Abstract
Keywords
Introduction
Hypnic headache (HH) is a rare primary headache disorder that is characterized by strictly sleep-related headache attacks, occurring almost every night and mainly affects older patients beyond the age of 50 (1). Only few studies deal with the underlying pathophysiological mechanisms in HH. The exclusive sleep relationship of headache attacks raised the question whether an alteration of the hypothalamus might be responsible for HH as this anatomic structure regulates sleep and controls circadian rhythms (2). This hypothesis has been supported by a recent voxel-based morphometry study that showed gray matter decrease in the anatomic area of the posterior hypothalamus (3). Another hypothesis postulated HH to be a rapid eye movement sleep-associated disorder but previous studies could not confirm this assumption (4–6). Sleep-associated breathing disorders (7,8) and nocturnal arterial hypertension (9) have also been accused to trigger HH but study results are inconsistent. In summary, the underlying pathophysiological mechanisms in this rare primary headache disorder are still unknown in most parts. However, due to its consistent occurrence every or almost every night in most patients and the mostly chronic course of disease, it could be hypothesized that HH shows central facilitation within the trigeminal system comparable to other chronic headache disorders.
Alterations of trigeminal processing in regard of central facilitation or sensitization are commonly observed in various headache disorders including medication-overuse headache (10,11), chronic migraine without medication overuse (12), chronic tension type headache (13), and during acute migraine attacks (14,15). Additionally, the phenomenon of impaired habituation plays a crucial role in the pathophysiology of migraine (16–19) and a habituation deficit was even suggested to be an endophenotypic marker for presymptomatic migraine in unaffected relatives of migraineurs (20–22). To which extent excitability changes of the trigeminal nociceptive system and alterations of habituation within this system are involved in the pathophysiology of HH remain to be investigated.
The nociceptive blink reflex (nBR) and pain-related evoked potentials (PREP) allow a quantitative analysis of the trigeminal system at brainstem as well as supraspinal level. For nociception specific stimulation, a special concentric copper platinum electrode is used, which is characterized by a small cathode anode distance and therefore provides a high current density with low current intensity. In turn, mainly nociceptive Aδ fibres are depolarized (23). The specificity of nBR and PREP for alterations of trigeminal nociceptive processing has been shown previously (14) and nBR habituation has been widely investigated (16,22,24).
The aim of this study was to evaluate the function of the trigeminal nociceptive system at brainstem as well as supraspinal level in 15 HH patients and 15 age- and gender-matched healthy controls to further illuminate the pathophysiology of this rare headache disorder.
Materials and methods
The study protocol was reviewed and approved by the Medical Ethics Committee of the University of Duisburg-Essen and written informed consent was obtained from all participants prior to investigation.
Patients and healthy controls
Demographic and clinical characteristics
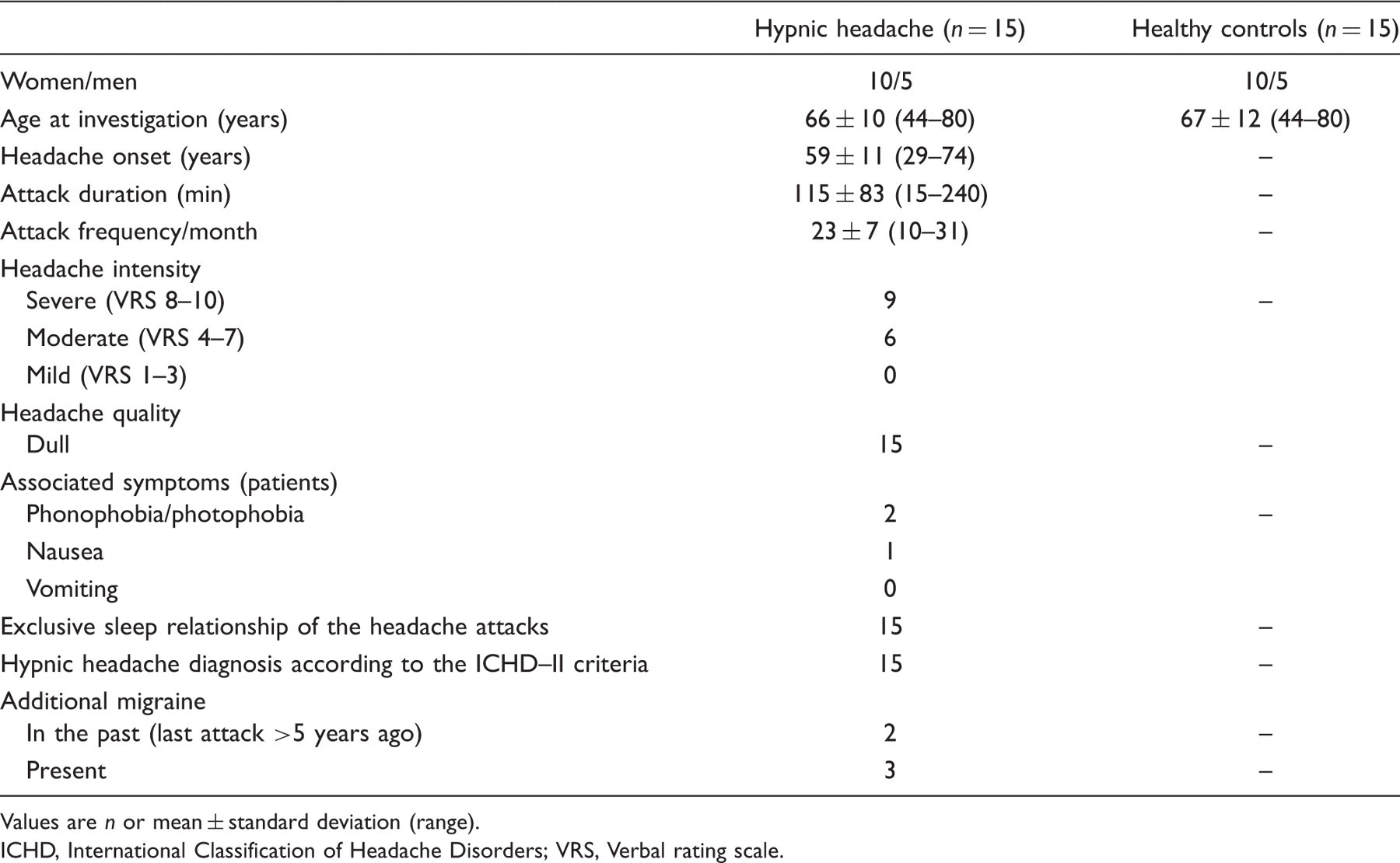
Values are
