Abstract
Background:
Analysis of habituation patterns in patients with primary headache disorders allows the detection of changes to the excitability level of the trigeminal nociceptive system. Previous studies demonstrated a habituation deficit to painful stimuli in migraine and it was suggested that similar observations could be made in cluster headache (CH).
Methods:
Habituation of the “nociceptive” blink reflex (nBR) (R2 response) was studied in 66 CH patients (18 episodic CH inside bout, 28 episodic CH outside bout, 20 chronic CH) as well as in 30 healthy controls in a case-control study design.
Results:
Habituation behaviour was similar in CH and healthy controls as well as in CH subtypes. No side-to-side differences of habituation between headache side and non-headache side were detected.
Conclusion:
Our results did not detect an altered habituation in CH patients. Despite clinical similarities, migraine and CH seem not to share the same pathophysiological mechanisms in this regard.
Introduction
Habituation is defined as a process of reduced responses to repeated stimulations over time. It is thought to be a form of non-associative learning and represents a basal expression of nervous system plasticity (1). Habituation seems to be a general phenomenon in both humans and animals, which can be observed in all sensory modalities. In regard to pain processing, it helps to keep a healthy balance between nociceptive and anti-nociceptive mechanisms.
Lack of habituation is a common observation in several neurological diseases, including migraine (2–6) and Parkinson’s disease (7). A habituation deficit was shown even for first-degree relatives of migraineurs and, therefore, this might be an endophenotypic marker for pre-symptomatic migraine (6). Cluster headache (CH) and migraine share several clinical characteristics and the prevalence of migraine in patients with CH is increased compared to the general population. This overlap of clinical presentation led to the assumption of a similar pathophysiological pathway involved in these two primary headache disorders. Up to now, there are only few studies investigating habituation pattern in CH. Previous research results reported a habituation deficit even more pronounced than in migraine using the regular blink reflex (8). In contrast, studies using event-related potentials did not detect a lack of habituation in CH (9,10). Due to these conflicting results, habituation pattern in CH remains to be determined.
Investigation of the nociceptive blink reflex (nBR) allows a direct exploration of the functional state of the trigeminal nociceptive system at the brain-stem level (11). In contrast to the classical innocuous blink reflex (BR), the nBR is elicited by a stimulation electrode that predominantly stimulates cutaneous superficial nociceptive Aδ-fibres without depolarizing non-nociceptive Aβ-fibres in deeper layers of the skin (12). It sensitively reflects changes in trigeminal activity (13). Analysis of habituation patterns of the nBR R2 response has evolved to be an important electrophysiological tool to assess central pain processing mechanisms.
Recently, we investigated trigeminal nociception in CH using the nBR and observed a distinct side shift within central pain processing between the headache side and the non-headache side, predominantly in CH inside bout (14). This analysis did not address the question of habituation of consecutive response behaviour. Here we present a secondary analysis of the data in order re-evaluate the habituation of the nBR R2 response in episodic CH inside bout, outside bout and chronic CH in comparison to normal habituation in healthy controls. Due to the strict unilaterality of CH pain, the question of side differences between headache side and non-headache side was illuminated.
Material and methods
The study was approved by the local ethics committee. Written informed consent was obtained from all patients and healthy controls prior to study inclusion. Patients were recruited from a tertiary headache centre between January 2009 and February 2010.
The study design, as well as demographic and clinical characteristics of CH patients, was reported elsewhere (14). Briefly, 66 patients with CH according to the ICHD-II (International Classification of Headache Diseases, second edition) classification (code 3.1.) were investigated in a case-control study design. Forty-six patients suffered from episodic CH (code 3.1.1.). Eighteen of these were inside bout at the time of recording (mean age: 43 ± 12 years; range: 19–60 years) with a mean attack frequency of 1.2 ± 1.0 per day; 28 were outside bout at the time of recording (mean age: 42 ± 11 years; range: 19–67 years). The remaining 20 patients had chronic CH (code 3.1.2.) (mean age: 47 ± 12 years; range 19–65 years) with a mean attack frequency of 3.1 ± 2.7. None of the patients had suffered from an attack within the last four hours prior to measurement. All patients had no concomitant migraine and a negative family history for migraine (first-degree relatives). Cluster-related acute and prophylactic medication was maintained and prescribed independently from study participation according to current therapy guidelines.
