Abstract
Objective: To describe a survey methodology to evaluate headache characteristics and burden and to present baseline characteristics for the international cohort of survey participants.
Methods: A targeted, web-based methodology was used to recruit and survey subjects with migraine in 10 countries. Based on reported symptoms, subjects meeting ICHD-2 criteria for migraine were included; eligible subjects were classified as chronic (≥ 15 headache days per month) or episodic (< 15 headache days per month). Outcomes included sociodemographic and clinical characteristics, resource utilization, disability, health-related quality of life, anxiety, depression and productivity.
Results: Of 23,312 survey respondents, 11,897 were eligible and 9715 (81.7%) completed the survey; subjects were 81.5% female; 5.7% (n = 555) had chronic migraine.
Conclusions: This is the first large international cohort of persons with chronic and episodic migraine studied using a web-based approach, a methodology well suited to the study of the burden of migraines.
Introduction
The burden of migraine to society remains significant (1,2). The World Health Organization (WHO) has identified migraine as an urgent public health priority as part of its global initiative to reduce the burden of headache (3). Episodic migraine (EM) has a one-year period prevalence that ranges from 5% to 8% in men and 11% to 16% in women (4–11). The substantial individual impact of migraine (12,13) is associated with reduced quality of life (14–17) as well as lost work (1,18,19) and decreased leisure time (15,20). The considerable direct costs are driven largely by medical consultations and use of pharmacotherapy for migraine management and prevention (1,19). Annual direct cost estimates have ranged from US$100 to more than US$7000 per person with migraine per year (21,22). Moreover, lost productive time (indirect costs) substantially exceed direct costs (21,22).
The International Classification of Headache Disorders (ICHD-2) (23) provides operational diagnostic criteria for the major types of migraine: migraine with aura, migraine without aura, and chronic migraine (CM). CM is included as a complication of migraine characterized by ≥ 15 headache days per month for at least 3 months, with at least eight headache days per month with one of two links to migraine (24). In addition, CM headaches must meet criteria for migraine without aura or respond to migraine-specific treatment. In the past, some patients with CM have been subsumed by the term transformed migraine, which refers to a change from EM to chronic daily headache (25). Although the best definition of CM is still debated, CM is the leading reason for headache consultations in headache subspecialty centers (26,27).
CM is a common condition, affecting approximately 1.4–2.2% of the general population (26). In addition, the observed rate of transformation from EM to CM ranges from approximately 2.5% in a large population study (11,25) to 14% per year in specialty clinics (28). Buse et al. showed that CM is associated with higher treatment costs and higher rates of comorbid medication and psychiatric conditions, including depression, anxiety, chronic pain and respiratory disorders, than EM in the USA (27). Despite evidence of the increased health burden among persons with CM, this subgroup of migraineurs has been studied much less frequently. Persons with CM are frequently excluded from clinical trials, which typically focus on EM, and few population studies have compared CM and EM directly. Though many studies have examined the burden of EM (29), population studies of CM are less common (26). Those studies comparing EM and CM have mostly been confined to single countries. International studies that comprehensively assess the burden of illness in CM using uniform methods are lacking, thus additional studies describing healthcare utilization patterns and patient-reported outcomes in this population are needed. Such studies will broaden the understanding of the clinical and health status of persons with CM and provide additional insight regarding the varying patterns of diagnosis, treatment, medical and psychiatric comorbidity, and occupational disability internationally.
To address this gap, novel and affordable methods that facilitate case-finding for EM and CM in large, geographically dispersed samples in multiple languages are required. These challenges can be approached using internet-based panel studies. By this method, panels of participants with access to the internet volunteer to participate in research. Initial sampling is not based on any particular condition (such as migraine) to reduce selection bias. Health panel participants are accessed through web-based survey designs, providing the opportunity to gather multinational data in a timely and cost-effective manner (30–32).
A web-based survey of health panel participants was employed in this study to explore burden among persons with migraine. The main objective of the International Burden of Migraine Study (IBMS) was to determine the relationship of migraine type (CM versus EM) and burden of illness outcomes in a large multinational cohort. Outcomes assessed included healthcare resource utilization, health-related quality of life (HRQoL), headache-related disability, lost work productivity, anxiety and depression. This paper describes the survey design and methodology employed, and presents baseline characteristics for the international cohort of subjects. Additional findings associated with specific outcome measures from the IBMS, including disability, HRQoL and healthcare resource utilization, have been reported elsewhere (33). Additional manuscripts reporting survey data, including the economic burden of EM and CM, are forthcoming.
