Abstract
Background: Migraine imposes significant burden on patients, their families and health care systems. In this study, we compared episodic to chronic migraine sufferers to determine if migraine status predicted headache-related disability, health-related quality of life (HRQoL) and health care resource utilization.
Methods: A Web-based survey was administered to panelists from nine countries. Participants were classified as having chronic migraine (CM), episodic migraine (EM) or neither using a validated questionnaire. Data collected and then analyzed included sociodemographics, clinical characteristics, Migraine Disability Assessment, Migraine-Specific Quality of Life v2.1, Patient Health Questionnaire and health care resource utilization.
Findings: Of the respondents, 5.7% had CM and 94.3% had EM, with CM patients reporting significantly more severe disability, lower HRQoL, higher levels of anxiety and depression and greater health care resource utilization compared to those with EM.
Interpretation: These results provide evidence that will enhance our understanding of the factors driving health care costs and will contribute to development of cost-effective health care strategies.
Introduction
Migraine is a common, disabling neurological disorder (1). Its prevalence generally ranges between 5% and 8% for men and 11% and 16% for women (2–9). Although migraine has a worldwide distribution, there are regional variations in migraine prevalence; rates are lowest in Africa and Asia, higher in Europe and South America and highest in North America (2). A recognized public health problem (10), migraine ranks 19th among all single causes for years lived with disability according to the World Health Organization (WHO) (2).
The WHO considers a day lived with severe migraine as disabling as a day lived with dementia, quadriplegia or acute psychosis and more disabling than blindness, paraplegia, angina or rheumatoid arthritis (11,12). Because migraine attacks are episodic and recurrent, assessing headache-related disability exemplifies the challenges presented by chronic disorders with episodic attacks (CDEAs) (13). CDEAs (e.g. migraine, epilepsy, asthma and angina) are characterized by symptomatic attacks superimposed on an enduring predisposition to attacks. As a group, CDEAs are highly prevalent and disabling; they present a unique set of challenges and opportunities (13).
Like other CDEAs, headache-related disability increases on days with attacks. Migraine attacks may necessitate complete bed rest and interfere with occupational and educational functioning, ability to do household work and chores as well as family responsibilities, and social and leisure activities (14,15). Headache-related disability can be measured with the Migraine Disability Assessment (MIDAS) questionnaire, which assesses lost work and personal time in the preceding three months due to migraine (16). MIDAS is used as an outcome measure and as a basis for treatment stratification in clinical trials (17,18) and treatment guidelines (19). Migraine sufferers also experience reductions in health-related quality of life (HRQoL) (13,14,20). Chronic migraine (CM) sufferers are significantly more likely to report depression, anxiety, chronic pain and respiratory disorders (21). The economic impact of migraine includes both increased health care costs and lost workplace productivity; the impact is most severe for those with CM (22,23).
Persons with migraine have been classified based on headache frequency: episodic migraine (EM; <15 headache days per month) and CM (≥15 headache days per month). In the second edition of the International Classification of Headache Disorders (ICHD-II), CM is listed as a complication of migraine (24). Although the specific criteria for CM have been debated, this term always describes migraine sufferers with headache on 15 or more days per month (25,26).
Most epidemiologic studies and clinical trials have focused on EM, often excluding the most disabled segment of sufferers, patients with CM. The small number of previously published epidemiologic studies have mainly focused on single regions or single countries. These studies vary in sampling strategies, methods and endpoints, making international comparisons difficult (27). Consequently, we know little about the international distribution of headache-related disability, clinical features that determine headache-related disability or consequences of headache-related disability on HRQoL and resource use, particularly for CM (28,29). Understanding the drivers and consequences of headache-related disability provides a foundation for the design of cost-effective interventions to reduce the individual and economic burden. Improving our understanding of migraine and taking steps to reduce headache-related disability have been identified as urgent public health priorities by the WHO in their global campaign to lift the burden of migraine (30). In addition, an effective approach to understanding the burden of migraine may provide insights into other chronic disorders with episodic attacks.
This study was undertaken by contemporaneously collecting data from a number of countries in Western Europe, North America and Asia/Pacific to facilitate direct international comparisons using the same methodology. The objective of this study was to determine the relationship between headache frequency (CM vs. EM) and its consequences for individuals (disability, HRQoL) and society (health care system burden). This paper will focus on the relationship between headache frequency and its consequences.