Additionally, the nBR habituation was investigated in 30 healthy controls (mean age: 44 ± 14 years; range 20–68 years) who did not suffer from any primary headache disorder and had a negative family history for headache disorders.
All smokers (episodic CH inside bout: 78%; episodic CH outside bout: 79%; chronic CH 50%; healthy controls: 13%) were instructed not to smoke at least four hours before examination. All subjects were not allowed to drink caffeine-containing beverages or any alcohol at the day of study participation.
Electrophysiolgical settings
Two planar concentric electrodes (FA Walter Graphtek GmbH, Lübeck, Germany; http://www.walter-graphtek.com) were attached to the skin 10 mm above the entry zone of the supraorbital nerve approximately 2 cm apart. Stimulation was applied in the following setting: triple pulse, monopolar square wave, duration 0.5 ms, pulse interval 5 ms, interstimulus interval 12–18 seconds, pseudo-randomized, intensity for stimulation two times the individual pain threshold. Individual pain thresholds were determined, with two ascending and descending sequences of successive current intensities between 0.2 and 2 mA in 0.2 mA intensity steps. One block of 15 stimuli was applied to the headache and non-headache sides in CH patients, or to the right and left head sides in healthy controls. Two randomly selected stimulation sequences, “headache/non-headache” or “non-headache/headache” were used in CH patients, and “right/left” or “left/right” in healthy controls, to avoid possible bias.
The nBR was recorded using surface electrodes placed infraorbitally referenced to the ipsilateral orbital rim. Recording parameters were bandwith 1 Hz–1 kHz, sampling rate 2.5 kHz, sweep length 300 ms (1401 plus, Signal, Cambridge Electronic Design, Cambridge, UK).
Signal analysis was performed by an investigator blinded to the diagnosis and study design. The first sweep of each block was excluded from further analysis to avoid contamination by a startle response. The area under the curve (AUC) of the R2 response was analysed in each sweep offline after demeaning, rectification and averaging between 27 and 87 ms. Each block was analysed separately. Mean values for each block were calculated. The regression coefficient for each block (habituation), as well as the mean regression coefficient (MRC), for each stimulation side (headache vs. non-headache) were calculated. MRC < 0 means habituation, MRC > 0 means augmentation.