ICHD, International Classification of Headache Disorders; VRS, Verbal rating scale.
All patients fulfilled the ICHD-II criteria of HH (code 4.5) but not all patients met all single diagnostic criteria. Three HH patients were younger than 50 years when the headache disorder started (29, 46, and 49 years), therefore did not fulfil criteria C3. Three patients suffered from either accompanying photophobia or nausea during the headache attacks, but none of them had more than one accompanying symptom. Four patients described headache attacks longer than 180 minutes (200, 240, 240, and 240 minutes), which is longer than the common HH attack duration of 15–180 minutes. However, the ICHD-II classification only requires a minimum duration. Longer HH attacks as in our patient population do not contradict the recent ICHD-II criteria of HH.
None of the patients described increased day-time sleepiness due to the sleep disturbances caused by their hypnic headache attacks.
No one took HH-specific prophylactic medication (e.g. lithium). Nine patients drank a cup of coffee, some of them before going to sleep, some of them when awaking with headache at night. All of these patients reported either reduction of attack frequency and/or severity. Twelve of the controls drank coffee on a regular basis. Patients and controls were told not to drink coffee within 10 hours before electrophysiological investigation to avoid influences of caffeine on the electrophysiological results.
One patient was taking indometacin as acute pain-relieving drug. Five patients took antihypertensive compounds for treatment of arterial hypertension (betablocker three patients, angiotensin-converting enzyme (ACE) inhibitors two patients, betablocker and ACE inhibitor one patient), one patient took
Five HH patients suffered from an additional episodic migraine despite the HH disorder. Three of them still had on average three migrainous headache days per month. Two of the migraineurs had their last migraine attack more than 5 years ago. All patients were able to clearly distinguish their HH attacks from their migraine.
Electrophysiological settings
For nociceptive stimulation, two planar concentric electrodes (Walter Graphtek, Lübeck, Germany, http://www.walter-graphtek.com/) were attached to the skin 10 mm above the entry zone of the supraorbital nerve approximately 2 cm apart. The left forehead and right forehead were stimulated 15 times per session in each patient and control (triple pulse, monopolar square wave, duration 0.5 ms, pulse interval 5 ms, interstimulus interval 12–18 seconds, pseudorandomized). An ascending and descending sequence of 0.2 mA intensity steps was used to determine perception and pain thresholds. The stimulus intensity was set at twice the individual pain threshold. Stimulation intensities above 2 mA were excluded to minimize the risk of Aβ fibre co-activation (23). Stimuli were delivered to each side of the forehead in pseudorandom order in terms of start site (i.e. left or right side of the forehead).
nBR and PREP were recorded simultaneously following trigeminal stimulation of the forehead. The nBR was recorded using surface electrodes placed infraorbitally referenced to the orbital rim. The recording parameters were bandwidth 1 Hz to 1 kHz, sampling rate 2.5 kHz, sweep length 300 ms (1401 plus, Signal, Cambridge Electronic Design, UK). PREP were recorded with electrodes placed at Cz referenced to linked earlobes (A1–A2) according to the international 10–20 system.