Methods
Study design and population
This was a cross-sectional, Web-based observational survey with participants from Australia, Canada, France, Germany, Italy, Spain, United Kingdom, Taiwan and the United States and was conducted from February to April 2009.
This study was approved by a central ethics review board (Institutional Review Board Services, Ontario, Canada). Potential participants were identified from panels maintained by Synovate Healthcare (Chicago, IL, USA) (or its partner companies); eligible panelists expressed willingness to complete Web-based health surveys and as part of a health screen were asked about previous natural diagnosis of a broad range of health problems. For the survey, invitees previously reported suffering from headaches or migraine (except in Taiwan, where potential participants were selected from the general pool). Participants completing the survey were offered an incentive for participation in the form of redeemable points and entry into a monthly sweepstakes.
All male and female potential participants in targeted countries were eligible for inclusion in the survey if they were ≥18 years of age; had an active email address at the time of study invitation; were able to read and understand the official language of their country of residence; and endorsed experiencing over the last three months headaches not associated with a cold, the flu, a head injury or a hangover.
Interested participants provided consent by “opting-in” using a Web link (provided in the email invitation) as a proxy for written informed consent. Validated screening questions (31) assessed the diagnostic features of migraine based on the ICHD-II criteria (24) and determined eligibility. Those who did not respond to the survey within three days of receipt were sent an email reminder (Figure 1). Participants were also asked to report the number of days in the last three months with a headache of any intensity.
Study participant disposition.
Eligible participants were categorized as either EM (<15 headache days per month) (23) or CM (≥15 headache days per month) (24) at the time of survey completion and enrollment continued until a minimum of 50 chronic migraineurs were identified in each country (100 in the United States), except in Taiwan, where data collected was terminated before the target goal was reached due the small number of available health panel members.
Data collection
Participants reported their age, gender, race, marital status, employment status, education, household income and height and weight. Household income was classified as less than or equal to the median or greater than the median for each country. Participants also reported the number of headache types they experienced and then detailed characteristics for their most severe and second-most severe headache type. The frequency of the following headache symptoms were reported using a four-point scale (“never”, “rarely”, “less than half the time” or “half the time or more”): moderate or severe pain, throbbing pain, pain that worsens with activity, worse pain on one side, nausea, photophobia and phonophobia. For each headache type, headache intensity was measured using the Visual Analog Scale (VAS) score on a 10-point scale and categorized as follows: mild (≤4), moderate (5 and 6), severe (7 and 8) and very severe (9 and 10). In addition, comorbid conditions reported were based on self-report of a physician diagnosis (SRPD). Conditions were assessed by respondent endorsement of the item “Have you been told by a doctor or any other health professional that you have any of the following health problems?” Conditions were divided into five categories (psychiatric, pain, vascular disease risk factors, vascular disease events and other).
Migraine Disability Assessment Questionnaire
The MIDAS questionnaire, used to assess headache-related disability, is the most frequently used disability instrument in migraine research and clinical practice (16,32,33). There is extensive evidence for reliability and validity of the MIDAS, and it has been translated into multiple languages (16,32–35). It is a self-administered questionnaire consisting of five items that assess days of missed activity or substantially reduced activity due to headache in three domains—schoolwork/paid employment, household work or chores, and non-work (family, social and leisure) activities. Because each day of headache could result in lost time in each of the three domains, the maximum MIDAS score per day is 3 and the maximum score over 3 months is 270. Responses to these items are summed for a total score, which can be categorized into one of four grades of headache-related disability: grade I, little or no disability (score of 0–5); grade II, mild disability (score of 6–10); grade III, moderate disability (score of 11–20) (36); and grade IV, severe disability (score of ≥21).
Review of MIDAS total scores, computed using the standard scoring method, revealed that some participants had scores above the usual range of 0–270. Some participants double counted disability days in a specific domain. For example, separate questions are asked about days of missed work and days with productivity at work reduced by half or more, and MIDAS instructions ask the participant not to report a day as both a missed workday and a reduced productivity day. A small proportion of participants appear to have reported days as missed and also as days of reduced activity, contributing to inflated MIDAS total scores. Total scores were recomputed to adjust for over-reporting. MIDAS responses were compared to the maximum number of days possibly impacted by headache based on total number of headache days reported at screening. That is, if a participant reports 10 headaches days, the maximum MIDAS score is 30. If they report 20 headache days, the maximum score is 60. Approximately 11% of MIDAS scores were adjusted to bring them into ranges consistent with their reported headache frequencies. Less than 5% of cases were deemed to be outliers (difference between their original MIDAS score and their capped score ≥18; N = 445), and in the absence of further information regarding the participant’s condition, these cases were excluded from analyses that included the MIDAS.
