Abstract
Headache syndromes often involve occipital and neck symptoms, suggesting a functional connectivity between nociceptive trigeminal and cervical afferents. Although reports regarding effective occipital nerve blockades in cluster headache exist, the reason for the improvement of the clinical symptoms is not known. Using occipital nerve blockade and nociceptive blink reflexes, we were able to demonstrate functional connectivity between trigeminal and occipital nerves in healthy volunteers. The R2 components of the nociceptive blink reflex and the clinical outcome in 15 chronic cluster headache patients were examined before and after unilateral nerve blockade of the greater occipital nerve with 5 ml prilocain (1%) on the headache side. In contrast to recent placebo-controlled studies, only nine of the 15 cluster patients reported some minor improvement in their headache. Six patients did not report any clinical change. Exclusively on the injection side, the R2 response areas decreased and R2 latencies increased significantly after the nerve blockade. These neurophysiological and clinical data provide further evidence for functional connectivity between cervical and trigeminal nerves in humans. The trigeminocervical complex does not seem to be primarily facilitated in cluster headache, suggesting a more centrally located pathology of the disease. However, the significant changes of trigeminal function as a consequence of inhibition of the greater occipital nerve were not mirrored by a significant clinical effect, suggesting that the clinical improvement of occipital nerve blockades is not due to a direct inhibitory effect on trigeminal transmission.
Introduction
Patients suffering from primary headache syndromes with typically frontal symptoms, such as migraine, tension-type headache or cluster headache (CH), often complain of accompanying neck pain, stiffness or tenderness, suggesting participation of trigeminal and cervical innervation in central pain processing mechanisms in these disorders (1). The most likely mechanism for this observation is ‘referred pain’ originating from structures in the neck and projecting to facial areas and vice versa at the level of second-order neurons in the brainstem, which receive convergent input from both trigeminal and cervical territories (2–4). Based on findings from experimental studies, electrical stimulation of the greater occipital nerve (GON) in animals has indeed been shown to have a facilitating effect on dural nociceptive stimulation (5–7), suggesting the subsequent induction of central sensitization on second-order neurons receiving cervical and trigeminal input.
We have recently shown that a reduction of sensory cervical input in humans inhibits the nociceptive transmission from the first division of the trigeminal nerve in healthy volunteers (8). Specifically, we found a decrease of the nociceptive blink reflex responses and an increase of the R2 latencies following GON blockade with Prilocain. These findings support the hypothesis of functional connectivity between trigeminal and cervical afferent pathways in humans (9).
Several studies have suggested that pain relief in migraine, cervicogenic headache and CH can be achieved by local injections of steroids, local anaesthetics, or a mixture of both in the area of the GON (10–15). However, the reason for the improvement of clinical symptoms after occipital nerve blockade in primary headache patients is unknown. Several hypotheses have been suggested, including inhibition of central pain-processing mechanisms at the brainstem level, a systemic steriod effect and placebo effects.
CH is an excruciatingly painful primary headache syndrome, characterized by attacks of severe, unilateral pain in the orbital, supraorbital and temporal areas, lasting from 15 to 180 min, recurring up to eight times daily and accompanied by ipsilateral autonomic symptoms (16). One in 10 subjects presents the chronic form (CCH), in which remissions are absent for at least 1 year or last <1 month. A significant proportion of CCH subjects are refractory to any pharmacological therapy and thus remain dramatically disabled (17). Blockade of the GON using suboccipital injections of steroids or local anaesthetics (or a combination of both) on the pain side is therefore justified before resorting to surgical procedures, as side-effects are rare (14, 18, 19).
In general, the success of any procedure or treatment for episodic CH has to be judged with great caution, as the natural course of the disease is a temporary remission. The purpose of this study was to assess the clinical effect of a single cervical cutaneous nerve blockade with local anaesthetics alone in otherwise chronic CH patients resistant to the usual treatment options. A further aim was to test whether the clinical outcome—in the case of a successful result—correlated with changes of trigeminal transmission, which would imply a modulatory role of the cervical innervation in these patients. We therefore examined with the same design as tested in healthy volunteers (8) the nociceptive blink reflex responses before and after GON blockade in a group of 15 CCH patients.
