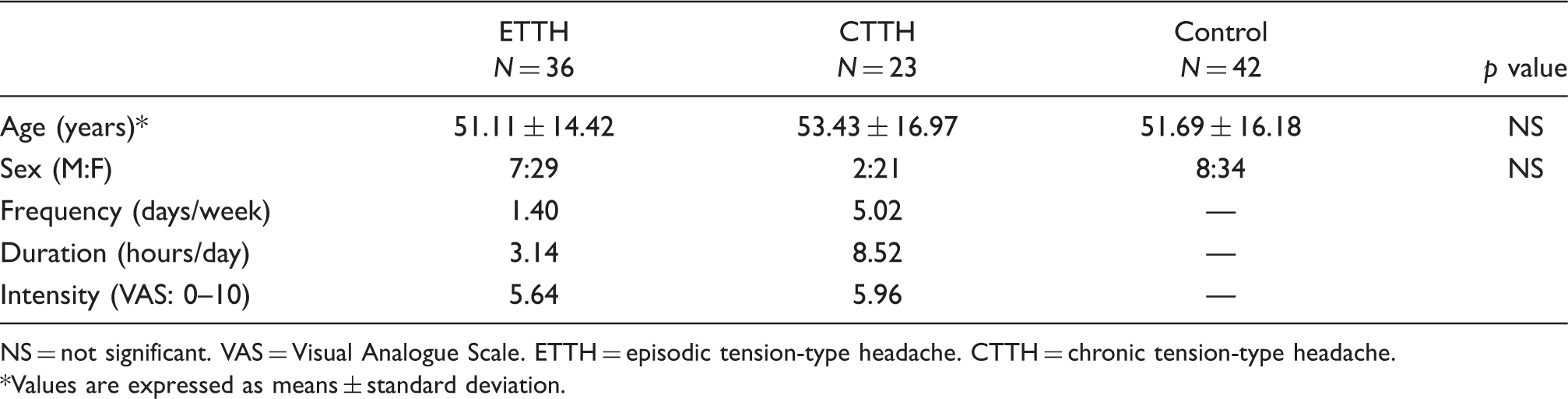
NS = not significant. VAS = Visual Analogue Scale. ETTH = episodic tension-type headache. CTTH = chronic tension-type headache. *Values are expressed as means ± standard deviation.
Myofascial trigger points
Myofascial trigger point measurements of each study group

SD = standard deviation. MTrP = myofascial trigger point. ETTH = episodic tension-type headache. CTTH = chronic tension-type headache. NS = not significant.
Distribution of subjects with myofascial trigger points in each study group

MTrp = myofascial trigger point. ETTH = episodic tension-type headache. CTTH = chronic tension-type headache.
*, ** Differences in the distribution of MTrPs between ETTH and CTTH groups. (*
Flexor head posture
The results of statistical analyses revealed significant differences in the C7-tragus angle between the CTTH and control groups (137.74 ± 7.68 vs. 133.31 ± 5.6, Flexor head posture measurements of each study group. *
Cervical range of motion
Range of motion for cervical mobility of each study group
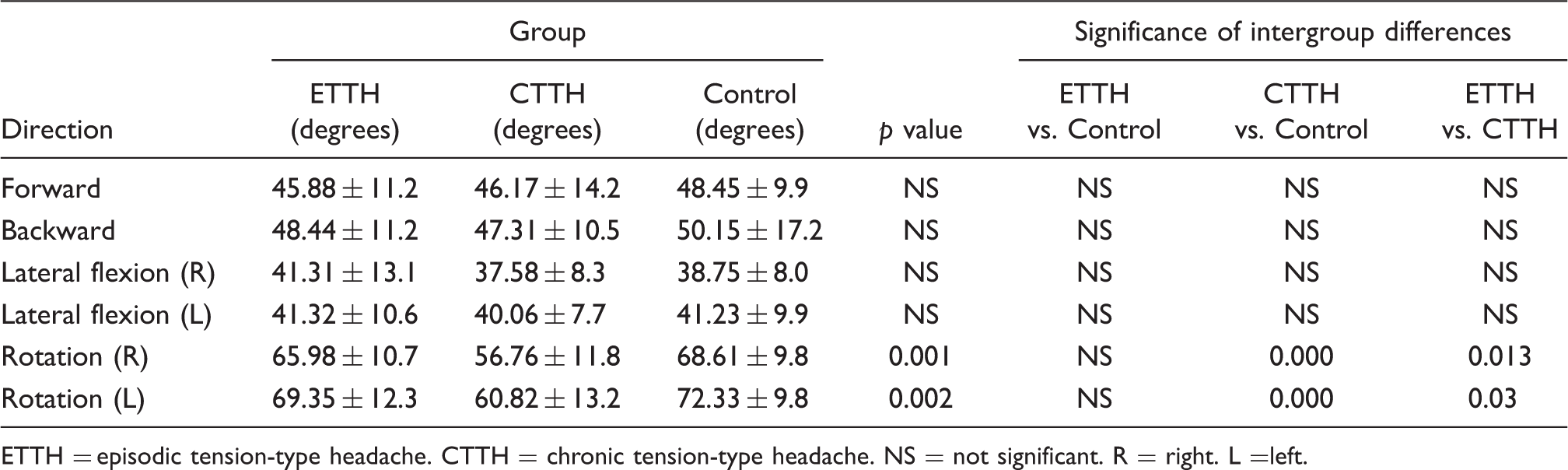
ETTH = episodic tension-type headache. CTTH = chronic tension-type headache. NS = not significant. R = right. L =left.
Correlation between headache parameters and musculoskeletal abnormalities
Correlation coefficients of headache characteristics from headache questionnaires and measured active MTrP numbers

ETTH = episodic tension-type headache. CTTH = chronic tension-type headache. VAS = Visual Analogue Scale. MTrPs = myofascial trigger points. NS = not significant. *
Discussion
The results of the present study indicated that the numbers of active and latent MTrPs in the ETTH and CTTH groups were significantly higher compared to the control group. In the CTTH group, the number of active MTrPs was significantly greater than in the ETTH group. In addition, patients with CTTH showed severe FHP and restricted neck mobility compared to the control group. Although the number of active MTrPs was correlated with the frequency and duration of each headache, no correlations were found between either FHP or neck mobility and any of the headache parameters.
