Abstract
Background: We investigated whether chronic headache related to medication overuse (MOH) is associated with changes in brain mechanisms regulating inhibitory cortical responses compared with healthy volunteers and episodic migraineurs recorded between attacks, and whether these changes differ according to the drug overused.
Subjects and Methods: We studied 40 MOH patients whose symptoms were related to triptans alone, non-steroidal anti-inflammatory drugs (NSAIDs) or both medications combined, 12 migraineurs and 13 healthy volunteers. We used high-intensity transcranial magnetic stimulation over the primary motor cortex to assess the silent period from contracted perioral muscles.
Results: In MOH patients the cortical silent period differed according to the type of headache medication overused: in patients overusing triptans alone it was shorter than in healthy volunteers (44.7 ± 14.2 vs. 108.1 ± 30.1 ms), but similar to that reported in migraineurs (59.9 ± 30.4 ms), whereas in patients overusing NSAIDs alone or triptans and NSAIDs combined duration of silent period was within normal limits (80.6 ± 46.4 and 103.8 ± 47.2 ms).
Conclusions: Compared with episodic migraineurs, MOH patients overusing triptans have no significant change in cortical inhibition, whereas those overusing NSAIDs have an increase in cortical inhibitory mechanisms. We attribute these changes to medication-induced neural adaptation promoted by changes in central serotonin neurotransmission.
Keywords
Introduction
Medication-overuse headache (MOH) is a chronic headache disorder (headache present on ≥ 15 days/month) that develops or markedly worsens during >3 months of medication overuse (analgesics or triptans or both) (1). Chronic headache is a disabling health problem that affects 2–5% of the general population and causes considerable long-term morbidity and disability (2). The proportion of patients with chronic migraine overusing symptomatic medication is 30–50% in general population samples (3) but reaches 80% in tertiary level headache centres (4). Among factors thought to promote the development of chronic pain are central sensitization and defective central pain control systems (5).
Experiments in animals show that chronic administration of analgesics facilitates the trigeminal nociceptive mechanism and increases cortical excitability, as studied with the cortical spreading depression model (6). The hyperexcitability of neurons in the cortex and trigeminal pathway coexists with changes in 5-HT receptor expression in the brain (7).
By changing neuron excitability, medication overuse in patients with chronic headache presumably alters the balance between inhibition and excitation in the cerebral cortex. One way to explore cortical inhibitory circuits is to use transcranial magnetic stimulation (TMS), a tool that can reveal functional abnormalities in the cerebral cortex and that is routinely used to study brain function non-invasively in various neurological diseases (8). A widely used inhibitory TMS variable is the cortical silent period (SP), an interruption in the voluntary motor activity induced by delivering a magnetic stimulus (9). The SP recorded from limb muscles reflects activation of spinal and cortical circuits (10,11) and is mediated mainly by gamma aminobutyric acid-B (GABA-B) receptors (12). In contrast, the SP recorded from facial muscles originates solely in the cortex (13,14).
Facial muscle SPs proved a highly sensitive neurophysiological variable that has already revealed abnormal cortical inhibitory mechanisms in patients with migraine. In migraineurs with and without aura studied between attacks we found a shortened facial SP, suggesting cortical inhibitory interneuronal hypoactivity (15). We attributed this functional abnormality to reduced thalamo-cortical drive on the motor cortex inhibitory interneurons reflecting the motor counterpart of the low pre-activation level in sensory cortices (16,17).
No study has evaluated whether medication overuse promotes specific changes in the brain mechanisms regulating inhibitory cortical responses in patients whose episodic migraine has evolved to MOH. Nor do we know whether cortical inhibitory responses differ according to the headache medication overused. Clarifying these concepts on the relationship between inhibitory mechanisms in the cortex and medication overuse would help in developing strategies for preventing or treating MOH.
In this neurophysiological study, seeking more information on cortical inhibitory mechanisms and prolonged pharmacological overstimulation, we investigated cortical excitability in patients with MOH. To do so we delivered high-intensity TMS over the primary motor cortex and recorded EMG responses from perioral muscles in patients and healthy controls. TMS variables measured to assess cortical excitability included the SP and the motor evoked potential (MEP). To investigate whether the duration of the SP responses differs according to the headache medication overused, we studied three sub-groups, patients overusing triptans, those overusing NSAIDs, and those overusing both medications combined. We then compared SPs responses recorded in the three sub-groups with those recorded in healthy volunteers and episodic migraineurs studied interictally.
