Abstract
Headache syndromes often suggest occipital and neck involvement, although it is still unknown to what extent branches of segment C1-C3 contribute actively to primary headache. Pain within the occipital area may be referred to the trigeminal territory. However, a modulation of trigeminal transmission by affecting cervical input in humans has not been elucidated so far. A convergence of cervical and trigeminal input at the level of the caudal part of the trigeminal nucleus in the brainstem has been suggested due to anatomical and neurophysiological studies in animals. We examined the R2 components of the nociceptive blink reflex responses in 15 healthy subjects before and after unilateral nerve blockade of the greater occipital nerve with 5 ml prilocain (1%). R2 response areas (AUC) decreased and the R2 latencies increased significantly after the nerve blockade only on the side of injection. AUC and latencies on the non-injection side remained stable. Thresholds for sensory or pain perception did not differ significantly between the repeated measurements on both sides. Our findings extend previous results related to anatomical and functional convergence of trigeminal and cervical afferent pathways in animals and suggest that the modulation of this pathway is of potential benefit in primary headache disorders.
Introduction
In clinical practice, many patients suffering from primary headache syndromes, such as migraine, tension-type headache or cluster headache, report neck symptoms, such as tenderness, stiffness or pain, which may accompany the headache or even precede it (1, 2). Some of these patients can provoke a frontal or generalized headache by external pressure to sensitive points in the neck or by turning their head. In particular, migraine patients complain of the neck as the starting point of their headache and as many as 50% of migraine patients complain of cervical tenderness (3). In these cases, the headache exceeds trigeminal and involves cervical innervation. However, a cervical contribution to the pain is usually not considered to be causally relevant and, with the exception of cervicogenic headache, has not been included in the diagnostic criteria of primary headache syndromes (Classification of Headaches, International Headache Society, 2004).
In contrast to this widely accepted view, a recently published Norwegian study reported that 75% of tractor drivers suffered from moderate to severe generalized headache only when working on their tractor, having their neck consecutively turned or twisted for many hours daily (4). The most likely mechanism for this observation is a ‘referred pain’ originating from structures in the posterior part of the head or from the neck to facial areas and vice versa at the level of second order neurons in the brainstem, which receive convergent input from both trigeminal and cervical territories (5–7). Afferent trigeminal nerves terminate to some extent in C2 spinal segments (8), where most of the afferents of the greater occipital nerve terminate as well (9). Activation of dural nociceptors increased the excitability to electrical stimulation of the GON in C-fibre responses in experimental rats and vice versa (6, 10–12). These results suggest the subsequent induction of a central sensitization of second order neurons receiving cervical and trigeminal input.
The blink reflex is a trigeminofacial brainstem reflex which can be used to delineate the topographical level of brainstem lesions and identify functional changes in central trigeminal nociception. After supraorbital stimulation, three components can be distinguished: an ipsilateral oligosynaptic pontine R1 component and two polysynaptic bilateral medullary components, R2 and R3 (13). Only R2 can also be elicited by selective activation of nociceptors (14). The use of a concentric surface electrode with low-stimulus intensities but electrode-dependent high current density allows preferential depolarization of superficial nociceptive Aδ-fibres (15). The nociceptive blink reflex has been established as a valid instrument for the examination of central trigeminal transmission (15–18).
Several studies have shown that electrical stimulation of the trigeminal nerve induces early and late reflex responses in the neck muscles, which led to the synonym ‘trigeminocervical reflex’(19–23). This suggests the existance of a functional connection between these two nerve territories in humans. However, these studies focus only the influence of trigeminal stimulation on cervical or upper limb structures. The purpose of this study was to determine whether a specific reduction of sensory cervical input in humans would influence the nociceptive transmission from the first division of the trigeminal nerve (in healthy volunteers without the diagnosis of a primary or secondary headache syndrome), which has not previously been studied systematically using non-invasive methods. Therefore, we examined the R2 components of nociceptive blink reflex responses in 15 healthy volunteers before and after a local anaesthesia of the GON of one side.
Subjects and methods
Subjects
Fifteen healthy volunteers (12 men, 3 women, age 19–31 years, mean age 24) were studied before and after occipital nerve blockade on one side. None of the volunteers had any history of primary or secondary headache syndromes.
Informed consent was obtained from all patients and the study was approved by the Ethics Committee of the University of Regensburg.