Statistical analysis
Analysis of covariance (ANOVA) was used for the analysis of regression coefficients (i.e. habituation) of CH patients and healthy subjects. The within-subject factor was side (headache side vs. non-headache side in CH patients and right vs. left side in healthy controls); between-subject factor was disease (episodic CH inside bout vs. episodic CH outside bout vs. chronic CH vs. healthy controls) . Bonferroni correction was used for post hoc analysis. The level of significance was set to p < 0.05. Pearson’s correlation was used to find correlations between regression coefficients and time from last attack. All statistics were calculated with SPSS 16 (SPSS Inc., Chicago, IL, USA).
Results
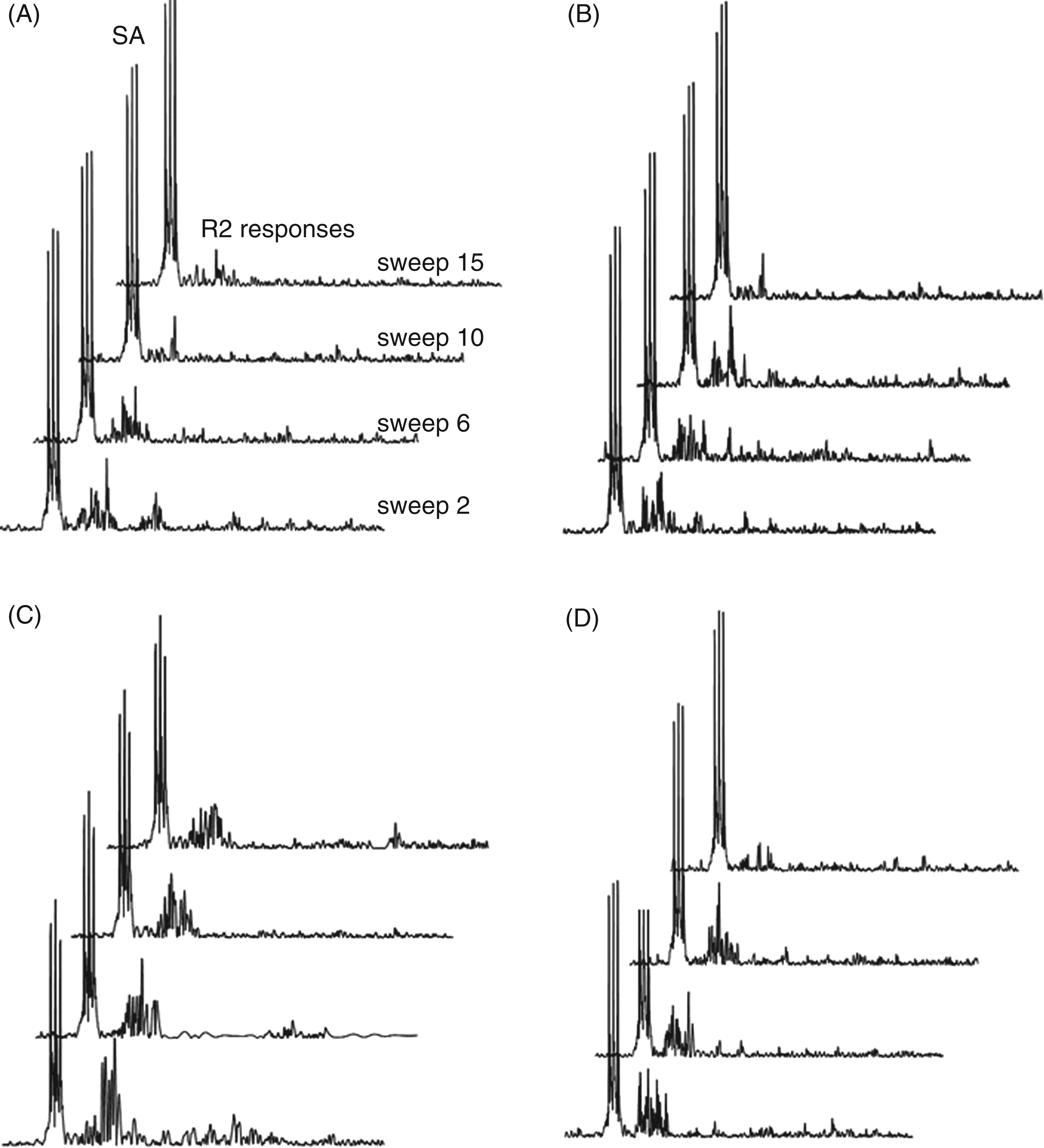
On all recordings, the R2 component of the nBR was clearly identified and there was no R1 component. In CH patients, nBR R2 responses habituated with repetitive stimulation (mean regression coefficient: episodic CH inside bout [MRC headache side = −2.2, MRC non-headache side = −1.2; p = 1.0]; episodic CH outside bout [MRC headache side = −1.4, MRC non-headache side = −1.2; p = 1.0]; chronic CH [MRC headache side = −1.4, MRC non-headache side = −1.4, p = 1.0]). No significant differences between headache and non-headache side stimulation were detected. No significant correlation between R2 component habituation and time from last attack was detected. In healthy controls, similar habituation was observed (MRC right-sided stimulation = −1.4, MRC left-sided stimulation = −1.8; p = 1.0). There were no significant difference of nBR response comparing CH and healthy controls (ANOVA: factor disease degrees of freedom (d.f.) 7; F 0.754; p = 0.626). Group mean regression lines are shown in Figure 1. Exemplary traces of single rectified nBR responses for each group are shown in Figure 2.
