Abstract
Background: Converging neuropsychological evidence suggests that in migraine executive functions (EF) may be affected during interictal periods.
Objective: To evaluate the functional connectivity of the fronto-parietal networks (FPN) known to be associated with EF, in migraine without aura (MwoA) patients, in the interictal period, in comparison to healthy controls (HC).
Methods: Using resting-state functional MRI (RS-fMRI), we compared functional connectivity within the FPN in 14 patients with MwoA versus 14 sex- and age-matched HC, and assessed the correlation between functional connectivity within FPN, clinical features of MwoA patients, and EF. We used voxel-based morphometry to assess whether between-group differences in functional connectivity were dependent on structural differences.
Results: Neuropsychological data revealed no significant executive dysfunction in MwoA patients. RS-fMRI showed that MwoA patients, compared to HC, had significant functional connectivity reduction within the right FPN and specifically in the middle frontal gyrus (MFG) and the dorsal anterior cingulate cortex. In addition, we found that MFG reduced connectivity was negatively correlated with the pain intensity of migraine attacks. There were no structural differences between the two groups.
Conclusions: Our data suggest that, even in the absence of clinically evident EF deficits, MwoA is associated with reduced FPN functional connectivity. This study provides further insights into the complex scenario of migraine mechanisms.
Introduction
Migraine is a common and disabling primary headache disorder in which episodic dysfunction of brain excitability may play an important role. Migraine might be considered as a chronic brain disease characterized by repeated migraine attacks (1); however, its pathophysiology is not completely understood and is a matter of ongoing research. Recent neuropsychological evidence suggests that migraineous mechanisms may affect cognitive functions both during the migraine attacks and interictal periods. Specifically, migraine correlates with ictal (2,3) and interictal (4,5) executive dysfunction, which presumably reflects frontal lobe abnormalities. Significant reduction of frontal lobe gray matter (GM) density has been reported in migraine patients (6). However, so far the relationship between disease mechanisms and cognitive impairments is not fully understood (4), and some authors have found no deficits in cognitive abilities in migraine patients (7,8).
Recently, the development of resting-state functional magnetic resonance imaging (RS-fMRI) has advanced assessment of brain functional connectivity. This new technique is based on analysis of the spatio-temporal distribution of the spontaneous coherent fluctuations of blood oxygen level-dependent (BOLD) signals within and between functionally linked cortical regions, and allows identification of the so-called resting-state networks (RSN) (9). A growing body of studies has documented the importance of RS-fMRI in depicting functional disconnection in psychiatric and neurological disorders (10). So far, only few studies have evaluated the abnormalities of RSN connectivity in migraine patients (11,12).
On the basis of emerging neuropsychological evidence pointing to a subclinical executive dysfunction in migraine patients (2,5), we aimed to explore the functional integrity of the fronto-parietal network (FPN) (13,14), a circuitry encompassing prefrontal, cingulated, and parietal cortices, which is considered to be strongly associated with executive functions (EF) (15,16), in migraine without aura (MwoA) patients. This was done by RS-fMRI, with an independent component analysis (ICA) (17) approach to avoid any a priori hypothesis about the source of a possible functional disconnection. We hypothesized that the functional integrity of FPN might be disrupted, in MwoA patients, in the interictal period, when compared to healthy controls (HC). In addition, we used voxel-based morphometry (VBM) (18,19) to assess whether between-group differences in functional connectivity were dependent on structural differences.
Methods
Patient population
Twenty consecutive episodic MwoA patients, according to the International Headache Society (ICHD-II) criteria (Headache Classification Subcommittee of the International Headache Society, 2004) were prospectively recruited from the migraine population referring to the outpatient headache clinic of the Department of Neurological Sciences at the Second University of Naples. Patients with hypertension, diabetes mellitus, heart disease, stroke, cognitive impairment, substance abuse, and chronic pain, as well as other neurological or psychiatric disorders were excluded. To avoid any possible migraine or pharmacologically related interferences, MwoA patients were both migraine-free and had not taken attack medications for at least 3 days before the MRI scan and they were not under any chronic prophylactic regimen for migraine prevention. MwoA patients were interviewed 7 days after the MRI scan to ascertain that they were also migraine-free in the week following the experimental day. By doing so, six enrolled MwoA patients were excluded and only 14 MwoA patients (mean age ± SE: 30.57 ± 8.41 years, seven/seven males/females) were included in the final analysis. Demographic data and the following clinical characteristics were obtained from the patients: age at disease onset, disease duration, migraine frequency (day/month), duration of migraine attacks, and mean pain intensity of migraine attacks. The latter was assessed by visual analogic scale (VAS), measured on a paper-based 10-cm non-graduated line. As previously suggested (20), to obtain an accurate assessment of patients’ headache-related disability, MwoA patients completed the MIDAS (Migraine Disability Assessment Scale) and the HIT-6 (Headache Impact Test).