Methods
This was a two-phase, web-based, cross-sectional panel survey conducted in 10 countries. In phase I, panelists were screened to identify individuals with CM or EM. In phase II, an online survey was administered to eligible participants.
Screening phase (phase I)
Web-based, cross-sectional survey data were collected from panelists in the following ten countries from February to July 2009: Australia, Brazil, Canada, France, Germany, Italy, Spain, Taiwan, the UK and the USA. Potential survey participants were identified by the global market research company Synovate Healthcare (Chicago, IL, USA) from a pool of registered panelists who expressed willingness to complete health surveys in general, without reference to headache. Panelists had previously participated in a health screen for symptom-based conditions including headache and migraine. Panelists who had reported headache or migraine in a previous survey were preferentially selected. Registered panelists received email invitations to participate in their country-specific official languages. Details of the Synovate Healthcare Panel can be found in the online supplementary information, Appendix 1.
Interested panelists who were 18 years of age or older, had an active email address at the time of study invitation and were able to read and understand the official language of their country of residence were asked to provide informed consent by ‘opting in’ via a web link in the email invitation. The study design and materials were approved by a central ethics review board (Institutional Review Board Services, Ontario, Canada).
In each country, panelists who previously reported migraine or headache were invited on an as-needed basis to permit close monitoring of eligibility and completion rates by country. This quota sampling method also allowed for precise control of the target sample size and timeline. Non-responders to the survey invitation were sent an email reminder after 3 days (online supplementary information, Figure 1). Respondents had the right to cancel their consent at any time by prematurely terminating the online survey.
Comorbidities by migraine group and total sample. Note: Percentages represent the proportion of respondents in each migraine group who reported having one or more of the conditions included in the comorbidity group. The five comorbidity groups were defined as follows: (1) Pain: chronic pain, rheumatoid arthritis/osteoarthritis, fibromyalgia. (2) Vascular disease risk factors: high blood pressure, obesity (derived from BMI), high cholesterol. (3) Vascular disease events: heart attack (myocardial infarction), stroke, transient ischemic attack (TIA), heart disease/angina (chest pain). (4) Psychiatric disorders: depression, anxiety/nervous or excessive worry. (5) Other conditions: irregular heart rhythms (palpitations or arrhythmias), PMS/menstrual problems, epilepsy/seizures/convulsions or ‘fits’, sinusitis or sinus infection, ulcers of the stomach or intestines, other.
A brief validated assessment was administered immediately following receipt of informed consent to screen out respondents who did not regularly suffer headache, and to classify those reporting migraine symptoms consistent with clinical criteria into one of two migraine subgroups: CM or EM. Screening began with respondents being asked whether they had headaches in the last 3 months not associated with a cold, the flu, a head injury, or a hangover. If yes, respondents were asked about the number of days in the last 3 months that they had: (a) a headache of any intensity and (b) headaches lasting 4 hours or longer. They were also asked how many different types of headache they had experienced and the number of headache days for each headache type (most severe, second most severe and other) they had in the past 3 months. Validated screening questions asked respondents to indicate symptoms they experienced during their most severe and second most severe headache types (34,35). The diagnostic features of migraine included in the screening questions were consistent with ICHD-2 (23) criteria: pain (moderate or severe pain, throbbing pain, pain that worsens with routine physical activity or unilateral pain), nausea, photophobia and phonophobia. Symptom frequency was collected using a 4-point scale of 0 = never, 1 = rarely, 2 = less than half the time or 3 = half the time or more. A 3-month time frame was implemented to achieve a stable estimate of monthly headache frequency and to be consistent with the definition of CM, which focuses on the previous 3 months (24).
In all countries, survey respondents were enrolled into the study if they reported:
at least 1 headache day in the last 3 months not associated with a cold, the flu, a head injury, or a hangover; and at least 1 day of any type of headache in the past 3 months; and responses of “Less than half the time” or “Half the time or more” for at least two of the four pain questions; and responses of “Less than half the time” or “Half the time or more” for either nausea or photophobia
Respondents meeting all of these criteria for migraine were deemed eligible for enrollment in phase II (online supplementary information, Figure 2), and were classified into one of two subgroups based on headache frequency: CM with ≥ 15 headache days per month and EM with < 15 headache days per month. The study targeted a minimum of 100 CM subjects in the USA and 50 in each of the remaining nine countries. EM respondents who fulfilled the study criteria were allowed to flow into the study naturalistically, but recruitment for all subjects was capped when the CM sample sizes were matched.