Habituation of the nociceptive blink reflex R2 response in cluster headache (CH). Dashed lines indicate individual regression lines (across 14 sweeps); bold lines indicate group mean regression lines. Dotted lines indicate the reference line (r = 0). (A) Healthy controls; (B) episodic CH, inside bout; (C) episodic CH, outside bout; (D) chronic CH. R: slope coefficient. Slopes of only one side are shown: headache side in CH patients and right side in controls. Habituation pattern of typical single rectified nociceptive blink reflex responses. Sweep 2, 6, 10 and 15 R2 responses are displayed in (A) healthy controls; (B) episodic cluster headache (CH), outside bout; (C) episodic CH, inside bout; and (D) chronic CH. The first sweep of each block is excluded from further analysis to avoid contamination by startle response. No significant difference of R2 habituation was observed between the several groups. Traces of only one side are shown; headache side in CH patients and right side in healthy control. SA: stimulus artefact.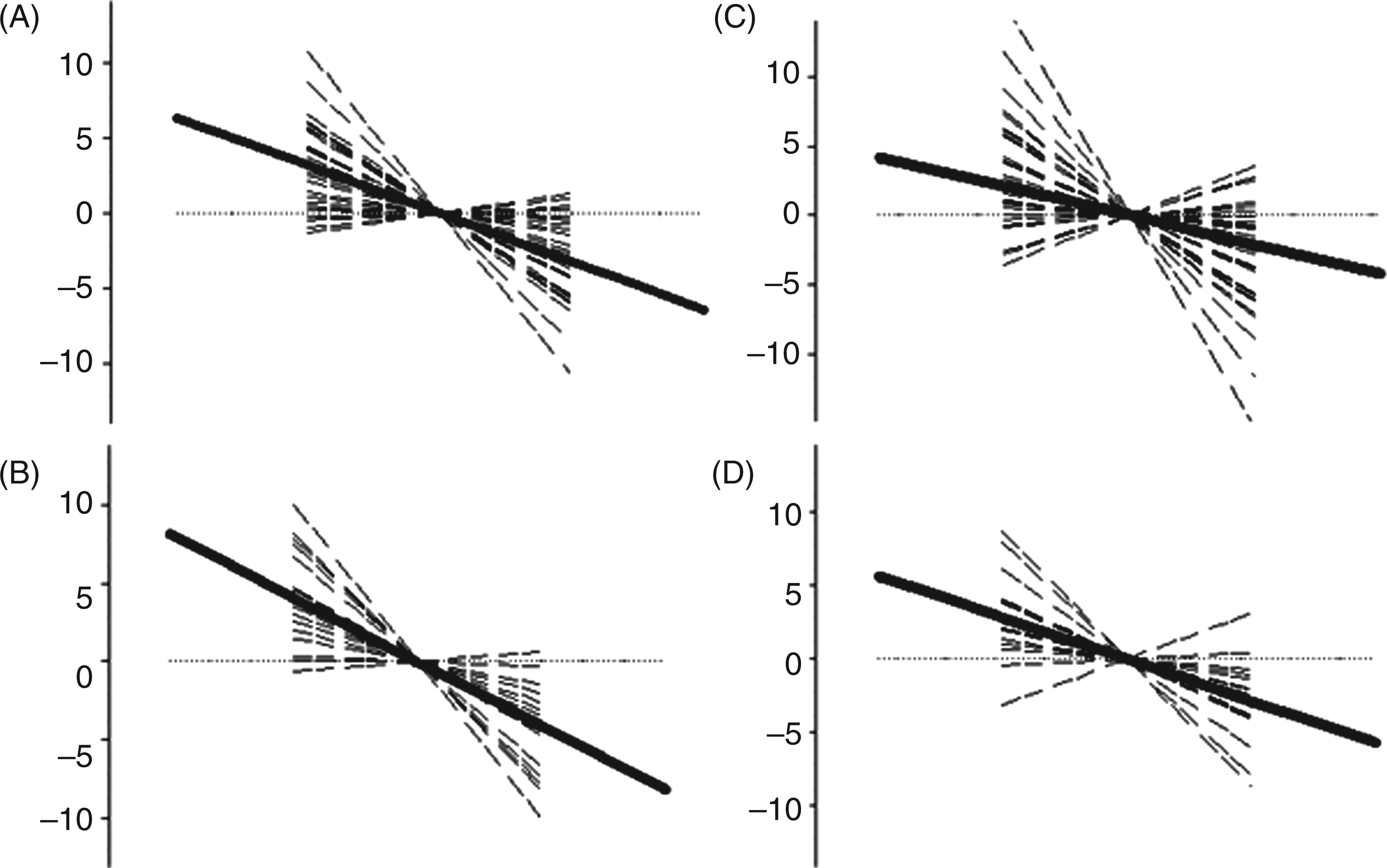
Discussion
Our results show no habituation deficit in chronic and episodic CH inside and outside bout compared with healthy controls. Furthermore, no significant side differences between headache side and non-headache side were detected in all groups. Subgroup analysis did not reveal differences of habituation responses comparing patients with CH in different stages of disease (inside bout vs. outside bout vs. chronic).