Subjects and methods
Subjects
Clinical and demographic data for healthy volunteers and patients with headache

HV: healthy volunteers; MO: patients with episodic migraine without aura; MOH: patients with medication overuse headache; Triptans: patients with MOH overusing triptans; NSAIDs: patients with MOH overusing NSAIDs; Both: patients with MOH overusing triptans and NSAIDs combined; n: number. Data expressed as mean ± SD.
Transcranial magnetic stimulation recordings from perioral muscles in healthy volunteers and patients with headache

HV: healthy volunteers; MO: patients with episodic migraine without aura; MOH: patients with medication overuse headache; Triptans: patients with MOH overusing triptans; NSAIDs: patients with MOH overusing NSAIDs; Both: patients with MOH overusing triptans and NSAIDs combined; n: number; EMG: electromyogram; MEP: motor evoked potential; SP: silent period; Area measured in mV x s. Data expressed as mean ± SD.
MEP variables recorded in patients were compared with those recorded in 13 age- and gender-matched healthy volunteers (HV, mean age: 36 ± 15; 8 women), having no personal or familial history (1st or 2nd degree relatives) of migraine, and no detectable medical condition.
Patients with MOH and healthy volunteers taking any regular medication except the contraceptive pill were excluded from the study. To avoid sex-hormone induced effects, we studied women during their follicular phase (days 8 to 10 from the first day of menstrual bleeding).
All participants were given a complete description of the study and gave their informed consent before taking part in the experiments. The study was approved by the institutional review board.
Transcranial magnetic stimulation
Stimuli were delivered with a Magstim Super-Rapid device connected to a figure-of-eight coil (outer diameter 90 mm) placed over the hot-spot for facial muscles. The coil was held with the handle pointing backward and was oriented to evoke the highest MEP size. The intensity of stimulation for evoking the SP was the highest the individuals could tolerate or the maximum stimulator output (range 95–100%).
Recording technique
Electromyographic (EMG) responses were recorded from surface electrodes placed over the left and right perioral muscles (18). The active electrode was placed 2 cm lateral to the midline in the mental region, and the reference electrode 0.5 cm lateral to the ipsilateral labial commissure. These electrode positions pick up EMG activity from perioral muscles (the orbicularis oris, risorius and triangularis muscles). Because we asked participants to ‘protrude their mouth vigorously’ the recorded EMG activity originated mostly from the orbicularis oris muscle.
EMG activity was recorded and stored for off-line analysis by means of a CED power1401 device (Cambridge Electronic Design Ltd, Cambridge, UK). EMG signals were amplified and filtered by a Digitimer D360 pre-amplifiers (Digitimer Ltd, UK) (band-pass 200–5000 Hz, gain 1000) and full-wave rectified. Background EMG activity was recorded for 200 ms before magnetic stimuli were delivered. Patients and controls were asked to contract the target muscles at their maximum strength by vigorously protruding their mouth.
Measurements
The neurophysiologists and other staff involved in data recording and analysis were blinded regarding the diagnostic status. As TMS variables reflecting cortical excitability we measured MEP latency and size and the duration of the perioral muscle SP. We also measured the background EMG activity in the 100 ms preceding TMS. All variables were measured in six single trials. Data were subsequently averaged to provide one mean value per variable per side and per individual for statistical analysis.
The background pre-stimulus EMG activity was calculated as the area (mV × s) under the signal in the 100 ms preceding the stimulus. The MEP was quantified as the area (mV × s) under the signal in the interval elapsing from the onset of the MEP (i.e. MEP latency) to the onset of the SP. The duration of the SP was taken from the end of the MEP to the latency at which the EMG activity returned to its mean pre-stimulus level.
Statistical analysis
All data had normal distribution and homogeneous variance, as checked by Levene's test. Background pre-stimulus EMG activity, MEP latency and area, and SP onset latency and duration data were tested in separate one-way between-groups analyses of variance (ANOVAs). For within-subjects analysis and comparison between patients’ headache type (MOH, MO), and overuser sub-groups (triptans vs. NSAIDs vs. both combined), separate one-way between-group ANOVAs were run for each variable. The Bonferroni test was used for all the comparisons between groups and sub-groups of MOH patients.
