Abstract
Background: Headache is a common and persistent symptom following traumatic brain injury (TBI). Headaches following TBI are defined primarily by their temporal association to injury, but have no defining clinical features. To provide a framework for treatment, primary headache symptoms were used to characterize headache.
Methods: Three hundred and seventy-eight participants were prospectively enrolled during acute in-patient rehabilitation for TBI. Headaches were classified into migraine/probable migraine, tension-type, or cervicogenic headache at baseline and 3, 6, and 12 months following TBI.
Results: Migraine was the most frequent headache type occurring in up to 38% of participants who reported headaches. Probable migraine occurred in up to 25%, tension-type headache in up to 21%, then cervicogenic headache in up to 10%. Females were more likely to have endorsed pre-injury migraine than males, and had migraine or probable migraine at all time points after injury. Those classified with migraine were more likely to have frequent headaches.
Conclusions: Our data show that most headache after TBI may be classified using primary headache criteria. Migraine/probable migraine described the majority of headache after TBI across one year post-injury. Using symptom-based criteria for headache following TBI can serve as a framework from which to provide evidence-based treatment for these frequent, severe, and persistent headaches.
Keywords
Introduction
The Centers for Disease Control and Prevention (CDC) estimates that between 1.4 and 1.8 m civilians in the USA sustain a traumatic brain injury (TBI) each year (1). Approximately 20% of Operation Enduring Freedom (OEF) and Operation Iraqi Freedom (OIF) veterans are thought to have experienced a TBI (2). In civilian and military populations, headache is not only one of the most commonly reported symptoms following TBI (3,4), but is also often persistent (5).
The reported prevalence of post-traumatic headache (PTH) in the literature has been broad (i.e. 30–90%), most likely due to patient sampling biases such as using patients treated in specialty clinics who have complicated recoveries versus prospectively studied representative TBI samples. In a 2006 review of the available literature, Lew et al. (3) found PTH to be often persistent, reporting that 18–22% of PTH lasted longer than one year. In a recent prospective longitudinal investigation of over 450 individuals with TBI, we found the prevalence of PTHs to be 47% following TBI, and prevalence remained consistently high at over 40% up to one year post-injury (5).
The study of headache was significantly advanced by the development of a classification system by the classification subcommittee of the International Headache Society in 1988, subsequently revised and titled the International Classification of Headache Disorders, 2nd edition (ICHD-2) criteria (6). The ICHD-2 divides headache disorders into primary and secondary headaches. Primary headache disorders, such as migraine, probable migraine (formerly migrainous headache), cluster headache, and tension-type headache among others, are thought to have no known underlying treatable cause and are primarily defined as syndromes using clinical symptoms such as severity, duration, pain characterization, and associated autonomic features for inclusion into a particular headache category. As secondary headaches, PTHs are defined based on a close temporal relationship with an antecedent TBI with severity, latency, and duration criteria, but with no defining clinical features as is found in the primary headache syndromes.
Classification into primary headache phenotypes has been used to characterize PTH (7) primarily in an effort to provide a framework for treatment decisions. The similarities in neurochemical changes reported after TBI with those found in migraine suggest shared pathophysiological pathways, possibly involving the trigeminovascular system that may allow the same effective treatments despite different causation (8,9). The primary purpose of this prospective, longitudinal study was to characterize and classify headache syndromes after TBI at multiple time points up to one year following injury.
Methods
Study participants
A total of 378 participants with a diagnosis of TBI, who had the cognitive and communication ability to give valid responses to questions, were enrolled during acute in-patient rehabilitation. Recruitment to the study occurred within the TBI Model Systems (TBIMS) from seven centers between February 2008 and June 2009. All follow-up assessments were completed by July 2010. Criteria for inclusion and exclusion were the same as for the TBIMS. Inclusion criteria required participants to:
present to the designated TBIMS acute care hospital within 72 h of injury; receive both acute medical and acute rehabilitation care within the same system; sustain a TBI with at least one of the following characteristics:
Glasgow Coma Scale (GCS) (10) score < 13 on emergency room admission (not due to intubation, sedation, or intoxication); loss of consciousness > 30 min (not due to sedation or intoxication); post-traumatic amnesia (PTA) > 24 h; or trauma-related intracranial abnormality on neuroimaging. be aged 16 years or older.