Subjects and methods
Subjects
Fifteen patients (14 men, one woman, age 23–64 years, mean 40 years) with active CCH according to the criteria of the International Headache Society (20) were studied before and after occipital nerve blockade ipsilateral to the headache side. The clinical and demographic characteristics are shown in Table 1. All patients were otherwise healthy, but had been suffering from daily CH attacks for many years, despite several preventative treatments. None had suffered an attack within the last 4 h prior to the first measurement taken outside of an attack.
The clinical and demographic characteristics of 15 chronic cluster headache patients

r, Right side; l, left side; M, male; F, female. Medication and attacks per day. Clinical outcome refers to the number of usual attacks (before the nerve blockade).
Written, informed consent was obtained from all patients and the study was approved by the Ethics Committee of the University of Regensburg.
Design
The response areas (AUC) and the latencies of the R2 components of the nociceptive blink reflexes and thresholds for sensory and pain perception were measured in 15 CCH patients. This was followed by recordings of the nociceptive blink reflexes as described below. Occipital nerve blocks were performed by injecting 5 ml of a local anaesthetic (Prilocain 1%; Astra Zeneca, Wedel, Germany) in the region of the GON ipsilateral to the headache side (11 right, four left). The GON was located in the nuchal line halfway between the mastoid process and the occipital protuberance, as described (8). Successful blockade was confirmed by testing sensory perception with a cotton swab. Finally, measurements of the nociceptive blink reflexes were repeated. The time interval between the two recordings was approximately 20 min. Patients were asked about the clinical outcome following the nerve blockade and about head pain and CH attacks on the evening of the same day, 1 day and 1 week after the procedure.
Electrophysiology
To study trigeminal nociception and transmission, the nociceptive blink reflex was elicited with a custom-built planar concentric electrode with a stimulation area of approximately 20 mm2 (21). It was placed on both sides of the forehead, 10 mm above the entry zone of the supraorbital nerve. Stimulus pulse width was 500 µs. Surface electrodes were placed infraorbitally below both eyes and at the root of the nose (22). Recording bandwidth was 50–2500 Hz. The patient's individual threshold for sensory detection (Ie) and pin prick like pain (Ip) were determined by applying two series of electrical pulses with increasing and decreasing stimulus intensities using stepwise increments of 0.1 mA. Nociceptive blink reflexes were elicited with stimulus intensities of 1.5 times the individual Ip at interstimulus intervals of 15–17 s (pseudorandomized).
The study was blinded in terms of the injection/headache side to the researcher who collected the blink reflex data.
The first sweep from each recording block of six sweeps was discarded to avoid contamination with EMG responses due to startle responses. From averages of five single stimuli, the AUC and the latencies were calculated offline after demeaning and rectification of the raw EMG signal. AUC were calculated between 30 and 90 ms (23). The values given are the means of four blocks of five single stimuli administered on each side.
PC-based, off-line analysis was done with custom-written software using Matlab 5.1 (Mathworks, Natick, MA, USA).
Statistics
R2 latencies and R2 AUC of the nociceptive blink reflex recordings and perception and pin prick-like pain thresholds of the 15 CH patients were proved for normal distribution by Kolmogorov–Smirnov tests and by q–q plots. Repeated measures
Results
Electrophysiology
Results are presented in mean and standard deviations.
Preblockade
In chronic CH patients the initial R2 latencies (in ms) on the injection/headache side were not significantly different from the non-injection/non-headache side (34.5 ± 3.9 vs. 35.3 ± 3.4, P = 0.453). Similarly, the R2 response AUC (in µVms) on the injection/headache side before the blockade did not differ from the non-injection/non-headache side (5786 ± 2604 vs. 5620 ± 1907, P = 0.694) (Fig. 1a,b). At baseline, patients with CCH showed no significant differences regarding the R2 onset latencies or the R2 response areas compared with healthy controls (8).

