Abstract
We studied the habituation of the ‘nociceptive’ blink reflex (nBR) in 15 healthy subjects and 17 migraine patients interictally as well as during unilateral migraine headache within six hours of onset and after treatment. In healthy volunteers the mean regression coefficient (MRC) was – 3.9 following right sided and – 4.9 left sided stimulation. This equals an amplitude loss of 19.5% (5 X −3.9) and 24.5% (5 X −4.9), respectively, across five consecutive sweeps. An augmentation of nBR responses was found in migraine patients interictally: MRC = 3.3 following stimulation of the headache side (HA) and MRC = 4.0 of the non-headache side (non-HA). The differences were statistically significant (ANOVA: d.f. = 1, F = 25.8, P < 0.001). During the migraine attack MRCs were negative both before (−5.0, HA and – 4.0, non-HA) and after treatment (−2.6, HA and −1.9 non-HA) and significantly differed from those outside the migraine attack (ANOVA: d.f. = 2, F = 12.4, P < 0.001). The demonstrated lack of habituation of the nBR responses indicates an abnormal trigeminal nociceptive processing in migraine patients outside the migraine attack.
Introduction
Migraine research has focused on the trigeminal nociceptive system as a pivotal player in the pathophysiology of migraine. One way of studying the trigeminal system is to examine blink reflex responses because the afferent part of the reflex arch is via the trigeminal nerve (1). However, previous blink reflex studies in migraine patients have produced contradictory results (2–4). In order to increase the sensitivity of the blink reflex to trigeminal pain processing, the ‘nociceptive’ blink reflex (nBR) has been established. The nBR is elicited by a novel concentric stimulating electrode, which by virtue of its new geometry predominantly stimulates cutaneous superficial nociceptive fibres without depolarizing more deeply lying non-nociceptive fibres (5). Therefore, the nBR is highly sensitive to changes in trigeminal nociception (6).
Recently, using the nBR we investigated trigeminal nociception in migraine patients and observed a facilitation of nBR responses during the acute migraine attack, predominantly on the headache side, which was not detectable in the pain free interval, indicating a temporary facilitation of trigeminal pain processing during the acute migraine attack (7). The analysis was based, however, on the mean of five consecutive nBR responses and did not address the question of habituation of consecutive responses behaviour. Here, we present a second analysis of the data in order to investigate the habituation of the ‘nociceptive’ blink reflex in comparison to habituation in healthy subjects.
The habituation of the blink reflex (R2 response) following application of repetitive stimuli has been established as an important electrophysiological tool to study the modified excitability of the blink reflex arc under the control of more rostral centres as demonstrated in disease conditions as idiopathic Parkinson's syndrome or Huntington's chorea (8–10). In cranial pain disorders, the habituation paradigm has been shown to be sensitive to detect functional pathological changes in the trigeminal system (11, 12).
Methods
Study design
The study was approved by the local ethics committee. Informed consent was obtained from all patients and volunteers.
The study design, as well as demographic and clinical characteristics of migraine patients were published previously (7). Briefly, 17 patients with migraine without aura (13) were investigated interictally, within the first six hours after onset of the acute attack and two hours after treatment (Drugs: lysine acetylsalicylate 1000 mg i.v. or zolmitriptan 5 mg p.o.). All patients had the last migraine attack at least three days before. In a follow up interview it was confirmed that none of the patients experienced a migraine attack within the next three days. The patients received no preventive or acute medication beside of the study medication.
The group of migraine patients consisted of 12 women and five men; median age 40 years, (range 24–56 years). Median illness duration was 20 years, (range 7–30 years), median attack frequency was three (range 2–5) attacks per months, which was equal to 8 (range 5–14) migraine days per month. All patients suffered from strictly unilateral moderate to severe migraine headache (median score: 7; range 6–9), on the verbal category scale (VCS): 0 = no pain, 10 = severe pain). Nine patients received zolmitriptan 5 mg p.o and seven patients received lysine acetylsalicylate 1000 mg i.v. Two hours after treatment the headache intensity was reduced to a median VCS score of 1, range (0–3), without significant differences between the treatment groups.