Division of MIDAS grade IV. MIDAS was originally developed and validated for use with a general headache sample. The standard MIDAS grading system groups patients with scores of 21 and above into a single category of “severe disability”. While this division works well for EM, a very high proportion of persons with chronic migraine fall into grade IV. Accordingly, for this study, we subdivided the most severe category of grade IV to allow for a finer examination of variation within the most severely disabled group. The new cutoff point for the highest grade was based on the distribution of scores in the CM and EM groups (approximately half of grade IV) to ensure sufficient sample of CM and EM participants within the highest category, and the highest category was divided into grade IV-A, severe disability (scores of 21–40) and grade IV-B, very severe (scores of 41–270).
Migraine-Specific Quality of Life Questionnaire, version 2.1
The MSQ v2.1 (37) is a 14-item questionnaire designed to measure how migraines affect and/or limit daily performance. It is divided into three domains: “Role Function-Restrictive” assesses how migraines limit a sufferer’s daily social- and work-related activities; “Role Function-Preventive” assesses how migraines prevent such activities; and “Emotional Function” assesses the emotions associated with migraine. Raw dimension scores are computed as a sum of item responses and rescaled to a 0–100 scale, where higher scores indicate better HRQoL.
Patient Health Questionnaire-4 Items
Depression and anxiety were measured using the Patient Health Questionnaire–4 Items (PHQ-4) (38)
Health care resource utilization
Participants were asked about the frequency of resources used over the previous three months for health care professional visits, use of emergency department or urgent care clinic, overnight hospital stay, diagnostic tests, and any headache-specific treatments used including pharmacologic and non-pharmacologic interventions.
Statistical analysis
Comparisons of sociodemographic and of clinical characteristics between EM and CM were evaluated based on Chi-square tests for categorical variables and two-sample t-tests for continuous variables.
A descriptive summary of the headache-related disability, HRQoL, and psychiatric comorbidities was examined in a similar way. Multivariate models (linear and ordinal logistic regression, as appropriate) were also examined to evaluate differences between EM and CM controlling for covariates of age, gender, country, education and comorbidities.
A descriptive summary of the number and proportion of participants using resources and their frequency of usage was performed. Poisson regression models were used to examine the relationships between frequency of emergency department visits, hospitalization, consultations with neurologists and primary care provider visits. Multivariate models included CM or EM status, age, gender, country, education and comorbidities. Because hospitalizations were relatively rare in the sample, these were combined with emergency department visits (by summing frequency of use of each resource) for multivariate analyses. Associations in these analyses were measured in terms of ratio of average rate of usage with 95% confidence intervals (CIs).
Role of the funding source
This study was funded by Allergan, Inc. The study sponsor was involved in the study design, data collection, data analysis, data interpretation and the writing of the article. AB, SV, TW and AK were involved in development of the Web-based questionnaire. The initial first drafts of the introduction and discussion sections of this paper were written by AB and SV. The initial first drafts of the methods and results sections were written by AK and TW. All authors had full access to all of the data. The corresponding author had final responsibility for submission of this paper.
Results
Sample characteristics
Frequency of chronic migraine and episodic migraine by country

Proportion is expressed as percent of overall CM and EM samples.
Sociodemographic characteristic of participants with chronic migraine and episodic migraine

CM = chronic migraine. EM = episodic migraine. SD = standard deviation. BMI = body mass index.
White/Caucasian group includes Arab and Middle Eastern participants.
Proportions are based on country-specific median income.
p values were based on Chi-square tests for categorical variables and two-sample t-tests for continuous variables.
The CM and EM groups differed in ethnic composition (p < .001), with a relatively greater number of Caucasian and Hispanic participants and a lower number of Asian participants meeting criteria for CM. The CM and EM groups also differed in employment status (p < .0001); fewer CM were employed full-time (34.9% vs. 49.0%) compared to EM. There were no meaningful differences between the groups in education (∼60% with more than a high-school education for both groups) or those with household income above the median (31.7% CM and 35.5% EM).