Design
Main outcome variables were the response areas (AUC) and the latencies of the R2 components of the nociceptive blink reflex. Initially, thresholds for sensory and pain perception were measured. This was followed by recordings of the nociceptive blink reflexes as described below. Occipital nerve blocks were performed in the patients by injection of 5 ml of a local anaesthetic (Prilocain 1%; Astra Zeneca, Wedel, Germany) in the region of the GON on one side (n = 8 on the right side, n = 7 on the left side, randomized). The GON was located in the nuchal line halfway between the mastoid process and the occipital protuberance. Successful blockade was confirmed by testing sensory perception with a cotton swab. Finally, measurements of the nociceptive blink reflex were repeated. The time interval between the two recordings was 15 min.
Electrophysiology
To study trigeminal brainstem nociception and transmission, the nociceptive blink reflex was elicited with a custom-built planar concentric electrode with a stimulation area of approximately 20 mm2 (15). It was placed on both sides of the forehead, 10 mm above the supraorbital groove. Stimulus pulse width was 500 µs. Surface electrodes were placed infraorbitally below both eyes and at the root of the nose (24). Bandwidth was 50–2500 Hz. The concentric electrode is selective for Aδ-fibres to approximately 95% (15). The remaining 5% of recruited fibres are Aβ-fibres. At very low intensities few of these fibres are depolarized and produce a minimal touch-like sensation. These we refer to as the threshold for sensory detection. The individual thresholds for sensory detection (Ie) and (pin prick-like) pain (Ip) were determined by applying two series of electrical pulses with increasing and decreasing stimulus intensity using stepwise increments of 0.1 mA. Nociceptive blink reflexes were elicited with stimulation intensities of 1.5 times the individual Ip at interstimulus intervals of 15–17 s (pseudo-randomized).
The first sweep from each recording block of six sweeps was discarded to avoid contamination with EMG responses due to startle responses. From averages of five single stimuli, the AUCs and the latencies were calculated offline after demeaning and rectification of the raw EMG signal. AUCs were calculated between 30 and 90 ms (25). The values given are the mean of four blocks of five single stimuli each on both sides.
PC-based off-line analysis was done with custom-written software (Matlab 5.1; Mathworks, Natick, MA, USA).
Statistics
Visual inspection of the raw distributions and q-q plots of all variables suggested no deviation from normal distribution. We used
Results
After the nerve blockade, all participants experienced a complete sensory loss at the posterior scalp lasting on average 4 h (range 1.5–7 h).
Preblockade
The initial R2 latencies (in ms) on the injection side were not significantly different from the non-injection side for ipsilateral (35.7 ± 2.9 vs. 35.4 ± 6.3, P = 0.87) and contralateral reflex responses (35.0 ± 4.5 vs. 37.2 ± 6.5, P = 0.28). Similarly, the response areas (in µVms) on the injection side before the blockade did not differ from the non-injection side for ipsilateral (6073 ± 2963 vs. 5626 ± 2346, P = 0.51) and contralateral reflex responses (5108 ± 3279 vs. 4838 ± 2473, P = 0.74).
Post blockade
Using
Effects of analysis of variance for R2 latencies and R2-response areas (AUC) of nociceptive blink reflexes
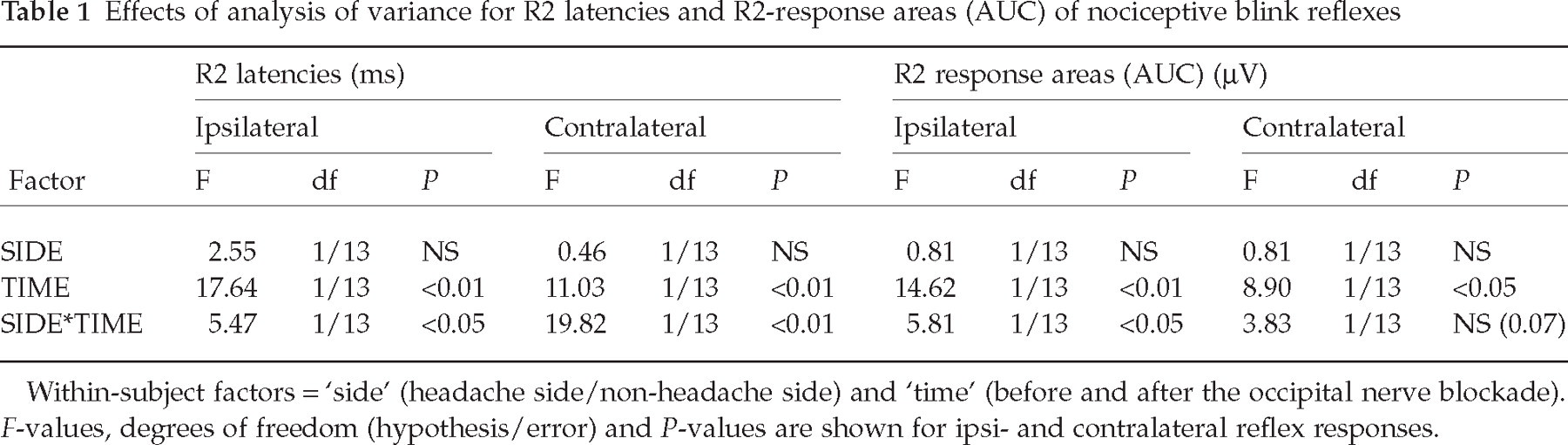
Within-subject factors =‘side’ (headache side/non-headache side) and ‘time’ (before and after the occipital nerve blockade). F-values, degrees of freedom (hypothesis/error) and P-values are shown for ipsi- and contralateral reflex responses.