Signal analysis was performed by an investigator blinded to the diagnosis. The first sweep was rejected to avoid contamination by startle response. The remaining 14 sweeps were averaged. For nBR onset latencies were rectified and analysed for each sweep separately. A mean value for each session was calculated. Areas under the curve were calculated between 27 and 87 ms (25). Concerning PREP, negative (N) and positive (P) peak latencies and peak-to peak (PPA) amplitudes were analysed. Mean values of the stimulation blocks for each subject and subsequent mean values for the group were calculated.
Additionally, areas under the curve (AUC) of the R2 nBR response were analysed in each sweep off-line. The data were normalized as a percentage deviation from the first value. Regression coefficient for each stimulation block of 14 consecutive sweeps (habituation) as well as a mean regression coefficient (MRC < 0 mean habituation, MRC > 0 mean augmentation) for each head side (right vs. left) were calculated.
Offline-analysis was performed with a custom-written PC-based software using Matlab version 7 (The MathWorks, Natick, MA, USA).
Statistical analysis
ANOVA was used for analysis of mean values for thresholds, latencies, amplitudes, AUC, and habituation (regression coefficients) of hypnic headache patients and healthy controls. The between-subject factor was ‘Disease’ (hypnic headache vs. healthy controls) and the within-subject factor was ‘Side’ (right side vs. left side). Bonferroni post-hoc correction for multiple comparisons was performed for post-hoc analysis. All statistics were calculated with SPSS version 16 (SPSS, Chicago, IL, USA). The level of significance was set to
Results
Nociceptive blink reflex
The nBR results are summarized in Table 2. An exemplary nBR recording of a HH patient as well as a healthy control is shown in Figure 1. Pain thresholds and stimulation intensities did not differ between HH patients and healthy controls (data not shown). No significant differences were observed comparing AUC (df 3, F = 0.285, Example pain-related evoked potential (PREP) and nociceptive blink reflex (nBR) in hypnic headache patients and healthy controls. Each curve represents recordings of one single hypnic headache (HH) patient or healthy control. A, PREP in a HH patient; B, PREP in a healthy control; C, nBR in a HH patient; D, nBR in a healthy control. Nociceptive blink reflex and pain-related evoked potentials PPA, peak-to-peak- amplitude.

nBR habituation
In healthy controls, nBR R2 responses habituated with repetitive stimulation (mean regression coefficient −2.7 after right-sided stimulation and −0.89 after left-sided stimulation, without significant differences after right- and left-sided stimulation) (Figure 2). This equals an amplitude loss of 37.8 % (14 × −2.7) and 12.5% (14 × −0.89), respectively, over 14 consecutive sweeps. In hypnic headache patients, nBR R2 responses with repetitive stimulation (MRC after right-sided stimulation and after left-sided stimulation, without significant differences between right- and left-sided stimulation). The differences between HH patients and healthy controls as well as stimulation side were insignificant (df 3, F = 0.920, Habituation in hypnic headache patients (B) compared with healthy controls (A) …, reference line (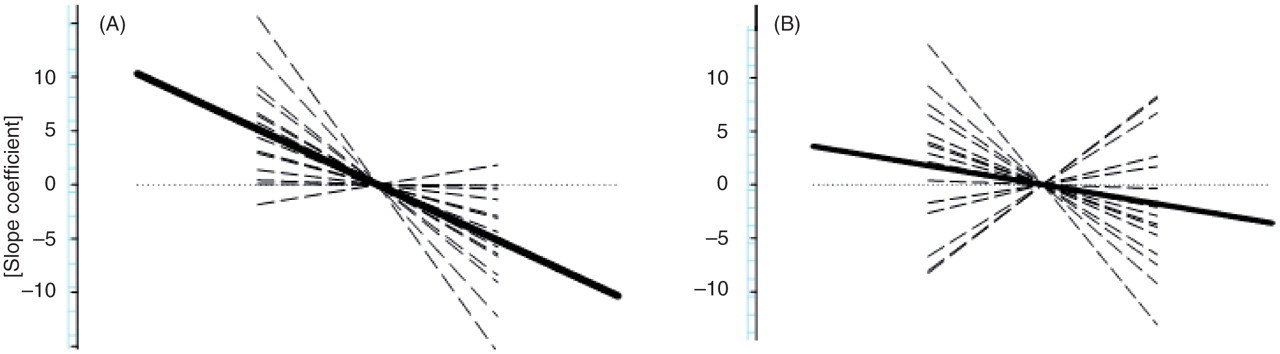
Pain-related evoked potentials
The pain-related evoked potential results are summarized in Table 2. An exemplary PREP recording of a HH patient and a healthy control is shown in Figure 1. Pain thresholds and stimulation intensities did not differ between HH patients and healthy controls (data not shown). No significant differences were observed comparing N2 latency (df 3, F = 0.568,
Discussion
No difference of trigeminal nociceptive processing in HH patients compared with healthy controls was detected, either at the brainstem level (nBR) or the supraspinal level (PREP). Moreover, we did not observe any changes of habituation in HH. So far, only structural brain changes in terms of hypothalamic gray matter decrease could be demonstrated in HH (3). Investigations on functional changes especially in regard to trigeminal pain processing have not been conducted until now, mainly due to the rareness of this disease. These results suggest that central facilitation and habituation deficit are not specific to chronic pain in general, but may be restricted to certain chronic pain conditions.