Comorbidities with chronic migraine and episodic migraine

CM = chronic migraine. EM = episodic migraine. SD = standard deviation.
p values were based on Chi-square tests for categorical variables and two-sample t-tests for continuous variables.
Mean represents mean number of comorbidities per participant reported for that group.
Clinical presentation
Clinical characteristics of participants with chronic migraine and episodic migraine


CM = chronic migraine. EM = episodic migraine. SD = standard deviation.
p values were based on chi-square tests for categorical variables and two-sample t-tests for continuous variables.
Based on MIDAS Question 7 (0–10 pain scale), where 0 = no pain at all and 10 = pain as bad as it can be.
MIDAS classification (headache disability)
The MIDAS classification of the participants indicated that more than a quarter (29.4%; N = 2432) met MIDAS grade I, with those classified as grade III, 23.1% (N = 1910), being the next highest. The remaining participants were classified in grade II, 20.8% (N = 1726); grade IV-A, 17.1% (N = 1420); or grade IV-B, 9.6% (N = 793). The distribution across the MIDAS quintiles varied considerably depending on the number of headache days (Figure 2). CM participants had significantly higher mean MIDAS scores (least-squares [LS] mean [standard error {SE}]: CM 72.57 [0.93] vs. EM 14.49 [0.23], p < .0001), indicating more severe disability compared to EM participants (Table 5). This finding remained even when accounting for age, gender, country, education and comorbidity differences between the groups (LS mean [SE]: CM 67.67 [1.04] vs. EM 13.57 [0.46], p < .0001). The proportion of participants (adjusting for covariates) with MIDAS scores above 21 (grade IV-A, severe or IV-B, very severe) were 78.0% for CM, compared to 23.3% for EM.
Percentage of respondents in Migraine Disability Assessment Scale (MIDAS) grades by monthly headache frequency. Impact and psychiatric comorbidities associated with chronic migraine and episodic migraine CM, chronic migraine; EM, episodic migraine; MIDAS, Migraine Disability Assessment; LS, least-squares; SE, standard error; MSQ v 2.1, Migraine Specific Quality of Life Questionnaire, version 2.1; PHQ-4, Patient Health Questionnaire-4 Items; CI, confidence interval. Unadjusted models include migraine group only. Adjusted models include migraine group and covariates: age, gender, country, education and comorbidities (pain, vascular disease [VD] risk factors, VD events, psychiatric disorders, other conditions). CM vs. EM comparison from linear regression models (analysis of covariance [ANCOVA]) for MIDAS, MSQ v2.1 and PHQ-4 were continuous scores and ordinal logistic regression models for MIDAS and PHQ-4 were categorical groupings. Frequency of headache-related disability over the previous 3 months using MIDAS. MIDAS quintiles were categorized as follows: 0-5 (grade I), 6-10 (grade II), 11-20 (grade III). 21-40 (grade IV-A) and 41-270 (grade IV-B). PHQ-4 scores were categorized as follows: 0-2 (normal), 3-5 (mild), 6-8 (moderate), 9-12 (severe). The adjusted model for PHQ-4 did not include psychiatric comorbidity.

Migraine-specific quality of life
CM was associated with consistently lower MSQ Role Function-Preventive, Role Function-Restrictive, and Emotional Function scores in both univariate and multivariate analyses (Table 5). Although adjustment for other covariates reduced the magnitude of differences in scores between CM and EM sufferers, these differences remained statistically significant (p < .0001). All else being equal, MSQ scores for chronic migraineurs were, on average, 6.0 points lower than episodic migraineurs on the Function-Preventive subscale, 7.9 points lower on Function-Restrictive, and 13.5 points lower on Emotional Function (Figure 3).
MSQ v2.1 adjusted and unadjusted subscale scores by migraine group. MSQ v2.1 = Migraine-Specific Quality of Life Questionnaire version 2.1.