Our results contradict prior data by Perotta et al., who investigated the habituation behaviour of the classical BR (R2 and R3 component) in 27 CH patients with inside bout, 22 migraineurs and 20 healthy controls. Results showed a significant habituation deficit in CH patients compared with healthy controls, which was even more pronounced than in migraine patients. The authors suggested that abnormal suprasegmental modulation of the BR might be responsible for this observation (8). One explanation for these differing results might rest in the different electrophysiolgical methods used in the two studies, notably the classical BR and the nBR, as they involve varying reflex arches. First of all, BR and nBR are elicited by activation of different populations of trigeminal fibres. For elicitation of the nBR, nociceptive Aδ-fibres are stimulated selectively with little contamination (< 10%) of somatosensory Aβ-fibers (12), while in contrast, for BR elicitation, Aβ-fibers are stimulated on a larger scale (15), leading to activation of other parts of the somatosensory and central processing systems. The medullary interneurons of the nBR are predominantly located in the subnucleus caudalis of the trigeminal spinal tract nucleus (STN); in contrast, the BR involves interneurons located more rostrally at the level of the inferior olive, for example, at the subnucleus interpolaris (16). This anatomic difference might contribute to the observed divergent results. Furthermore, one can hypothesise that noxious and innocuous trigeminal stimuli are modulated by different suprasegmental or segmental influences. Additionally, the stimulation mode used was different. Perotta el al. delivered the electrical stimuli in five different frequencies (0.2, 0.3, 0.5, 0.7 and 1 Hz) (8), which all were much faster than in our experiment, where the interstimulus interval (ISI) was between 12 and 18 seconds. In contrast to the nBR, the classical BR does not habituate to ISI intervals as low as 15 s (4,7). Most likely, there is an influence of the stimulation frequency on habituation, and an alteration might be more pronounced for higher stimulation frequencies, as already suggested by the authors themselves (8). In which regard an increase of stimulation frequency may have influenced our results will be subject to future studies. However, our stimulation paradigm is very similar to the one used by Karsarava et al., who detected a significant habituation deficit in migraine patients (2). Therefore, the paradigm used seems to be able to detect habituation pattern in headache. A different, very simple explanation of habituation observed by Perotta et al. might be that some of the investigated CH patients also possibly had migraine or first relatives with migraine and were, therefore, prone to have a habituation deficit. Different disease phases as well as drug therapy might also be significant confounders responsible for the divergent study results.
Our finding of normal habituation in CH is in line with studies on event-related potentials in this patient group (9,10). Evers et al. investigated visually evoked event-related potentials (ERPs) in 50 episodic CH patients inside bout and 11 chronic CH patients. There was no cognitive habituation deficit in CH as has been detected in migraineurs. The authors hypothesised that pathophysiological mechanisms of mitochondrial energy metabolism and serotonergic pathways dysfunction, which are thought to contribute to a lack of habituation in migraine patients, do not play a relevant role in the pathophysiology of CH (10).
Reduced habituation is a commonly observed phenomenon in migraine patients. Habituation deficit was detected using several electrophysiological study methods, including nBR (2,5), classic BR (3,4), visually evoked potentials (17,18), auditory evoked potentials (18,19) and contingent negative variation (CNV) (20). A habituation deficit was also shown in first-degree relatives of migraineurs who were not affected by the disease (6). Interestingly, the nBR habituation deficit is detected predominantly interictally. During the acute migraine attack, the habituation normalises and is comparable with that of healthy controls (2). Additionally, previous studies showed that nBR habituation in migraineurs increases with increasing attack frequency (5,6). Our observed CH patients, namely inside bout and chronic patients, were apparently more likely to be recorded in a closer temporal relationship to a headache attack, which might contribute to the observation of a normal habituation pattern in these CH subgroups. However, this does not explain regular habituation in CH outside bout. A regular habituation was also shown in hypnic headache, a rare primary headache disorder that shares some clinical features (e.g. nocturnal headache attacks) with CH (21). Therefore, a lack of habituation is not a general phenomenon in headache diseases in general but seems to be a more specific electrophysiological feature in distinct headache entities such as migraine.