This study differed from earlier studies in that we simultaneously evaluated cervical musculoskeletal dysfunction in ETTH and CTTH. In addition, we incorporated a control group that was carefully age- and sex-matched with the patients who had ETTH and CTTH.
Gerwin has suggested that the referred pain from MTrPs and MTrPs themselves are important in the patients’ perception of TTH (34). A headache is most likely due to referred pain from MTrPs in the head, neck and shoulder muscles, mediated through the spinal cord and the brain stem trigeminal nucleus caudalis, rather than direct tenderness of the pericranial muscles themselves. Our results support the hypothesis that MTrPs in the head, neck and shoulder muscles play an important role in the etiology of TTH. Specifically, the active MTrPs are estimated to be the most important contributing factors in causing, maintaining or perpetuating TTH.
Previous studies have indicated that active MTrPs in the upper trapezius, sternocleidomastoid, temporalis and suboccipital muscles were also present in ETTH (27,35), to a similar degree as in CTTH (36). However, the results of the present study were different from those of previous studies. The number of latent MTrPs was similar in the ETTH and CTTH groups (3.57 vs. 3.74). The difference between the ETTH and CTTH groups was in the number of active MTrPs, which was significantly greater in the CTTH group (0.5 vs. 2.43,
Our results indicated more active and latent MTrPs in all headache groups compared to European studies, even taking into account differences in study design and methods. This may have been due to differences in sociocultural background and lifestyle. Furthermore, racial differences seem to exist regarding the number of MTrPs, with MTrPs being more prevalent in Asians than in Europeans. Differences in expression, interpretation of pain and actual symptoms may account for these racial differences. In addition, a study has suggested that the likelihood of developing pain-producing active MTrPs increases in middle age (20). Thus, the increased number of MTrPs in the present study may have been a secondary effect of the older mean age of the study population.