Anxiety and depression
Anxiety and depression, as measured by the PHQ-4, was worse for participants with CM compared to EM (LS mean scores [SE]: CM 5.63 [0.14] vs. EM 3.76 [0.04], p < .001). Again, although adjustment for other covariates reduced the magnitude of differences in scores between CM and EM, these differences remained statistically significant (p < .001) (Table 5). Moderate to severe anxiety and depression (PHQ-4 scores >5) was more prominent for CM compared to EM (47.0% vs. 25.1%, adjusted model).
Resource utilization
Resource utilization for participants with chronic migraine and episodic migraine
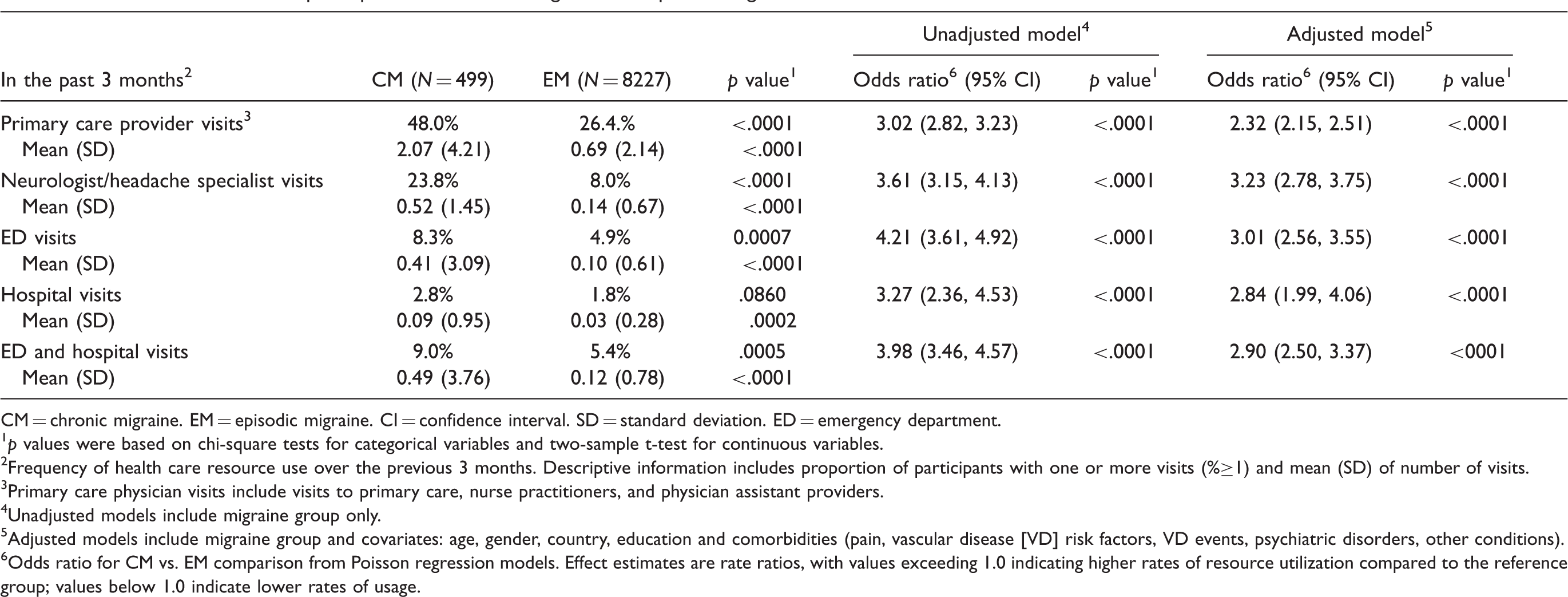
CM = chronic migraine. EM = episodic migraine. CI = confidence interval. SD = standard deviation. ED = emergency department.
p values were based on chi-square tests for categorical variables and two-sample t-test for continuous variables.
Frequency of health care resource use over the previous 3 months. Descriptive information includes proportion of participants with one or more visits (%≥1) and mean (SD) of number of visits.
Primary care physician visits include visits to primary care, nurse practitioners, and physician assistant providers.
Unadjusted models include migraine group only.
Adjusted models include migraine group and covariates: age, gender, country, education and comorbidities (pain, vascular disease [VD] risk factors, VD events, psychiatric disorders, other conditions).
Odds ratio for CM vs. EM comparison from Poisson regression models. Effect estimates are rate ratios, with values exceeding 1.0 indicating higher rates of resource utilization compared to the reference group; values below 1.0 indicate lower rates of usage.