Data collection in phase II
In each country, until the CM target sample sizes were attained, all eligible respondents were enrolled and linked automatically from the screening section to the study questionnaire. All data, including a patient-reported confirmation of a physician diagnosis of migraine, were collected directly from respondents via the web-based survey. The recall period for survey questions varied from 1 week to 3 months, depending on the referent time frame used in validated questionnaires and in consideration of appropriate recall periods for specific migraine outcomes and resources. Survey questions were designed for ease of completion, and employed various question formats. Where possible, yes/no gatekeeper questions with skip patterns were programmed to reduce the survey length for respondents. In addition, question groups with multiple items that used the same Likert-type response scale were organized into a grid and combined on one screen, thereby reducing repetition of instructions and web page load time for respondents.
Respondents who completed the survey received a participation incentive in the form of redeemable points and monthly online sweepstakes. Details of the incentive program can be found in the online supplementary information, Appendix 1. Subjects unable to complete the survey in one sitting had the option of returning at a later date, until the survey was closed, and continuing from the last question completed. The core English survey was translated and adapted for each country, incorporating tailored language and questions related to sociodemographic characteristics and pharmacotherapies. When translation was necessary, local experts reviewed the survey language for readability and interpretation; minor modifications were made as appropriate. If a validated country-specific translation for a clinical or quality of life measure was unavailable, data for the instrument were planned and treated as missing by design; no data imputation was performed.
Study variables
Sociodemographic variables included age, gender, race, marital status, employment status, education, income, height and weight. Clinical history included headache frequency and pain severity, frequency of headache symptoms (moderate or severe pain, throbbing pain, pain that worsens with routine physical activity, pain that is worse on just one side of the head for part of the headache, nausea, photophobia and phonophobia), headache status at time of survey completion and comorbid conditions [pain, vascular disease (VD) risk factors, VD events, psychiatric disorders and other conditions]. Comorbidity assessment was based on self-report of a physician diagnosis in response to the question, “Have you ever been told by a doctor or any other health professional that you have any of the following health conditions?” A comprehensive list of 17 common medical and psychiatric conditions comorbid with migraine was provided to respondents and they were instructed to check all that apply. Authors and findings from the American Migraine Prevalence and Prevention (AMPP) study (20,36) contributed to the creation of this list of common comorbid conditions. Additionally, depressive and anxious symptomatology in the preceding 2 weeks was assessed by means of the Patient Health Questionnaire (PHQ-4) (37).
The survey also collected detailed healthcare resource utilization data, including headache-related visits to healthcare professionals, emergency room or urgent care clinics, diagnostics tests and treatments over the preceding 3-month time period. Subjects also reported over-the-counter and prescription medications consumed for the treatment or prevention of headaches in the preceding 4 weeks.
Country-specific lists of over-the-counter and prescription medications used for the prevention or acute treatment of headache were compiled. The medication list employed in the survey was adapted to the local markets in each country by a key opinion leader or local affiliate familiar with the prescribing practices and medications available in that country. Both acute and preventive medications were included. Acute medication classes included antipsychotics (such as olanzapine administered for its tranquilizer/sedative properties), combination pain medications with and without opiates/barbiturates, dopamine blockers, ergotamines, muscle relaxants, narcotics/opiates, non-steroidal anti-inflammatory drugs (NSAIDs), simple analgesics, triptans and triptan combinations. Preventive medication classes included antidepressants, anti-epileptics, cardiovascular drugs (beta-blockers, calcium channel blockers and others) and serotonin agonists. Subjects were also given the option to select “other”, without specifying a response. Subjects reported the number of days they took each of the medications over the preceding 4-week period.
The Headache Impact Test (HIT-6) (38) was administered to assess the impact of headache on subjects′ lives in the preceding 4 weeks across six domains: pain, role functioning (ability to carry out usual activities), social functioning, energy or fatigue, cognition and emotional distress. Headache-related disability was quantified by means of the Migraine Disability Assessment questionnaire (MIDAS) (39), which assesses the number of days that migraine prevented or limited activities in the preceding 3 months. This questionnaire was supplemented with additional questions pertaining to work or school attendance and non-attendance. Self-reported health status was measured by means of the EuroQol five-dimensions (EQ-5D) (40), a widely used measure to assess current health status across five dimensions (mobility, self-care, usual activities, pain/discomfort, and anxiety/depression) and generate health utilities. The Migraine-Specific Quality of Life Questionnaire (41), version 2.1 (MSQ v2.1) was used to measure how migraines affected and/or limited subjects’ daily performance in the preceding 4 weeks across three domains: Role Function Restrictive, Role Function Preventive and Emotional Function.