Pearson’s coefficient was used to test correlations between neurophysiological (pre-stimulus EMG, MEP size and SP duration) and clinical variables (disease duration, days with headache, monthly tablet intake).
Results
SP recordings were obtained from all patients and controls participating in the study (Figure 1). All groups studied had similar age (p > 0.05) and gender distribution (chi-squared p = 0.736).
Raw data from a representative participant in each study group and sub-group. Grand-average of six traces from the right and left perioral muscles. The magnetic stimulus is delivered at 200 ms. Arrows indicate the silent period (SP).
Preliminary data analysis across the recording side (left and right) revealed no main effect for factor side, indicating that within both groups the values of the TMS variables studied remained unchanged across the side of the face. In subsequent analyses we therefore used for each participant the mean value from the pooled data from the two sides of the face.
No difference was found in MEP latencies in patients with MOH or MO and healthy controls (F(2,62) = 0.84, p = 0.56). The background EMG (F(2,62) = 1.27, p = 0.28) and MEP areas (F(2,62) = 0.49, p = 0.63) were similar in all participants studied, as were the onset latencies of the SP (F(2,62) = 1.30, p = 0.29).
ANOVA testing SP duration showed a main effect for factor group (F(2,62) = 4.25, p = 0.01) (Figure 2). Post hoc analysis showed that whereas SP duration was reduced in patients with MO (p = 0.01), no difference was found between patients with MOH and controls (p = 0.14; Figure 2).
Bar chart of the SP duration in facial muscles in the study groups and sub-groups. HV healthy volunteers, MOH patients with medication overuse headache, MO patients with migraine without aura.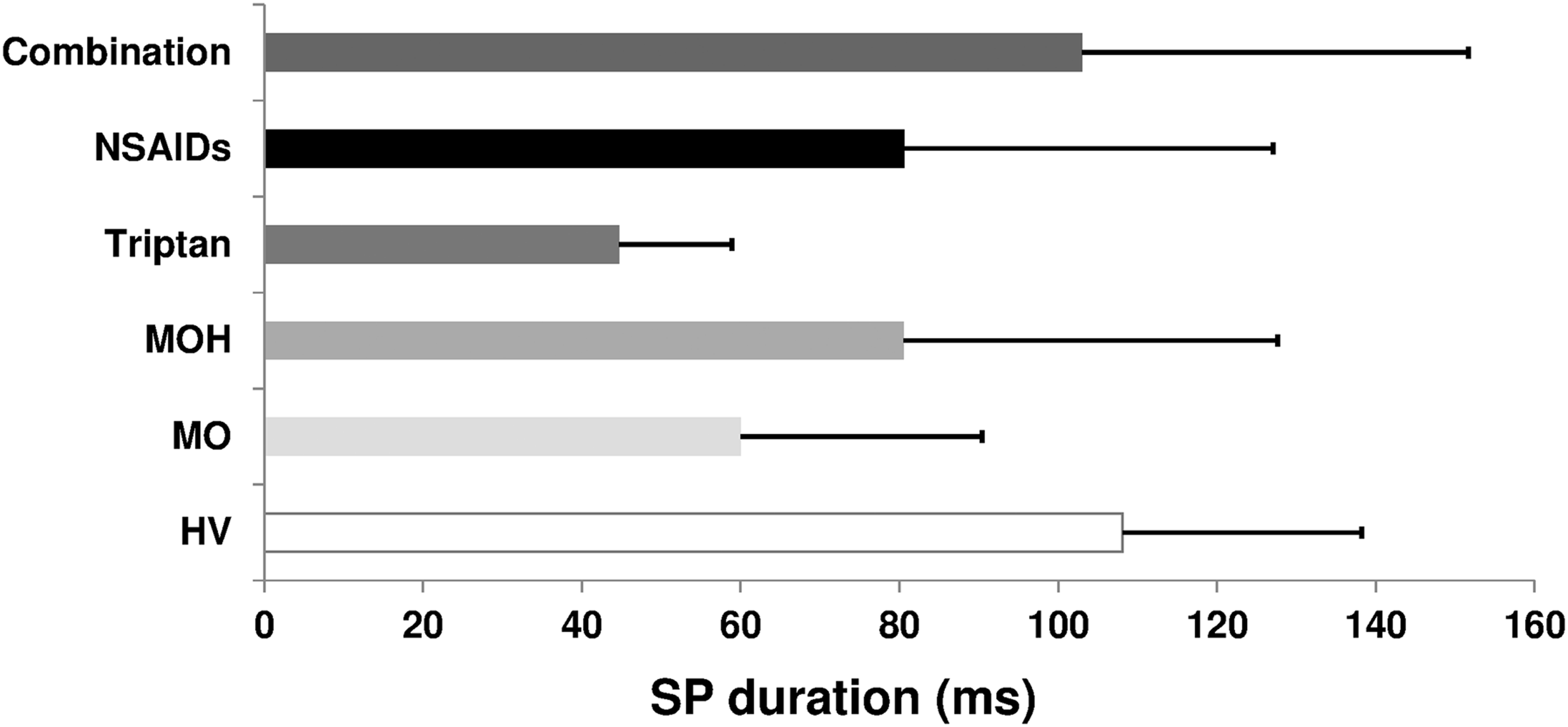
Within MOH sub-groups ANOVA showed that the SP duration was shorter in patients overusing triptans (p = 0.002 vs. controls) than in those who overused NSAIDs (p = 0.664 vs. controls) or triptans and NSAIDs combined (p > 0.05 vs. controls).