Hyperexcitability of the trigeminal system is a common observation in chronic headache disorders and has been extensively investigated in chronic migraine (CM) and medication overuse. de Tommaso et al. (12) performed event-related potentials following CO2−laser thermal stimulation (LEPs) of hand and facial regions in 25 CM patients and compared them with 15 patients with episodic migraine without aura and 15 normal control subjects. LEP amplitude variation was significantly reduced in CM in comparison with both control groups. These data suggest an abnormal cortical processing of nociceptive inputs in CM patients. Ayzenberg et al. (10) investigated 15 triptan-overuse headache patients, 14 analgesic-overuse headache patients, 16 patients with episodic migraine, and 15 healthy controls using nBR and PREP. In medication-overuse patients, central facilitation was mainly observed on supraspinal level after somatic (upper limb) as well as trigeminal (forehead) nociceptive stimulation. After withdrawal of medication and reduction of headache frequency in parallel, trigeminal PREP results normalized eventually. The degree of facilitation or sensitization seems to be positively correlated with the amount of headache days (26). Interestingly, although the HH patients on average had 23 headache attacks per month and therefore fulfilled the diagnostic criteria of a chronic pain condition (>15 headache days per month and >3 months (1)), we were unable to find a similar pattern of central facilitation in these patients. Chronicity of disease does not seem to be the only predictor for electrophysiologically conceivable cerebral overexcitability.
Habituation is defined as a decrease of strength of cortical responses when applying a stimulus repeatedly. It is thought to be a basic kind of learning and helps to avoid sensory overload (27,28). In regard to the ‘dual-process theory’, habituation results from a dynamic balance between decremental and incremental processes (27,28). A deficit of habituation is a lack of response reduction after repeated stimulation and is commonly observed in migraineurs in different evoked potential studies including the nociceptive blink reflex (16). Interestingly, the most pronounced habituation deficit is observed interictally and tends to normalize just before and during a migraine attack (29). On one hand it is possible that the high attack rate in HH patients is responsible for the normal habituation pattern as the last attack is only few hours away and the next only few hours to come. On the other hand habituation might just simply be unaffected and normal in HH. However, it must be mentioned that this might be due to a methodological limitation, as several studies tested more stimulation blocks to evaluate habituation (22,24). This surely increases the sensitivity of the method und might reveal more subtle changes regarding habituation. Additionally it must be emphasized that three HH patients and one healthy control did show slight augmentation of habituation (MRC > 0), which implies a lack of habituation (Figure 2). Accompanying migraine can just partly explain this observation as only one of these HH patients was a migraneur. For all other patients and the healthy controls, this habituation deficit remains unexplained as no further clinical features were predictive for this observation (e.g. duration or frequency of the HH attacks, headache intensity).
In addition to primary headache disorders, a lack of habituation has also been observed in other chronic conditions like chronic back pain (30), tinnitus (31), schizophrenia (32), Parkinson’s disease (33), and fibromyalgia (34). Stankewitz et al. (35) proposed in this context the ‘unifying thesis’, signifying that the observed neuronal excitability changes are more likely due to the process of chronicity itself rather than being specific for a distinct disease. Our data do not support this hypothesis as HH patients do not show any changes of habituation despite the obvious chronic character of this disorder. Other factors beyond attack frequency and duration of pain history also seem to contribute to the pathological mechanisms of excitability and habituation changes.