Habituation is a classic phenomenon of polysynaptic activity (22). There is only sparse information on the exact mechanisms associated with habituation. It is known that classical BR habituation is modulated by dopamine, as a lack of habituation was observed in Parkinson’s disease, which was found to be reversible under dopaminergic therapy (23). It remains to be determined if this pathway is also involved in the habituation of the nBR in primary headache disorders. Further hypotheses of habituation modulation include segmental control mechanisms, such as pre- or post-synaptic inhibition, homosynaptic depression and membrane desensitization (1,24–28), but suprasegmental influences mediated by descending modulatory nociceptive/anti-nociceptive pathways were also discussed as being involved. Previous functional neuroimaging studies showed an increased activity of the rostral anterior cingulate cortex (rACC) during habituation to pain (29,30), supporting its crucial role in regard to anti-nociceptive mechanisms in the habituation phenomenon. Schoenen et al. hypothesised that “ceiling effects” of neuronal activation might explain reduced habituation in migraineurs. According to this pathophysiological theory, migraine patients have a lower pre-activation level of the sensory cortex compared with healthy controls. After repetitive stimulation, migraineurs, therefore, have a larger range for activation up to the ceiling and thus lack proper habituation, whereas healthy controls reach the top of response activity faster and thus show appropriate habituation in response to painful potentially damaging stimuli (20). Our previous data already suggested that cortical excitability and especially efficiency of inhibitory top-down circuits are not altered in CH (14). This might explain why habituation remains unaffected in CH compared with migraine.
Some important limitations of this study have to be addressed. Prophylactic and acute pain medication might influence our results and account for differences between CH and healthy controls. To what extent medication intake affects habituation and in what manner has not yet been investigated. Unfortunately, separation into smaller, medication-adjusted groups is virtually impossible because of the large number of different medications and medication combinations, which would lead to very small group sizes without enough statistical power. Nicotine intake has to be considered as additional confounder. There was a between-group difference in smoking habits between CH and healthy controls. More patients than healthy controls were smokers, so an influence of smoking on our results cannot be ruled out completely. To minimize its possible effects, all patients and healthy controls were instructed not to smoke for at least four hours before study participation. Studies on the effects of nicotine on nBR habituation are lacking. However, we did not determine any significant correlation between smoking frequency and habituation behaviour, suggesting that its impact might be rather small. Additionally, in this study, habituation inside and outside bout was measured in different patients, which might influence the study results significantly. To confirm our data, a longitudinal investigation with measurement of the same patients inside and outside bout is already planned.
In summary, our data show normal habituation of the nBR R2 response to repetitive nociceptive stimuli in episodic as well as chronic CH. Different mechanisms, such as central sensitization, seem to play a more crucial role in the pathophysiology of this disease than alteration of habituation in CH. These results do not support the hypothesis that CH and migraine share the same pathophysiological mechanisms in regard to neuronal adaptation processes such as habituation, despite a considerable overlap in clinical characteristics.
Footnotes
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of interest
Dagny Holle has received a research grant from Grünenthal.
Silke Zillessen reports nothing to disclose.
Charly Gaul has received research grants and honoraria from Deutsche Gesellschaft für Muskelkranke, Roux-Program of the University of Halle-Wittenberg, MSD, Berlin Chemie, Medtronic and Böhringer Ingelheim.
Steffen Naegel reports nothing to disclose
Holger Kaube has received research grants and honoraria from MSD, Boston Scientific, St. Jude Medical, Medtronic Medical, Linde AG, Berlin-Chemie and Grünenthal.
Hans-Christoph Diener has received honoraria for participation in clinical trials, and has contributed to advisory boards or lectures for Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, Coherex Medical, CoLucid, Böhringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen-Cilag, Lilly, La Roche, 3M Medica, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, SanofiAventis and Weber & Weber. He has received research support from Allergan, Almirall, AstraZeneca, Bayer, Galaxo-Smith-Kline, Janssen-Cilag, and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF) and the European Union.
Zaza Katsarava has received research grants and honoraria from Allergan, Bayer, Biogen and Merck, and is an advisory board member for Allergan. Headache research at the Department of Neurology in Essen is supported by the DFG, the BMBF and the European Union.
Mark Obermann has received scientific grants by the German Federal Ministry of Education and Research BMBF 01EM 0513.