Exclusion criteria were participant incarceration or no telephone access for follow-up. Informed consent was obtained as approved by the institutional review boards of all seven institutions. Initial data were collected on headache during or within 1 week after discharge from in-patient rehabilitation. Follow-up interviews were conducted by telephone by trained research assistants using structured interviews at 3, 6, and 12 months after injury.
Measures
Demographic data included age at injury, sex, race (white, black, Asian/Pacific Islander, native American, Hispanic, and other), education (completed high school/did not complete high school), and cause of injury (moving vehicle-related, violence, sports, fall, hit by object, and other). Prospectively assessed PTA duration was used as a marker of brain injury severity [categorized as mild (PTA < 24 h), moderate (PTA 1–7 days), severe (PTA > 8 days] and unknown. A headache survey included detailed questions about current headache at initial evaluation during in-patient rehabilitation, history of headache prior to injury, and headache at follow-up time points. Questions were asked specifically about headache intensity, location, duration, disability and associated symptoms such as nausea, vomiting, and neurological accompaniments including visual disturbance, disability, and neck pain to classify each headache by its symptoms.
Headache characterization
Headaches were classified into the following categories for primary headache disorders: migraine, probable migraine, tension-type headache and cervicogenic headache (a symptom-defined secondary headache). If the headaches did not fit into one of these categories at a given time point, they were deemed unclassifiable. Specific decisions for classification were based on answers to the headache questionnaire, ICHD-2 criteria, and expert review (KB, SL) using an ordered decision tree. A headache was considered to be a migraine if the pain rating was moderate to severe and at least two of the following criteria were met:
the headache had a significant disabling impact; the headache was unilateral; the headache was throbbing/pulsating; the headache was worsened by movement or physical activity.
Finally, each participant had to endorse nausea and/or vomiting or sensitivity to light and sound. A headache was classified as probable migraine if the participant endorsed moderate to severe pain, but met only one of the other criteria for migraine headache classification. A headache was classified as tension-type if it was bilateral, vice-like, squeezing or tight, and the pain was rated as mild to moderate. A headache was classified as cervicogenic if it was unilateral, neck pain was endorsed, and the headache did not meet the criteria for migraine, probable migraine, or tension-type headache. All headaches that did not fit into the four classifications were considered unclassifiable. Two random samples of headaches (N = 10 and 18 respectively) defined as unclassifiable were reviewed by experts (KB, SL) when initial classification was started and prior to final data analysis. All reviewed cases were missing at least one or more symptoms to meet diagnostic classification in one of the four headache types.