Clinical characteristics of MwoA patients and HC.
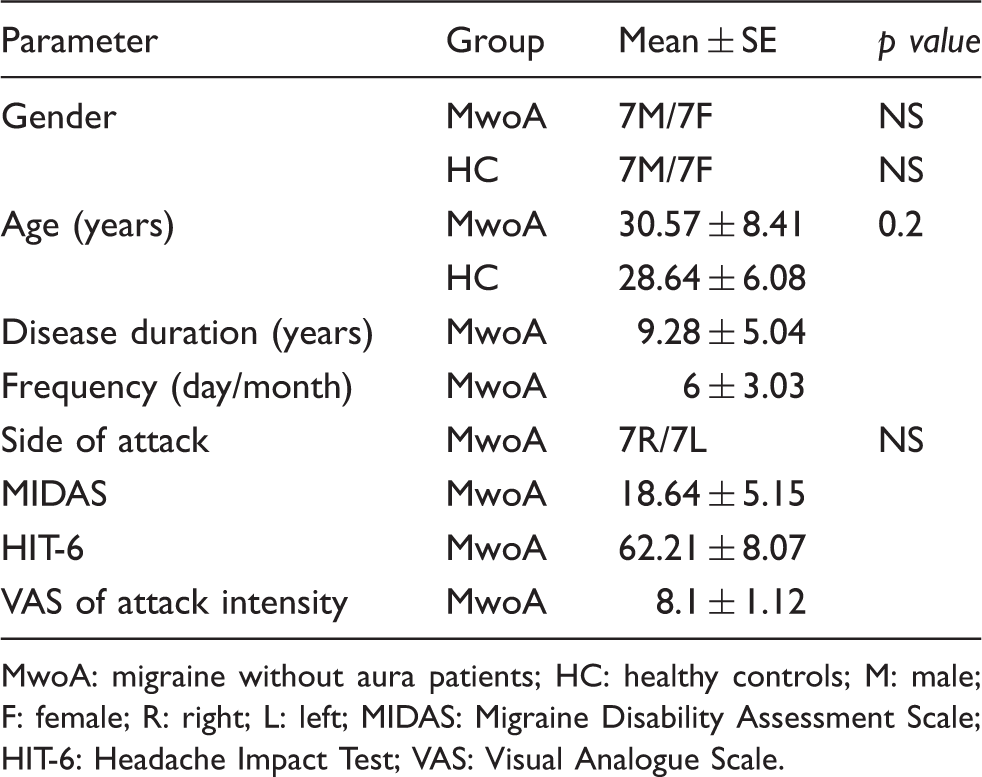
MwoA: migraine without aura patients; HC: healthy controls; M: male; F: female; R: right; L: left; MIDAS: Migraine Disability Assessment Scale; HIT-6: Headache Impact Test; VAS: Visual Analogue Scale.
All patients and subjects underwent preliminary MRI examination before entering the present study, to exclude any relevant structural abnormality. In particular, no patients had visible T2-weighted hyperintensities in deep white matter (WM).
Standard protocol approvals, registrations, and patient consents
The study was approved by the local ethics committee, and written informed consent was obtained from all subjects.
Neuropsychological test
General intellectual ability of patients was screened using the Mini-Mental State Examination (MMSE). As in a previous study (8), the following battery of standardized tests was administered to evaluate EF.
Trail Making Test Part A and B. Part A (TMTa) assesses visuomotor tracking and selective attention. Part B (TMTb) evaluates divided attention and set-shifting.
Wisconsin Card Sorting Test (64 card modality) (WCST). A widely used test to measure cognitive flexibility, that is the ability to alter a behavioral response mode in the face of changing contingencies (set-shifting). Scores adopted in the study included the numbers of categories achieved (WCSTc) and the number of perseverative errors (WCSTp).
Phonemic Fluency (PF). This is a word generation task based on a phonological cue, sensitive to frontal dysfunction.
Moreover, we also administered the Frontal Assessment Battery (FAB) (21) and Raven PM 47 (22).
FAB. This is a short cognitive and behavioral test battery for the screening of a global executive dysfunction extensively used thanks to its ease of administration and claimed sensitivity.
Raven PM 47. This test assess the ability to form comparisons, deduce relationships, correlates, and reason by analogy.