For all validated questionnaires, missing data were scored per developer scoring instructions for each instrument.
Data management
Data validation rules and electronic edit checks were programmed to enhance data quality at the point of data entry. Given logistical constraints, follow-up data queries could not be issued to subjects after survey completion, but variables were cross-checked within each survey to verify subgroup assignment, participant disposition, point of exclusion, and type of error detected in the record, if any. The survey was also programmed to prevent subjects from leaving questions blank. Data management features were designed to enhance data quality and obtain a complete data set that required minimal data cleaning; customized error messages and automated data validations aimed to facilitate survey completion. Post-database lock, quality control measures were also taken to ensure that records with inconsistent reports of age or gender were discarded. In addition, any records where the pattern of responses indicated that the subject may have responded carelessly were discarded.
Statistical analysis
The data were stratified by CM, EM, and total sample for each country individually to report subject recruitment and disposition. Countries were also pooled to describe overall characteristics of the sample. In relation to subgroups of interest, descriptive statistics were reported for the sociodemographic and clinical data. For continuous variables, mean, standard deviation, and minimum and maximum values were calculated. For categorical variables, frequencies and percentages were reported.
Separate comorbidities collected in the survey instrument were pooled into five categories for summary reporting purposes; percentages represented the proportion of subjects in each migraine group who reported having one or more of the conditions included in each of the comorbidity categories.
Incomplete survey data due to the participant terminating the survey prior to completion were not included in analyses. SAS version 9.1 (SAS Institute, Cary, NC) was used to perform all analyses.
Role of the funding source
This study was funded by Allergan, Inc. United BioSource Corporation (UBC) was contracted by Allergan, Inc., to design a burden of illness survey for administration to patients with EM or CM. Synovate was subcontracted by UBC to host and administer the web-based survey designed by UBC. The study sponsor was involved in the study design, data collection, data analysis, data interpretation, and writing of the article. KAP, AKK, KY, TKW, DCB, RBL, PJG, and AMB were compensated by the sponsor for consulting time on this study.
Results
Screening phase (phase I)
Response rates and proportions of subjects with chronic migraine or episodic migraine by country

Represents the proportion of the total sample by country that falls into each of the subgroups.
Represents the proportion of invited panelists who responded to some or all of the screening questions.
Represents the proportion of the total international sample that originates from each of the individual countries.
A total of 23,312 individuals responded to email invitations. The average response rate among the 10 countries was 32.4% (online supplementary information, Figure 1; Table 1). Response rates varied by country: from a low of 22.4% (Spain) to a high of 68.1% (USA). Among those who responded, 21,545 completed the survey, for a completion rate of 92.4%. The median survey completion time was 16 minutes. Of those who completed the screening survey, 11,897 (55.2%) met the criteria for CM or EM and were eligible for the phase II online survey. Use of electronic survey data collection made it possible for raw data to be available for analysis by country within 3 to 5 days after the study closed in the country.
Summary of screening form by migraine group



NA (not applicable) includes those who do not have second type of headache.
CM = chronic migraine; EM = episodic migraine.