The classification was ordered in that it gave precedence to migraine headache with or without aura at a given time point if the criteria were met. Only if the criteria were not met for migraine would probable migraine, followed by tension-type, and finally cervicogenic, be the primary given headache for a participant.
Statistical analysis
Descriptive statistics are the statistics we used in the manuscript (summarizing what we found in our study sample) rather than testing a hypothesis (inferential statistics). In our paper, the descriptive statistics we use are means (SD), percentages, and counts which are all descriptive.
Results
Demographics
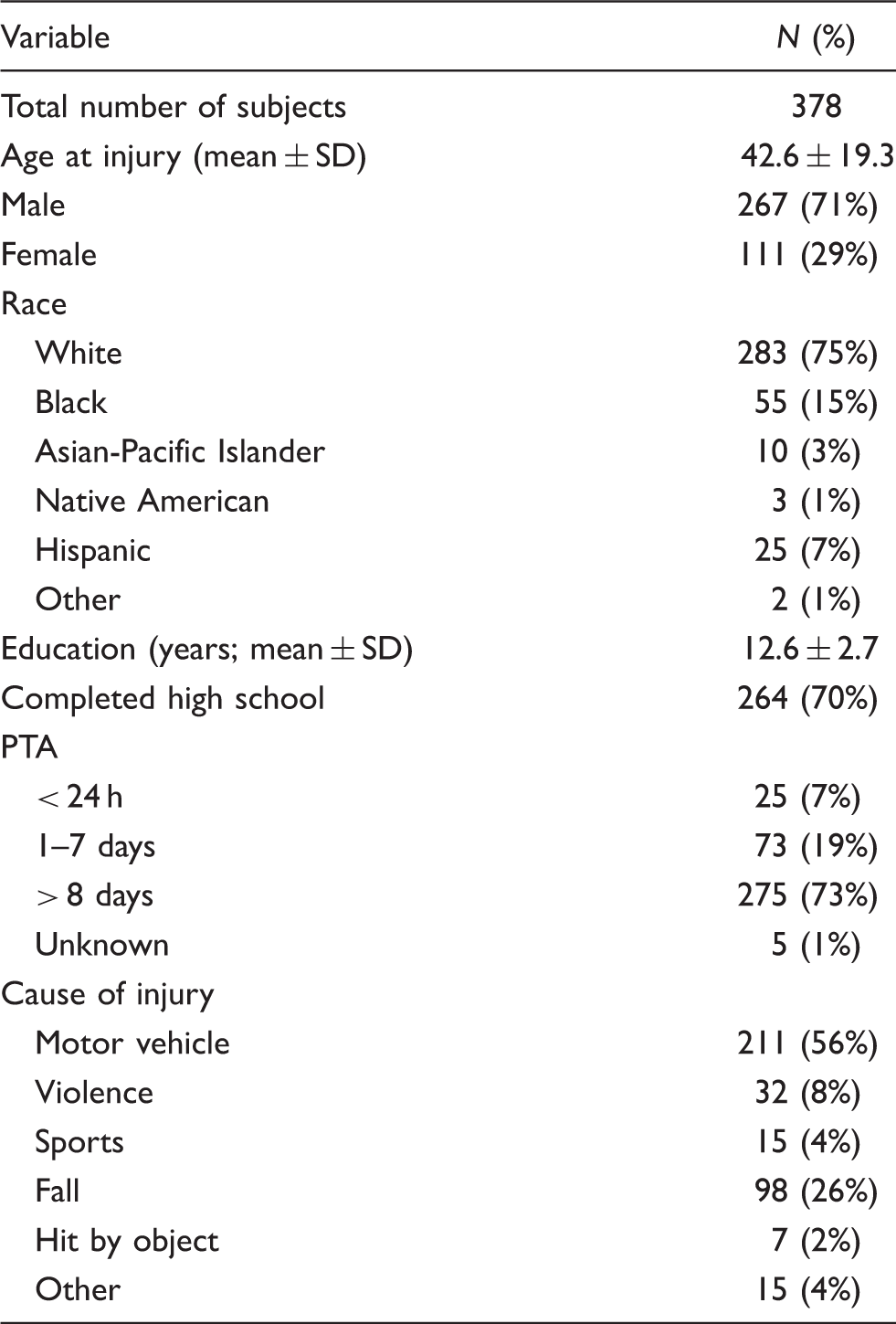
Headache characteristics
Headache classification over the first year following traumatic brain injury

Headache classification over the first year after traumatic brain injury in individuals without a history of headache
Classification of migraine/probable migraine by sex across all time points

Headache frequency was higher for those who endorsed migraine or probable migraine over other headache types after injury (Figure 1). Participants whose headaches were classified as migraine were more likely to have frequent headaches (several days per week or daily) than did those whose headaches met the criteria for other headache phenotypes.
Headache frequency distribution by classification.
Discussion
The most frequent primary headache phenotype found in this study was migraine headache, occurring at baseline in 31% of individuals with headache after TBI. Probable migraine was next in frequency with a baseline occurrence of 21% of headache types. These headache types were consistent over one year post-injury indicating that moderate to severe headaches of migraine phenotype are common and persistent following TBI. Unclassifiable headaches tended to become more ‘classifiable’ largely because the number of tension-type headaches increased from the unclassifiable pool. Despite this, tension-type headaches were described in only about one-fifth of persons with headache after TBI. Cervicogenic headaches were also infrequent, a surprising finding given the high incidence of vehicle accidents as the cause of injury.
Our findings are similar to the work of others using primary headache criteria to characterize headache after TBI. Theeler et al. (11) in a questionnaire-based study of US soldiers with a history of concussion during deployment in Iraq or Afghanistan found that 58% of PTH had a migraine phenotype according to the ICHD-2 criteria remarkably similar to headache characterization in our civilian population. A later retrospective, chart-based review by Theeler and Erickson (12) in a mild TBI population sustaining head or neck trauma while deployed to Iraq, found that 78% of all headaches presenting to a specialty neurology clinic met the criteria for migraine with and without aura or probable migraine, and only 18% met criteria for tension-type headache.