Patients with TTH have been reported to show greater numbers of active or latent MTrPs compared to healthy subjects. However, this study did not investigate the prevalence of TTH in which muscle MTrPs were more frequent (22). Within a cohort of 15 ETTH sufferers, the most prevalent MTrPs were found in the temporalis (active MTrP, 86%; latent MTrP, 60%), followed by the upper trapezius (active MTrP, 46%; latent MTrP, 80%) and sternocleidomastoid (active MTrP, 33%; latent MTrP, 60%) muscles. Furthermore, active MTrPs of the suboccipital muscle were found in 6 of 10 patients (60%) with ETTH (27,35). In addition, another study with a cohort of 25 CTTH sufferers showed similar MTrP prevalence rates in the temporalis (active MTrPs, 68%; latent MTrPs, 72%) and upper trapezius (active MTrPs, 60%; latent MTrPs, 72%), followed by the sternocleidomastoid (active MTrPs, 44%; latent MTrPs, 68%) muscles. Moreover, active MTrPs of the suboccipital muscle were seen in 13 of 20 patients (65%) with CTTH (36,37).
These findings correspond to the results of the present study, in which significant differences were found in the distributions of both active and latent MTrPs for most muscles analyzed in patients with ETTH and CTTH compared to controls. However, our results partly contradict those of a previous study with regard to different affected muscle distribution of MTrPs. However, these results were based on a relatively small sample size; large controlled clinical studies are required. Our results indicated that active MTrPs are not only significantly prevalent on the upper trapezius and suboccipital muscles based on analysis of each muscle distribution, but are also greater in number in CTTH than in ETTH. Therefore, these results may support the importance of active MTrPs in CTTH.
In addition, TTH subjects with active MTrPs in the upper trapezius, sternocleidomastoid, temporalis and suboccipital muscles showed greater headache frequency and longer headache duration, but not headache intensity, compared to those with latent MTrPs in the same muscles. Active MTrPs were reported to show greater headache duration and intensity than latent MTrPs in patients with CTTH (36). However, within the ETTH group, none of the headache clinical parameters were related to the activity of MTrPs, that is, active or latent (35). Nevertheless, our results showed that the number of active MTrPs was significantly and positively correlated with the headache parameters in both ETTH and CTTH groups. These results were similar to those reported previously for CTTH groups but different for ETTH. In addition, in multiple regression analysis, only active MTrPs were significantly related to the frequency and duration of headaches in the CTTH group. These results therefore indicated that active MTrPs were the most important factor in triggering and sustaining headaches.
Within our findings, the intensity of headache was not correlated with the musculoskeletal abnormalities in the TTH groups. We used a headache questionnaire pertaining to the previous four weeks that was completed on the day of examination instead of a headache diary. We may have collected more sufficient and precise information about the required content of headaches if we had used a headache diary instead of a headache questionnaire.