Phase II survey
Sociodemographic characteristics by migraine group
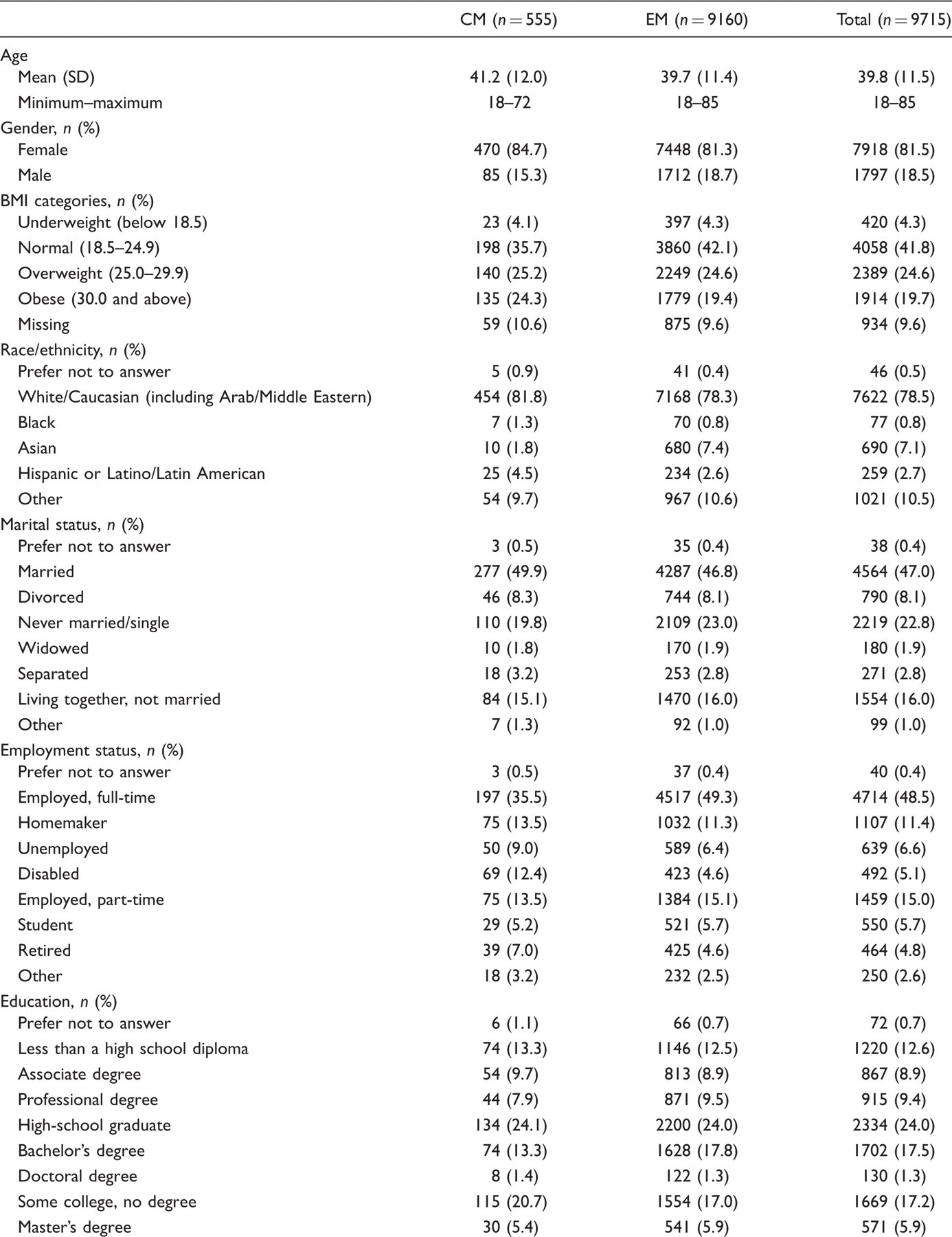

CM = chronic migraine, EM = episodic migraine, SD = standard deviation.
Clinical characteristics by migraine group


CM = chronic migraine, EM = episodic migraine, SD = standard deviation.
Respondents were asked to report the severity of the pain they experience when their most severe type of headache is at its worst.
CM subjects were slightly older, more likely to be Caucasian or Hispanic, and less likely to be Asian. CM was also associated with higher rates of unemployment due to occupational disability or retirement (Table 3). Although 65.0% of total completers reported having a physician-confirmed diagnosis of migraine, this proportion was slightly larger in the CM group (CM: 78.6% versus EM: 64.2%) (Table 4).
Rates of comorbidity were higher in those with CM than EM for all comorbidities evaluated in this study (Table 4). The number of comorbidities per participant was higher for CM (2.8) than for EM (1.7). Evaluating the specific comorbidity groups, subjects with CM were more likely than those with EM to have pain (38% versus 18.2%), VD risk factors (41.6% versus 33.7%), psychiatric disorders (45.6% versus 29.7%) and ‘other’ conditions (51.5% versus 40.3%) (Figure 1).
The majority reported that they have ‘severe’ headache pain with migraine (78.8%), particularly among individuals with CM (Table 4).