Finally, to assess levels of depression and anxiety, at the end of the cognitive session, MwoA patients performed the Hamilton Depression Rating Scale (HDRS) and the Hamilton Anxiety Rating Scale (HARS).
Imaging parameters
MRI was performed on the 3 Tesla GE (General Electric, Minneapolis, MN, USA) Medical System whole-body scanner “Signa HDxt” equipped with an eight-channel parallel head coil. fMRI data consisted of 240 volumes of a repeated gradient-echo echo planar imaging T2*-weighted sequence (TR = 1508 ms, axial slices = 29, matrix = 64 × 64, field of view = 256 mm, thickness = 4 mm, interslice gap = 0 mm, 10 discarded scans at the beginning).
During the functional scan, subjects were asked to simply stay motionless, awake, and relaxed, and to keep their eyes closed; no visual or auditory stimuli were presented at any time during functional scanning. Three-dimensional high-resolution T1-weighted sagittal images (GE sequence IR-FSPGR, TR = 6988 ms, TI = 1100 ms, TE = 3.9 ms, flip angle = 10, voxel size = 1 × 1 × 1.2 mm3) were acquired for registration and normalization of the functional images as well as for atrophy measures and VBM analysis.
Statistical analysis of clinical data
Demographic and clinical features of MwoA patients and HC were compared using a t-test for independent samples or χ2, as appropriate.
RS-fMRI pre-processing and statistical analysis
Standard image data preparation and pre-processing, statistical analysis, and visualization were performed with the BrainVoyager QX software (Brain Innovation BV, The Netherlands). Nuisance signals (global signal, WM and cerebro-spinal fluid (CSF) signals and motion parameters) were regressed out from each data set (23). Single-subject and group-level ICA were carried out, respectively, using the fastICA and the self-organizing group ICA (sogICA) algorithms. For each subject, 25 independent components (corresponding to one-sixth of the number of time points) were extracted. All single-subject component maps from all subjects were then “clustered” at group level, resulting in 25 single-group average maps that were visually inspected to recognize the main physiological RSNs. The selection of the RSN components entailed identifying in each group component map the presence of anatomically relevant areas that reproduced the layouts of the main physiological RSNs as already reported in earlier RS-fMRI studies conducted with identical or similar ICA methodologies (15,24).
A group-level random effects (RFX) analysis was carried out treating the individual subject map values as random observations at each voxel (10,25). Single-group contrasts were used to map the whole-brain distribution of the FPN components (p = 0.05, cluster-level corrected). Then, an inclusive mask was created from these maps to define the search volume for within-network group comparisons (MwoA patients versus HC). These entailed calculating two-group contrasts at each voxel of the mask and accepting regional differences surviving a threshold of p = 0.05 (cluster-level corrected). The resulting statistical maps were overlaid on the standard “Colin-27” brain T1 template.
To correct for multiple comparisons in all RFX maps, regional effects were only accepted for clusters surviving the joint application of a voxel- and cluster-level statistical threshold chosen with a non-parametric randomization approach. Namely, an initial voxel-level threshold was set to p = 0.05 (uncorrected), and a minimum cluster size was estimated after 1000 Montecarlo simulations that protected against false-positive clusters up to 5%.
Individual ICA z-scores were extracted from regions identified in the above analyses and used in linear correlation analyses with headache-related disability, intensity of pain in the course of a migraine attack, and frequency of migraine. In fact, ICA z-scores express the relative modulation of a given voxel by a specific ICA component activity and hence reflect the amplitude of the correlated fluctuations within the corresponding functional connectivity network.
VBM
Data were processed and examined using SPM8 software (Wellcome Trust Centre for Neuroimaging, London, UK; http://www.fil.ion.ucl.ac.uk/spm), in which we applied VBM implemented in the VBM8 toolbox (http://dbm.neuro.uni-jena.de/vbm.html) with default parameters incorporating the DARTEL toolbox, which was used to obtain a high-dimensional normalization protocol (18). Images were bias-corrected, tissue-classified, and registered using linear (12-parameter affine) and non-linear transformations (warping) within a unified model (18). Subsequently, the warped GM segments were affine-transformed into Montreal Neurological Institute (MNI) space and were scaled by the Jacobian determinants of the deformations to account for the local compression and stretching that occurs as a consequence of the warping and affine transformation (modulated GM volumes) (19). Finally, the modulated volumes were smoothed with a Gaussian kernel of 8-mm full-width at half maximum (FWHM). The GM volume maps were statistically analyzed using the general linear model based on Gaussian random field theory. Statistical analysis consisted of an analysis of covariance (ANCOVA) with total intracranial volume (TIV) and age as covariates of no interest. We assessed whole-brain regional differences, as well as differences over region of interest (ROI) based on the results of the whole-brain between groups RS-fMRI analysis. Statistical inference was performed at the voxel level, with a family-wise error correction for multiple comparisons (p < 0.05).