Monthly tablet intake was significantly higher in MOH patients overusing triptans and NSAIDs combined than triptans (p = 0.026) or NSAIDs (p = 0.014) alone. The number of days with headache and duration of headache history were similar in all groups (F(2,36) = 0.65, p = 0.52; F(2,40) = 1.64, p = 0.20, respectively).
In patients with MOH, correlation analysis revealed various relationships between clinical and neurophysiological variables. Among clinical variables, the number of days with headache correlated with monthly tablet intake (R = 0.315, p = 0.048), this correlation being stronger in triptan overusers than in NSAID overusers (R = 0.897, p < 0.001). The SP duration correlated positively with the monthly tablet intake in all MOH patients (R = 0.506, p = 0.001; in NSAID overusers (R = 0.537, p = 0.05) and in combination drug overusers (R = 0.476, p = 0.05, Figure 3). In triptan overusers, correlation analysis showed inverse relationships between SP duration and monthly tablet intake (R = –0.714, p = 0.02) and the number of days with headache (R= – 0.734, p = 0.01, Figure 3).
In the all MOH patients group, as well as in the NSAID and combination sub-groups, the SP duration correlated positively with the monthly tablet intake. In triptan overusers, correlation analysis showed an inverse relationship between SP duration and monthly tablet intake.
In the control experiment, performed in 18 patients after 3 weeks off medication, recordings in the eight triptan overusers elicited similar-duration SPs before and after withdrawal (52.9 ± 48.3 ms before, 54.1 ± 48.5 ms after, paired sample t-test p = 0.610; Figure 4). Recordings in ten patients overusing triptans and NSAIDs combined showed a shortening of SPs after withdrawal (76.8 ± 36.7 ms before, 56.3 ± 47.9 ms after, paired sample t-test p = 0.035) within the duration range of episodic migraineurs (Figure 4).
Bar chart of the SP duration in facial muscles in the triptan (n = 8) and combination-overusers (n = 10) sub-groups before and after 3 weeks of drug withdrawal.
Discussion
In this study we provide new evidence showing that, in patients with MOH, the duration of the cortical SP in response to TMS differs according to the headache medication overused. Overusing triptans leaves the cortical SP almost unchanged compared with that reported in episodic migraineurs between attacks (15), whereas NSAIDs lengthen and thus normalize the SP. After withdrawal, SP duration returned to abnormal low values in patients overusing triptans and NSAIDs combined, whereas it remained unchanged in those overusing triptans alone.
We took several precautions to ensure that the shortened SP recorded in triptan overusers arises from disease-related mechanisms and not from variations in the study protocol tested. Most importantly, we carefully monitored background EMG muscular contraction levels, stimulation intensity, and MEP size. By excluding participants in the menstrual or perimenstrual period we also controlled for variability due to hormonal changes in female patients (19).
Ample information from studies in healthy volunteers (13,14) and patients with migraine (15) shows that the cortical SP recorded in perioral muscles, unlike limb SPs, reflects changes in intracortical inhibitory interneuron excitability alone, uncontaminated by interference from segmental mechanisms. Facial motoneuron excitability tested at various time points during the TMS-evoked SP remains unchanged, indicating that activating cortical inhibitory mechanisms has no effect on the reflex activation of the brainstem centres controlling facial muscles in healthy volunteers or migraineurs.