Analyses based on the frequency of use for these health care resources revealed a similar pattern to the other outcomes. In univariate analyses for each health care resource type (adjusting for sociodemographic variables and comorbidities), the odds ratio of experiencing primary care provider (PCP) visits was 2.32 (95% CI: 2.15–2.51) for CM vs. EM, 3.23 (2.78–3.75) for neurologist visits, 3.01 (2.56–3.55) for emergency department visits and 2.84 (1.99–4.06) for hospitalizations (Table 6).
Discussion
Migraine is a common condition associated with high levels of headache-related disability. Our study in 8726 people with migraine confirms the burden of both EM and CM in many countries around the world. In addition, the demographic and comorbidity data in this study show that CM patients are more likely than EM patients to be overweight, unemployed and more depressed and anxious. This multinational study suggests that CM is significantly more disabling than EM, impairing HRQoL and imposing a greater burden on the individual and the health care system. This is consistent with country-specific findings from previous clinic- and population-based studies, in which CM patients sufferered greater impairment in occupational, educational, family and social aspects of life, a poorer quality of life (42), more medical and psychiatric comorbidities (21), and greater perceived frustration and burden due to migraine compared to episodic migraineurs (28,43). Headache intensity and headache features, such as pain intensity, have been shown in other studies to predict headache-related disability (44). In a French population-based survey, HRQoL scores were correlated with headache frequency, severity and headache-related disability (45). In two US studies and one UK study, people who reported “moderate”, “severe” and “very severe” migraines (based on pain intensity) scored significantly lower (worse state) on the HRQoL scale than the general population (46,47).
Relieving and/or preventing pain and headache-related disability are the primary goals of treatment and as expressed in treatment guidelines (48). As a consequence, high levels of headache-related disability reflect unmet treatment needs. In this study, we found that participants with CM reported higher levels of headache-related disability and health care resource utilization (e.g. physician visits and emergency care), far greater than those with EM. This finding is consistent with previous country-specific reports (23,28,49).
One of the important considerations when establishing treatment goals is clinical presentation. In this study, in addition to headache frequency, participants with CM report headaches of longer duration, more severe pain and greater pain intensity. Additionally, headache features of moderate to severe pain, throbbing, photophobia and phonophobia occur more often. Thus, associated symptoms are more prominent in the CM than the EM population. Similar findings were present in the American Migraine Prevalence and Prevention (AMPP) study (22). This suggests that as migraine chronification occurs, clinical features of the individual attack still remain prominent. Identification of these headache parameters during the clinical assessment may provide more insight for establishing a treatment plan.
It has been demonstrated that information about the disability associated with migraine attacks (or lack of) influences health care providers perceptions of illness severity, diagnosis, and treatment; and that fully assessing this impact improves migraine treatment (50). The five-grade MIDAS scoring system developed for this study allowed us to better characterize headache-related disability, particularly in the CM group. The traditional cutoff for “severe” disability (MIDAS grade IV- 21 points) may mask the worsening disability resulting from greater frequency of headaches. Further subdivision of the scale at a score of 41 points revealed an increasing proportion of respondents with scores above this threshold who experienced 12–19 headache days per month. This suggests that this revised MIDAS grading system using quintiles is closely aligned with the definition of CM and EM proposed by the ICHD-II. In addition, due to the greater number of days with headache, CM patients tended to double report on the MIDAS questions leading to falsely elevated scores. There was no double reporting associated with EM patients in this study or previous studies as the total number of days with disability is much fewer, so errors of this type are not detected.
This study has several potential limitations. Despite a moderate participation rate of 31%, the minimum sample of CM participants was reached in all countries except Taiwan. The low recruitment of Taiwanese participants may have been due the fact that participants were contacted from a general pool of survey participants rather than prescreened for headache or migraine. Electronic collection of the data may also introduce a participation bias because individuals lacking appropriate computer skills and/or access to resources would not have been contacted. Despite this, our results are broadly compatible with other studies with higher participation rates. The clinical information in this study including headache frequency, symptomology, and medical and psychiatric comorbidities was not based on medical records or health care provider diagnosis; rather it was based on respondent self-report. However, in clinical practice much of that data is collected as self-report, and we used validated tools for assessing headache-related disability and HRQoL.