In addition, the habituation of nBR in a comparable group of healthy subjects (11 women, 4 men; median age 35 years; range 27–47 years) was studied. None of them received any acute or chronic medication other than oral contraception. All subjects were nonsmokers, fasted in the morning and were not allowed caffeine containing beverages or any alcohol on the previous night.
Electrophysiological stimulation and recording setup
The ‘nociceptive’ blink reflex was elicited as described previously (5). Interictal recordings in migraine patients as well as recordings in controls were performed between 0900 and 1100h. All subjects first attended an introductory recording session some days before the actual measurements to reduce anxiety and familiarize them with the procedure and experimental setup.
Stimulation: monopolar square wave pulse, duration 0.3 ms, interstimulus interval 15–17 s (pseudo-randomized), intensity for stimulation 1.5 times individual pain threshold. Individual pain thresholds were determined with two ascending and descending sequences of successive current intensities between 0.2 and 2mA in 0.1mA increments (mean pain threshold: migraine patients: 1.04 ± 0.23mA, subjects: 1.09 ± 0.15mA; mean stimulus intensity: migraine patients: 1.55 ± 0.55mA, subjects: 1.64 ± 0.22 mA). Two blocks of six stimuli were applied to the headache and non-headache sides in migraine patients, or to the right and left head side in controls. Two randomly selected stimulation sequences ‘headache/non-headache/non-headache/headache’ or ‘non-headache/headache/headache/non-headache’ were used in migraine patients, or similarly ‘right/left /left/right’ or ‘left/right/right/left’ in controls to avoid a possible bias.
Recordings
Bilateral surface electrodes placed infraorbitally and at the bridge of the nose, bandwidth 1Hz-1 kHz; digitization: sampling rate 2.5 kHz, sweep length 150 ms (1401plus, Signal, Cambridge Electronic Design, Cambridge, UK).
Analysis
PC-based offline analysis was computed with custom written software (Matlab 5.3, MathWorks, Natick, MA, USA). The first sweep of each block was excluded from further analysis to avoid contamination with a startle response. Areas under the curve (AUC) of the ipsilateral R2 response were analysed in each sweep off-line after demeaning, rectification and averaging between 27 and 87 ms (5). Each block was analysed separately. Mean values for each block were calculated. The data were normalized as a percentage deviation from the mean value of five consecutive sweeps and the regression coefficient for each block (habituation) as well as a mean regression coefficient (MRC < 0 means habituation, MRC > 0 means augmentation) for each stimulation side (HA vs. non HA) were calculated.
Statistical analysis
ANOVA was used for the analysis of habituation (regression coefficients) of migraine patients interictally and healthy subjects. Within subject factor SIDE (headache side vs. non-headache side in migraine patients and right side vs. left side in healthy subjects). Between subject factor: disease (healthy subjects vs. migraine patients interictally vs. healthy subjects).
ANOVA for repeated measurements was used for the analysis of habituation (regression coefficients) in migraine patients interictally, during migraine attack and after treatment. Within subject factors: time (baseline – migraine attack – drug treatment) and side of stimulation (headache – non-headache side). Between subject factor: drug (lysine acetylsalicylate – zolmitriptan). Correction of degrees of freedom was applied when necessary (Greenhouse-Geisser). Bonferoni corrected t-tests were used for posthoc analysis. Factors and interactions with error probabilities P < 0.05 were considered significant.