Additionally, it has been suggested that facilitation and reduced habituation might be characteristic for so-called ‘sine materia’ pain syndromes (36) such as fibromyalgia (34) and cardiac X syndrome (37). Garcia Larrea et al. (36) hypothesized that this enhancement after pain stimulation might reflect an attentional bias in these allegedly nonorganic pain syndromes and point to psychophysiological factors to be involved in the pathophysiology of these disorders. However, our data do not support this concept because HH headache does not share this model of ‘sine materia’ chronic pain although no organic correlate can be detected in this headache.
Five of the investigated HH patients did also have an additional migraine headache, which might have influenced the electrophysiological results. In contrast to investigations in migraineurs, HH patients did not show any hyperexcitability or deficient habituation. Therefore, migraine associated changes of trigeminal pain processing presumably did not dominate in our HH patients.
Nine of the investigated HH patients were drinking coffee for headache treatment. There were no significant differences between the caffeine-consuming patients and the patients who did not drink coffee. Additionally, in the healthy control group, 12 subjects were also drinking coffee on a regular basis. We tried to reduce confounding influences of caffeine intake by caffeine abstinence of all study participants within 10 hours before examination. Sample sizes were too small to perform reliable comparisons between caffeine-consuming patients and non-caffeine-consuming patients (nine HH patients vs. six HH patients). Further investigation of non-caffeine-consuming patients is needed for a closer evaluation of the influence of caffeine in this headache entity.
Interestingly, none of the HH patients described increased day-time sleepiness despite obvious nocturnal sleep disturbances due to the nocturnal headache attacks. The reduced sleep requirement in this rather aged population might contribute to this observation. Affected young patients would probably report a larger impact on their day-time performance in regard to the headache. However, it cannot be excluded that headache-induced sleep disturbances have influenced the electrophysiological study results.
One methodological limitation due the relatively low number of investigated patients has to be considered when interpreting the results. Very small differences between groups might have not been detected due to lack of statistical power. We tried to compensate these restrictions by accurate age and gender matching of the HH patients and healthy controls. These data will need reconfirmation with larger patient numbers, which is quite difficult considering the rareness of the disease. It seems reassuring, however, that many other electrophysiological studies (i.e. medication-overuse headache) included subgroup analyses with 15 patients or less and were still able to find significant differences in this regard.
In summary, trigeminal excitability and habituation does not seem to be affected in HH at least in regard to the investigated specific modalities (nBR, PREP). These observations underline that HH is a distinct headache disorder that can be distinguished from other primary headaches such as migraine or cluster headache not only by its clinical characteristics but also by electrophysiological differences. Our data suggest that the chronic course of a primary headache disorder does not automatically determine the presence of changes in cortical excitability and alterations of the habituation phenomenon.
Footnotes
Funding
This work was supported by the IFORES grant 2010 of the University of Duisburg-Essen.
Conflicts of interest
Dagny Holle has nothing to disclose.
Charly Gaul has received research grants and honoraria from Deutsche Gesellschaft für Muskelkranke, Roux-Program of the University of Halle-Wittenberg, MSD, Berlin Chemie, Medtronic and Böhringer Ingelheim.
Sarah Krebs has nothing to disclose.
Steffen Naegel has nothing to disclose.
Hans-Christoph Diener has received honoraria for participation in clinical trials, contribution to advisory boards or lectures from Addex Pharma, Allergan, Almirall, AstraZeneca, Bayer Vital, Berlin Chemie, Coherex Medical, CoLucid, Böhringer Ingelheim, Bristol-Myers Squibb, GlaxoSmithKline, Grünenthal, Janssen- Cilag, Lilly, La Roche, 3M Medica, Minster, MSD, Novartis, Johnson & Johnson, Pierre Fabre, Pfizer, Schaper and Brümmer, SanofiAventis, and Weber & Weber; received research support from Allergan, Almirall, AstraZeneca, Bayer, Galaxo-Smith-Kline, Janssen-Cilag, and Pfizer. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF), and the European Union.
Holger Kaube has nothing to disclose.
Zaza Katsarava has received research grants and honoraria from Allergan, Bayer, Biogen and Merck, and is an advisory board member for Allergan. Headache research at the Department of Neurology in Essen is supported by the German Research Council (DFG), the German Ministry of Education and Research (BMBF), and the European Union.
Mark Obermann has nothing to disclose.