A number of factors make this study unique. The strengths include the strong statistical power, achieved by its large sample size and multinational subject pool with participants from nine countries across North America, Europe and the Asia/Pacific region. Additionally, this study applied uniform methodology across study countries, using well-validated headache-related disability and HRQoL instruments and strong analytics. The results from this study extend our understanding of the international burden of chronic migraine, and could be used as a foundation for health policy assessment of health care management strategies for CM at a global level and allow for more direct country-to-country comparisons. The tools used to gather data in this study were shown to have universal application, and the data in this study could be generalized to other countries. Thus, countries with fewer resources for research can extrapolate from extensive study data that already exists or has been collected here, as well as from data that are likely to emerge in the future. Countries could use these data to make determinations about headache-related disability, HRQoL, medical and psychiatric comorbidities, health care resource allocation and health care management protocols. Finally, this study introduced a revised scoring system for the MIDAS, allowing for greater sensitivity in its use with CM. Further analysis of the data relating to disability and resource use may afford a clearer and more objective indication of the inflection point at which migraine chronification occurs.
The identification of disease-specific needs can be the first step in a broader plan to develop cost-effective health care management strategies. Multinational research studies such as this one can have a broad international impact on health care management and planning by shedding light on the needs of patients with migraine worldwide. Future work will include more detailed examination of country-specific differences, which may be in part due to health care systems, resource availability and other cultural factors.
Footnotes
Funding
This study was funded by Allergan, Inc., Irvine, CA, USA.
Disclosure
The sponsor and corresponding authors directed the research design and reviewed all major research decisions (i.e. study instruments, sampling and analyses). The authors had full access to all data and had final responsibility for the decision to submit for publication.
Contributors
AB participated in planning the study and interpreting the results. SV participated in planning the study, collecting the data and interpreting the results. TW and AK participated in planning the study, collecting the data, doing the analyses and interpreting the results. AM, PG and RL participated in planning the study and interpreting the results. AB, SV, TW and AK wrote the initial first draft of the article; the initial drafts of the introduction and discussion were written by AB and SV and the methods and results by TW and AK. All authors were involved in planning the article, critical review and editing of the first draft and subsequent revisions of the paper. All authors approved the manuscript before submission.
Conflict of interest statement
SV and AM are employees of Allergan, Inc. AB has received research grants from Allergan, Pfizer, GlaxoSmithKline (GSK) and Medtronic, has received honoraria from Allergan, Pfizer, GSK, Merck, Ortho-McNeil-Janssen Pharmaceuticals (OMP) and Medtronic, and has consultancy agreements with Allergan, Pfizer and GSK. TW and AK are employees of United BioSource Corporation. DB has received honoraria from Endo, Merck, Iroko, and MAP Pharmaceuticals, and has consultancy agreements with Endo and Allergan. PG has received research grants from MSD, Neuralieve, GSK and Johnson & Johnson and has received honoraria from and has consultancy agreements with Allergan, Almirall, BosSci, Eli Lilly, Endo, MAP, Medtronic, Minster, MSD, Neuralieve, Neuraxon and NeuroTherapeutics Pharma. RL has received research grants from Advanced Bionics, Allergan, GSK, Minster, Merck, Neuralieve, Novartis, OMP, Pfizer and ProEthics/KOWA, has received honoraria from Allergan, AstraZeneca, GSK, Merck, Novartis, OMP and Pfizer, and has consultancy agreements with Advanced Bionics, Allergan, Boehringer Ingelheim, Bristol-Myers Squibb, Cierra, Endo, Neuralieve, Novartis, OMP, Pfizer, Pozen and ProEthics/KOWA.
Acknowledgements
The authors wish to acknowledge Krista Payne (UBC) for her leadership contributions to design and execution and Dr Jack Ishak (UBC) for his contribution to data analysis and interpretation. The authors are also grateful to Karen Yeomans (UBC) for her ongoing study management and support on data collection forms design and implementation; Ren Yu (UBC) and Ye Tan (formerly with UBC) for data analysis and statistical support; Caroline Burk (Allergan) for her ongoing study management; Manasee V. Shah (formerly with Allergan) for study management and input on data collection forms; Synovate for study management and data collection form design; and IntraMed Educational Group (New York, NY, USA) for editorial support in the preparation and styling of this manuscript.