Discussion
This targeted cross-sectional, web-based, two-phase panel survey of nearly 10,000 persons with migraine from North America, South America, Europe and Asia has collected data on the burden of migraine from a large multinational sample of episodic and chronic migraineurs. It provides a uniform international data set with detailed patient-level information on clinical features, economics and quality of life outcomes. CM sample size targets were met in all but one of the countries surveyed. These data provide a foundation for examining inter-country variations in clinical, quality of life and other health economic outcomes as well as patterns of care.
Two national benchmark studies in migraine produced similar results: the AMPP study, focused on the USA (20,36), and the German Headache Consortium (GHC) study, focused on a single European country (42,43). However, given differences in design and methodology, outcomes from each study are not directly comparable. The AMPP study was based on the methods used by the American Migraine Studies 1 and 2 (20,36) and was a two-phase, 6-year longitudinal study consisting of a screening phase to identify cases of self-reported severe headache from a stratified random sample of 120,000 US households (257,399 household members) in 2004, and a longitudinal, annual, follow-up phase of questionnaires distributed annually between 2005 and 2009 to 24,000 persons with severe headache identified in the screening phase (5,44). The AMPP study was designed to evaluate prevalence and changes in headache diagnosis, symptomatology, headache-related disability and impact, medical and psychiatric conditions, treatment and resource utilization.
The GHC study mailed questionnaires to a random sample of 15,705 individuals between 18 and 65 years of age in Germany between 2003 and 2005 (42,43). Non-responders were mailed a reminder after 2 weeks; where possible, telephone interviews were performed to collect questionnaire responses. The study was designed to describe the epidemiology profile of responders and estimate CM prevalence in this responder population using demographic, personal and lifestyle characteristics data.
To some degree, the findings from the present web-based study were consistent with GHC and AMPP results when comparison of either sociodemographic or clinical characteristics was possible. Subjects with CM across all studies were generally older than patients with EM (AMPP 2009 survey: CM = 47.7 years versus EM = 46.0, p = 0.03 (27); GHC: CM = 46.3 versus EM = 40.3, p < 0.001). Additionally, subjects with CM from all three studies (IBMS, GHC, and AMPP) reported higher BMI. Although IBMS was not population-based, the mean rate of CM in the surveyed migraine population was 5.7%, only slightly higher than the rate of CM observed in the AMPP cohort (5.5%) (27). In addition, the observed eligibility rate of 55% was higher than the migraine prevalence estimated from population-based studies (6–8% of men and 15–25% of women in Western countries) (45) because the majority of panelists in all countries except Taiwan had been pre-screened for self-reported headache or migraine. These consistencies among our web-based study, AMPP, and GHC provide confirmatory evidence suggesting that selection bias did not preclude recruitment of a representative sample of persons with migraine.
This study is not without limitations. Data collection was complex in that there were three levels of ascertainment: panel pre-screening, phase I screening, and phase II assessment. Also, the study was not designed to create a population-based survey sample, but rather used targeted methods to recruit and survey a specific subset of persons with migraine (subjects with CM). The potential for selection bias may have been augmented as a result of web-based data collection methodology (30). Individuals who do not actively engage in computer-based communications may not have been contacted. Moreover, patient-reported data may not produce accurate results for certain types of questions, which may have affected the accuracy of the healthcare resource utilization data, including medication use. No medical records or healthcare providers were queried. However, the patient-reported outcomes in the rest of the survey were specifically designed for patients to complete, and validated instruments were used where possible (e.g. PHQ-4).
If the patients did not accurately respond to the screening questions related to migraine diagnosis, this may have inadvertently created a selection bias. This bias may have impacted eligibility rates (46) and clinical characteristics, particularly in the context of a 31% rate of response and survey completion, although this is unlikely due to some of the consistencies seen in relation to results from other cohort studies. More specifically, registered panelists may have intentionally responded to the screening questions in an attempt to qualify for the survey and thereby receive the incentive; however, given the complexity of the screening logic and the fact that respondents were unaware of the two-phase structure of the survey, this is unlikely. Furthermore, had respondents attempted to give ‘worst-case’ symptom-related responses, the rate of CM would likely have been much higher than that observed in other research, whereas it is in fact only 0.2% higher than rates previously reported in US population data (27).
Another challenge was that all data on diagnosis and prescription and over-the-counter medication use were based solely on patient report, and could not be confirmed through physician records. Inter-group differences in frequencies of comorbid health conditions, particularly for the pain category, may also have been confounded with the headache-related pain experienced by persons with migraine.