Thresholds for sensory and pin prick-like pain perception
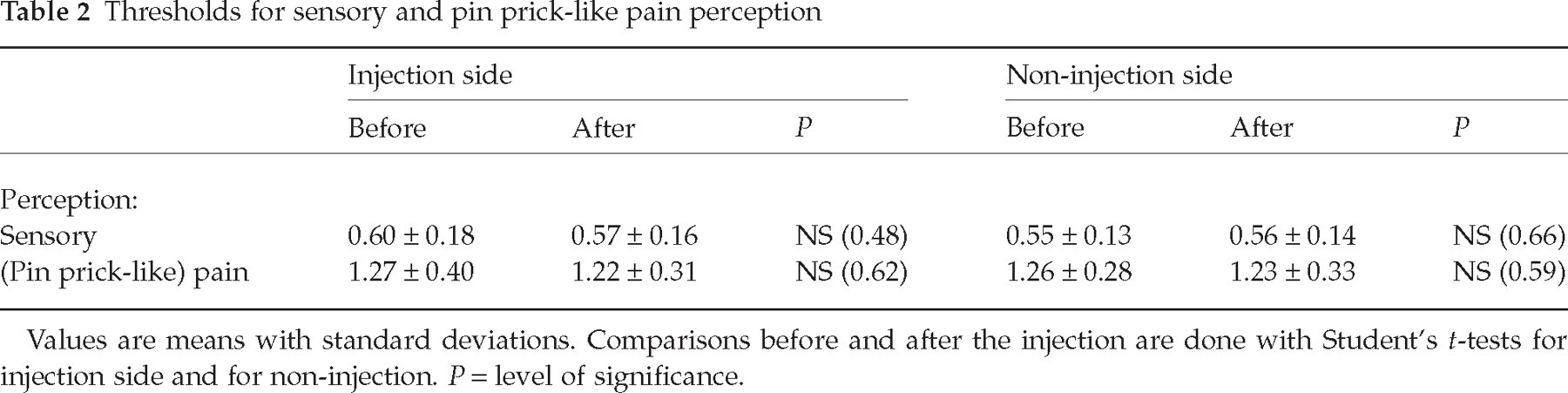
Values are means with standard deviations. Comparisons before and after the injection are done with Student's t-tests for injection side and for non-injection. P = level of significance.
Post hoc analyses, after eliciting the blink reflex on the injection side, revealed a significant increase of R2 latencies for ipsilateral (before 35.7 ± 2.9, after 40.6 ± 2.7; P ≤ 0.001) and contralateral (before 35.0 ± 4.5, after 41.0 ± 2.8, P ≤ 0.001) reflex responses. Similarly, on the non-injection side we found a significant decrease of R2-response areas for ipsilateral (before 6073 ± 2963, after 3738 ± 1884; P ≤ 0.01) and contralateral (before 5108 ± 3279, after 3496 ± 2403; P ≤ 0.01) reflex responses.
After eliciting the nociceptive blink reflex responses on the non-injection side, we found that R2 latencies and R2 response areas were stable over time and did not show any significant differences regarding either ipsilateral or contralateral reflex responses (Figs 1 and 2).

Error bars represent the ipsilateral and contralateral R2 latencies of nociceptive blink reflexes as means with 95% confidence interval before and after the occipital nerve blockade. ∗Significant at P < 0.001.

Error bars represent the ipsilateral and contralateral R2 response areas (AUC) of nociceptive blink reflexes as means with 95% confidence interval before and after the occipital nerve blockade. ∗Significant at P < 0.01.