Overall, the results obtained in triptan overusers suggest that these medications left the SP unchanged from values in patients with episodic migraine. The inverse correlation we found between the monthly triptan tablet intake and SP duration suggests, however, that triptans oppose cortical inhibition in favour of excitatory mechanisms. This interpretation finds support in several studies testing the neurophysiological effects of triptans. In healthy volunteers a single dose of zolmitriptan decreases short-interval intracortical inhibition of the MEP, but leaves the motor threshold and SP unaltered (20). In episodic migraineurs and healthy volunteers a single dose of zolmitriptan increases the intensity dependence of cortical auditory evoked potentials (21). In healthy women a single dose of rizatriptan increases the amplitude of the contingent negative variation to a choice task paradigm (22). Patients with MOH overusing triptans have larger-amplitude pain-related cortical evoked responses than healthy controls (23). Collectively, these studies show that triptans change cortical responsiveness, probably favouring excitatory mechanisms.
The most striking finding in our study is the dose-related NSAID-induced increase in the cortical SP bringing it to values comparable to those recorded in healthy volunteers. In patients whose episodic migraine has become chronic, NSAID overuse normalizes the short-lasting SP recorded interictally (15). Although no study has investigated how NSAIDs influence cortical excitability in humans, the NSAID-induced SP increase we describe in patients with MOH fits in well with current knowledge from the two available studies in animals. Whereas fenamate significantly prolonged inhibitory post-synaptic currents in Purkinje cells recorded in mouse cerebellar slices (24), flufenamic acid, another NSAID, suppressed epileptiform activity in rat hippocampal slices by decreasing glutamatergic excitatory transmission (25).
The precise mechanism through which NSAIDs prolong cortical motor inhibition is unclear, but they could do so by modulating central serotonin transmission. Considerable evidence shows that serotonin is involved in motor function in animals and humans (26,27). A single dose of selective serotonin reuptake inhibitors (SSRIs) – drugs that enhance central 5-hydroxytryptamine (5-HT) neurotransmission and postsynaptic serotonin – modulates motor performance and functional magnetic resonance imaging cerebral activation in healthy volunteers and post-stroke patients (28,29). TMS studies in healthy volunteers show that both single (30,31) and chronic SSRI doses (32) change various measures of cortical excitability.
How NSAID overuse alters cortical excitability as measured by the cortical SP remains conjectural. Current pharmacological knowledge shows that NSAIDs influence central serotoninergic transmission and act on 5-HT receptors (33,34). In particular, aspirin down-regulates 5-HT2 receptors (35). Unlike the 5-HT1 receptor family, which primarily exerts an inhibitory effect, 5-HT2A receptor activation increases neuronal excitability ultimately by raising intracellular calcium (36). In the cerebral cortex of rats chronically exposed to paracetamol, 5-HT2A receptor upregulation coexists with increased frequency cortical spreading depression (7), an electrophysiological depolarization also reported in 5-HT-depleted rats (37). Patients with MOH have increased platelet 5-HT2A receptor density attributed to the reduced 5-HT level (38-40). Medication withdrawal reverses these changes, and normalizing platelet HT2A receptor density correlates with clinical improvement. Because central 5-HT2 receptors are located post-synaptically and exert an excitatory action on projection neurons and local GABAergic neurons (41), 5-HT2A receptor up-regulation could be one mechanism by which NSAIDs overuse prolongs the SP duration, thereby normalizing cortical inhibitory mechanisms.
Although excessive intake of triptans and NSAIDs can both lead to migraine chronification, the clinical features are not entirely overlapping. Unlike patients who suffer from MOH following analgesic overuse, those overusing triptans did not describe the typical tension-type daily headache, but rather a migraine-like daily headache. Moreover, triptans overusers typically show a faster chronification of migraine but a milder and shorter duration of withdrawal headaches (42,43). Whether the difference here we have observed between the drug classes with regard to cortical inhibition is related to these clinical observations remains to be determined.
In conclusion, although the sample size should be acknowledged as a limitation of the present study, compared with episodic migraineurs in patients with MOH, triptan overuse leaves cortical inhibition unchanged, whereas NSAIDs intensify cortical inhibitory mechanisms. We attribute these changes to medication-induced neural adaptation promoted by changes in central serotonin neurotransmission.