Error bars represent the ipsilateral and contralateral R2 areas under the curve (a) and latencies (b) of nociceptive blink reflexes as means with 95% confidence interval before and after the occipital nerve blockade in 15 chronic cluster headache patients. ∗Significant at P < 0.01.
Post blockade
In comparing measurements from before and after the nerve blockade, significantly decreased AUC and increased latencies of the R2 components were found in the patients exclusively on the injection side. Inclusion of thresholds for sensory and pin prick-like pain perception did not explain more of the variance. Correlations between thresholds and R2 latencies/R2 response areas before and after the nerve blockade were not significant in patients or in controls. The thresholds were therefore not considered in the final model (Table 2).
Analysis of variance for repeated measures (General linear model)
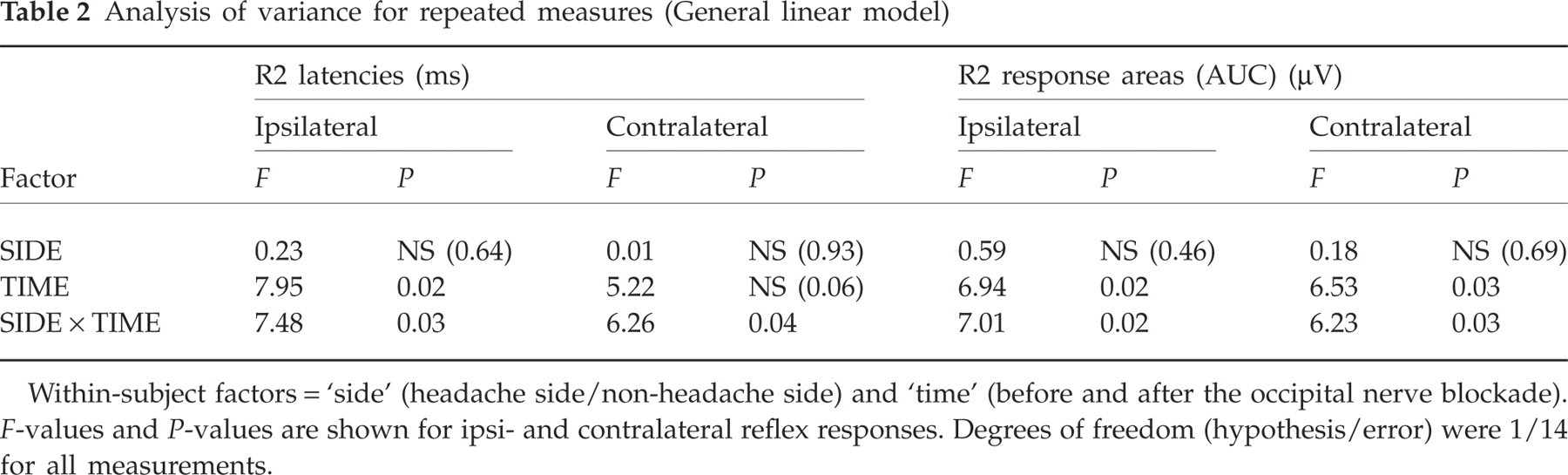
Within-subject factors = ‘side’ (headache side/non-headache side) and ‘time’ (before and after the occipital nerve blockade). F-values and P-values are shown for ipsi- and contralateral reflex responses. Degrees of freedom (hypothesis/error) were 1/14 for all measurements.
Performing post hoc analyses, a significant increase of R2 latencies (before 34.5 ± 3.9, after 38.9 ± 3.3; P = 0.001) and a significant decrease of R2 response areas (before 5786 ± 2604, after 4046 ± 2067; P = 0. < 001) were found on the injection/headache side. On the non-injection/non-headache side, neither the latencies (before 35.3 ± 3.1, after 36.4 ± 3.9; P = 0.297) nor the AUC (before 5620 ± 1907, after 4842 ± 2979; P = 0.153) were changed significantly, although there was a tendency towards longer R2 onset latencies and decreased AUC after the nerve block on the headache side (Fig. 1a,b).