Baseline thresholds for sensory perception (and pin prick-like) pain perception were not different between the two sides of the head in our subjects. After occipital nerve blockade on one side, we found no significant changes of any of the thresholds (Table 2).
Discussion
After occipital nerve blockade in 15 healthy volunteers without a primary or secondary headache syndrome we found a significant decrease of the ipsi- and contralateral nociceptive blink reflex responses areas and an increase of the ipsi- and contralateral R2 latencies only on the injection side. The altered trigeminal transmission after inhibiting occipital nerve structures gives support to the hypothesis of a functional connectivity between these two nerves, which has been suggested in experimental animals (6, 10–12, 26).
On the side without the blockade, latencies increased and the response areas decreased to some degree, but these changes were only small and did not reach statistical significance. These changes of both sides—in favour of the injection side—suggest bilateral inhibition following the unilateral nerve blockade. We think the inhibitory effect could be due to a modulation of the heterosynaptic convergent entrance on second order neurons or interneurons in deeper layers of the dorsal horn, from the caudal trigeminal nucleus to the upper cervical segments (9, 11, 27–30). These second order neurons are believed to receive ipsilateral as well as contralateral input from the great occipital nerve (6). Therefore, it can be expected that any modulation via ipsilateral second order neurons will influence the R2 responses on both sides to some extent. It is difficult to conceive how modulation of the sensory arc of this trigeminal reflex could alter efferent outflow but not sensor perception. This may be due to lack of power to detect subtle differences in sensory thresholds or a modulatory effect, that rather influences gain than thresholds. Changes of sensory intensity were not monitored in our study.
Our design was not placebo-controlled, because there is no placebo equivalent for the hypaesthesia that occurred in our subjects after the blockade in the distribution of the GON. Therefore, one would require a perceptible stimulus in any of a sham device, which may on his part provide neurostimulation (31).
Fatigue, loss of attention or other mechanisms contributing to response habituation may have contributed to the recording of lower response areas and longer latencies after the blockade. However, in a study with repeated measurements of nociceptive blink reflex recordings, habituation was found to be minimal with interblock intervals of 7 min (16). Therefore, we consider that the significant interaction of the factors SIDE and TIME suggests a side-specific effect of the GON injection.
The blockade effect may be due to a general reduction of afferent excitatory input in the trigeminocervical complex, as suggested in the literature (26). The dysfacilitation suggests a functional connectivity between the cervical and the trigeminal system in the trigeminocervical complex of the brainstem.
As we did not record conventional blink reflex responses, our data cannot indicate whether the inhibitory modulation occurs only for nociceptive or for any somatosensory input. However, we do not expect the normal blink reflex responses to be abnormal, because previous studies clearly demonstrated that the conventional blink reflex was not influenced by subtle functional changes in nociceptive processing in the trigeminocervical complex, whether it was examined in healthy volunteers, in migraine patients or in patients with sinusitis (17, 18).
The response areas and the R2 latencies of the blink reflex responses were our main outcome variables. However, to determine the stimulus intensity, we further examined the individual sensitivity and pain thresholds that did not show any significant changes after the nerve blockade. We do not think that the small changes in thresholds have influenced the recordings of the R2 latencies or response areas of the blink reflex, because the stimulus intensity was kept fixed at 1.5 times the individual pain perception threshold as described above. In addition, including them into the model could not explain more of the variance. It should be noted that our study population was rather small and the threshold findings need to be studied in more detail and with a greater number of subjects.
In summary, our neurophysiological data provide evidence for a functional connectivity between the sensory occipital (C2-) segments and the ophthalmic branch of the trigeminal nociceptive system in humans. The occipital nerve probably has an excitatory influence on trigeminal circuits, that can be reduced by selective inhibition of that nerve due to a anaesthetic blockade. Our method of eliciting predominantly nociceptive blink reflex responses proves to be sufficient in detecting changes of trigeminal transmission due to the occipital blockade. This may be of relevance for our understanding of clinical observations in many patients suffering from primary headache syndromes, such as migraine and tension-type headache, who complain about cervical discomfort or pain in the course of their headache. This ‘cross-talk’ between the cervical and the trigeminal sensory/nociceptive/parasympathetic system in the trigeminocervical complex needs to be studied thoroughly, if we are ever to understand the complex mechanisms at work in head pain.
Footnotes
Acknowledgements
The authors thank Tina Schneider for technical support. The study was supported by a University grant (ReForM-A) to V.B.