Results
Typical nBR responses are shown in Fig. 1. In healthy subjects nBR R2 responses habituated with repetitive stimulation (mean regression coefficient: − 3.9 right sided and − 4.9 left sided stimulation, with no significant differences between right and left sided stimulation). This equals an amplitude loss of 19.5% (5 ×−3.9) and 24.5% (5 ×−4.9), respectively, across five consecutive sweeps. In migraine patients, however, a lack of habituation of nBR responses was observed interictally: MRC = 3.3 following stimulation of HA side, and MRC = 4.0 on the non-HA side. The differences between migraine patients interictally and healthy subjects were statistically significant (ANOVA: factor disease d.f. = 1, F = 25.8, P < 0.001, factor side not significant; Fig. 2).
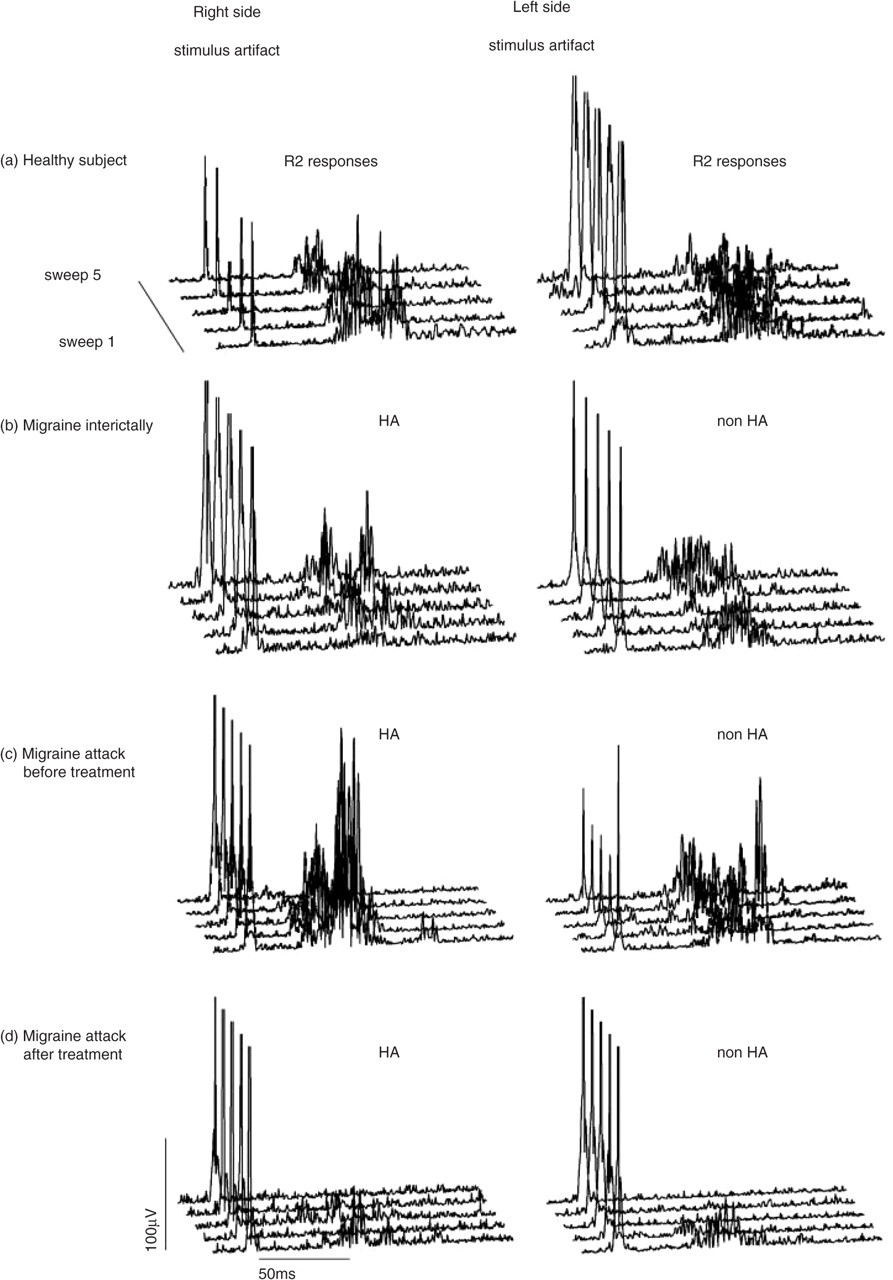
Typical traces of single rectified nBR responses elicited in trains (ISI 15–17 s) of five sweeps. Normal habituation is shown in a healthy subject. This pattern of habituation is suspended in migraine patients outside the acute attack. During the attack habituation is normal but the R2 responses are significantly increased.