Results
Clinical evaluation
The two groups enrolled in the study (14 MwoA patients and 14 HC) did not differ in age and male/female ratio. See Table 1 for further details on demographic and clinical features.
Neuropsychological data
Neuropsychological evaluation in MwoA patients.

MMSE: mini mental state examination; HARS: Hamilton anxiety rating scale; HDRS: Hamilton depression rating scale; TMT A: Trail Making Test Part A; TMT B: Trail Making Test Part B; WCST: Wisconsin Card Sorting Test; PF: phonemic fluency; FAB: frontal assessment battery; Raven PM 47: Raven Standard Progressive Matrices.
Values are relative to published normative data (see references for further details).
RS-fMRI and VBM
As also reported by Damoiseaux et al. (24), we identified two lateralized FPN components. Of these, the right FPN exhibited statistically significant regional differences in the groups comparison between MwoA patients and HC.
As illustrated in Figure 1 for the two groups separately, the right FPN corresponded well with the typically reported executive RS network (14,15), involving prefrontal and parietal cortical areas in both groups. The two-sample t-tests revealed significant group differences in the right middle frontal gyrus (MFG) (Talairach coordinates x,y,z: 27, 3, 46; BA 6) and in the right dorsal anterior cingulated cortex (dACC) (Talairach coordinates x,y,z: 4, 33, 35; BA 32), indicating that these regions had reduced component time course-related activity in MwoA patients compared to HC (Figure 2(a) and (b)). Post-hoc correlation analyses revealed that individual ROI averaged ICA scores in the rMFG were negatively correlated with pain intensity of migraine attack (r = −0.60, p = 0.03) (see Table 3 for details on correlation analysis).
Group level FPN connectivity in MwoA patients (a) and HC (b) (p < 0.05, cluster-level corrected). (a) T-map of statistically significant differences within the FPN between MwoA patients and HC groups (p < 0.05, cluster-level corrected) overlaid on the standard “Colin-27” brain T1 template. Talairach coordinates (x,y,z): top: right MFG = 27, 3, 46; bottom: right dACC = 4, 33, 35. (b) Bar graphs of the ROI-averaged ICA z-scores (±SD) for MwoA patients and HC groups. Top: right MFG (MwoA patients: 0.83 ± 0.16; HC: 1.96 ± 0.37; p = 0.001). Bottom: right dACC (MwoA patients: 0.74 ± 0.30; HC: 2.11 ± 0.31; p = 0.003). Correlation analysis between FPN connectivity and clinical parameters in MwoA patients. FPN:; MwoA: migraine without aura patients; MFG: middle frontal gyrus; ACC: anterior cingulate cortex; MIDAS: Migraine Disability Assessment Scale; HIT-6: Headache Impact Test; VAS: Visual Analogue Scale.


There were no differences in global GM, WM, and CSF volume between the two groups (GM: MwoA patients = 681.33 ± 73.80; HC = 688.47 ± 72.45; p = 0.72; WM: MwoA patients = 507.03 ± 60.62; HC = 498.38 ± 75.91; p = 0.72; CSF: MwoA patients = 200.87 ± 24.38; HC = 207.31 ± 49.41; p = 0.64; total atrophy: MwoA patients = 1389.23 ± 129.48; HC = 1394.06 ± 176.51; p = 0.93). Moreover, both the whole-brain and ROI-based analyses of regional volume differences did not reveal any differences in local GM between MwoA patients and HC, neither at a statistical threshold corrected for multiple comparisons (p = 0.05) nor at an uncorrected threshold (p < 0.001; cluster size:100).
Discussion
Here we report for the first time a reduced functional connectivity within the right FPN, at the level of the MFG and dACC, in MwoA patients with no clinically detectable EF deficits, during interictal periods.
RS-fMRI allows exploration of whole-brain functional connectivity with minimal bias towards a specific motor or cognitive function. In fact, the BOLD signals are topographically organized in highly reproducible functional networks, called RSN (15,16,24). The most commonly reported RSN are the default mode network (DMN) (25), the FPN (or executive network) (14), the sensorimotor network (SMN) and the visual and auditory networks (15,24). As previously reported (15,16), FPN seems to represent the neuronal correlates of EF.
EF are multifaceted, high-order cognitive processes that require the coordination and goal-orientation of several subprocesses, including cognitive flexibility, decision making, planning, feedback utilization, working and long-term memory (5,26). These complex operations, that are involved in daily living experiences (27), require direction of attention to pertinent stimuli, while behavioral choices are weighed against shifting conditions, background homeostatic demands, and context (16).