Web surveys do offer some significant advantages, including a high level of control in terms of programming, automated data checks for quality control, and use of email invitations to reach target goals, with access to a large pool of panelists (31). Interviewer bias was eliminated as a result of standardized online questionnaire administration, thereby providing a final cohort of migraineurs identified by a uniform set of screening questions.
The main advantage of health panels is the sheer number and geographic dispersion of the potential pool of pre-registered panelists who have expressed interest in completing surveys. Target sample sizes can be reached with relatively low programming costs and without the need to invest in clinic-based recruitment efforts. Web-based research methods grant access to populations that might not typically be found in a clinical setting. If necessary, web surveys of registered panelists can provide the opportunity to pre-screen a subset of the panel for specific criteria prior to survey launch, thus providing a rough estimate of the condition’s prevalence and the approximate number of invitations required to reach target sample sizes. Ability to pre-screen can be both time- and cost-effective, particularly for rare indications or difficult-to-locate populations. In addition, panel providers have the ability to partner with other providers to supply supplemental panelists if the eligibility or response rates fall below anticipated levels.
Other main strengths of this survey include its statistical power, its multinational scope, and the implementation of headache-related disability and HRQoL patient-reported outcome measures with strong psychometric properties. Data were collected from almost 10,000 subjects in just a few months, which would not have been possible using traditional paper-based approaches. Finally, electronic data collection, where participants are guided through the survey with streamlined branching logic and customized error messages, reduces burden on participants and minimizes data entry errors.
Despite the gold standard of clinical trial-based evidence, surveys remain an important epidemiologic tool for capturing cross-sectional or longitudinal data and providing fundamental insights into health and disease (47). Currently, participation in internet panels is high, with panelists participating in surveys on every continent. In light of society’s increasing reliance on the internet and web-based technology, applying this methodology to academic research interests and the collection of patient-reported outcomes can help improve our science and global reach. A primary goal of this web survey is for results of these data to contribute to a more in-depth understanding of the unique clinical and psychological needs of distinct subgroups of persons with migraine. Results of the relationship of headache-related disability to burden of illness in this cohort have already been reported through various media, including one publication (33); additional publications reporting the clinical determinants of health economic and quality-of-life outcomes in EM and CM are forthcoming.
Footnotes
Acknowledgements
The authors would like to thank the following individuals for their contributions to this study: CC Turkel (Allergan, Inc., Irvine, CA, USA); JE Hansen (Allergan, Inc., Irvine, CA, USA); MV Shah (formerly with Allergan, Inc.); L Allen, M-P Desrosiers, Dr KJ Ishak, I Proskorovsky, R Yu (United BioSource Corporation, Bethesda, MD, USA); Y Tan (formerly with United BioSource Corporation); Dr C Burk (Outcomes Research Consultant, Laguna Beach, CA, USA); and Synovate Healthcare (Chicago, IL, USA).
Funding
This analysis was supported by Allergan, Inc., Irvine, CA.
Conflict of interest statement
KP, AK, KY, and TW are employees of United BioSource Corporation, the research organization contracted by Allergan, Inc. to design and execute the survey. SV and AM are employees of Allergan, Inc. AB has received research grants from Allergan, Pfizer, GlaxoSmithKline (GSK), and Medtronic, has received honoraria from Allergan, Pfizer, GSK, Merck, Ortho-McNeil-Janssen Pharmaceuticals (OMP), and Medtronic, and has consultancy agreements with Allergan, Pfizer, and GSK. DB has received honoraria from Endo, Merck, Iroko, and MAP Pharmaceuticals, and has consultancy agreements with Endo and Allergan. PG has received research grants from MSD, Neuralieve, GSK, and Johnson & Johnson and has received honoraria from and has consultancy agreements with Allergan, Almirall, BosSci, Eli Lilly, Endo, MAP, Medtronic, Minster, MSD, Neuralieve, Neuraxon, and NeuroTherapeutics Pharma. RL has received research grants from Advanced Bionics, Allergan, GSK, Minster, Merck, Neuralieve, Novartis, OMP, Pfizer, and ProEthics/KOWA, has received honoraria from Allergan, AstraZeneca, GSK, Merck, Novartis, OMP, and Pfizer, and has consultancy agreements with Advanced Bionics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Cierra, Endo, Neuralieve, Novartis, OMP, Pfizer, Pozen, and ProEthics/KOWA.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