Individual regression lines (across five consecutive sweeps);
The lack of habituation in migraine patients was not present on either side during the acute migraine attack (MRC =−5.0 for HA sided stimulation, and MRC =−4.0 for non-HA sided stimulation) and after drug treatment (MRC =−2.6 for HA sided stimulation, and MRC =−1.9 for non-HA sided stimulation). The differences in habituation of nBR responses interictally and during migraine attacks were statistically significant (ANOVA: factor time d.f. = 2, F = 12.4, P < 0.001, factors stimulation and drug as well their combinations not significant; Fig. 2).
Discussion
The main finding of the study is a significant difference in habituation expressed by regression coefficients between the migraineurs and healthy subjects. An abnormal modulation of R2 and R3 responses in migraine patients was reported recently 12. These findings are also in parallel to previous investigations on other modalities. Studies on visually or auditory evoked potentials as well as of the contingent negative variation (CNV) revealed deficient cortical habituation and increased intensity dependence in migraine patients compared to healthy subjects (14–16). One recent study, however, failed to confirm these findings Moreover, similar CNV abnormalities were found in subjects with a positive family history of migraine (17, 18). Thus, a deficit in habituative processes in migraine patients previously observed at a cortical level also seems to include systems at brain stem level such as the trigeminal nociceptive system. This globally deficient habituations detectable on a cortical level as well as in the trigeminal nociceptive system might serve as a trait marker for migraine patients and could be responsible for the increased susceptibility of migraine patients. This may explain the vulnerability of migraine patients to chemical challengers such as glyceroltrinitrate (19). Only 14 of 17 migraine patients showed an augmentation of nBR responses interictally. In contrast, a habituation of nBR responses was observed in 12 of 15 studied healthy subjects. Based on the data of this relatively small groups of migraineurs and healthy subjects we, however, can not explain the different behaviour of three migraine patients and three healthy volunteers. None of the studied controls had a first grade relative suffering from migraine, which could be a possible explanation for the observed habituative behaviour (18). Further, larger studies are needed to evaluate the specificity of this phenomena for migraine patients.
Interestingly, during the acute migraine attack habituation returned to normal as observed in healthy subjects. These findings are also in line to the previous findings of other groups. In a longitudinal study using CNV modality the maximal cortical susceptibility was observed on the day one before the acute migraine attack which returned to normal during further two or three days (20). In another study on event related potentials the impaired habituation continuously increased during the pain free interval and returned to normal during the acute migraine attack (21). A similar behaviour was observed using visual and auditory evoked cortical potentials (22).
In our study no differences were observed between the habituation of nBR responses following stimulation of the headache- and non-headache side despite of the strictly unilateral headache in all patients. As reported previously, both, lysine acetylsalicylic acid and zolmitriptan suppressed the nBR in parallel to the headache relief (7). The actual analysis of habituation pattern revealed, that neither acetylsalicylic acid nor zolmitriptan affect the habituation processes during the attack. It is a methodological drawback of this study that treatment was achieved in a not blinded fashion. The main study subjects, however, were the comparison of trigeminal transmission in and outside the attack and during the attack before and after relief of headache. These findings suggest, that the inhibition of the nBR R2 response was a consequence of the two potent migraine drugs. The observed normalization of habituation of nBR responses, however, has to be considered as specific for the migraine attack itself and not a consequence of drug application.
Footnotes
Acknowledgements
We thank Prof PJ Goadsby for his helpful discussion and review of the original manuscript.