Other studies attempting to classify headache following mild head injury have not found the higher prevalence of migraine that this and other studies found (11,12). Haas (13) found that 75% of a clinic sample of patients, over 80% with mild TBI, met the criteria for chronic tension-type headache and only 21% met the criteria for migraine without aura using the ICHD-1 criteria. This study was based on a select clinic sample, did not include patients with exacerbations of pre-existing headaches, and the median time from injury to consultation was 15 months. In a medical record-based retrospective study of 53 patients in a headache specialty clinic using the ICHD-2 criteria for chronic PTH classification, up to 62% endorsed chronic or episodic tension-type headache, but 29% had migraine or probable migraine either as a single headache type or coexisting with other headache types (14). Comparison between studies is difficult because analgesic overuse was documented in 42% of the patient sample and this could have affected headache severity and frequency (14).
In another cohort of 66 patients with acute PTH after mild head injury, migraine or probable migraine was reported to occur in approximately 35% using the ICHD-2 criteria in a prospective observational study. Those with pre-existing chronic headache or a whiplash type neck injury were excluded from analysis (15). The latter two studies evaluated only headaches which stayed within the latency requirements for PTH (occurring within 7 days of injury). Stovner and colleagues (16) followed a prospective cohort of 217 patients enrolled for approximately 3 months after mild head injury and an age- and sex-matched orthopedic control cohort. They found no significant difference in headache prevalence between these two groups at one year post-injury and noted that tension-type headaches were the most frequent classification of headache. Comparison to the current study is difficult as the majority of controls (72%) and 44% of those with mild head injury reported a pre-injury history of headache compared to 18% in our sample of moderate to severely injured patients. Valid comparisons between studies showing higher migraine or probable migraine phenotypes after brain injury (our work and 11,12) versus those studies with higher tension-type headache (13–15) cannot be made due to significant differences in sample selection and methodological differences in the way occurrence of headache and headache symptoms were used to determine headache type. The difficulties in classification using a symptom-based scheme beg for further studies with standardized protocols and the use of representative cases of patients with TBI.
As described earlier, prior history of headache was a risk factor for the development of headache after TBI in our cohort (5). Most participants who had a history of migraine or probable migraine before injury and endorsed headache after injury continued to have the same headache types post-injury (51–67%). Prevalence of migraine or probable migraine was also high at all time points, even for those who did not have a history of headache pre-injury (52–58%; Table 3). This highlights the importance of assessing pre-injury headache history as a risk factor for headaches following TBI. Females were found to have higher rates of headache than males at every time point post-injury (5), similar to the higher rates in females seen in the general headache literature. Furthermore, we found that migraine and probable migraine were more common in females than males following brain injury at all time points, whether or not there was a history of pre-injury headache.
In this study, any headache reported following TBI was evaluated whether or not it met the latency (i.e. time from injury) criteria for PTH. Prevalence rates using the ICHD-2 criteria requiring PTH to occur within 7 days after injury or regaining consciousness, likely underestimates the scope of the problem. As noted by Evans (7), this requirement would eliminate those who have severe brain injuries and are unable to complain of headache within the first week after injury. Previously reported data on those with civilian TBIs shows that 28% of new cases of PTH are not initially reported until after 3 months post-injury (5). In a study of returning war veterans, only 27% of new headaches were reported to develop within a week of the injury (12). Given this data, adherence to the ICHD-2 criteria for PTH may result in underdiagnosis and possibly undertreatment of headache after TBI. This is important because large numbers of those with headache not only met the criteria for migraine or probable migraine, by definition a moderate to severe headache, but also reported a headache frequency occurring several times a week or daily. This study supports the findings of Couch et al.(17), who found through a population-based telephone interview that any history of head or neck trauma is significantly associated with chronic daily headache in men and a similar association in women.
The current study has several limitations. First, the assessment was not conducted by a physician. Instead, information was collected by trained examiners using a standardized questionnaire with in-person initial assessment followed by telephone follow-up at 3, 6, and 12 months following the injury. However, the questionnaire was designed to collect data which would allow classification into headache type similar to a structured headache interview conducted by a health-care provider in a headache specialty clinic. This could impact the diagnosis of cervicogenic headache that requires assessment of clinical signs implicating the neck as the source of pain. Second, we did not ask whether participants experienced more than one type of headache within the previous 3 months. Therefore, even if some participants may have experienced multiple types of headache, they were categorized into only one type. Third, we chose to categorize headaches based on whether symptoms met the classification criteria for the most severe type (migraine) at each time period. Lastly, even though patients may have had migraine with or without aura, for the current study we chose to group migraine with or without aura into a single category.