Patients with CTTH showed greater FHP in the sitting position than headache-free subjects. However, no significant difference in FHP was observed between the ETTH and control groups. Our data showed that both the TTH and control groups had a larger sagittal C7-tragus angle, suggesting more severe FHP compared to previous studies (24,27,28,32,37). Several reasons exist for this result. The first is age: previous investigations demonstrated that the relationship between the sagittal C7-tragus angle and age was statistically significant (
In the measurement of neck mobility, six directions of cervical motion showed a smaller angle in our study compared to previous reports (39–42). Measurement of a normative cervical ROM lacks a scientific consensus due to the use of a wide array of techniques and a lack of standardized procedures for its evaluation. Cervical spine mobility has been shown to decrease with age because of degenerative changes. However, correlation analyses showed a weak and inconsistent association. Therefore, age is not strongly associated with limited mobility. Zwart found no differences in neck mobility between patients with TTH and controls. However, the episodic and chronic patterns of headache were not distinguished in this study (29), whereas the Fernandez-de-las-Peñas et al. indicated that a group of patients with CTTH showed less neck mobility than healthy controls (43). Our results were in agreement with the previous data. Compared to controls, individuals with CTTH had a lower ROM in all six directions of cervical motion and significant reductions in two directions (right and left rotation). However, individuals with ETTH and control subjects showed no differences in ROM in all six directions. Previous reports suggested that a reduction in neck mobility was likely a consequence of the protective posture against pain. In addition, kinesiophobia of the cervical segment may have occurred given that head movements intensified pain during an attack (28,44). Muscles with active MTrPs have a restricted passive (stretch) ROM because of pain, as reported previously (45). The limitation of stretch due to pain is not as great with active movement as with passive lengthening of the muscle, at least partly due to reciprocal inhibition. Therefore, more restricted neck mobility may have occurred, as MTrPs were more prevalent in the CTTH group. Head movement can worsen a headache, and this may induce kinesiophobic behavior of the neck that may be manifested by reduced cervical ROM (46). The long duration of headaches in the CTTH group may affect cervical mobility due to the daily or near-daily kinesiophobic behavior. Furthermore, in cervical motion, rotation showed large ranges of neck motion compared to lateral flexion, forward flexion or extension. Therefore, the limitation of cervical motion may be accentuated to a greater extent in both rotation directions in patients with CTTH. However, restricted neck mobility did not affect the patients with ETTH due to the intermittent nature of the condition. Furthermore, previous studies showed that FHP could affect cervical mobility. FHP may lead to excessive compression of the facet joints and posterior surfaces of the vertebral bodies, thus affecting the biomechanics of the head and the neck (47,48).
In conclusion, the active MTrPs may be the contributing factor in causing and maintaining TTH. Although the FHP is suggested to activate MTrPs because it is related to the shortening of the head and neck muscles, the FHP may be the result of CTTH. The differences in neck mobility are also caused by abnormal head posture or chronic pain.
We showed that active MTrPs in the pericranial and neck–shoulder area were present in both ETTH and CTTH groups, and more active MTrPs were found in patients with CTTH. In addition, active MTrPs were related to headache clinical parameters. The results suggested that temporal and/or spatial summation of peripheral stimuli may have a role in people predisposed to the pathophysiological mechanisms of ETTH. The increased nociceptive input to a supraspinal structure may result in supraspinal sensitization. The central neuroplastic changes may affect the regulation of peripheral mechanism and lead, for example, to increments in pericranial muscle activity or release of neurotransmitters in the myofascial tissues. Our study suggested that more active MTrPs in the CTTH group were the consequence of central sensitization.
This study had some limitations. The sample size was small, and only TTH was included. In addition, patients with cervicogenic headaches may have been included in the TTH group because of ambiguous diagnostic criteria. Some difficulties remain in differential diagnosis, as considerable symptomatic overlap exists between cervicogenic headache and TTH. Therefore, evaluating changes in the headache clinical parameters, such as intensity after performing joint or nerve block, may be meaningful considering the possibility that cervicogenic headache may have been included in the group diagnosed with TTH.
Taken together, the results suggest that musculoskeletal abnormalities in the craniocervical region can be one of the causes of TTH, and abnormalities in both posture and neck mobility may be the result of chronic headache. Specifically, active MTrPs in the pericranial–neck–shoulder muscles play important roles in triggering or maintaining TTH. Therefore, inactivation of MTrPs in CTTH sufferers would be expected to reduce headache symptoms, and trigger point-specific physical therapy would be an effective treatment in patients with TTH. In addition, clinicians should be aware of the relationship between chronic headache conditions and poor craniocervical posture. Postural correction and re-education of cervical posture should be integral parts of both prevention and management of patients with CTTH. Therefore, treatment methods tailored to the individual patient should be developed, focusing on myofascial and postural abnormality in the chronic headache treatment protocol.