Thresholds for sensory and pin prick-like pain perception are shown in Table 3. Thresholds of pin prick-like pain perception were significantly increased on the headache compared with the non-headache side (1.43 ± 0.33 vs. 1.23 ± 0.29, t = 4.61, P < 0.001) and with healthy controls (8). This difference continued after the blockade (1.39 ± 0.27 vs. 1.20 ± 0.27, t = 3.59, P < 0.01). However, there was no significant change of these thresholds after the occipital nerve blockade on both sides (Table 3).
t-test comparison of thresholds before and after the greater occipital nerve blockade

Values are means with standard deviations for patients. Comparisons before and after the injection are done with Student's t-tests for injection side and for non-injection side. The injection side in patients was the side of their headache.
Thresholds for sensory perception showed no initial differences between headache and non-headache side in our patients and no changes after the nerve blockade on both sides (Table 3).
Clinical outcome
After the nerve blockade, all patients experienced complete sensory loss in the posterior scalp lasting between 2 and 11 h (mean 4 h).
Detailed information about the clinical outcome is given in Table 1. Because of our primarily neurophysiological approach, data regarding the clinical outcome are only descriptive. Generally, nine out of 15 patients reported some improvement of the headache attacks, but only to a minor degree. Seven patients reported no attacks for the rest of the day, one patient for the following 3 days. One day after the blockade, only one patient continued to keep pain free, whereas the remaining 14 patients experienced the same number of attacks as before the blockade. One week after the nerve blockade, no therapeutical benefit was reported. The pain intensity of single attacks was also reduced in four patients, but these changes were not statistically analysed, e.g. using rating scales.
One patient was completely pain free for 3 days after the occipital nerve block. Two patients experienced a significant reduction in the intensity and frequency of their attacks for 2 days. Four patients reported a feeling of ‘light-headedness’ and did not have any attacks for the rest of the day. One patient reported complete cessation of his interval headaches, which is a feature in some patients. One patient, although still suffering from pain attacks, observed no autonomic symptoms for 3 days during his acute cluster attack. The remaining six patients did not report any clinical changes following the nerve block.
No major adverse events were observed after the nerve block, but one patient reported hypoaesthesia of all three trigeminal divisions of the ipsilateral face lasting for about 4 days. None of the other patients complained of any facial sensory loss.
Discussion
Clinical effect
After occipital nerve blockade, nine of 15 patients with CCH presented some general improvement, but only to a small degree, including one patient with reduced autonomic symptoms and one with an arrested interval headache for about 3 days after the nerve block. Taking a possible placebo effect into account (24), the specific effect size of the treatment in our study was even smaller.
In other studies, the clinical effect of GON blockades has been reported higher (12) and in the only placebo-controlled trial so far, a single suboccipital steroid injection completely suppressed attacks in >80% of CH patients (14). However, most of these patients were episodic cluster patients. Furthermore, in all these studies, cortisone or a mixture of cortisone and local anaesthetics were used. Therefore, the data cannot answer the question, whether the steroid is acting systemically and whether the suboccipital injection site does not provide any advantage (19). Our results are in accordance with those of Anthony, who found that suboccipital injections of local anaesthetic alone have neither a beneficial nor harmful effect on CH attacks (18).
The absence of a uniform clinical reaction in our patients may in part be explained by anatomical and physiological variations of the main course of the GON between the midline and the nerve along the intermastoid line (25). However, all of our patients experienced anaesthesia in the GON area. Moreover, the mean reduction of R2 response areas and increase in R2 onset latencies were significant, suggesting a sufficient nerve block.
Electrophysiology
Following the injection, a significant decrease of the ipsi- and contralateral nociceptive blink reflex R2 areas and an increase of the ipsi- and contralateral R2 latencies exclusively on the side of the nerve block were found. This altered trigeminal transmission provides evidence for the existence of functional connectivity between the occipital and trigeminal nerve structures, not only in healthy volunteers (8), but also in CH patients, a finding which has been suggested in experimental studies (2, 26). The similar results in patients and controls suggest a general ‘cross-talk’ between the two afferent nerve territories, which is independent of an underlying diagnosis of a primary CH syndrome.