This prospective study was based on a large representative sample of those with moderate to severe TBI recovering from their injuries. This may reflect more accurate post-injury type, severity, and prevalence estimates of PTHs compared to selected samples who present to specialty clinics.
To date, with no defining clinical features of PTH, the diagnosis of PTH itself has limited usefulness in determining effective treatment. As a secondary headache, PTH is thought to have a structural or physiological basis that, if corrected or resolved, results in resolution of the headache. TBI may result not only in structural neuronal damage, direct vascular trauma, and impaired autoregulation, but also neurochemical changes that may be similar to those seen in primary headache disorders. There are similarities between TBI and migraine in biochemical changes found in the brain. Increased extracellular potassium and intracellular sodium, calcium, and chloride have been reported (8). Both entities show excessive release of excitatory amines such as glutamate and aspartate. Intracellular and total brain magnesium has been found to decline in mild TBI and is postulated to be low during and between migraine attacks. There is a change in the calcium/magnesium ratio in both conditions and nitric oxide, while potentially leading to tissue injury through free radical formation following TBI, is postulated to be involved in migraine as it is a potent vasodilator at the vascular endothelium (8). These similarities between neurochemical changes of TBI and migraine suggest a shared causation for headache occurrence, by way of a ‘final common path’ (9). Since the migraine pain pathway is thought to include the activation of the trigeminovascular system and the release of inflammatory neuropeptides causing a neurogenic inflammation, it is plausible that direct physical force from TBI causes the biochemical changes briefly reviewed above that may result in similar activation of the trigeminovascular pathway and neurogenic inflammation.
In summary, our data show that a majority of those with headache after TBI may be classified using symptom-based, primary headache criteria. Migraine and probable migraine described the majority of headache cases after TBI at all time points up to one year post-injury. For those who had frequent headaches (multiple times per week or daily), the most common phenotype was migraine or probable migraine implying that the majority of headaches experienced post-TBI are both moderate/severe and frequent. This represents a significant societal burden on health-care demand in civilian and also active-duty and veteran military populations sustaining TBI.
This study strongly supports moderate to severe head injury as a risk factor for the development of headache syndromes, particularly migraine, in those without prior headache history. This is compelling, given that our population was predominantly male and older than expected when compared to age- and sex-specific incidence rates of migraine in the general population (18). The low proportion of tension-type and cervicogenic headaches, especially given the high proportion of vehicle accidents as cause of head injury in this study, supports migraine as the predominant headache phenotype in PTH. These findings and the relationship between head trauma and chronicity of headache in our study and others (17) have substantial implications for the treatment of headache following TBI. Evidence-based treatments using defined clinical syndromes such as those found in primary headache disorders may provide a practical and useful diagnostic framework on which to base management of a common, frequent, and persistent symptom of TBI.
Footnotes
Funding
This study was funded by the Department of Education, National Institute on Disability and Rehabilitation Research, TBI Model Systems: University of Washington Traumatic Brain Injury Model System H133A070032, Virginia Commonwealth Traumatic Brain Injury Model System H133A070036.
Conflict of Interest statement
Dr Lucas received funding from NIDRR and Wadsworth Foundation for this study. Other research funding has been from Merck, GSK, Map, Nupathe, St. Jude Medical (formerly AGA), BiogenIdec, Sanofi-Aventis, John L. Locke Foundation and the National Headache Foundation. Honoraria, advisory or consulting fees have been received from Merck, GSK, BiogenIdec, EMD Serono-Pfizer, Zogenix, Map, Novartis and Allozyne. Dr Hoffman receives research funding from NIDRR. Dr Bell receives research funding from NIDRR. Dr Walker receives research funding from NIDRR. Dr Dikmen receives research funding from NIDRR.
Acknowledgements
This study was conducted as part of the Department of Education, National Institute on Disability and Rehabilitation Research, TBI Model Systems module projects. Data were contributed by seven centers: University of Washington Traumatic Brain Injury Model System H133A070032; Rocky Mountain Regional Brain Injury System H133A070022; Mayo Clinic Traumatic Brain Injury Model System H133A070013; UAB TBI Model System H133A070039; North Texas Traumatic Brain Injury Model system H133A0252604; The Moss Traumatic Brain Injury Model System H133A070040; and the Virginia Commonwealth Traumatic Brain Injury Model System H133A070036.