The neural substrates of EF were assumed to be located in the frontal lobes (28). However, several fMRI studies have revealed that EF can be attributed to distinct cortical regions. Specifically, the prefrontal cortex (PFC) is considered to have an essential role in performance monitoring and error detection (27,29). Likewise, other EF have been ascribed to ACC, a multi-integrative structure, showing considerable heterogeneity in terms of both functional and anatomical connectivity (30). In particular, fMRI studies considered dACC to be involved in attention-for-action/target selection, competition monitoring, anticipation, reward assessment and novelty, performance monitoring, conflict and error monitoring (31,32). Furthermore, converging fMRI evidence showed that dACC and PFC (BA 32 and 6) exhibited a strong functional connectivity with parietal cortex (near or along the intraparietal sulcus) (33), and that such a circuitry constitutes the FPN, which as a whole represents the underlying functional neuronal correlates of EF (13–16).
Our results are in line with a recent fMRI study showing functional changes of PFC and ACC during trigeminal repetitive stimulation in migraine patients (34). More recently, a RS-fMRI study showed regional abnormalities in cortical areas also related to pain processing, including PFC and right ACC in migraine patients (11).
Previous VBM studies in patients with chronic painful conditions have revealed different patterns of brain tissue loss in several regions known to be involved in pain processing. Specifically, MFG atrophy has been demonstrated in patients suffering from migraine, chronic daily headache, cluster headache, chronic back pain, and chronic pain due to primary hip osteoarthritis (32). The MFG, a multi-integrative structure known to be involved during the experience of pain, showed a tissue volume loss independently of the primary origin of the pain, suggesting that patients with chronic pain may share a common pattern of GM injury. Furthermore, in one study, right MFG decreased volume was associated with executive dysfunctions in migraine patients (4). Other VBM studies (6,31) have demonstrated a significant GM reduction in other regions such as ACC as well as parietal cortices in migraine patients.
Nevertheless, it is noteworthy that our RS-functional connectivity data are not related to structural differences as revealed by our VBM results, in agreement with a previous VBM study that revealed no change in the structure of the brain in MwoA patients compared to HC (35).
In migraine patients the presence of cognitive deficits remains under debate (2). Some neuropsychological studies demonstrated impaired performances in EF tests (3–5). On the contrary, our neuropsychological data revealing no cognitive deficits in EF, during interictal periods in MwoA patients, are in agreement with previous studies (7,8), which did not reveal any cognitive impairments in migraine patients.
Multiple factors, including sample sizes, different patient populations (i.e. migraine with and without aura, chronic migraine) and methodological issues (i.e. migraine patients with MRI WM lesions, different toolbox or SPM version) may have contributed to apparent differences in both VBM (1,6,33) and neuropsychological (2–5,7,8) results from previous studies.
In the present study, there were no correlations between FPN functional connectivity disturbances and clinical features such as age of disease onset, disease duration, frequency (day/month) and duration of migraine attack. Nevertheless, post-hoc analyses revealed that functional connectivity in the MFG was negatively correlated with the intensity of pain of migraine attacks (i.e. the higher the pain intensity, the lower the functional connectivity within FPN).
In conclusion, taken together our imaging and neuropsychological data revealed a regional reduced FPN functional connectivity, which is independent from structural abnormalities, in MwoA patients without cognitive disturbances in tests exploring EF integrity.
The interpretation of the present findings is not free from some limitations. First, the possible mechanisms underlying reduced FPN functional connectivity in MwoA patients are currently unknown and we can only speculate on the nature of these functional changes. Second, we do not know whether the differences in FPN between MwoA patients and HC are a primary phenomenon due to the hereditary liability or a secondary phenomenon as a result of repetitive migraine attacks. In fact, it is well known that cognitive and pain networks partially overlap and MFG and dACC are critically involved not only in EF but also in pain modulation and perception (34).
In conclusion, based on the detected reduced functional connectivity within FPN, we believe that MwoA patients, although free from clinically evident EF deficits, could experience some degree of difficulty in daily living. Indeed, daily living involves a number of complex executive processes including the maintenance of current goals, allocation of attentional resources, performance monitoring, inhibition of irrelevant stimuli or responses, detection of errors and the subsequent adjustment of behavior (27).
Future fMRI activation studies exploring EF in migraine patients are needed to further elucidate the complex relationship between migraine, cerebral FPN connectivity and patients’ susceptibility to external stressors.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
The authors confirm that there are no conflicts of interest.