At baseline, the patients with CCH showed no significant differences regarding R2 onset latencies or R2 response areas compared with healthy controls (8). This suggests that there is no facilitation of trigeminal nociceptive transmission in the nucleus caudalis in patients with CCH outside the acute attack. Additionally, there was no correlation between clinical and electrophysiological outcome in the cluster patients. Among those who reported clinical improvement after the occipital nerve blockade, no particular changes in blink reflex responses could be found. Our data suggest therefore that the physiological ‘trigemino-cervical cross-talk’ as such does not play a pivotal role in the clinical manifestation (frequency or duration of attacks) of CH. In view of the positive effects of occipital stimulation in CH patients (27), it is remarkable that one patient described a cessation of his constant background headache, which is known to occur in some patients with CCH (8). In view of these results, it seems unlikely that the therapeutic effect of a mixture of steroids or local anaesthetics (18) is solely due to direct inhibition of occipital input.
The pain thresholds at baseline were higher in our cluster patients ipsilateral to the headache compared with the non-headache side and compared with both sides of healthy controls (8). Our data are in agreement with the literature concerning significant increases in thermal threshold on the pain side of cluster patients (28). New data from QST studies in cluster patients have shown either impaired (29) or increased thermal thresholds for warmth and thermal sensory limen in cluster patients regardless whether they were inside or outside the bout (30). An increase of pin prick-like threshold does not necessarily mean an inhibition of the trigemino-cervical circuit itself. As discussed in the literature (31–33), the inhibition of thresholds may be an impairment of descending pain control systems. The nociceptive blink reflex is known to be oligosynaptic and poorly influenced by (supratentorial) descending pathways. This could be the explanation for our observation of decreased pin prick-like perception on the headache side but the same reflex response regarding AUC integral and latencies compared with the non-headache side in our cluster patients. Other studies have reported a decrease in pain thresholds (31, 34). Interestingly, one patient reported the absence of autonomic symptoms for several days after the nerve blockade, though his headache was not clinically meaningfully reduced. This may be explained by the anatomical pathways of parasympathetic fibres passing from the superior salivatory nucleus via the greater petrosal nerve to the sphenopalatine ganglion, which mediates the parasympathetic features associated with trigeminally mediated pain (35). Previous studies on symptomatic headaches involving cervical innervation have also reported cranial autonomic symptoms resembling CH (36, 37). In one case report, an aneurysm of the basilar artery was reported to have caused a cluster-like headache in a female patient. Both the pain and the autonomic features were significantly reduced after the angiographic coiling of the aneurysm, providing evidence for some ‘cross-talk’ between the cervical and the trigeminal sensory/nociceptive/parasympathetic systems in the trigeminocervical complex (38).
It is noteworthy that one patient reported a facial hypoaesthesia of all three trigeminal branches, lasting for 3 days after the GON blockade on the ipsilateral side. This observation is similar to the case report (39), providing further clinical evidence for a cervico–trigeminal interaction (37).
In summary, our neurophysiological and clinical data provide further evidence for the existence of functional connectivity between the sensory occipital segments and the ophthalmic branch of the trigeminal nociceptive system in humans. This cross-talk is independent of whether the subjects suffer from CH or are healthy controls. The trigeminocervical complex does not seem to be facilitated in CH, suggesting a more centrally located pathology of the disease. The impressive changes of trigeminal function due to cervical inhibition are not mirrored by a significant clinical effect of an occipital injection of local anaesthetics alone, suggesting that the clinical improvement in some cases is not due to a direct inhibitory effect on the trigeminal transmission. In refractory CH patients, who failed to respond to occipital nerve blockade, it may be necessary to block the more proximal C2 nerve roots, to use longer acting anaesthetics (40) or to use local cortisone injections (11, 18, 41).
Acknowledgements
The authors thank Tina Schneider for technical support. The study was supported by a University grant (ReForM-A) to V.B.
